Modified Technique for Coronary Artery Ligation in Mice
Summary
The surgical procedure used to induce experimental myocardial infarction in mice begins with left thoracotomy between the third and the fourth ribs in order to visualize the anterior surface of the heart and left lung. The left coronary artery is ligated, the chest is closed and the mouse is allowed to recover spontaneously.
Abstract
Myocardial infarction (MI) is one of the most important causes of mortality in humans1-3. In order to improve morbidity and mortality in patients with MI we need better knowledge about pathophysiology of myocardial ischemia. This knowledge may be valuable to define new therapeutic targets for innovative cardiovascular therapies4. Experimental MI model in mice is an increasingly popular small-animal model in preclinical research in which MI is induced by means of permanent or temporary ligation of left coronary artery (LCA)5. In this video, we describe the step-by-step method of how to induce experimental MI in mice.
The animal is first anesthetized with 2% isoflurane. The unconscious mouse is then intubated and connected to a ventilator for artificial ventilation. The left chest is shaved and 1.5 cm incision along mid-axillary line is made in the skin. The left pectoralis major muscle is bluntly dissociated until the ribs are exposed. The muscle layers are pulled aside and fixed with an eyelid-retractor. After these preparations, left thoracotomy is performed between the third and fourth ribs in order to visualize the anterior surface of the heart and left lung. The proximal segment of LCA artery is then ligated with a 7-0 ethilon suture which typically induces an infarct size ~40% of left ventricle. At the end, the chest is closed and the animals receive postoperative analgesia (Temgesic, 0.3 mg/50 ml, ip). The animals are kept in a warm cage until spontaneous recovery.
Protocol
1. Protocol Text
- Before proceeding to surgery, anesthetize a male mouse (C57BL/6) that is 6 weeks of age or older by inhalation of 2-5% isoflurane. Perform a toe pinch to confirm that the animal is sufficiently anesthetized.
- The unconscious mouse is placed on its back on a warm pad to maintain a constant temperature of 37 °C. The precordial chest on the left side is shaved.
- Secure the mouse to an intubation device with an isoflurane-filled chamber. Insert a tube of 0.2 inner diameter into the mouse’s trachea through mouth, then place the intubated mouse on an aseptic surgical area (37 °C by a temperature controller) and connect the endotracheal tube to a ventilator (rate of 120 per min, pressure of 4-6 mmH2O); disinfect the area with betadine and 75% ethanol .
- Apply ophthalmic ointment to the mouse’s eyes. Place standard ECG electrodes on the paws and connect the mouse to the ECG recorder.
- Use sterile scissors to make a 1.5 cm incision along the mid-axillary line. Scissors are used rather than a scalpel to avoid injury to the underlying tissue in mice. Then, use blunt-tip vessel scissors to dissect and retract the precordial muscle from the chest wall.
- Perform a left thoracotomy between the third and fourth ribs to visualize the anterior surface of the heart and left lung
- Further retract the thoracic wall using a suitable retractor in order to improve visualization and accessibility. Remove the pericardium by using toothed forceps and blunt-tipped scissors.
- Ligate the left coronary artery by placing the needle (attached with 7-0 ethilon suture) beneath the artery with a band of myocardium between the ligature and the artery. A successful occlusion of coronary artery is verified by hypokinesis/akinesis of the anterior left ventricular wall and by the alterations of ECG recording (e.g. ST segment elevation, QRS broadening)
- Close the chest by placing two stitches (6-0 ethilon suture) on the third and fourth ribs.
- Use 5-0 ethilon suture to close the skin.
- For post-operative analgesia, administer a single intraperitoneal injection of buprenorphine (0.05 mg/kg). The animals will spontaneously recover within 2-5 min after stopping isoflurane anesthesia. Finally, place the mouse in a warm cage and provide 0.6 mg/ml of buprenorphine in the drinking water to prevent post-operative distress. Closely monitor the mouse for the first 24 hours after surgery.
- Echocardiography is performed to evaluate cardiac function 3 days after myocardial infarction. Successful procedure is verified by akinesia in the anterior left ventricular wall (see the enclosed video in Results). At the end of the experiment, the heart will be collected and stored in -80 °C for further investigations. i.e. gene expression.
Representative Results
| Instrument | Function description |
| Blunt-tip scissors | Used to cut skin, intercostal chest muscle, and sutures |
| Eye retractor | Used to expose surgical view |
| Blunt-tip forceps | Used to grasp skin and muscle |
| Needle holder | To hold a needle |
| 5-0 suture | Used to suture skin |
| 6-0 suture | Used to suture chest muscle |
| 7-0 suture | Used to ligate coronary artery |
Table 1. Instruments used in the procedure of myocardial infarction.
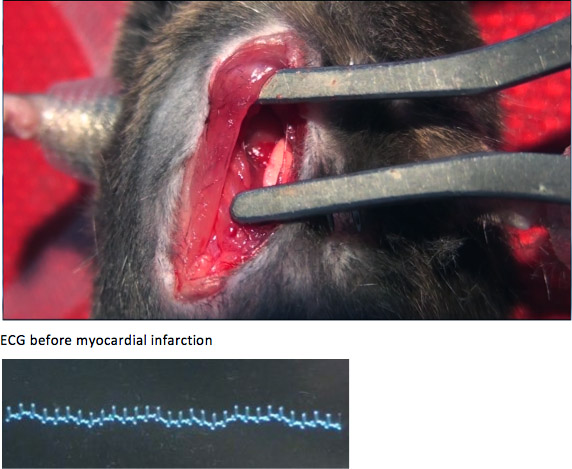
Figure A. Before coronary artery ligation, the left atrium, left lung, and anterior wall of left ventricle were exposed. The QRS complexes shown in ECG were narrow and short.
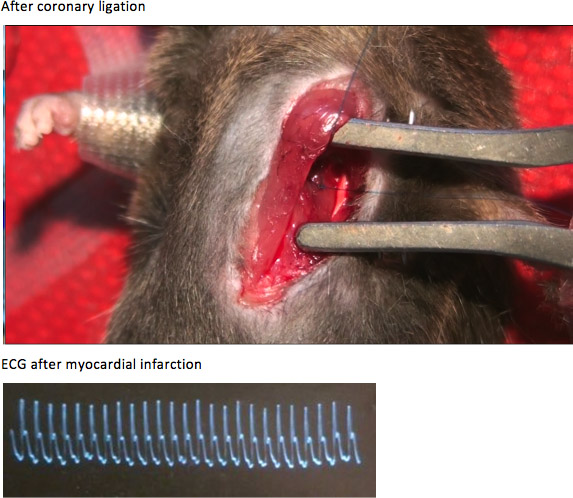
Figure B. After coronary artery ligation, the anterior wall of left ventricle developed cyanosis and diskinesia. The QRS complexes became significantly wider and the ST segments were obviously elevated, compared with those before coronary artery ligation.
Discussion
The surgical procedure for LCA ligation as demonstrated in this report is a reliable and reproducible method for induction of experimental MI in mice3, 6. The ligation of LCA according to this protocol induces myocardial ischemia engaging typically ~40% of left ventricle3. This induces significant ECG changes in ST segment7 and visible pallor on the anterior surface of the heart. Due to the large myocardial infarction, the mortality can be up to 30%8, 9. A note should be made that infarcted myocardial area and post-MI mortality may be different among various animal strains. The method has several advantages. Placement of skin incision along mid-axillary line rather than on anterior chest wall facilitates the later investigation with echocardiography. The stiches in the operative wound do not interfere with ultrasound imaging of the anterior segments of the heart. Moreover, the intubation of mice allows the operator to have adequate time to perform the surgery. The ligation of the LCA “in situ” inside the chest is more “physiological” as opposed to ligation of the LCA after externalization of the as this will induce severe bradycardia and global hypoxia10. This approach also holds advantages over the cryoinjury-based approach as the latter is associated with pathophysiological processes distinct from those of ischemia-induced myocardial infarction which may confound mechanistic studies of ischemic cardiomyopathy. We believe that this video and the accompanying instructions will be of useful complement to the existing techniques for experimental MI in mice.
Troubleshooting:
- To get the best visualization of important anatomical structures (left atrium, left anterior ventricular wall, and left lung), the left thoracotomy is usually performed between the third and the fourth rib.
- Be aware that accidental puncture of the left atrium results in severe bleeding that may lead to death of the animal.
- Lung contusion should be avoided as it may cause increased mortality. If left lung is covering the heart after opening of the chest, the lung tissue should be retracted gently by using e.g. cotton-stick.
- High concentration of isoflurane should be avoided. Typically 2% of isoflurane is optimal for the achievement of proper anaesthesia in the present surgery.
Disclosures
The authors have nothing to disclose.
References
- Tabrizchi, R. Beta-blocker therapy after acute myocardial infarction. Expert Rev. Cardiovasc. Ther. 11, 293-296 (2013).
- Pell, S., Fayerweather, W. E. Trends in the incidence of myocardial infarction and in associated mortality and morbidity in a large employed population, 1957-1983. N. Engl. J. Med. 312, 1005-1011 (1985).
- Ramunddal, T., Gizurarson, S., Lorentzon, M., Omerovic, E. Antiarrhythmic effects of growth hormone–in vivo evidence from small-animal models of acute myocardial infarction and invasive electrophysiology. J. Electrocardiol. 41, 144-151 (2008).
- Thom, T., Haase, N., Rosamond, W., Howard, V. J., Rumsfeld, J., Manolio, T., Zheng, Z. J., Flegal, K., O’Donnell, C., Kittner, S., et al. Heart disease and stroke statistics–2006 update: A report from the american heart association statistics committee and stroke statistics subcommittee. Circulation. 113, 85-151 (2006).
- van den Bos, E. J., Mees, B. M., de Waard, M. C., de Crom, R., Duncker, D. J. A novel model of cryoinjury-induced myocardial infarction in the mouse: A comparison with coronary artery ligation. Am. J. Physiol. Heart Circ. Physiol. 289, 1291-1300 (2005).
- Omerovic, E., Ramunddal, T., Lorentzon, M., Nordlander, M. Effects of neuropeptide y2 receptor blockade on ventricular arrhythmias in rats with acute myocardial infarction. Eur. J. Pharmacol. 565, 138-143 (2007).
- Ahn, D., Cheng, L., Moon, C., Spurgeon, H., Lakatta, E. G., Talan, M. I. Induction of myocardial infarcts of a predictable size and location by branch pattern probability-assisted coronary ligation in c57bl/6 mice. Am. J. Physiol. Heart Circ. Physiol. 286, 1201-1207 (2004).
- Lindbom, M., Ramunddal, T., Camejo, G., Waagstein, F., Omerovic, E. In vivo effects of myocardial creatine depletion on left ventricular function morphology and lipid metabolism: Study in a mouse model. J. Card Fail. 14, 161-166 (2008).
- Omerovic, E., Bollano, E., Basetti, M., Kujacic, V., Waagstein, L., Hjalmarson, A., Waagstein, F., Soussi, B. Bioenergetic, functional and morphological consequences of postinfarct cardiac remodeling in the rat. J. Mol. Cell. Cardiol. 31, 1685-1695 (1999).
- Gao, E., Lei, Y. H., Shang, X., Huang, Z. M., Zuo, L., Boucher, M., Fan, Q., Chuprun, J. K., Ma, X. L., Koch, W. J. A novel and efficient model of coronary artery ligation and myocardial infarction in the mouse. Circ. Res. 107, 1445-1453 (2010).

