Electrode Positioning and Montage in Transcranial Direct Current Stimulation
Summary
Transcranial direct current stimulation (tDCS) is an established technique to modulate cortical excitability1,2. It has been used as an investigative tool in neuroscience due to its effects on cortical plasticity, easy operation, and safe profile. One area that tDCS has been showing encouraging results is pain alleviation 3-5.
Abstract
Transcranial direct current stimulation (tDCS) is a technique that has been intensively investigated in the past decade as this method offers a non-invasive and safe alternative to change cortical excitability2. The effects of one session of tDCS can last for several minutes, and its effects depend on polarity of stimulation, such as that cathodal stimulation induces a decrease in cortical excitability, and anodal stimulation induces an increase in cortical excitability that may last beyond the duration of stimulation6. These effects have been explored in cognitive neuroscience and also clinically in a variety of neuropsychiatric disorders – especially when applied over several consecutive sessions4. One area that has been attracting attention of neuroscientists and clinicians is the use of tDCS for modulation of pain-related neural networks3,5. Modulation of two main cortical areas in pain research has been explored: primary motor cortex and dorsolateral prefrontal cortex7. Due to the critical role of electrode montage, in this article, we show different alternatives for electrode placement for tDCS clinical trials on pain; discussing advantages and disadvantages of each method of stimulation.
Protocol
1. Materials
- Check if you have all materials needed (Table 1, Figure 1).
TDCS devices should be battery-driven and function as a constant current stimulator with a maximum output in the miliAmps range. In some devices, the batteries can be chargeable. Constant voltage (voltage controlled) stimulators are not appropriate for tDCS. Using electrical outlets to power the device is not convenient or appropriate as mal-functioning devices might deliver large intensities of electrical currents with no warning. - Electrodes used for tDCS generally consist of a metal or conductive-rubber electrodes enclosed in a perforated sponge pocket which is saturated with an electrolytes (fluid with salt). Another possibility is the use of rubber electrode with conductive gel. Prolonged passage of direct current current across metallic electrode (where electrons from the stimulator are converted to ions carried through the body8) can produce undesired electrochemical products such as pH changes. The sponge pocket may act to physically separate, and thus buffer, the skin from electrochemical changes.
- For this reason, metal or rubber electrodes should never be placed on the skin during tDCS. Similarly during stimulation user should be vigilant against sponge dehydration and movement. A further related consideration is the durability and re-usability of tDCS electrodes. Our experience is that, especially when the polarity of electrodes is rotated, and proper stimulation conditions consistently maintained, rubber and metal electrodes can be re-used. The choice of electrolyte is discussed further below. From operating experience, it is recommended to use flat, and not too coarse perforated sponges, as they best absorb the electrolyte conduction solution and provide uniform skin contact8.
- There is the possibility to apply topical anesthetics. Especially for short-lasting stimulation, when ramping is not possible, it might prevent somatosensory perception and uncomfortable sensation arising from tDC stimulation. Another reason for using topical application of local anesthetics is to create best comparability between sham and active tDCS conditions, since no subject would feel if current is flowing or not and optimal blinding situation would be guaranteed. This approach is particular vulnerable when planning to use larger intensities as blinding might be less effective in this situation7. Though sensation/pain and skin irritation are not always correlated, excessive use of topical anesthetics might mask serious adverse effects such as burning.
In this guide we illustrate the most typical tDCS set-up for pain management: using conductive rubber electrodes, pocket type perforated sponges, both placed on the head, with no topical anesthetic.
2. Measurements
- Make sure the subject is seated comfortably.
- The area of stimulation will be found through the measurement of the scalp. Usually the convention of the EEG 10/20 system is used7. The site of stimulation depends on your experimental approach.
- Find localisation of the Vertex (Figure 2):
Measure the distance of nasion to inion and mark halfway using a skin marker.
Nasion – point between the forehead and nose, at the junction of the nasal bones (Figure 3).
Inion – most prominent point of the occipital bone (Figure 3).
Measure the distance between the pre-auricular points and mark halfway. Mark both halfway spots to find the Vertex. -
- To locate the primary motor cortex, or M1, use 20% of the auricular measurement and use this measurement from Cz through auricular line (to the side of the Vertex) (Figure 4). This spot should correspond to C3/C4 EEG location. This method of localization is enough given the focality of traditional large electrodes tDCS. For more focal tDCS, other methods of cortical localization might be needed.
- To locate the dorsolateral prefrontal cortex (DLPFC)9,10: One practical method is to measure five centimetres forward from the M1 location or to use the 10/20 EEG system. This should correspond to the F3 or F4 EEG location, as seen here (Figure 5). This method of determining the stimulation site is sufficient when using traditional tDCS electrodes. For more focal tDCS, other methods of cortical localization may be needed, such as neuronavigation.
3. Skin Preparation
- Inspect the skin for any pre-exiting irritation, cuts, or lesions – avoid stimulating over damaged skin and over skull lesions.
- To increase conductance, move hair away from the site of stimulation and clean the surface of the skin to remove any signs of lotion, dirt, etc. and allow it to dry. For subjects with thick hair, use of conductive gel may be necessary.
- If using re-usable electrodes, inspect the rubber insets and sponges for wear. Inspect the rubber insets and sponges for wear. If there is any evidence of deterioration, throw out the dirty components and use a new electrode.
4. Position electrodes
- After finding the site of stimulation and skin preparation you should place one of the elastic or rubber head straps around the head circumference. The elastic head strap should be placed under the inion as to avoid movement during stimulation. The elastic straps should be made of non-conducting material (or they will function as electrodes) and non-absorbent material (to avoid the straps absorbing fluid from the sponges).
- Each side of the sponges should be soaked with saline solution. For a 35 cm2 sponge, approximately 6 mL of solution per side may suffice (total of 12 mL per sponge). Be careful not to over soak the sponge (not excessively wet- there should be no water leaking; but also not dry as to have a good electrode contact). Avoid fluid leaking across the subject. You can use a syringe to add more solution if needed.
There is evidence that electrolyte solutions with lower NaCl concentrations(15 mM) are perceived as more comfortable during tDCS than those solutions with higher NaCl concentrations (220 mM)11,12. Since the ionic strength of deionised water is much less than that of all NaCl solutions, there is a significantly larger voltage required to carry current across the electrode and through the skin compared to NaCl solutions. Thus, it is recommended the use of solutions with moderate NaCl concentration, in the range 15 mM to 140 mM, as tDCS at these concentrations is more likely to be perceived as comfortable, requires moderately lower voltage while still allowing good conduction of current.11 The use of gels (adapted from applications such as EEG) has also been considered – a main limitation is the increased hassle of set-up clean-up following stimulation, without proven benefit regarding outcome when using perforated sponge electrodes. - Connect the cables to the device.
Consult with your stimulator operating manual on if the stimulator should be powered on before or after connecting positioned electrodes to the stimulator. Using all stimulators, the electrodes should not be disconnected or connected when current flow has been initiated. Ensure the connection polarity is correct as the effects of tDCS are highly polarity specific (typically, red indicates the Anode electrode, and black or blue indicates the Cathode electrode; this is the convention- but check with your device). Note that in the context of tDCS (and more broadly electrical stimulation in general), “Anode” always indicated the relative positive terminal where positive current flows intro the body, while “Cathode” indicates the relative negative terminal where the positive current then exits the body. - Insert the connector cord pin securely into the opening of the receptacle on the conductive rubber inset.
- Slide the conductive rubber inset into the sponge. The insulated portion of cable will protrude from the sponge-pocket opening. Ensure the entire conductive rubber inset is covered by the sponge and that there is no part of the connector cord pin is visible.
- Place one sponge electrode below the elastic head strap. Ensure that excessive fluid is not ejected from the sponge unto the scalp during this process as this will spread current flow across the scalp and deplete the sponge of fluid.
- Connect the second elastic head strap to the first elastic head strap according to the electrode montage you want to use (Table 2). Other elastic head straps can be used.
- Place the second sponge electrode on the head under the second elastic head strap. Make sure you place it onto the marked area you want to stimulate.
- The path from one device terminal, through one electrode, across the body, through the second electrode, and back to the second device terminal forms a circuit – the total resistance of which (sum of the electrodes and body resistance) can be measured. If the overall resistance is abnormally high, this may indicate improper electrode set-up. If your device measure resistance – this would be recommended – the indication field should display appropriate electrode contact. Ideally, one should aim to have the impedance under 5k Ohms. Some devices indicate the voltage across the path rather than resistance – in this case resistance can be calculated simply using ohms law (Resistance = Indicate Voltage / Current applied). Many devices continue to provide an indication of resistance during the course of stimulation, which provides a useful way to detect a potentially hazardous situation (such as a drying electrode). In some cases, device will automatically terminate stimulation or reduce stimulation intensity if resistance increases beyond a certain threshold.
5. Start tDCS
- Prior to start the procedure, screen subjects for any contraindications (see discussion).
- The subject should be relaxed, comfortable and awake during the procedure. Uncontrolled interference with current cortical activity during tDCS should be avoided. For stimulation to the motor cortex area, it has been demonstrated that intensive cognitive effort unrelated to the target area, as well as massive activation of the motor cortex by prolonged muscle contraction abolishes the effects of tDCS13.
- Adjust the settings on the tDCS stimulator that you want to stimulate with, including the intensity, time, and if applicable to your device, the sham condition setting (Figure 10). Note that some stimulators have to be switched on before the contact between the electrodes and the skin is made to avoid electrical shocks.
- Now initiate the tDCS. To reduce any adverse effects start current flow by ramping up the current. Many commercial devices include features to automatically ramp current on and off. One point that should be noted is that subjects usually continue feeling some local sensation even after current is discontinued.
- Some subjects may experience discomfort during the initial tDCS period. In such cases the current may be moderately decreased for a temporary period, for example by 50%, as the subject adjusts, then gradually increased back up to the desired level. This feature may depend on the device being used.
- At the beginning of stimulation, the majority of subjects will perceive a slight itching sensation, which then fades in most cases. Similarly, rapid changes of the stimulating circuit immediately might induce peripheral nerve firing. The subject can notice it as brief retinal phosphenes with electrodes near the eyes. These effects can be largely avoided by ramping the current up and down at the beginning and end of treatment. This might also prevent the dizziness or vertigo occasionally reported when current is suddenly increased or decreased.7
- After the stimulation, current flow should be ramped off as well. Note on High-Definition tDCS (HD-tDCS): TDCS with electrodes smaller then approximately 2 cm2 is called HD-tDCS and often uses array of electrode (more than two) to guide current through the brain for specific applications14. This methods paper focused only on conventional tDCS (using larger sponge electrodes), and it is important to emphasize that HD-tDCS requires specific electrodes15, skin preparation, and stimulator hardware. It is not recommended to apply tDCS using 1-2 mA of sponge electrodes14,15.
6. After the procedure
- To evaluate transcranial DC stimulation regularly and to record the safety of this technique over a long period of time, it is recommended to use a questionnaire of adverse effects.
- Such a questionnaire should include any possible adverse effects associated with tDCS. The most common adverse effects are tingling, itching and burning sensations, headache and discomfort. You can find an example for such a questionnaire in the article of Brunoni et al. (2011)16. It is also recommended to collect quantitative on the adverse effects like a 1 to 5 or 1 to10 grade scale.
- One should also use this adverse effect questionnaire after the condition of sham stimulation to reveal a better comparability between the two stimulation situations. There is evidence that sham stimulation causes a comparable amount of itching and tingling sensations as active stimulation.
7. Representative Results:
With proper setup, the tDCS device should display that either current is flowing during active tDCS situation, or the device should display sham mode when running a sham stimulation procedure (Figure 10).
Of note, even with the device indicating that current is flowing through the system, current might actually being shunted through the skin. In order to avoid this effect, it is recommended to have sufficient distance between electrodes. According to modeling studies we recommend that to be of at least 8cm when using 5x7cm electrodes17.
Furthermore, it is recommended to consult computer head models14 and neurophysiological studies. These additional steps would ensure that a specific montage is associated with significant changes in cortical excitability in the area that is being investigated.
Representative for anodal stimulation is an increase of brain excitability, whereas cathodal stimulation leads to a reduction of the cortical excitability. Robust evidence for this has been revealed in trials targeting the primary motor cortex (Figure 6).
The variation of the electrode size leads to a variation of focal effects. With a decrease of the diameter of the electrode, a more focal stimulation can be achieved. This can be proven by using TMS over the motor cortex. On the other hand by increasing electrode size it is possible to have a functionally ineffective electrode (Figure 8).
With session duration of 20 minutes or more and with multiple sessions over consecutive days, the after-effects of tDCS will last longer. Example for this is the treatment of pain syndromes.
One important point is the location of the reference electrode. If an extracephalic position is chosen, investigator should be aware of current distribution as the reference electrode might displace the peak of induced current and modify the effects of tDCS.
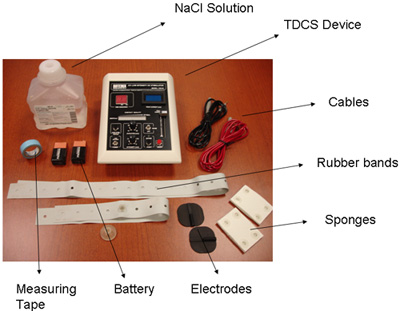
Figure 1. Materials
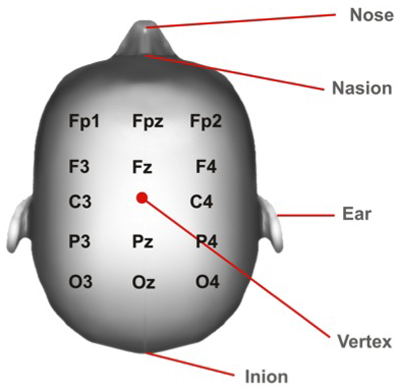
Figure 2: Vertex Position. Cortical areas marked according to the 10/20 system.
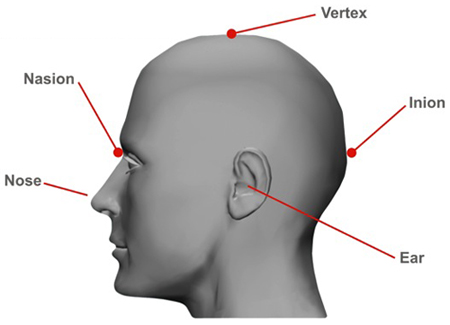
Figure 3: Nasion and Inion Position
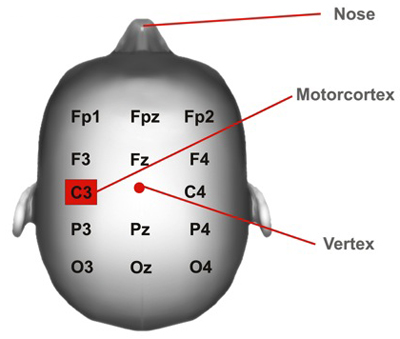
Figure 4: Motor cortex Position. Cortical areas marked according to the 10/20 system.
Figure 5: DLPFC Position. DLPFC= dorsolateral prefrontal cortex. Cortical areas marked according to the 10/20 system.
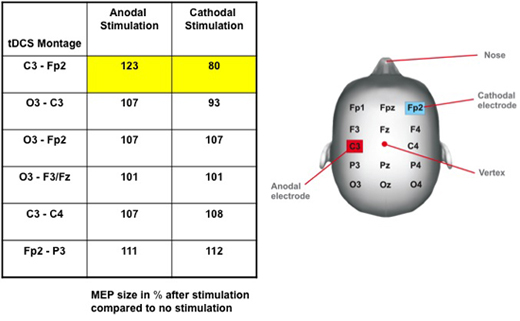
Figure 6: Change in cortical excitability due to current polarity and tDCS montage. Table: Induced effects of tDC stimulation on the size of the motor evoked potential (MEP), assessed by transcranial magnetic stimulation (TMS). MEP amplitudes after stimulation are given in percent of MEP without stimulation. Note that only the motor cortex (M1) – contralateral supra-orbital (Fp2) montage setting leads to a significant increase of MEP size after anodal and a decrease of MEP amplitude after cathodal stimulation. There are no significant effects on MEP amplitude in other tDCS montages. Figure: Electrode Placements6 (modified from Nitsche 2000).

Figure 7: Electrode Sizes
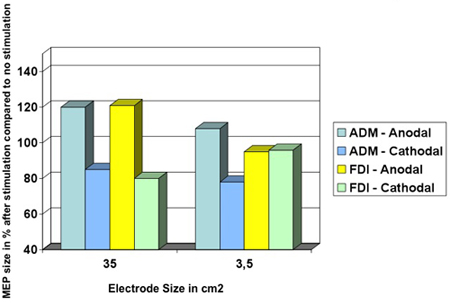
Figure 8: Decreasing the size of the electrode leads to a more focally effect of tDCS. Muscle-evoked potential (MEP) amplitude sizes of the abductor digiti minimi (ADM) and of the first dorsal interosseus muscle (FDI) during anodal or cathodal tDCS. Using the condition of a 35 cm2 electrode, anodal and cathodal tDCS influence the MEP amplitude size of the ADM and the FDI to a similar extent. At this montage, both hand muscle representation areas are located underneath the stimulating electrode. In the case of a smaller electrode, which is only placed over the representational area of the ADM, the effects of MEP amplitude changes of the cortical FDI representation are not reproducible (see yellow column)18 (modified from Nitsche 2007).
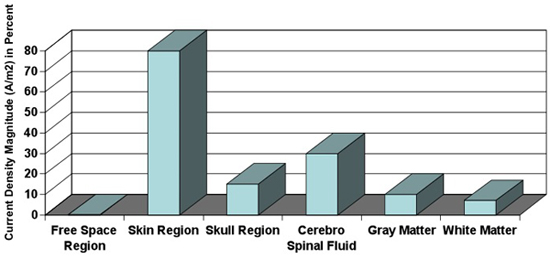
Figure 9: Tissue-depended current density. Current densities calculated in different tissues. Magnitude of current density is dependent on the conductivity of tissue. Note that approximately 10% of current density reaches the Gray Matter19 (modified from Wagner 2007a).

Figure 10: Different stimulation conditions: active vs. sham. Some tDCS devices provide set ups for active and sham condition. Usually applicable stimulation is indicated with a light signal.
| Material |
| TDCS device |
| 9V Battery (2x) |
| Two rubber head bands |
| Two conductive rubber electrodes |
| Two sponge electrodes |
| Cables |
| NaCl solution |
| Measurement Tape |
Table 1. Materials
| Anode Electrode Positioning | Cathode Electrode Positioning | Observations | Caveats |
| Primary Motor cortex (M1) | Supra-Orbital | This is the most used montage. It has been proven that the cortical excitability can be changed up to 40%6 (Figure 6). Anodal stimulation results in neuronal depolarisation and increasing neuronal excitability while cathodal stimulation has opposite results6. | Only one motor cortex is stimulated – might be a problem for bilateral pain syndromes. Also the confounding effect of the supra-orbital electrode needs to be considered. |
| Primary Motor cortex (M1) | Primary Motor cortex | – Interesting approach when there is a bi-hemispheric imbalance between motor cortices (such as in stroke) – Can be used with two anodal stimulation electrodes (see sixth row), where cathodal electrode is placed in the supraorbital area for instance. |
Electrodes might be too close to each other- issue of shunting. A decrease of the area of the electrodes will increase the degree of shunting along the skin 19 Therefore shunting might be related not only to electrode positioning but also to electrode size. The relative resistance of the tissues is dependent upon the electrode position and size- the overall resistance on which the current flows is dependent upon the electrode properties19. |
| Dorsolateral Prefrontal Cortex (DLPFC) | Supra-Orbital | Most used for DLPFC stimulation – positive results for treatment of depression20 and also chronic pain3. | Only unilateral DLPFC stimulation situation is possible with this montage. |
| Dorsolateral Prefrontal Cortex | Dorsolateral Prefrontal Cortex | – Interesting approach when there is a bi-hemispheric imbalance. – Can be used for a two anodal stimulation situation (see sixth row), where cathodal electrode is placed in the supraorbital area for instance. |
Electrodes might be too close to each other- issue of shunting 19. (Please see second row, fourth column). |
| Occipital | Vertex | Interesting active control for chronic pain trials or for modulation of visual cortex. | When used as active control, reference electrodes are placed in different locations- problem of comparability between intra- and inter- experimental approaches. |
| Two anodal electrodes, e.g. both Motor cortices | Supra-Orbital | Simultaneous change in cortical excitability | Transcallosal inhibition might add a confounding factor21 |
| One electrode over a cortical target, e.g. Primary Motor cortex (M1) | Extra-Cranial | Avoid the confounding effect of two electrodes with opposite polarities in the brain7. | Depending on intended target, current distribution might not be optimal and therefore induce ineffective stimulation22 |
Table 2. Electrode Positioning7
Note: It is possible that the differences between various electrode positions might be the activation of different neuronal populations due to different electrical field orientations.
Discussion
Critical Steps:
Aspects to be checked prior to starting procedure:
- First of all, patients should be screened for whether there are any contraindications for tDCS – these contraindications may be application specific. This includes questions such as presence of severe or frequent headache, chronic skin disorder, or adverse reactions to a previous tDCS treatment. If he or she has any metal in the head or had a serious brain injury, the anatomical changes may modify current flow23,24. History of seizure, pregnancy and history of a stroke are usually not strict contraindications – and indeed, might be inclusion criteria in some clinical trials.
- Check for any lesions on the scalp- additionally subjects should be specifically interviewed and inspected for the existence of skin diseases. If there are any lesions, tDCS procedure should be avoided or, if appropriate, ensured that stimulation will not be conducted directly over or across the lesion. Stimulation of a different site can be considered. It is reported that repeated daily tDCS causes clinically significant skin irritation under the electrodes in some patients7. There is evidence of tDCS induced lesions according to skin integrity. For instance, it has been shown extensive redness and brown crusty intracutaneous changes with irregular round shapes due to tDC stimulation at an intensity of 2 mA for a period of 2 weeks including five sessions each week25. If tDCS is strongly indicated or has to be conducted, it is possible to take into consideration to stimulate with a lower intensity such as 0.5- 1.0 mA, but it is not guaranteed that this will prevent skin irritations or lesions. Thus, the condition of the skin under the electrodes should be inspected before and after tDCS7.
- Check connectors of cables for electrolysis. Use another pair if evident. It is recommended to check the cables after approximately two months of use.
During both- active or sham- tDCS always ask whether subject still feels comfortable and is able to continue procedure.
Possible modifications:
- There are many varieties of electrode positioning7 (Table 2).
- There are many varieties of electrode sizes26 (Figure 7). For a given applied current, electrode size influences the current density18 and influences the focality of brain modulation (Figure 8). Clinical studies have suggest the smaller the size of the electrode the larger the current density26, however modelling studies suggest that the relationship between electrode size and area of brain modulation may be more complex27. Furthermore, the effects of small electrodes can differ qualitatively due to differential shunting of current in the scalp, and greater edge effect relative to the overall electrode area7. There were drastically superior levels of shunting for small electrode sizes reported than for the larger electrode schemes19.
- High-Definition tDCS (HD-tDCS) is a technology that improves spatial focality but requires special hardware and procedural controls15.
- The electrode montage (electrode position and size) along with the applied current determine the generated electrical field strength in the brain which, in turn, determines the efficacy of tDCS. The use of just electrode current density, defined by the ratio between the current strength and the electrode size, has been proposed to normalize clinical outcomes – but modelling studies suggest this may only apply over a limited range and that overall electrode montage design determines outcome. Generally, increasing current intensity (or current density) for any given montage result in stronger effects. It is important to note that the current density at the skin surface is much higher than that in the brain19 (Figure 9).
- The position of the “return” (“reference”) electrode may influence the overall current flow pattern through the brain, and thus even influence brain modulation under the active electrodes22. Thus the potion of both electrodes should be considered.
- The duration of the stimulation depends on the aim of the experimental approach. An increase of duration of the stimulation is associated with the occurrence and a longer duration of after-effects3,4. However at least one study reported a reversal of effects directions when stimulation duration was increased, suggesting that more intensity does not necessarily translate into a more robust clinical outcome. Though tDCS within published parameters is considered safe and well tolerated, the potential for undesired side-effects increases with increasing intensity (time, duration, or repetition rate/number).
- Orientation of the electric field: defined by the electrodes positions and polarity. Cathodal stimulation typically decreases cortical excitability, whereas anodal stimulation typically increases the cortical excitability2,3.
- Placebo: For sham-tDCS the same protocol above is used. However, the current will be applied for 30 seconds. This is one of the advantages of tDCS compared to other non-invasive brain stimulation methods. Since the arising sensations resulted from active-tDCS tend to occur only at early stages of application, this sham method makes it difficult for the patient to distinguish the placebo from active tDCS application. This initial and brief stimulation is a reliable method of placebo28.
- Note that the technique can also be applied when using other transcranial electric therapies such as tACS29 or tRNS30.
Rationale for using tDCS in chronic pain:
The fact that multiple therapeutic pharmacological modalities provide only modest relief for chronic pain patients raises the possibility that the cause for the persistence of this debilitating disorder may lie within plastic changes in pain related neural networks. Interestingly, modulation of cortical activity can be achieved non-invasively by tDCS, as described earlier, which has been reported to produce lasting therapeutic effects in chronic pain due to changes in cortical plasticity.
Clinical effect of tDCS in chronic pain:
It has been shown that tDCS applied to the motor cortex changes the local cortical excitability (Figure 6)6. More precisely, anodal stimulation results in an increase of neuronal excitability, whereas cathodal stimulation has opposite results6. Indeed, anodal tDCS application over M1 leads to a greater improvement in visual analogue scale (VAS) pain ratings than sham tDCS. This therapeutic effect on pain following M1 stimulation, although transient, was reproduced in several groups of patients with neuropathic pain syndromes like trigeminal neuralgia, poststroke pain syndrome31, back pain and fibromyalgia32. Interestingly, clinical trials in neuropathic pain, due to spinal cord injury, stimulation of the motor cortex by tDCS showed pain improvement and cumulative analgesic effect that lasted two weeks after the stimulation. There is also evidence of its analgesic effect in fibromyalgia patients33 that is still significant after three weeks of follow-up for anodal tDCS of the M1 compared with sham stimulation, and as well as stimulation of the DLPFC33. Although the effects of anodal tDCS over DLFPC for pain improvement have not been explored extensively, it was shown it can be used to modulate pain thresholds in healthy subjects34. Nevertheless, stimulation of this brain area is a reliable technique for enhancing working memory 10, increasing performance on memory tasks in Alzheimer disease9 and reducing cue-provoked smoking craving significantly35 for instance; therefore it is also conceivable that this might be a useful strategy to modulate affective-emotional cognitive networks associated with pain processing in patients with chronic pain.
Disclosures
The authors have nothing to disclose.
Acknowledgements
DaSilva AF received funding support from CTSA high-tech funding grant, University of Michigan to complete this review. Volz MS is funded by a grant scholarship from Stiftung Charité.
References
- Fregni, F., Pascual-Leone, A. Technology insight: noninvasive brain stimulation in neurology-perspectives on the therapeutic potential of rTMS and tDCS. Nat Clin Pract Neurol. 3 (7), 383-383 (2007).
- Wagner, T., Valero-Cabre, A., Pascual-Leone, A. Noninvasive human brain stimulation. Annu Rev Biomed Eng. 9, 527-527 (2007).
- Fregni, F., Freedman, S., Pascual-Leone, A. Recent advances in the treatment of chronic pain with non-invasive brain stimulation techniques. Lancet Neurol. 6 (2), 188-188 (2007).
- Lefaucheur, J. P., Antal, A., Ahdab, R., Ciampi de Andra, D., Fregni, F., Khedr, E. M., Nitsche, M., Paulus, W. The use of repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current stimulation (tDCS) to relieve pain. Brain Stimul. 1 (4), 337-337 (2008).
- Antal, A., Paulus, W. Transcranial magnetic and direct current stimulation in the therapy of pain. Schmerz Apr. 24 (2), 161-161 (2010).
- Nitsche, M. A., Paulus, W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J Physiol. 527 (Pt 3), 633-633 (2000).
- Nitsche, M. A., Cohen, L. G., Wassermann, E. M., Priori, A., Lang, N., Antal, A. Transcranial direct current stimulation: state of the art. Brain Stimul. 11, 642-642 (2008).
- Merrill, D. R., Bikson, M., Jefferys, J. G. Electrical stimulation of excitable tissue: design of efficacious and safe protocols. J Neurosci Methods. 141 (2), 171-171 (2005).
- Boggio, P. S., Khoury, L. P., Martins, D. C., Martins, O. E., de Macedo, E. C., Fregni, F. Temporal cortex direct current stimulation enhances performance on a visual recognition memory task in Alzheimer disease. J Neurol Neurosurg Psychiatry. 80 (4), 444-444 (2009).
- Fregni, F., Boggio, P. S., Nitsche, M., Bermpohl, F., Antal, A., Feredoes, E., Marcolin, M. A., Rigonatti, S. P., Silva, M. T., Paulus, W., Pascual-Leone, A. Anodal transcranial direct current stimulation of prefrontal cortex enhances working memory. Exp Brain Res. 166 (1), 23-23 (2005).
- Dundas, J. E., Thickbroom, G. W., Mastaglia, F. L. Perception of comfort during transcranial DC stimulation: effect of NaCl solution concentration applied to sponge electrodes. Clin Neurophysiol. 118 (5), 1166-1166 (2007).
- Minhas, P., Datta, A., Bikson, M. Cutaneous perception during tDCS: Role of electrode shape and sponge salinity. Clin Neurophysiol. 11, (2010).
- Antal, A., Terney, D., Poreisz, C., Paulus, W. Towards unravelling task-related modulations of neuroplastic changes induced in the human motor cortex. Eur J Neurosci. 26 (9), 2687-2687 (2007).
- Datta, A. Gyri-precise head model of transcranial direct current stimulation: improved spatial focality using a ring electrode versus conventional rectangular pad. Brain Stimul. 2 (4), 201-201 (2009).
- Minhas, P., Bansal, V., Patel, J., Ho, J. S., Diaz, J., Datta, A., Bikson, M. Electrodes for high-definition transcutaneous DC stimulation for applications in drug delivery and electrotherapy, including tDCS. J Neurosci Methods. 190 (2), (2010).
- Brunoni, A. R. A Systematic Review on Reporting and Assessment of Adverse Effects associated with Transcranial Direct Current Stimulation. Int J Neuropsychopharmacol. , (2011).
- Wagner, T. Transcranial direct current stimulation: a computer-based human model study. Neuroimage. 35 (3), 1113-1113 (2007).
- Nitsche, M. A., Doemkes, S., Karaköse, T., Antal, A., Liebetanz, D., Lang, N., Tergau, F., Paulus, W. Shaping the effects of transcranial direct current stimulation of the human motor cortex. J Neurophysiol. 97 (4), 3109-3109 (2007).
- Wagner, T., Fregni, F., Fecteau, S., Grodzinsky, A., Zahn, M., Pascual-Leone, A. Transcranial direct current stimulation: a computer-based human model study. Neuroimage. 35 (3), 1113-1113 (2007).
- Boggio, P. S., Rigonatti, S. P., Ribeiro, R. B., Myczkowski, M. L., Nitsche, M. A., Pascual-Leone, A., Fregni, F. A randomized, double-blind clinical trial on the efficacy of cortical direct current stimulation for the treatment of major depression. Int J Neuropsychopharmacol. 11 (2), 249-249 (2008).
- Williams, J. A., Pascual-Leone, A., Fregni, F. Interhemispheric modulation induced by cortical stimulation and motor training. Phys Ther. 90 (3), 398-398 (2010).
- Lang, N., Nitsche, M. A., Rothwel, J. C., Williams, J. A., Lemon, R. N. Effects of transcranial direct current stimulation over the human motor cortex on corticospinal and transcallosal excitability. Exp Brain Res. 156 (4), 439-439 (2004).
- Bikson, M., Datta, A., Rahman, A., Scaturro, J. Electrode montages for tDCS and weak transcranial electrical stimulation: role of “return” electrode’s position and size. Clin Neurophysiol. 121 (12), (1976).
- Bikson, M., Fregni, F. Transcranial direct current stimulation in patients with skull defects and skull plates: high-resolution computational FEM study of factors altering cortical current flow. Neuroimage. 52 (4), 1268-1268 (2010).
- Datta, A., Baker, J. M., Bikson, M., Fridriksson, J. Individualized model predicts brain current flow during transcranial direct-current stimulation treatment in responsive stroke patient. Brain Stimulation. , (2011).
- Palm, U., Keeser, D., Schiller, C., Fintescu, Z., Nitsche, M., Reisinger, E. Padberg Skin lesions after treatment with transcranial direct current stimulation (tDCS). Brain Stimul. 1 (4), 386-386 (2008).
- Datta, A., Elwassif, M., Bikson, M. Bio-heat transfer model of transcranial DC stimulation: comparison of conventional pad versus ring electrode. Conf Proc IEEE Eng Med Biol Soc. , 670-670 (2009).
- Miranda, P. C., Faria, P., Hallett, M. What does the ratio of injected current to electrode area tell us about current density in the brain during tDCS?. Clin Neurophysiol. 120 (6), 1183-1183 (2009).
- Gandiga, P. C., Hummel, F. C., Cohen, L. G. Transcranial DC stimulation (tDCS): a tool for double-blind sham-controlled clinical studies in brain stimulation. Clin Neurophysiol. 117 (4), 845-845 (2006).
- Antal, A. Comparatively weak after-effects of transcranial alternating current stimulation (tACS) on cortical excitability in humans. Brain Stimul. 1 (2), 97-97 (2008).
- Terney, D. Increasing human brain excitability by transcranial high-frequency random noise stimulation. J Neurosci Methods. 28 (52), 14147-14147 (2008).
- Fregni, F., Boggio, P. S., Mansur, C. G., Wagner, T., Ferreira, M. J., Lima, M. C., Rigonatti, S. P., Marcolin, M. A., Freedman, S. D., Nitsche, M. A., Pascual-Leone, A. Transcranial direct current stimulation of the unaffected hemisphere in stroke patients. Neuroreport. 16 (14), 1551-1551 (2005).
- Antal, A., Terney, D., K hnl, S., Paulus, W. Anodal transcranial direct current stimulation of the motor cortex ameliorates chronic pain and reduces short intracortical inhibition. J Pain Symptom Manage. 39 (5), (2010).
- Fregni, F., Gimenes, R., Valle, A. C., Ferreira, M. J., Rocha, R. R., Natalle, L., Bravo, R., Rigonatti, S. P., Freedman, S. D., Nitsche, M. A., Pascual-Leone, A., Boggio, P. S. A randomized, sham-controlled, proof of principle study of transcranial direct current stimulation for the treatment of pain in fibromyalgia. Arthritis Rheum. 54 (12), (2006).
- Boggio, P. S., Zaghi, S., Lopes, M., Fregni, F. Modulatory effects of anodal transcranial direct current stimulation on perception and pain thresholds in healthy volunteers. Eur J Neurol. 15 (10), 1124-1124 (2008).
- Fregni, F., Liguori, P., Fecteau, S., Nitsche, M. A., Pascual-Leone, A., Boggio, P. S. Cortical stimulation of the prefrontal cortex with transcranial direct current stimulation reduces cue-provoked smoking craving: a randomized, sham-controlled study. J Clin Psychiatry. 69 (1), 32-32 (2006).

