Intravital Video Microscopy Measurements of Retinal Blood Flow in Mice
Summary
Intravital microscopy can be used in animals to visualize and measure retinal vascular diameters, bloodstream velocities, and total retinal blood flow.
Abstract
Alterations in retinal blood flow can contribute to, or be a consequence of, ocular disease and visual dysfunction. Therefore, quantitation of altered perfusion can aid research into the mechanisms of retinal pathologies. Intravital video microscopy of fluorescent tracers can be used to measure vascular diameters and bloodstream velocities of the retinal vasculature, specifically the arterioles branching from the central retinal artery and of the venules leading into the central retinal vein. Blood flow rates can be calculated from the diameters and velocities, with the summation of arteriolar flow, and separately venular flow, providing values of total retinal blood flow. This paper and associated video describe the methods for applying this technique to mice, which includes 1) the preparation of the eye for intravital microscopy of the anesthetized animal, 2) the intravenous infusion of fluorescent microspheres to measure bloodstream velocity, 3) the intravenous infusion of a high molecular weight fluorescent dextran, to aid the microscopic visualization of the retinal microvasculature, 4) the use of a digital microscope camera to obtain videos of the perfused retina, and 5) the use of image processing software to analyze the video. The same techniques can be used for measuring retinal blood flow rates in rats.
Introduction
The retina is one of the most metabolically active tissues in the body, and consequently requires a generous blood supply. Two separate circulations meet this demand: the choroidal circulation for the outer portion of the retina, and the retinal circulation for the inner portion of the retina. Investigations of retinal perfusion are imperative for understanding the pathological mechanisms and consequences of diabetic retinopathy, oxygen-induced retinopathy, retinal artery or vein occlusion, and stroke. Several methods have been employed to quantify retinal blood flow, with each technique having its advantages, disadvantages, limitations, and assumptions. Among these techniques are infusion of 7-8 μm diameter microspheres that lodge in precapillary arterioles1,2, quantitative autoradiography3,4, optical microangiopathy-optical coherence tomography5,6, magnetic resonance imaging7,8, and intravital video microscopy9-16. Advantages of the latter include direct live visualization of retinal vessels and flow, a dependence on only a few minor assumptions, and affordability for labs having a fluorescence microscope with an attached video camera. In previous studies of intravital video microscopy9-16, fluorescent dextran has been used as a plasma marker, and fluorescently labeled red blood cells (from a donor animal) have been used as velocity markers. In the current protocol, 1.9-μm diameter fluorescently labeled microspheres, instead of red blood cells, are used to measure velocity, with this alteration negating the need for a blood cell donor.
Protocol
The procedures involving the use of animals were reviewed and approved by the Institutional Animal Care and Use Committee of LSUHSC-S and performed according to the criteria outlined by the National Institutes of Health.
1. Preparation of Perfusion Solutions
- Sonicate a 1% (by weight) stock solution of 1.9 µm diameter microspheres.
- Withdraw ~25-30 x 106 green fluorescent microspheres (that is, ~10 µl of the stock solution) into a 300 µl syringe. Dilute the microspheres by withdrawing an additional 240 µl of saline into the syringe.
- Prepare a 50 µl solution of 2 x 106 molecular weight fluorescent dextran dissolved in sterile saline, with the injected dose in the range of 3-10 mg/kg. Keep the dextran covered in aluminum foil until time for its infusion.
2. Animal Anesthesia and Vascular Cannulation
- Anesthetize the animal for surgical insertion of vascular cannulas and for stationary positioning under a microscope objective. Many choices of anesthetic protocols are available, with various advantages and disadvantages, and potential effects on systemic parameters. Among the many different choices are a combination of ketamine (100-150 mg/kg), xylazine (10 mg/kg), and acepromazine (2 mg/kg), or a combination of ketamine (50 mg/kg) and pentobarbital (50 mg/kg). The described procedures are acute and conclude with euthanasia induced by an overdose of pentobarbital (150 mg/kg) followed by a thoracotomy; inclusion of an analgesic is not necessary for the terminal procedure.
- Maintain a heating pad underneath the animal for the remainder of the experiment.
- Keep the eyes moist with phosphate buffered saline during the subsequent surgical insertions of vascular cannulas.
- Prepare a vascular cannula for subsequent infusions of fluorescent tracers. A 20 cm length of polyethylene tubing (for example, 0.28 mm inside diameter; 0.61 mm outside diameter) is placed on the tip of a syringe needle, with the sharp end of the needle broken off beforehand by bending back and forth with a hemostat.
- Fill a syringe and tubing with heparinized (25 U/ml) saline.
- Shave one side of the lower abdomen and make an incision to expose the femoral vein.
- To cannulate the femoral vein, tie off the vessel with a 5.0 suture, make a partial incision of the vessel (approximately one-half the vessel diameter), and insert the tubing into the vessel, securing the tubing with the suture tied around the cannulated section.
3. Preparation for Intravital Microscopy
- Dilate the pupil of the eye with a drop of 1% tropicamide ophthalmic solution followed by a drop of 2.5% hypromellose ophthalmic solution.
- Cover the eye with a 5 mm glass coverslip.
- Place the animal on a plexiglass board, which will be positioned underneath the microscope objective of an upright microscope. Strategically place surgical gauze underneath the head to allow a direct axis through the objective to the central retina and optic disk.
- Focus through the microscope on the optic disk, finding the disk with a low power 4X objective and using a fluorescein filter.
4. Infusion of Fluorescent Microspheres to Measure Velocities
- Using a 4X objective, focus on the retina with the optic disk in the center of the field of view.
- Infuse the fluorescent microsphere solution at a rate of 250 µl/min/kg.
- Video-record the viewable retina around the optic disk, using a camera exposure time of ~8 msec.
- Stop the microsphere infusion when a sufficient duration of video has been recorded (1-2 min); some microsphere solution likely will remain unused.
5. Infusion of a Fluorescent Plasma Marker to Measure Diameters
- Using a 4X objective, focus on the retina with the optic disk in the center of the field of view.
- Infuse the prepared solution of fluorescent dextran into the femoral vein as a bolus over ~5 sec, while video-recording. This video will help determine the identity of the arterioles vs venules, with the arterioles being perfused first. Perform this step with the camera exposure time set to ~8 msec, although the precise exposure time is not critical.
- Switch to a 10X objective and 20-40 msec exposure time for improved resolution to video each of the 4-7 arterioles and 4-7 venules in the superficial retina. Focus on (and video-record) approximately one quadrant of the viewable retina at a time, with the center of the optic disk in one corner of the field of view. An attempt should be made to minimize the time that the microscope light illuminates the tissue to reduce the possibility of light/dye-induced phototoxicity17.
6. Video Analysis
- Using a micrometer scale, calibrate the video system for a conversion of µm/pixel.
- With image processing software, play back the recorded video.
- From the video of the bolus infusion of fluorescent dextran, identify arterioles vs venules.
- For each of the 4-7 arterioles and 4-7 venules recorded with the 10X objective, measure the diameters filled with fluorescent dextran, averaging 5 measures of diameter per vessel along the viewable length.
- For each of the same arterioles and venules, measure the streak length of the fluorescent microspheres, using 10 successive microsphere streaks in each vessel to avoid selection bias. Divide the streak lengths by the exposure time to calculate the microsphere velocity, for example, 160 µm/8 msec = 20 µm/msec = 2 cm/sec. Average the 10 velocities per vessel.
- Calculate the volumetric flow rate for each vessel with the following equation: flow = mean velocity × π × diameter2/4.
- Sum the arteriolar flow rates, and separately the venular flow rates, to obtain total retinal blood flow.
Representative Results
Figure 1 shows single frames of video from an experiment, with panels A-D and F-I showing fluorescent microsphere streaks captured with a 4X objective and 8 msec exposure time (and binning 2 x 2 pixels to reduce the video file size). Figure 1E shows the orientation of the retinal vessels in the other panels of the figure. Not every frame of video will have a fluorescent streak in focus; however, some frames may have multiple streaks for use in the analysis (e.g. panels C, D, G, H, and I). In this experiment, most of the fluorescent streaks (captured during an 8 msec exposure time) were in the range of 120-170 µm in length, which corresponds to velocities of 1.5-2.1 cm/sec.
Figure 2 shows nine frames of video from the bolus infusion of 2 x 106 molecular weight fluorescein isothiocyanate-labeled dextran, with 0.071 sec (twice the frame capture rate) between the selected frames. The length of time between the first and final panels of Figure 2 is ~0.57 sec. The vessels filling first with the fluorescent dextran (shown in panels A-D) are arterioles, in this case corresponding to “clock” positions of ~1:30, 4:30, 7:00, and 10:00. After a short delay, the venules (in positions of ~12:00, 3:30, 5:30, and 9:00) are filled after the dye makes its transit through the capillary beds.
Figure 3 shows a 10X view of two of the vessels (in the 9:00 and 10:00 positions), with this and similar pictures of the other vessels used to measure diameters. In this experiment, the arteriolar diameters were measured to be in the range of 47-55 µm, and the venules measured to be 50-65 µm in diameter.
The velocities and diameters are inserted into a volumetric flow calculation, flow = mean velocity × π × diameter2/4. For example, 2.0 cm/sec × π × (55×10-4 cm)2/4 = 4.8×10-5 cm3/sec = 48 nl/sec. Summing the 4 arterioles (or the 4 venules) together provides a calculation of the total retinal blood flow rate. In the mouse eye of this example experiment, the sum of arteriolar flow was 138 nl/sec and the sum of venular flow was 172 nl/sec.
Figure 4 shows flow measurements from an experiment in which both fluorescent microspheres and fluorescent red blood cells (as described in our previous studies9-16) were injected into the same mouse, to determine if similar vessel velocities and flows would be found. In this experiment, the mouse retina had 6 arterioles and 6 venules, with measured velocities and flows very similar (p=0.75 for arterioles; p=0.38 for venules).
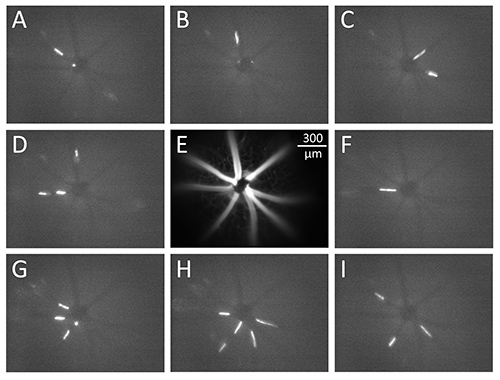
Figure 1. Video frames captured during an intravital microscopy experiment. Fluorescent microspheres 1.9 µm in diameter flow through retinal vessels (panels A-D and F-I), with the appearance of the microspheres as streaks during the camera exposure time of 8 msec. Panel E shows the orientation of the blood vessels in the superficial layer of the retina. The scale bar in panel E corresponds to all images in Figure 1.

Figure 2. Infusion of high molecular weight dextran through the retinal circulation. Bolus infusion of fluorescent dextran, with 0.071 sec between the selected frames. The vessels filling first with the fluorescent dextran (panels A-D) are arterioles, with the venules filled after the dye makes its transit through the capillary beds. The scale bar in panel E corresponds to all images in Figure 2.
Click here to view larger image.

Figure 3. A retinal arteriole and venule at higher magnification. A 10X objective was used to capture images of the superficial retinal vessels for measurements of diameter.
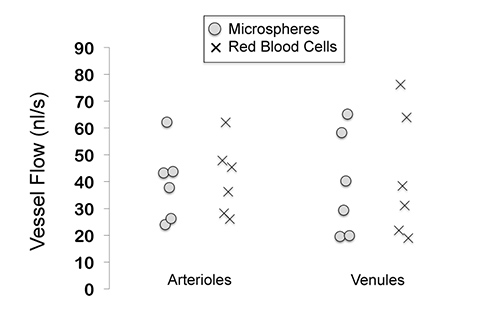
Figure 4. Retinal microvessel flows measured with the use of fluorescent microspheres and fluorescent red blood cells. No statistical differences between the two techniques were found (using a paired t-test). In this experiment, the flows were measured in 6 arterioles and 6 venules of a mouse retina.
Click here to view larger image.
Discussion
This technique of intravital video microscopy can be applied not only to mice, but also to rats. This protocol relies on only several assumptions, but is limited to use under anesthesia and with pupil dilation as we have described it. The other assumptions and limitations are as follows:
1. Assumption of minimal optical magnification error resulting from the refractive nature of the eye. As described by others18-20, the method of filling the space between the cornea and coverslip with an ophthalmic solution fluid layer counteracts the refractive power of the cornea. Any unaccounted magnification would affect measurements of length and diameter.
2. Limitation of optical resolution. We typically use a 10X objective (working distance = 1.05 cm) for our diameter measurements, with this objective having a numerical aperture of 0.25 and a nominal resolution of ~1.2 µm (approximately the uncertainty in the measurement). The overall resolution is not limited by the camera/image processor resolution at this magnification, equal to 0.47 µm/pixel with no pixel binning and equal to 0.94 µm/pixel with 2 x 2 binning. Higher numerical aperture objectives can be used for this protocol to improve resolution, as long as the working distances are sufficient: the axial lengths of the mouse and rat eyes are ~3.4 mm and ~6.3 mm, respectively21-23.
3. Assumption of dextran filling the entire vascular lumen. The endothelial glycocalyx may largely exclude the 2 x 106 molecular weight dextran over the short time frame involved in the measurement protocol. Therefore, this technique likely defines the inner vessel diameter as beginning at the lumen edge of the glycocalyx, which is typically estimated to extend 0.1-0.5 µm from the endothelial cell membrane24. This thickness is smaller than the optical resolution of the video collection system, but contributes to an uncertainty in the diameter measurement.
4. Assumption of right circular cylindrical vessels. Previous measurements of retinal blood flow using intravital video microscopy have found slightly higher values of venular flow compared to arteriolar flow11-13,15, with this discrepancy likely due to an elliptical shape of the venular diameter. The venular diameter may have its longer radius perpendicular to the optical axis, and therefore calculations of venular flow may be slightly overestimated. We have previously documented similar geometrical effects on intravital microscopic measurements of flow in intestinal submucosal venules of mice25.
5. Assumption that the sampled velocities represent the mean. The microspheres flowing closer to the vessel wall will travel more slowly that those in the centerline. Our assumption in past studies (using fluorescently labeled RBCs) is that 10 velocity measurements are sufficient to obtain a reasonably accurate distribution of velocities whose mean approximates the true average red blood cell velocity. However, measuring more than 10 velocities per vessel is likely to provide even more precision in the average.
This protocol was written specifically for use in the retina; however, the procedures can be applied to other tissues that are accessible via intravital microscopy. The protocol requires minimal troubleshooting, with the main step that requires some practice being the alignment of the mouse's head in the correct position to allow a direct optical path through the objective to the center of the optic disk. If the retina is not easily seen through the iris, visually confirm that the pupil has been sufficiently dilated by the tropicamide solution. In addition, the arteriolar and venular microspheres will not always be in focus simultaneously, as the arterioles and venules are slightly offset from each other: therefore, collecting the video requires some adjustments up and down in the focus. Several steps in the procedure can be modified to variations in the available equipment. For example, a digital camera (although helpful) is not required: we have used a similar protocol with a nondigital camera that used standard frame rates, and with our previous method of using fluorescently labeled red blood cells or platelets from donor animals instead of microspheres10,16. The critical steps are simply to be able to record the diameters of the fluorescently filled blood vessels and to record the velocity of fluorescently labeled bloodstream markers.
Disclosures
The authors have nothing to disclose.
Acknowledgements
Funded by NIH EY017599 (NRH).
Materials
| Name of the Reagent/Equipment | Company | Catalog Number or Product | Comments/Description |
| Fluorescent microspheres | Bangs Laboratories | FS04F/10584 (green) | |
| High molecular weight fluorescent dextran | Molecular Probes | D-7137 (green); D-7139 (red) | |
| Microscope system | Nikon | Eclipse E600FN + attachments | |
| 4X objective | Nikon | Plan Fluor 4X | numerical aperture 0.13; working distance 17.2 mm |
| 10X objective | Nikon | Plan 10X | numerical aperture 0.25; working distance 10.5 mm |
| Tropicamide ophthalmic solution | Bausch & Lomb | 1% Tropicamide | |
| Hypromellose ophthalmic solution | HUB Pharmaceuticals | 2.5% Goniovisc | |
| Image processing software | University of California San Francisco Vale Lab | Micro-Manager | |
| Digital video camera for microscopy | Photometrics | CoolSnap ES | 1392 x 1040 pixel resolution; pixel size 6.45 x 6.45 μm |
References
- Wang, L., Fortune, B., Cull, G., McElwain, K. M., Cioffi, G. A. Microspheres method for ocular blood flow measurement in rats: size and dose optimization. Exp. Eye Res. 84, 108-117 (2007).
- Wang, L., Grant, C., Fortune, B., Cioffi, G. A. Retinal and choroidal vasoreactivity to altered PaCO2 in rat measured with a modified microsphere technique. Exp. Eye Res. 86, 908-913 (2008).
- Pouliot, M., Hetu, S., Lahjouji, K., Couture, R., Vaucher, E. Modulation of retinal blood flow by kinin B(1) receptor in Streptozotocin-diabetic rats. Exp. Eye Res. 92, 482-489 (2011).
- Pouliot, M., et al. Quantitative and regional measurement of retinal blood flow in rats using N-isopropyl-p-[14C]-iodoamphetamine ([14C]-IMP). Exp. Eye Res. 89, 960-966 (2009).
- Zhi, Z., et al. Volumetric and quantitative imaging of retinal blood flow in rats with optical microangiography. Biomed. Opti. Express. 2, 579-591 (2011).
- Zhi, Z., Cepurna, W. O., Johnson, E. C., Morrison, J. C., Wang, R. K. Impact of intraocular pressure on changes of blood flow in the retina, choroid, and optic nerve head in rats investigated by optical microangiography. Biomed. Opti. Express. 3, 2220-2233 (2012).
- Li, G., De La Garza, B., Shih, Y. Y., Muir, E. R., Duong, T. Q. Layer-specific blood-flow MRI of retinitis pigmentosa in RCS rats. Exp. Eye Res. 101, 90-96 (2012).
- Muir, E. R., Renteria, R. C., Duong, T. Q. Reduced ocular blood flow as an early indicator of diabetic retinopathy in a mouse model of diabetes. Invest. Ophthalmol. Vis. Sci. 53, 6488-6494 (2012).
- Wright, W. S., Harris, N. R. Ozagrel attenuates early streptozotocin-induced constriction of arterioles in the mouse retina. Exp. Eye Res. 86, 528-536 (2008).
- Lee, S., Harris, N. R. Losartan and ozagrel reverse retinal arteriolar constriction in non-obese diabetic mice. Microcirculation. 15, 379-387 (2008).
- Wright, W. S., Messina, J. E., Harris, N. R. Attenuation of diabetes-induced retinal vasoconstriction by a thromboxane receptor antagonist. Exp. Eye Res. 88, 106-112 (2009).
- Wang, Z., Yadav, A. S., Leskova, W., Harris, N. R. Attenuation of streptozotocin-induced microvascular changes in the mouse retina with the endothelin receptor A antagonist atrasentan. Exp. Eye Res. 91, 670-675 (2010).
- Yadav, A. S., Harris, N. R. Effect of tempol on diabetes-induced decreases in retinal blood flow in the mouse. Curr. Eye Res. 36, 456-461 (2011).
- Wang, Z., Yadav, A. S., Leskova, W., Harris, N. R. Inhibition of 20-HETE attenuates diabetes-induced decreases in retinal hemodynamics. Exp. Eye Res. 93, 108-113 (2011).
- Wright, W. S., Yadav, A. S., McElhatten, R. M., Harris, N. R. Retinal blood flow abnormalities following six months of hyperglycemia in the Ins2(Akita) mouse. Exp. Eye Res. 98, 9-15 (2012).
- Lee, S., Morgan, G. A., Harris, N. R. Ozagrel reverses streptozotocin-induced constriction of arterioles in rat retina. Microvasc. Res. 76, 217-223 (2008).
- Rumbaut, R. E., Sial, A. J. Differential phototoxicity of fluorescent dye-labeled albumin conjugates. Microcirculation. 6, 205-213 (1999).
- Tadayoni, R., Paques, M., Gaudric, A., Vicaut, E. Erythrocyte and leukocyte dynamics in the retinal capillaries of diabetic mice. Exp. Eye Res. 77, 497-504 (2003).
- Walsh, M. K., Quigley, H. A. In vivo time-lapse fluorescence imaging of individual retinal ganglion cells in mice. J. Neurosci. Methods. 169, 214-221 (2008).
- Shahidi, M., Wanek, J., Blair, N. P., Mori, M. Three-dimensional mapping of chorioretinal vascular oxygen tension in the rat. Invest. Ophthalmol. Vis. Sci. 50, 820-825 (2009).
- Hughes, A. A schematic eye for the rat. Vis. Res. 19, 569-588 (1979).
- Remtulla, S., Hallett, P. E. A schematic eye for the mouse, and comparisons with the rat. Vis. Res. 25, 21-31 (1985).
- Schmucker, C., Schaeffel, F. In vivo biometry in the mouse eye with low coherence interferometry. Vis. Res. 44, 2445-2456 (2004).
- Mulivor, A. W., Lipowsky, H. H. Inflammation- and ischemia-induced shedding of venular glycocalyx. Am. J. Physiol. Heart Circ. Physiol. 286, 1672-1680 (2004).
- Harris, N. R., Whatley, J. R., Carter, P. R., Specian, R. D. Venular constriction of submucosal arterioles induced by dextran sodium sulfate. Inflamm. Bowel Dis. 11, 806-813 (2005).

