Induction of Petite Colonies in Candida glabrate via Rose Bengal-Mediated Photodynamic Therapy
Summary
The significance of petite colonies in Candida spp. drug resistance has not been fully explored. Antimicrobial photodynamic therapy (aPDT) offers a promising strategy against drug-resistant fungal infections. This study demonstrates that rose bengal-mediated aPDT effectively deactivates Candida glabrata and induces petite colonies, presenting a unique procedure.
Abstract
Facing a 40% mortality rate in candidemia patients, drug-resistant Candida and their petite mutants remain a major treatment challenge. Antimicrobial photodynamic therapy (aPDT) targets multiple fungal structures, unlike antibiotics/antifungals, potentially thwarting resistance. Traditional methods for inducing petite colonies rely on ethidium bromide or fluconazole, which can influence drug susceptibility and stress responses. This study investigated the application of green light (peak 520 nm) and rose bengal (RB) photosensitizer to combat a drug-resistant Candida glabrata isolate. The findings revealed that aPDT treatment significantly inhibited cell growth (≥99.9% reduction) and effectively induced petite colony formation, as evidenced by reduced size and loss of mitochondrial redox indicator staining. This study provides initial evidence that aPDT can induce petite colonies in a multidrug-resistant C. glabrata strain in vitro, offering a potentially transformative approach for combating resistant fungal infections.
Introduction
Fungal infections, particularly those caused by Candida albicans and increasingly drug-resistant Candida glabrata, pose a serious global threat1. These infections can be deadly, especially for hospitalized patients and those with weakened immune systems. Rising antifungal resistance threatens the control of invasive candidiasis, a severe fungal infection with high mortality, especially from Candida albicans2. Resistant strains hinder effective treatment, potentially increasing both complexity and death rates. In Alameda County, California, USA, C. glabrata has become the most prevalent invasive species3. This shift in the prevalence and distribution of Candida species may be influenced by local healthcare practices, patient demographics, the utilization of antifungal agents, and the prevalence of risk factors for Candida infections.
Petite mutants in Candida, lacking functional mitochondria, reveal how this organelle affects drug response, virulence, and stress resistance4,5. C. glabrata readily forms these colonies, gaining sensitivity to polyenes while losing it to azoles6. Azole sensitivity and respiratory function are intricately linked, with diminished respiration leading to resistance via mitochondrial DNA loss7. Petite colonies of C. glabrata with azole resistance have been isolated from human stool samples from a bone marrow transplant recipient undergoing fluconazole treatment8 and from blood culture bottles of patients with bloodstream infections9. Their potential implications in drug resistance, virulence, and stress response highlight their clinical significance. Additionally, their distinct properties make them valuable tools for investigating fundamental questions in mitochondrial biology5. As research into petite mutants continues, their applications in both clinical and basic research are likely to expand.
This study discovered that photodynamic therapy (PDT) can induce petite colonies in C. glabrata, expanding the range of methods beyond the traditional techniques of exposing C. glabrata to ethidium bromide or fluconazole.
Protocol
1. Culturing of C. glabrata
NOTE: A multidrug-resistant C. glabrata (C2-1000907) that is resistant to most antifungal agents, including fluconazole, is used for the experiments. The experimental conditions may need to be adapted to the specific strain, as variations may exist among different strains. All experiments used log-phase Candida grown at 25 °C (mimicking natural infection) for consistency. C. glabrata's lack of hyphae simplifies quantification compared to C. albicans, which forms hyphae at 37 °C10.
- To prepare a log-phase culture of C. glabrata, pick a single colony from an agar plate and transfer it to a glass test tube with 3 mL of sterile yeast peptone dextrose (YPD) medium (see Table of Materials). Incubate for 14-16 h at 25 °C with shaking (155 rpm, 45° angle to increase air transmission to the medium).
- After incubation, dilute the culture with fresh YPD to an OD600 of 0.1 using sterile technique. Incubate at 25 °C for 6 h with shaking at 155 rpm. Verify the log phase of C. glabrata by measuring the OD600. Aim for 0.65-1.00, which corresponds to 1 × 107-1.5 × 107 cells/mL.
2. Induction of petite colonies by ethidium bromide, fluconazole and photodynamic therapy
- Ethidium bromide petite colony induction
- Adjust a yeast suspension to an OD600 of 0.1 (5 × 106 cells/mL) with YPD to a final volume of 3 mL, then add 30 µL of ethidium bromide stock (10 mg/mL) (see Table of Materials) for a final concentration of 100 µg/mL.
- Incubate the yeast suspension overnight (16-18 h) at 25 °C, 155 rpm, 45° angle. Adjust to an OD600 of 0.65 (1 × 107 cells/mL). Perform a 10-fold dilution series in a 96-well plate by adding 20 µL of the adjusted yeast suspension to 180 µL of PBS per well. This creates six dilutions ranging from 10-1 to 10-5.
- Select 4 dilution factors (e.g., 100, 101, 102, and 103) and plate 3 drops (20 µL) each on 4 quadrants of YPD agar plates (triplicate).
- Incubate plates overnight at 37 °C. Select quadrants with 5-80 colonies from the previously chosen dilutions for triphenyltetrazolium chloride (TTC) staining11 (see step 3). Scan plates at 1200 dpi using a 48-bit full-color optical scanner.
- Fluconazole petite colony induction
NOTE: To expedite the process, use T3 cells (a mixture of normal and petite cells obtained after 3 RB-PDT treatments; see step 2.3) for fluconazole-induced petite colony formation. This is because standard treatment typically results in slower formation.- Prepare and adjust a yeast cell suspension as described in step 2.1.1.
- Incubate overnight (16-18 h) at 25 °C. Adjust the OD600 to 0.1 again.
- Transfer 1 mL of the adjusted suspension to a sterile 5 mL tube.
- Dip a sterile cotton swab (15 cm long, with a 0.9 cm x 2.6 cm tip, see Table of Materials) in the yeast suspension, ensuring contact with the tube bottom and twisting to remove excess liquid from the tube wall.
- Prepare the plates in the hood using Mueller-Hinton agar (see Table of Materials).
- Swab the cotton back and forth on the agar, rotate 60° twice, then swab the perimeter for even coverage.
- Sterilize forceps over a flame for 1-2 s, allowing them to cool briefly, then use them to pick up the blank disks.
- Divide the plate into three equal sectors using a marker. Place one blank disk in the center of each sector.
- Add 12.5 µL fluconazole stock (2 mg/mL, see Table of Materials) to each disk (25 µg/disk), mix thoroughly to avoid bubbles, and incubate at 37 °C for 20-24 h.
- Perform the TTC staining (see step 3). Scan plates at 1200 dpi using a 48-bit full-color optical scanner.
- PDT petite colony induction
NOTE: Different fungi may produce different numbers of petite colonies after PDT. Some strains may not produce any at all.- Prepare the aPDT system.
NOTE: The setup procedure for the aPDT system device follows the method reported by Hung et al12. Briefly, the aPDT system consists of a green LED array with a peak at 520 nm (see Table of Materials), shining light from the bottom with good alignment with each well of a 96-well plate. - Adjust the yeast cell suspension in the YPD medium to an OD600 of 0.65 (about 1 × 107 cells/mL).
- Mix 1 mL of the yeast suspension with 111 µL of 2% rose bengal (RB, Figure 1) (see Table of Materials) in a round-capped tube to give a final RB concentration of 0.2%. Incubate the mixture at 25 °C for 15 min, rotating it at a 45° angle at 155 rpm.
- Transfer the mixture to a 1.5 mL microcentrifuge tube and centrifuge at 16,100 x g for 2.5 min (at room temperature).
- Discard the supernatant and gently scrape the tube five times on the hood floor to resuspend the pellet.
- Wash the suspension with 1x PBS four times to remove all the RB. Each wash is followed by resuspension of the pellet with brief vortexing or pipetting for thorough resuspension.
- After the final wash, add 1000 µL of PBS. The solution is light pink in color. Transfer 200 µL of the washed RB-loaded yeast suspension to each of three wells in a 96-well plate (triplicate).
- Position the 96-well plate on the photodynamic light system with LED bulbs shining green light from below. Turn off the room lights to ensure uniform illumination and prevent interference. Activate the LED light system to deliver the PDT dose of 4.38 J/cm2 over 2 min; ensure the system is properly aligned with the wells.
- After irradiation, dilute the yeast suspension in a 96-well plate. Add 20 µL of yeast suspension to a well containing 180 µL PBS, creating a 10-fold dilution. Repeat for the remaining dilutions to achieve a range of 10-1 to 10-5.
- Choose four dilution factors (e.g., 10-2 to 10-5) based on the yeast cell concentration adjustment.
- For each dilution factor, plate 3 drops (20 µL each) per quadrant on YPD agar plates.
- After the yeast suspension is completely absorbed, around 10 min by the agar plates, invert and incubate overnight at 37 °C.
- Define T0 as the control parental C. glabrata without PDT treatment. Prepare T0 fungi in the log growth phase as described above and expose them to 4.38 J/cm2 green light in the presence of 0.2% RB. This PDT condition consistently inhibits 3 to 3.5 logs of fungal growth.
- Denote the survived fungi as T1 and expose them to the same PDT dose again after they are expanded in vitro to a log growth phase. The survived fungi after the second PDT are denoted as T2 and so forth.
- On the next day, choose quadrants with colony counts between 5 and 80. Count the number of colonies in each quadrant and calculate the titer with the following formula: Colony-forming unit (CFU)/mL = Numbers of colonies (average of triplicate) × Dilution factor × 50.
- Perform the TTC staining (see step 3). Scan the plate as described earlier (step 2.2).
- Prepare the aPDT system.
3. Mitochondrial function analysis (TCC staining test)
NOTE: TTC is a redox indicator and an electron acceptor. It turns red when white compounds are broken by electrons11. Note that not all cells necessarily form visible colonies within 24 h after culture on an agar plate. Colonies with functional mitochondria turn red, while those with non-functional mitochondria remain white. This allows differentiation between colonies with different mitochondrial functionality.
- After treatment, dilute Candida suspensions on YPD agar plates according to the desired cell density to obtain distinct colonies for easy visualization and staining.
- After 24 h of growth at 37 °C, visible colonies are formed from a single cell. Pipette 20 µL of 20% TTC directly onto the center of each colony.
- After complete absorption, around 10 min of TTC by the colonies, incubate at 37 °C for 30-40 min.
4. Growth kinetics of normal and petite C. glabrata
NOTE: Three strains of yeast were compared: C. glabrata C2-1000907 T0 (clinical isolate without PDT treatment), T3n (C. glabrata C2-1000907 after 3 consecutive RB-PDT exhibiting colonies with an average diameter of 1.5 ± 0.8 mm similar to the parental cells), and T3p (petite colonies of C. glabrata C2-1000907 after 3 consecutive RB-PDT).
- Adjust the yeast cell suspension in YPD medium to an OD600 of 0.1 (5 × 106 cells/mL) in a 3 mL tube.
- Automatically measure the OD600 of the statical culture every 20 min using the multi-mode microplate reader (see Table of Materials) for 24 h.
Representative Results
The data is presented as the mean with ± standard error and was obtained from three independent experiments, with at least triplicates in each group. Experimental data, including colony counts, OD600 measurements, and TTC staining results, were graphed and statistically analyzed using graphing and statistical software (see Table of Materials). One-way ANOVA or t-test was used to analyze the data, and a p-value <0.05 was considered significant. Scanning was performed with a 48-bit full-color optical scanner at a resolution of 1200 dpi, and subsequent measurements of the colony diameters were conducted using ImageJ software.
As depicted in the growth curves in Figure 2, petite mutants of C. glabrata C2-1000907 (T3p) exhibited slower growth compared to the untreated parental fungi of regular size (T0). When C. glabrata was mixed with 0.2% RB for 15 min and exposed to 4.38 J/cm2 green light (PDT treatment), the titer of the fungi decreased from 107.5 CFU/mL to 104.5 CFU/mL (at least 3 logs, Figure 3) compared with the control group (RB only), which had a cell density of around 107 CFU/mL. Additionally, after aPDTs, petite colonies were observed. These petite colonies had an average size of 0.4 mm ± 0.25 mm instead of the usual size of 1.5 mm ± 0.8 mm (Figure 4A). Small-size colonies with mitochondrial dysfunction can be identified by their size and color after staining with 20% TTC. The petite colonies that maintained a white color (Figure 3B, white arrows) exhibited mitochondrial dysfunction, while some smaller red-colored colonies and normal-sized colonies retained normal mitochondrial function (Figure 4B). The traditional DNA intercalating agent ethidium bromide (Figure 4C) and fluconazole (Figure 4D) can effectively induce petite mutants of Candida. Disk diffusion is a simple method to define drug susceptibility in fungi. In Figure 4D, a clear circular zone of no growth surrounded the fluconazole disk containing 25 µg fluconazole in the T0 culture (without PDT), indicating less drug resistance in this parental fungus. Nonetheless, the C. glabrata C2-1000907 is still defined as a resistant strain by minimal inhibition concentration toward fluconazole (data not shown) and the diameter of the clear zone. In the T3 culture, many petite colonies formed near the disk (arrow, lower panel), suggesting drug resistance after three repeated PDT treatments.
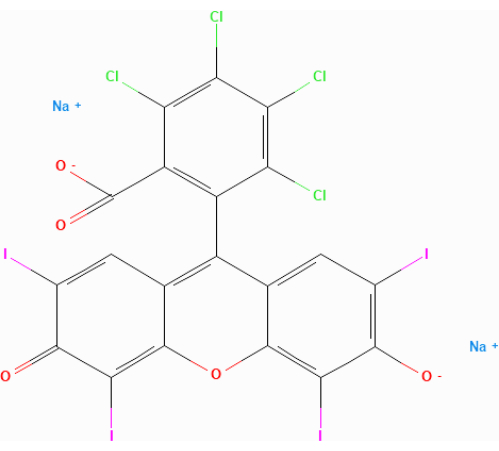
Figure 1: Molecular structure of rose Bengal (RB). Please click here to view a larger version of this figure.
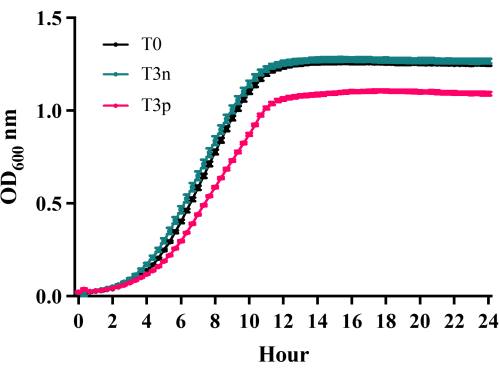
Figure 2: Growth Curves of C. glabrata C2-1000907 24 h following repetitive RB-PDT. Growth curves were compared for three strains: C. glabrata C2-1000907 T0 (naïve parental cells), T3n (colonies with normal size after 3 repeated treatments), and T3p (petite colonies after 3 repeated treatments). Compared to T0 and T3n, T3p colonies exhibited a significantly slower growth rate (n = 3, p < 0.05). All cells are grown in YPD medium. Please click here to view a larger version of this figure.
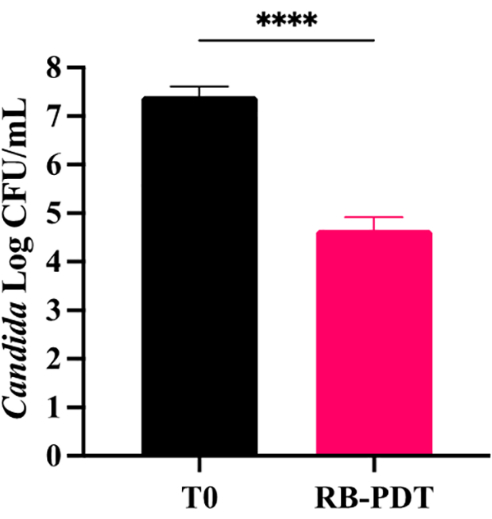
Figure 3: Growth Inhibition of C. glabrata C2-1000907 following aPDT. A single aPDT treatment with a 4.38 J/cm² light dose and 0.2% RB significantly inhibited C. glabrata C2-1000907 growth by 3 logs compared to the naïve control (p < 0.0001, unpaired t-test). Error bars represent mean ± SEM, data is pooled from 3 independent experiments. Please click here to view a larger version of this figure.

Figure 4: Mitochondrial dysfunction analysis using triphenyltetrazolium chloride (TCC) staining. (A) C. glabrata C2-1000907, 24 h following aPDT treatment (0.2% RB, 4.38 J/cm² green light), displayed a bimodal colony size distribution, with large (1.5 mm ± 0.8 mm) and small (petite) colonies (0.4 mm ± 0.25 mm). (B) TCC staining revealed that the large colonies were functional, as they turned red, while the petite colonies were non-functional, as they remained white. (C) Petite colonies were also induced by ethidium bromide (100 µg/mL for 30-40 min) and (D) fluconazole (25 µg/mL) treatment. A clear circular zone of no growth surrounded the fluconazole disk containing 25 µg fluconazole in the T0 culture (without PDT), indicating susceptibility. In the T3 culture, many petite colonies formed near the disk (arrow, lower panel), suggesting drug resistance after three repeated PDT treatments. Scale bars: 1 mm. Please click here to view a larger version of this figure.
Discussion
This study unveils PDT as the first reported method to induce petite colony formation in Candida, surpassing the established effects of ethidium bromide and fluconazole. This novel observation necessitates further exploration to unravel its implications for both fungal eradication by decreasing virulence and the emergence of resistance mechanisms.
RB-mediated PDT effectively inhibits the growth of C. glabrata, suggesting a potential alternative treatment approach for Candida infections. As a light-activated photosensitizer, RB produces singlet oxygen and reactive oxygen species (ROS) when exposed to a specific wavelength. This oxidative stress causes damage to essential cellular components such as lipids, proteins, and nucleic acids13, leading to mitochondrial dysfunction, as shown in the present study. This makes it difficult for Candida to develop resistance through conventional mechanisms observed with standard antifungal drugs.
The three yeast strains used in this study, T0, T3n, and T3p, exhibit distinct growth patterns. T0 remains untreated, whereas T3n and T3p undergo RB-PDT three times. T3n displays normal-sized colonies, whereas T3p exhibits petite colonies. The slower growth of the petite colonies may indicate alterations in cellular energy and metabolism. The petite phenotype, often associated with mitochondrial dysfunction4, could lead to impaired aerobic respiration, consequently influencing the overall growth kinetics. The impaired aerobic respiration in petite colonies might affect their virulence, drug susceptibility, or ability to persist in different environments. This connection holds potential clinical implications and provides avenues for future research directions.
Ethidium bromide, a DNA intercalating agent, inhibits mitochondrial DNA synthesis and degrades existing mitochondrial DNA, transforming Candida into petite colonies with mitochondrial dysfunction14. Fluconazole, commonly used to treat Candida infections, can induce drug resistance and petite colony development in C. glabrata6. Petite mutants may exhibit azole drug resistance through the activation of the transcription factor PDR1 and its regulation of genes CDR1 and CDR215. Moreover, due to the partial or complete loss of mitochondrial DNA, mitochondrial function is compromised16.
PDT has demonstrated efficacy in reducing bacterial virulence17, inducing susceptibility to antibiotics18, and reducing biofilm formation19 in bacteria. Studies on PDT against fungal infections are limited20. This discovery raises intriguing questions about how RB-PDT affects mitochondria in C. glabrata. To fully comprehend how RB-PDT impacts yeast cells, understanding the molecular mechanisms causing growth variations is crucial. However, petite mutants induced by PDT are currently understudied, and their detailed mechanistic underpinnings remain unclear. Further investigation is needed to determine if there are any differences between small colonies created by various methods. Understanding how petites form in Candida species is important for studying the relationship between mitochondrial function and pathogenicity. Furthermore, petite mutants can serve as a model for exploring antifungal drug resistance mechanisms.
The current study, while providing valuable insights into the effects of RB-mediated PDT on C. glabrata and the emergence of petite colonies, has certain limitations that should be considered. Firstly, the study primarily focused on in vitro experiments, and the translation of these findings to clinical settings warrants further investigation. Additionally, the specific molecular mechanisms underlying the formation of petite colonies following RB-PDT and their potential implications for antifungal drug resistance require more in-depth exploration. Furthermore, while the study compared the growth patterns of different yeast strains, additional molecular and genetic analyses could provide a deeper understanding of the metabolic and genetic changes associated with petite colony formation.
Moreover, the potential variability in response to RB-PDT among different clinical isolates of C. glabrata was not addressed in this study, highlighting the need for broader strain-specific investigations. Future studies addressing these limitations will be crucial for a more comprehensive understanding of the implications of RB-mediated PDT and the emergence of petite colonies in the context of antifungal treatment strategies.
In summary, this investigation reveals that aPDT induces C. glabrata (C2-1000907) petite mutants with impaired mitochondrial function. This unique mechanism might offer a new method for antifungal studies. The precise mechanism of action underlying PDT-induced petite colonies remains to be elucidated, and exploring potential differences from other methods will be crucial for optimizing therapeutic strategies.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work has received funding from the Ministry of Science and Technology, Taiwan [MOST 110-2314-B-006-086-MY3], National Cheng Kung University [K111-B094], [K111-B095], National Cheng Kung University Hospital, Taiwan [NCKUH-11204031], [NCKUMCS2022057].
Materials
| 0.22 μm filter | Merck, Taipei, Taiwan | Millex, SLGVR33RS | |
| 1.5 mL microfuge tube | Neptune, San Diego, USA | #3745 | |
| 20% Triphenyltetrazolium chloride (TTC) | Sigma-Aldrich, MO, USA | T8877 | |
| 5 mL polypropylene round bottom tube | Corning, AZ, USA | 352059 | |
| 5 mL round-bottom tube with cell strainer cap | Corning, AZ, USA | Falcon, #352235 | |
| 96-well plate | Alpha plus, Taoyuan Hsien, Taiwan | #16196 | |
| Agar | BRS, Tainan, Taiwan | AG012 | |
| Blank disk | Advantec, Tokyo, Japan | 49005040 | |
| Centrifuge | Eppendorf, UK | 5415R | |
| Ethidium bromide solution | Sigma-Aldrich, MO, USA | E1510 | |
| Fluconazole, 2 mg/mL | Pfizer, NY, USA | BC18790248 | |
| GraphPad Prism | GraphPad Software | Version 7.0 | |
| Green light emitting diode (LED) strip | Nanyi electronics Co.,Ltd, Tainan, Taiwan | 5050 | Excitation wave: 500~550 nm |
| Low Temperature. shake Incubators | Yihder, Taipei, Taiwan | LM-570D (R) | |
| Mouth care cotton swabs | Good Verita Enterprise, Taipei, Taiwan | 161357 | |
| Muller Hinton II agar | BD biosciences, California, USA | 211438 | |
| Multimode microplate reader | Molecular Devices | SpectraMax i3x | |
| OD600 spectrophotometer | Biochrom, London, UK | Ultrospec 10 | |
| Rose Bengal | Sigma-Aldrich, USA | 330000 | stock concentration 40 mg/mL = 4%, prepare in PBS, stored at 4 °C |
| Sterilized glass tube | Sunmei, Tainan, Taiwan | AK45048-16100 | |
| Yeast Extract Peptone Dextrose Medium | HIMEDIA, India | M1363 |
References
- Soriano, A., et al. Invasive candidiasis: current clinical challenges and unmet needs in adult populations. J Antimicrob Chemother. 78 (7), 1569-1585 (2023).
- Pappas, P. G., Lionakis, M. S., Arendrup, M. C., Ostrosky-Zeichner, L., Kullberg, B. J. Invasive candidiasis. Nat Rev Dis Primers. 4, 18026 (2018).
- Meyahnwi, D., Siraw, B. B., Reingold, A. Epidemiologic features, clinical characteristics, and predictors of mortality in patients with candidemia in Alameda County, California; a 2017-2020 retrospective analysis. BMC Infect Dis. 22 (1), 843 (2022).
- Whittaker, P. A. The petite mutation in yeast. Subcell Biochem. 6, 175-232 (1979).
- Hatab, M. A., Whittaker, P. A. Isolation and characterization of respiration-deficient mutants from the pathogenic yeast Candida albicans. Antonie Van Leeuwenhoek. 61 (3), 207-219 (1992).
- Defontaine, A., et al. In-vitro resistance to azoles associated with mitochondrial DNA deficiency in Candida glabrata. J Med Microbiol. 48 (7), 663-670 (1999).
- Brun, S., et al. Relationships between respiration and susceptibility to azole antifungals in Candida glabrata. Antimicrob Agents Chemother. 47 (3), 847-853 (2003).
- Bouchara, J. P., et al. In-vivo selection of an azole-resistant petite mutant of Candida glabrata. J Med Microbiol. 49 (11), 977-984 (2000).
- Badrane, H., et al. Genotypic diversity and unrecognized antifungal resistance among populations of Candida glabrata from positive blood cultures. Nat Commun. 14 (1), 5918 (2023).
- Shantal, C. -. J. N., Juan, C. -. C., Lizbeth, B. -. U. S., Carlos, H. -. G. J., Estela, G. -. P. B. Candida glabrata is a successful pathogen: An artist manipulating the immune response. Microbiol Res. 260, 127038 (2022).
- Gamarra, S., Mancilla, E., Dudiuk, C., Garcia-Effron, G. Candida dubliniensis and Candida albicans differentiation by colony morphotype in Sabouraud-triphenyltetrazolium agar. Rev Iberoam Micol. 32 (2), 126-128 (2015).
- Hung, J. H., et al. Rose bengal-mediated photodynamic therapy to inhibit Candida albicans. J Vis Exp. (181), e63558 (2022).
- Cardoso, D. R., Franco, D. W., Olsen, K., Andersen, M. L., Skibsted, L. H. Reactivity of bovine whey proteins, peptides, and amino acids toward triplet riboflavin as studied by laser flash photolysis. J Agric Food Chem. 52 (21), 6602-6606 (2004).
- Hall, R. M., Trembath, M. K., Linnane, A. W., Wheelis, L., Criddle, R. S. Factors affecting petite induction and the recovery of respiratory competence in yeast cells exposed to ethidium bromide. Mol Gen Genet. 144 (3), 253-262 (1976).
- Chen, X. J., Clark-Walker, G. D. The petite mutation in yeasts: 50 years on. Int Rev Cytol. 194, 197-238 (2000).
- Piskur, J. Inheritance of the yeast mitochondrial genome. Plasmid. 31 (3), 229-241 (1994).
- Wong, T. W., Wang, Y. Y., Sheu, H. M., Chuang, Y. C. Bactericidal effects of toluidine blue-mediated photodynamic action on Vibrio vulnificus. Antimicrob Agents Chemother. 49 (3), 895-902 (2005).
- Wong, T. W., et al. Indocyanine green-mediated photodynamic therapy reduces methicillin-resistant Staphylococcus aureus drug resistance. J Clin Med. 8 (3), 411 (2019).
- Warrier, A., Mazumder, N., Prabhu, S., Satyamoorthy, K., Murali, T. S. Photodynamic therapy to control microbial biofilms. Photodiagnosis Photodyn Ther. 33, 102090 (2021).
- Hung, J. H., et al. Recent advances in photodynamic therapy against fungal keratitis. Pharmaceutics. 13 (12), 2011 (2021).

