Mechanical Testing of Mouse Carotid Arteries: from Newborn to Adult
Summary
Passive mechanical testing of mouse carotid arteries is described, with special consideration for adapting to different specimen ages. The procedures include determining the in vivo length of the artery, mounting it in a pressure myograph, recording data, measuring the unloaded dimensions and analyzing the resulting data.
Abstract
The large conducting arteries in vertebrates are composed of a specialized extracellular matrix designed to provide pulse dampening and reduce the work performed by the heart. The mix of matrix proteins determines the passive mechanical properties of the arterial wall1. When the matrix proteins are altered in development, aging, disease or injury, the arterial wall remodels, changing the mechanical properties and leading to subsequent cardiac adaptation2. In normal development, the remodeling leads to a functional cardiac and cardiovascular system optimized for the needs of the adult organism. In disease, the remodeling often leads to a negative feedback cycle that can cause cardiac failure and death. By quantifying passive arterial mechanical properties in development and disease, we can begin to understand the normal remodeling process to recreate it in tissue engineering and the pathological remodeling process to test disease treatments.
Mice are useful models for studying passive arterial mechanics in development and disease. They have a relatively short lifespan (mature adults by 3 months and aged adults by 2 years), so developmental3 and aging studies4 can be carried out over a limited time course. The advances in mouse genetics provide numerous genotypes and phenotypes to study changes in arterial mechanics with disease progression5 and disease treatment6. Mice can also be manipulated experimentally to study the effects of changes in hemodynamic parameters on the arterial remodeling process7. One drawback of the mouse model, especially for examining young ages, is the size of the arteries.
We describe a method for passive mechanical testing of carotid arteries from mice aged 3 days to adult (approximately 90 days). We adapt a commercial myograph system to mount the arteries and perform multiple pressure or axial stretch protocols on each specimen. We discuss suitable protocols for each age, the necessary measurements and provide example data. We also include data analysis strategies for rigorous mechanical characterization of the arteries.
Protocol
1. Pressure myograph set-up
- Summarized from the manufacturer instructions (Danish Myotechnology). A schematic of the complete system with optional accessories is shown in Figure 1.
- Turn on the air tank and myograph control boxes and start the MyoView software (Danish Myotechnology). In the software, load the appropriate diameter calibration file, inflation protocol file and name the file for saving data for the first protocol. Recommended inflation protocols for each age are given in Table 1.
- Choose the required cannulae and suture size for the test system depending on the mouse age. Dimensions of the custom-designed stainless steel cannulae for young and adult mice are shown in Figure 2. Place the bath under the dissecting microscope and mount the cannulae in the test system, being careful not to apply excessive force (> 1 N) to the force transducer.
- Fill the inflow bottle to 150 mL with physiological saline solution (PSS) (Table 2)8. Fill the bath and inflow tubing with PSS. Fill the outflow tubing halfway with PSS. Leaving the outflow bottle empty eliminates having to clean this side of the system. However, if PSS enters this side of the system, clean and flush after use following the manufacturers instructions.
- When the temperature reaches 37.5 °C, place the filled bath on the inverted microscope stage and zero the force transducer using the MyoView software. The force transducer drifts with temperature and location, therefore it is important to zero it in the condition it will be used for testing.
2. Carotid dissection
- Sacrifice the mouse using approved euthanasia methods (we use CO2 asphyxiation). All methods shown in this protocol have been approved by the Institutional Animal Care and Use Committee.
- Secure the mouse by taping the hands and feet to a dissecting board and place under a dissecting microscope. Open the chest to expose the right and left carotid arteries using dissecting scissors and curved forceps. Both arteries can be used for mechanical testing to increase numbers or to provide an internal control for different treatment protocols.
- Carefully clear fat and nearby tissue using curved forceps and fine tweezers until the full length of both carotids from the aorta to the common carotid bifurcation can be visualized (Figure 3). Make sure to keep the arteries moist with PSS during the dissection procedure.
- Cut a piece of 7-0 suture to the desired length using the digital calipers and dissection scissors. Suggested lengths for each age are given in Table 1. Lay the suture on the vessel and use an 18 G needle dipped in activated charcoal to mark the ends of the suture on the vessel (Figure 4A).
- Make sure that the complete length of the artery is lying in a single horizontal plane, otherwise the in vivo length measurements may be incorrect. The head of the mouse may have to be tilted up to accomplish this. Take a picture of each carotid using a camera connected to the dissection microscope. Using known calibrations, measure the in vivo length using Image J software. The length is measured from the image rather than the suture length because the charcoal markers for the cut sites often cannot be applied directly at the ends of the suture.
- Use a syringe attached to an 18 G needle to inject PSS into the left ventricle and clear all blood out of the carotid arteries. Cut the carotids at the charcoal markers using micro-scissors.
- Place the cut carotids in a petri dish with a small amount of PSS. Remove any fat from the outside of the arteries and let equilibrate for 2 – 3 minutes. Take a picture of each artery (Figure 4B) and use Image J software and known calibrations to measure the ex vivo length. Calculate the in vivo stretch ratio using the in vivo and ex vivo lengths.
- Place the carotids in a microfuge tube filled with PSS until ready for testing. They can be stored in the refrigerator for up to 3 days with no changes in passive mechanical behavior9.
3. Mounting an artery in the test system
- If the arteries have been in the refrigerator, remove and allow to come to room temperature.
- Place the heated, filled bath under the dissection microscope. Use the micrometer to bring the cannulae tips together until they are just touching and record this value. This is the “zero length” of the micrometer.
- Place a small loop of suture tied with a loose overhand knot on each end of the cannulae. Using fine tweezers, grasp the end of an artery and place it in the bath. Do not use the tweezers on any other part of the artery as the tips may puncture the wall.
- Use two tweezers to hold one end of the artery and slip it over one cannula. For mice ≥ 21 days, tighten the suture knot and repeat the process for the other side. If it is difficult to mount the second side, apply a slight pressure to open up the artery. For mice < 21 days it may be easier to slide the vessel completely onto one side, bring the cannulae together until they are touching, slide the vessel back over the second cannula, separate the cannulae and tighten the suture knot. Avoid twisting the artery during mounting.
- Calculate the necessary micrometer reading for the artery to be at the measured ex vivo length. We tie our arteries onto the cannulae with about 0.6 mm on each side, so our total unstretched artery length can be calculated from the increase in the micrometer distance from the zero length + 1.2 mm. The artery length that will be stretched in the mechanical test protocols does not include the extra 1.2 mm beyond the suture ties. The distance beyond the suture ties may need to be adjusted for different ages.
- Turn the micrometer to set the total artery length to the measured ex vivo distance and note the appearance and the axial force readings. Check the ex vivo length using the digital calipers. The artery should look unstretched (Figure 5) and the force should be close to zero. We have found that setting the artery at the measured ex vivo length is easier than trying to estimate the unloaded length based on zero force measurements or visualization alone.
- Calculate the necessary micrometer reading for the artery length between the suture ties to be at the measured in vivo stretch ratio. Turn the micrometer to set the artery at this length and begin testing protocols. This is the “in vivo testing length” of the artery.
4. Testing protocols
- Diameter tracking. Place the bath on the inverted microscope stage, position the central portion of the artery over the lens and focus on the edges of the artery wall. Start the diameter tracking in MyoView and check tracking of the outer diameter (Figure 6). The inner diameter will not track well for the carotid artery and must be calculated in the data analysis. Tracking problems can be remedied by ensuring no fat is on the artery wall, adjusting the contrast and brightness of the image and eliminating all bubbles from the fluid tubing.
- Pressure preconditioning. Biologic tissues must be preconditioned to obtain repeatable mechanical loading curves10. We have found that mouse arteries show repeatable behavior after 3 preconditioning cycles. With the artery at the in vivo testing length, manually increase and decrease the pressure 3x from zero to the maximum pressure for the specimen age (Table 1). The artery is pressurized by fluid flowing in from both cannulae. When the artery is at the in vivo stretch ratio, the axial forces should decrease slightly as the pressure is increased11.
- Axial stretch preconditioning. Manually set the pressure to 1/3 of the maximum pressure and stretch the artery axially 3x by manually turning the micrometer from the in vivo stretch ratio to the maximum axial stretch. The maximum axial stretch should be 1.2 x the in vivo testing length for mice < 21 days and 1.4 x the in vivo testing length for mice ≥ 21 days. In preliminary tests, this was the approximate maximum stretch that could be applied without any permanent creep of the artery in the axial direction.
- Pressure protocols. After preconditioning, three pressure and three axial stretch protocols are performed on each artery. With the vessel at the in vivo stretch ratio, run the appropriate inflation protocol for the specimen age (Table 1) to automatically increase pressure from zero to the maximum pressure 3x and record the data. Stretch the artery to half the maximum axial stretch and repeat, recording the data in a new file. At this length, the axial force should remain approximately constant as the pressure increases. Stretch the artery to the maximum axial stretch and repeat. At this length, the axial force should increase as the pressure increases.
- Axial stretch protocols. The Danish Myotechnology myograph system does not have automated axial stretch capabilities. Therefore, this is done manually with care taken to apply the correct stretch distance and a constant stretch rate during each protocol. Set the pressure to 1/3 the maximum pressure, manually stretch the artery to the maximum axial stretch 3x at approximately 10 μm/sec, recording the data in a new file. Set the pressure to 2/3 of the maximum pressure and repeat. Set the pressure equal to the maximum pressure and repeat.
5. Unloaded dimensions and opening angles
- Remove the artery from the bath and place in a petri dish with PSS.
- Under the dissection microscope, hold one end of the artery with fine tweezers and cut 3 – 5 rings about 0.5 mm thick using a disposable scalpel. Use the tweezers to gently open up each ring into a circle. Take pictures of the most circular rings for unloaded dimension measurements (Figure 7A). Use the micro-scissors or a scalpel to cut each ring at one location and allow it to open into an arc. After 10 minutes to equilibrate to their final shape, take a picture of the open rings for opening angle measurements12 (Figure 7B).
- If desired, fix the remainder of the carotid artery in 10% formalin for histology processing.
6. Data analysis
- We use custom Matlab (Mathworks Inc.) scripts to plot the pressure, diameter and force versus time for the 3 cycles of each protocol (Figure 8). The software records all data at 1 Hz. The third cycle is used for analysis unless there is something wrong with the data, i.e. the system lost the automatic diameter tracking. Because the axial stretch is manual, we enter the starting and final stretch values in Matlab and assume a constant stretch rate.
- We isolate the loading portion for a single cycle and save the measured pressure, diameter and force data, as well as the calculated axial stretch ratios (Figure 9). For the inflation cycles, the average pressure, diameter and force is calculated for each pressure step. The recorded data can be used to determine differences in the mechanical behavior between arteries.
- The deformed outer diameter, unloaded dimensions and axial stretch ratio are necessary to calculate the deformed inner diameter based on conservation of volume11. Additional calculations include compliance, stiffness, average stress and average strain3. The opening angle measurement is necessary for calculating stress and strain distributions through the wall thickness12. The complete set of six test protocols and the opening angle can be used to fit constitutive equations and fully characterize the mechanical behavior of each artery13-16.
7. Clean up
- Flush and clean the myograph system according to the manufacturers instructions.
- If there is a suspected clog in a cannula, clear it by inserting fine wire into the cannula tip. Do not try to force fluid through a clogged tip, as this will overload the pressure transducers.
8. Representative Results
All results are shown for C57BL6J 3-day-old mouse carotid arteries. Example closed and open rings are shown in Figure 7 for calculating unloaded dimensions and opening angles12. Example raw pressure, diameter and force data versus time are shown in Figure 8. Single loading cycles with no artifacts are isolated from this data for each protocol (Figure 9), which can then be used for further calculations, analyses and modeling to determine differences in the mechanical behavior between arteries3,11,15,16.
| Approximate age (days) | Max pressure (mmHg) | Wait time (sec) | Pressure steps (mmHg) | Deflation time (sec) | Suture length (mm) |
| 3 | 90 | 8 | 9 | 90 | 3 |
| 7 | 120 | 9 | 12 | 90 | 3.5 |
| 14 | 140 | 10 | 14 | 90 | 4.5 |
| 21 | 160 | 11 | 16 | 90 | 5 |
| 30 and above | 180 | 12 | 18 | 90 | 6 |
Table 1. Recommended automatic myograph inflation protocols and suture lengths for different age specimens. The maximum pressure avoids damage to the artery from overinflation, while capturing the nonlinear mechanical behavior, and ranges from 1.5 to 3 times the systolic pressure for each age3. The wait time provides enough time for the system to stabilize at each pressure and allows operator intervention if necessary to correct tracking problems. The pressure steps provide ten steps for each inflation cycle and an overall rate of 1 – 2 mmHg/sec from a starting pressure of 0 mmHg. This is considerably slower than the physiologic loading rate in an adult mouse artery (about 330 mmHg/sec for a 40 mmHg pulse pressure at 600 bpm), but soft biologic tissues are generally insensitive to loading rates over about three orders of magnitude10. Preliminary data showed no differences between mechanical behavior of mouse arteries when pressurized at the maximum rate of the system (approximately 7 mmHg/sec) and the rates listed here11. The deflation time allows the artery to fully return to the starting dimensions, while minimizing total cycle time. The suture length is approximated from the available carotid artery length at each age.
| Chemical | Conc (mM) | For 1 Liter (g) |
| NaCl | 130 | 7.6 |
| NaHCO3 | 15 | 1.25 |
| Dextrose | 5.5 | 1 |
| KCl | 4.7 | .35 |
| MgSO4-7H2O | 1.2 | .29 |
| KH2PO4 | 1.2 | .16 |
| EDTA | .026 | .01 |
| CaCl2 solution, pH 7.2 | 1.6 | 1.6 mL of 1M solution |
Table 2. Recipe for physiological saline solution (PSS). All chemicals from Sigma. PSS can be stored in the refrigerator for up to 3 days.
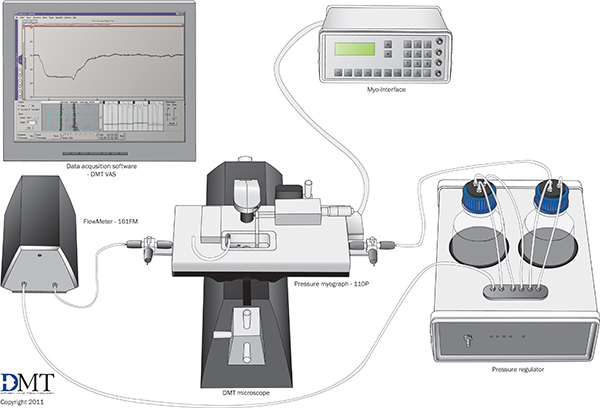
Figure 1. Schematic of the mechanical test system with optional accessories. Used with permission from Danish Myotechnology.
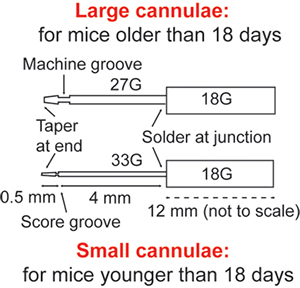
Figure 2. Dimensions of the custom-made cannulae for different aged specimens. All parts are made from 316 stainless steel hypodermic tubing (Small Parts). 7-0 suture should be used to secure the arteries to the large cannulae and 10-0 suture should be used for the small cannulae.
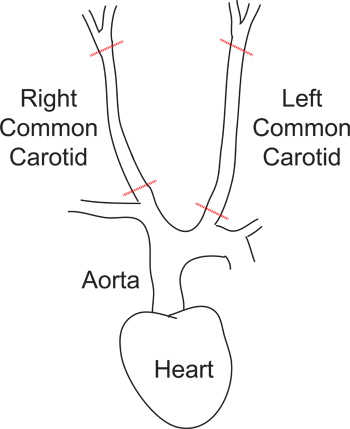
Figure 3. Diagram of the carotid artery dissection. The heart, aorta, right and left common carotid arteries and cut locations for the arteries are shown.
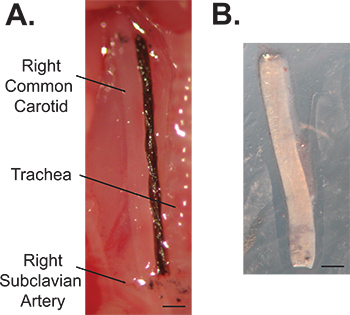
Figure 4. Images of a 3-day-old right common carotid artery in vivo (A) and ex vivo (B). The reference length is determined with a suture and the cut locations are marked with carbon particles. Note the decrease in length upon dissection. Scale bar = 0.25 mm.
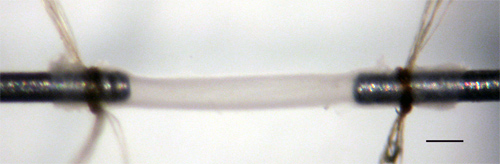
Figure 5. 3-day-old mouse carotid artery mounted on the cannulae in the myograph bath at its unstretched, ex vivo length. Scale bar = 0.2 mm.

Figure 6. Screen shot of the MyoView software showing outer diameter tracking of a 3-day-old carotid artery stretched to 1.2 times the in vivo length and pressurized to 0 mmHg (A) and 90 mmHg (B).
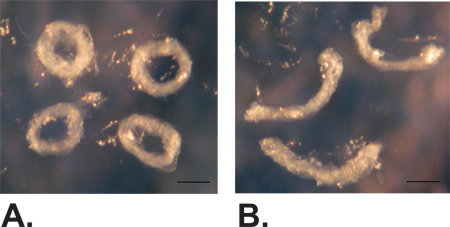
Figure 7. Example closed (A) and open (B) rings of a 3-day-old carotid artery cut to measure unloaded dimensions and opening angle. Scale bar = 0.1 mm.
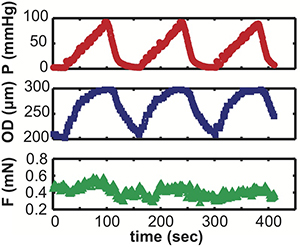
Figure 8. Example raw pressure, diameter and force data versus time for a single inflation protocol for a 3-day-old carotid artery.
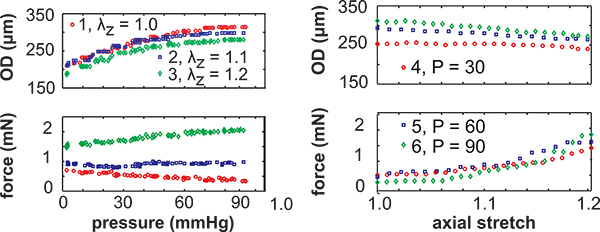
Figure 9.Example isolated loading cycles showing the recorded pressure, diameter, force and calculated axial stretch for all six mechanical test protocols for a 3-day-old carotid artery. The left panels show the inflation protocols at constant axial stretch (λz) with respect to the in vivo length and the right panels show the axial stretch protocols at constant pressure (P, mmHg). The manual axial stretch protocols are performed at a faster rate than the automated inflation protocols, so less data points are recorded.
Discussion
The protocol presented here provides a straightforward and repeatable method for characterizing the passive mechanical behavior of mouse carotid arteries. Although smooth muscle cells and endothelial cells are critical to the function of smaller, muscular arteries and capillaries, they do not contribute significantly to the mechanical behavior of large elastic arteries. Poisoning the cells with KCN has no significant effect on the pressure-diameter behavior of mouse carotid arteries17. Passive mechanical characterization is critical for determining the effects of changes in the matrix composition of large elastic arteries due to development, aging, disease or injury and the subsequent effects on cardiac and cardiovascular function. Mechanical characterization can be used to better understand the developmental process and recreate tissue-engineered arteries with appropriate mechanical properties. It can also be used to test disease treatments aimed at reversing mechanical changes that compromise human health.
Commercial myograph systems have been used to obtain pressure-diameter relationships for large, elastic arteries8 and to investigate contractility and smooth muscle cell function in arterioles18 from adult mice. More complete mechanical characterization of these vessels has been prevented by the use of traditional glass cannulae that do not allow substantial axial stretch of the vessel. Axial stretch is important for duplicating the in vivo configuration of the arteries and some large elastic arteries, such as the abdominal aorta, have in vivo stretch ratios of up to 1.711. We have designed custom stainless-steel cannulae that can be used for mechanical testing of carotid arteries from mice aged 3 days to adult3. The cannulae allow substantial axial stretch and are less prone to breakage than glass. The smallest size can get clogged by residual salts or vessel pieces, but these can be removed by insertion of a fine wire into the cannula tip and mostly prevented by thorough cleaning after each experiment. Limitations of the Danish Myotechnology myograph system used in this protocol include the lack of automated axial stretch and that only step changes in pressure can be programmed into the software.
The data gathered in this protocol, namely in vivo axial stretch, unloaded dimensions, opening angle and pressure, diameter, force and axial stretch for multiple inflation and stretch protocols, provides the required information to rigorously characterize the passive arterial mechanical behavior. The protocol takes more time and effort than a single pressure-diameter or contractility protocol, but provides enough information to compare the behavior for any loading condition and may highlight subtle differences in mechanical behavior that are not apparent in traditional pressure myograph experiments. Microstructurally-based constitutive equations can be used to correlate the mechanical behavior with measured differences in matrix protein amounts or organization13-16.
The protocol characterizes the in vitro passive mechanical behavior of carotid arteries, hence care must be taken to extrapolate any results to the in vivo condition. In vivo, the loading rate will be higher, there will be blood flowing through the artery, the artery will be tethered by surrounding tissue, and the artery will be exposed to a variety of chemical stimuli. The protocol is applied to large, elastic arteries, but can be modified to any vessel type suitable for the cannulae. Different buffers and bath additives may be used to isolate passive and active behavior for the carotid artery and for more muscular, contractile vessels. We have used the protocol for vessels ranging in inner diameter from 150 – 1000 μm and 1.5 – 8 mm in length. As the vessels decrease in length, end effects due to constraints at the suture ties will affect the results. We have performed a finite element study with estimated nonlinear mechanical properties to show that the stresses are homogeneous over at least 60% of the length when the length:diameter ratio is at least 2.519.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was funded, in part, by NIH grants HL087653 and HL105314. Some of the methods described in this work were developed in the laboratory of Dr. Robert Mecham at the Washington University School of Medicine.
Materials
| Name of the reagent/equipment | Company | Catalogue number | Comments |
| Air tank and regulator | Airgas Mid America | UN3156 | For pressurizing myograph |
| Pressure myograph and software | Danish Myotechnology | 110P, MyoView | With custom cannulae (Figure 2) |
| Inverted microscope, 5x lens and camera | Zeiss | Axiovert 40C | For tracking artery diameter |
| Physiological saline solution (PSS) | Chemicals from Sigma | Recipe and details in Table 2 | |
| Surgical tape | Various suppliers | For securing the mouse during dissection | |
| Dissection board | Fisher Scientific | 09-002-24A | For securing mouse during dissection |
| Dissecting microscope with camera | Zeiss | Stemi 2000-C |
For arterial dissection and mounting |
| Dissecting scissors | Fine Science Tools | 14058-11 | For cutting skin and opening the chest |
| Fine tweezers (2) | Fine Science Tools | 11200-14 | For grasping artery ends |
| Curved forceps | Fine Science Tools | 11274-20 | For clearing tissue and exposing carotid arteries |
| Micro-scissors | Fine Science Tools | 15005-08 | For precise cutting of arteries |
| 7-0 and 10-0 silk suture | Various suppliers | For estimating length and fastening arteries on cannulae | |
| Digital calipers | Fisher Scientific | 806-93-111 | For measuring suture length and checking artery length |
| Disposable scalpel | Feather | No. 15 | For cutting artery rings |
| Activated charcoal | Sigma | C4386-500G | For marking cut locations on vessels |
| 18G Needle | Beckton-Dickinson | 305136 | For applying activated charcoal to vessels, clearing blood and filling myograph tubing |
| 20 mL syringe | Various suppliers | For clearing blood and filling myograph tubing | |
| Petri dish | Fisher Scientific | 08-757-13B | For inserting vessels after dissection and testing to take pictures |
| Microfuge tube | Fisher Scientific | 02-682-550 | For storing vessels before testing |
| Fine wire | California Fine Wire Company | 100192 | For clearing clogged cannula |
| ImageJ software | National Health Institute | www. rsbweb.nih.gov/ij | Open-source image processing program developed by NIH |
| Matlab software | Mathworks | Useful for analyzing data and fitting constitutive equations |
References
- Dobrin, P. B., Sumpio, B. E., Sidawy, A. N., DePalma, R. G. Chapter 3: Physiology and Pathophysiology of Blood Vessels. The Basic Science of Vascular Disease. , 69-105 (1997).
- Wagenseil, J. E., Mecham, R. P. Vascular extracellular matrix and arterial mechanics. Physiol. Rev. 89 (3), 957-989 (2009).
- Le, V. P., Knutsen, R. H., Mecham, R. P., Wagenseil, J. E. Decreased aortic diameter and compliance precedes blood pressure increases in postnatal development of elastin-insufficient mice. Am. J. Physiol. Heart Circ. Physiol. , (2011).
- Pezet, M. Elastin haploinsufficiency induces alternative aging processes in the aorta. Rejuvenation Res. 11 (1), 97-112 (2008).
- Dye, W. W., Gleason, R. L., Wilson, E., Humphrey, J. D. Altered biomechanical properties of carotid arteries in two mouse models of muscular dystrophy. J. Appl. Physiol. 103 (2), 664-672 (2007).
- Ma, X. Upregulation of elastase proteins results in aortic dilatation in mucopolysaccharidosis I mice. Mol. Genet. Metab. 94 (3), 298-304 (2008).
- Eberth, J. F. Importance of pulsatility in hypertensive carotid artery growth and remodeling. J. Hypertens. 27 (10), 2010-2021 (2009).
- Faury, G. Developmental adaptation of the mouse cardiovascular system to elastin haploinsufficiency. J. Clin. Invest. 112 (9), 1419-1428 (2003).
- Amin, M., Kunkel, A., Le, V. P., Wagenseil, J. E. Effects of Storage Duration on the Mechanical Behavior and Morphology of Mouse Carotid Artery. Journal of Biomechanical Engineering. 133, (2011).
- Fung, Y. C. . Biomechanics : Mechanical Properties of Living Tissues. , (1993).
- Wagenseil, J. E. Effects of elastin haploinsufficiency on the mechanical behavior of mouse arteries. Am. J. Physiol. Heart. Circ. Physiol. 289 (3), H1209-H1217 (2005).
- Chuong, C. J., Fung, Y. C. On residual stresses in arteries. J. Biomech. Eng. 108 (2), 189-192 (1986).
- Fonck, E. Effect of elastin degradation on carotid wall mechanics as assessed by a constituent-based biomechanical model. Am. J. Physiol. Heart. Circ. Physiol. 292 (6), H2754-H2763 (2007).
- Rezakhaniha, R., Fonck, E., Genoud, C., Stergiopulos, N. Role of elastin anisotropy in structural strain energy functions of arterial tissue. Biomech. Model Mechanobiol. 10 (4), 599-611 (2011).
- Eberth, J. F., Taucer, A. I., Wilson, E., Humphrey, J. D. Mechanics of carotid arteries in a mouse model of Marfan Syndrome. Ann. Biomed. Eng. 37 (6), 1093-1104 (2009).
- Wan, W., Yanagisawa, H., Gleason, R. L. Biomechanical and microstructural properties of common carotid arteries from fibulin-5 null mice. Ann. Biomed. Eng. 38 (12), 3605-3617 (2010).
- Faury, G. Relation between outer and luminal diameter in cannulated arteries. Am. J. Physiol. 277 (5 Pt. 2), H1745-H1753 (1999).
- Sonveaux, P. Endothelin-1 is a critical mediator of myogenic tone in tumor arterioles: implications for cancer treatment. Cancer Res. 64 (9), 3209-3214 (2004).
- Mulcahy, C., Amin, M., Wagenseil, J. E. Creation of a finite element model of a mouse artery to examine circumferential stress distribution. Biomedical Engineering Society Annual Meeting. , (2010).

