A High Yield and Cost-efficient Expression System of Human Granzymes in Mammalian Cells
Summary
We describe here a cost-efficient granzyme expression system using HEK293T cells that produces high yields of pure, fully glycosylated and enzymatically active protease.
Abstract
When cytotoxic T lymphocytes (CTL) or natural killer (NK) cells recognize tumor cells or cells infected with intracellular pathogens, they release their cytotoxic granule content to eliminate the target cells and the intracellular pathogen. Death of the host cells and intracellular pathogens is triggered by the granule serine proteases, granzymes (Gzms), delivered into the host cell cytosol by the pore forming protein perforin (PFN) and into bacterial pathogens by the prokaryotic membrane disrupting protein granulysin (GNLY). To investigate the molecular mechanisms of target cell death mediated by the Gzms in experimental in-vitro settings, protein expression and purification systems that produce high amounts of active enzymes are necessary. Mammalian secreted protein expression systems imply the potential to produce correctly folded, fully functional protein that bears posttranslational modification, such as glycosylation. Therefore, we used a cost-efficient calcium precipitation method for transient transfection of HEK293T cells with human Gzms cloned into the expression plasmid pHLsec. Gzm purification from the culture supernatant was achieved by immobilized nickel affinity chromatography using the C-terminal polyhistidine tag provided by the vector. The insertion of an enterokinase site at the N-terminus of the protein allowed the generation of active protease that was finally purified by cation exchange chromatography. The system was tested by producing high levels of cytotoxic human Gzm A, B and M and should be capable to produce virtually every enzyme in the human body in high yields.
Introduction
The Gzms are a family of highly homologous serine proteases localized in specialized lysosomes of CTL and NK cells1. The cytotoxic granules of these killer cells also contain the membrane-disrupting proteins PFN and GNLY that are released simultaneously with the Gzms upon recognition of a target cell destined for elimination2,3. There are five Gzms in humans (GzmA, B, H, K and M), and 10 Gzms in mice (GzmA – G, K, M and N). GzmA and GzmB are the most abundant and extensively studied in humans and mice1. However, more recent studies have begun to investigate the cell-death pathways as well as the additional biological effects mediated by the other, so called orphan Gzms in health and disease4.
The best known function of the Gzms, in particular of GzmA and GzmB, is the induction of programmed cell death in mammalian cells when delivered into the target cells by PFN5,6. However, more recent studies also demonstrated extracellular effects of the Gzms with profound impact on immune regulation and inflammation, independently of cytosolic delivery by PFN7,8. The spectrum of cells that are killed efficiently after cytosolic entry of the Gzms was also recently widened from mammalian cells to bacteria9,10and even certain parasites11. These recent discoveries opened up a whole new field for Gzm researchers. Therefore, a cost-efficient, high-yield mammalian expression system will significantly ease the way for those future studies.
Native human, mouse and rat Gzms have been successfully purified from the granule fraction of CTL and NK cells lines12-14. However, in our hands the yield of such purification techniques is in the range of less than 0.1 mg/L cell culture (unpublished observation and12). Furthermore, the chromatographic resolution of a single Gzm without contamination by other Gzms and/or proteins that are also present in the granules is challenging (unpublished data and12,14). Recombinant Gzms were produced in bacteria15, yeast16, insect cells17and in even in mammalian cells such as HEK 29318,19. Only the mammalian expression systems bear the potential to produce recombinant enzymes with posttranslational modifications identical to the native cytotoxic protein. Posttranslational modifications have been implicated with the specific uptake by endocytosis and the intracellular localization of the protease within target cells20-22. Therefore, by using pHLsec23(a kind gift of Radu Aricescu and Yvonne Jones, University of Oxford, UK) as the plasmid backbone for Gzm expression, we established a simple, time- and cost-efficient system for high-yield protein production in HEK293T cells. pHLsec combines a CMV enhancer with a chicken Β-actin promoter; together, these elements demonstrated the strongest promoter activity in various cell lines24. In addition, the plasmid contains a rabbit Β-globin intron, optimized Kozak and secretion signals, a Lys-6xHis-tag and a poly-A signal. Inserts can be cloned conveniently between the secretion signal and the Lys-6xHis-tag (Figure 1) ensuring optimal expression and secretion efficiency for proteins lacking appropriate N-terminal domains. For the expression of the Gzms, we replaced the endogenous secretion signal sequence with the secretion signal provided by the vector followed by an enterokinase (EK) site (DDDDK) so that EK treatment activated the secreted Gzms (active Gzms start with the N-terminal amino acid sequence IIGG25). Additionally in favor for this method, HEK293T cells grow rapidly in low priced medium, such as Dulbecco’s Modified Eagle’s Medium (DMEM), and are well suited for cost-efficient calcium-phosphate transfection method.
Protocol
1. Production of the expression plasmid pHLsec-Gzm
- Prepare total RNA from human NK cells (primary cells prepared as in26or the NK cell line NK-92 expressing all five human Gzms) using a suitable RNA isolation method and reverse transcribe using a first-strand cDNA synthesize kit, following the manufacturer`s recommendations. Amplify Gzm cDNA using PCR and clone into pHLsec as described in23(kind gift of Radu Aricescu and Yvonne Jones, University of Oxford, UK) using the AgeI and KpnI sites (Figure 1).
NOTE: Use the following primers indicated in Table 1. - Confirm correct inserts by sequencing. Expand the expression plasmids in DH5α cells and purify using an endotoxin-free plasmid isolation kit and follow the manufacturer’s instructions.
- Resuspend the purified plasmids in endotoxin-free, sterile water at a concentration of 2 mg/ml and store at -20 °C until use.
2. Expression of Gzms in HEK293T cells
- Preparation of reagents
- Prepare standard culture medium. To Dulbecco's Modified Eagle Medium (DMEM) containing high glucose (25 mM), GlutaMax (4 mM), sodium pyruvate (1 mM) add 10% of standard fetal calf serum (FCS), penicillin (100 units/ml), streptomycin (100 μg/ml).
- Prepare transfection medium. To culture medium without penicillin-streptomycin add 25 μg/ml chloroquine (add freshly on the day of transfection from 1,000x stocks in PBS, stored at -20 °C).
- Prepare serum-free culture medium: To serum-free medium for HEK293 cells add glutamine (4 mM), penicillin (100 units/ml), streptomycin (100 μg/ml), and 2.5mg/L amphotericin B.
- Prepare the solutions described in Table 2 and filter with a 0.45 μm filter before use:
- Expanding HEK293T cells
- Grow HEK293T cells in 20 ml culture medium using 150 x 25 mm tissue culture dishes.
- Split cells at 80% confluency (split-ratio of 1:4, usually every 3rd day). Cells loosely attach, they can be mechanically detached without trypsinization by rigorously pipetting up and down.
- Plate cells the night before transfection to give 60-70% confluency at the day of transfection (seeding density ~2 x 105cells/ml). A usual preparation size consists of 20 to 25 culture dishes.
- Calcium-phosphate transfection
- 1 hr prior to transfection, change the standard culture medium to 20 ml transfection medium. Near the end of this 1 hr incubation step, mix 400 μg of plasmid DNA with 10.95 ml ddH20 and 1.55 ml of 2M CaCl2 in 50 ml tubes (1 tube/5 dishes).
- 1 to 2 min prior to the transfection, add 12.5 ml 2x HBS to 12.5 ml of the DNA-Calcium solution, mix by inversion of the tubes and incubate for 30 sec at RT.
- Add the mixture prepared in 2.3.2 directly to the cells (5 ml per dish), drop-wise into the medium. Sprinkle evenly over the entire area, turning the medium to a slightly orange color.
- Incubate the culture dishes for 7 to 11 hr in a tissue culture incubator (37°C, 5% CO2 in humidified air). After the incubation, a fine precipitate is visible. Remove the transfection medium, rinse carefully with pre-warmed PBS and add 20 ml serum-free culture medium prepared in 2.1.3. Incubate for 72 hr in a tissue culture incubator.
- Analyze the transfection and expression efficiency by running a sample (~20 μl) of the cell supernatant on SDS-PAGE and staining with Coomassie Brilliant Blue to visualize a granzyme band.
- Purification of Gzms from culture supernatant by Nickel-IMAC
- After the incubation, decant the cell culture supernatants into 250 ml tubes and clear by centrifugation. Carry out a first spin (400 x g, 10 min, 4 °C) that will clear the medium from detached cells. Transfer the supernatant in fresh 250 ml tubes and spin at 4,000 x g, 30 min at 4 °C to remove any remaining cell debris.
- Add 5 ml of 5 M NaCl, 6.25 ml of 2 M Tris-Base (pH 8) and 1 ml of 0.25 M NiSO4per 250 ml of cleared supernatant. Filter the supernatant using a 500 ml Vacuum filter unit (0.45 mm).
- Equilibrate a Nickel-IMAC column with His-binding buffer A (10 column volumes, CV) using a suitable FPLC system.
- Apply the cleared supernatant to the equilibrated column at a flow rate of 0.5 ml/min at 4 °C.
- After the supernatant was applied, wash column with His-binding buffer until UV absorbance baseline is reached (usually 10 CV).
- Elute Gzms with a linear 20 ml-gradient (0 to 250 mM imidazole) at a flow rate of 0.5 ml/min while collecting 2 ml-fractions. Analyze the elution fractions by SDS-PAGE and Coomassie staining.
- Enterokinase (EK) treatment and final clean-up by cation exchange chromatography
- Pool Gzm containing fractions in a dialyzing tube with ≤10 MWCO. Store a small sample at -20 °C as pre-EK control.
- Add EK at 0.02 unit/50 ml of initial supernatant directly to the pooled fraction in the dialysis tube and dialyze O/N (at least 16 hr) at RT in EK-buffer (4 L, change buffer once).
- The next day, analyze EK treated Gzms in comparison to the pre-EK control by SDS-PAGE and Coomassie staining.
- When N-terminal processing is complete, change dialysis buffer to S buffer A (4 L) and dialyze for another 4 hr at RT. Filter dialysate (0.45 μm).
- Equilibrate a S-column with S buffer A. Load the sample on the column with at a flow rate of 0.5 ml/min at 4 °C.
- After sample loading, wash the column with S buffer A until UV absorbance baseline is reached (about 10 CV). Elute the Gzms with a 30 ml linear gradient (150 to 1,000 mM NaCl). GzmA elutes at ~650 mM NaCl, GzmB at ~700 mM NaCl and GzmM at ~750 mM NaCl.
- Analyze elution fractions by SDS-PAGE and colorimetric assays (see below). Pool fractions containing Gzms and concentrate (about 30-fold, to a concentration of about 100 μM) in spin filters (15 ml, 10 MWCO). Aliquot the concentrated Gzm preparations and store at -80 °C.
3. Testing Gzm activity
- Preparation of reagents
- Prepare colorimetric assay buffer: 50 mM Tris-Base, pH 7.
- For the measurement of GzmA, add N-a-CBZ-L-Lysine thiobenzyl ester (S-Bzl) ( BLT) to the assay buffer to a final concentration of 0.2 mM and 5,5′-dithio-bis(2-nitrobenzoic acid) (DTNB) to a concentration of 0.22 mM. For GzmB, include 0.2 mM Boc-Ala-Ala-Asp-S-Bzl ( AAD) and 0.22 mM DTNB. For GzmM, include 0.2 mM Suc-Ala-Ala-Pro-Leu-p-nitroanilide (pNA) ( AAPL). Synthetic peptides are added from appropriate stock solutions (in DMSO, stored at -20 °C) to the buffer freshly before the assay.
- Prepare cleavage assay buffer: 100 mM NaCl, 50 mM Tris-Base, pH 7.5
- Prepare colorimetric assay buffer: 50 mM Tris-Base, pH 7.
- Colorimetric assays
- Distribute small Gzm samples (<5 μl) from elution fractions or concentrated stocks in the 96-well flat-bottom plates. Include a positive control (tested Gzm preparations or crude NK cell lysates) and a negative control (buffer only).
- Add 100 μl of assay buffer to each well (using a multi-channel pipette). Incubate for 10 min at 37 °C. Measure OD at 405 nm in a microplate reader.
- Cleavage assay
- For cleavage assays using purified substrates, co-incubate a protein substrate (native or recombinant, 100 – 400 nM; a few examples of efficient Gzm substrates are presented in the result section and in the note at the end of this section) with serial dilutions of a Gzm (starting at 400 nM) in assay buffer for various times. Analyze by SDS-PAGE and Coomassie or silver staining; or by western blotting using specific antibodies.
- For cleavage assays using cell lysates, suspend 106 HeLa cells (or any available tumor cell line) in 1 ml of assay buffer and lyse by freezing in an ethanol/dry ice bath and thawing at 37 °C three times. Remove cell debris by centrifugation (15,000 x g for 10 min at 4 °C).
- Co-incubate the cleared lysate with Gzms at various concentrations and times as above. Cleavage is detected by immunoblotting using appropriate antibodies.
NOTE: A few examples of bona fide substrates for which commercial antibodies are available: Bid (BH3-interacting domain death agonist) and caspase 3 cleaved by GzmB27,28; multiple heterogeneous nuclear ribonucleoproteins (hnRNPA1, A2 and U) cleaved by GzmA29; cytomegalovirus phosphoprotein 71 by GzmM30.
Representative Results
In the following section we will present a complete documentation of a GzmA preparation to illuminate the method. We also successfully purified GzmB and GzmM, producing similar results in regard to purification efficiency and activity. However, from those later preparations we will only show a few selected pieces of data. GzmB preparations from 293T cells following the current protocol were used in several published studies, highlighting their activity in various biological assay systems9,29,31-34.
A typical GzmA purification is shown in Figure 2A. Efficient transfection and expression after 72 hr was indicated by a detectable Gzm band on a Coomassie-stained SDS-PAGE gel without further concentration of the culture supernatant (~37 kDa for GzmB and GzmM under reducing conditions; ~60/34 kDa for GzmA under non-reducing/reducing conditions). After the IMAC purification the samples were treated with enterokinase (EK). A visible shift of the EK treated sample (post-EK) compared to the pre-EK control was observed. The final purification on the S-column resulted in highly pure Gzm preparations (GzmA in Figure 2A; GzmB and GzmM in Figure 2B) with a single band appearance on Coomassie-stained protein gels. The pooled fractions were concentrated to a final concentration of ~100 μM. Typically, we obtained yields of about 0.5 to 1 mg Gzms per 100 ml culture supernatant. To demonstrate efficient glycosylation, a few initial preparations were treated with Endoglycosidase (Endo) H, which removes high mannose oligosaccharides from N-linked glycoproteins. Native GzmA has a N-linked glycosylation site at Asn-142, to which a high mannose oligosaccharide is bound35. EndoH treatment induced an obvious mobility shift in the Gzm A, as well as GzmB, preparations from HEK293T cells but not in bacterially generated GzmA as analyzed by SDS-PAGE and Coomassie staining (Figure 2C).
Routinely, every Gzm batch was tested for enzymatic activity in colorimetric assays. This rapid and reliable test demonstrates proteolytic activity of the Gzms by the cleavage of small synthetic peptides that is indicated spectrophotometrically by a color change of the assay buffer. Due to different preferences concerning the amino acid residues (P1 position) after which these proteases cleave, the choice of a specific peptide varies among the Gzms. GzmA has trypsin-like activity, cleaving after the basic residues arginine (Arg, R) and lysine (Lys, K). GzmB cleaves preferentially after aspartic acid residues (Asp, D), and GzmM cleaves after leucine (Leu, L) and methionine (Met, M)1. Our preferential peptide choice is BLT-S-Bzl to measure GzmA activity, AAD-S-Bzl for GzmB and AAPL-pNA for GzmM.
The major difference between the thiobenzylester substrates (AAD and BLT) and the p-nitroanilide substrate (AAPL) is the specific chemistry, in that Gzm mediates cleavage of thiobenzylester substrates releases benzyl-mercaptan, which only triggers a chromogenic reaction in its downstream reaction with the chromophore DTNB. Therefore, in the reaction buffers of thiobenzylester substrates DTNB has to be present, while the Gzm-mediated release of p-nitroanilide is sufficient for the colorimetric detection. The detailed method of the colorimetric assays was recently published36. Figure 3A shows the representative BLT and AAPL esterase activity in the elution fractions from the S-column of a GzmA and GzmM preparation, respectively. Colorimetric assay was also used to compare the activity of recombinant to native GzmB preparations. As shown in Figure 3B, recombinant 293T GzmB cleaved the chromogenic substrate with similar efficiency compared to native GzmB purified from a human NK cell line, as recently described12.
To more specifically test Gzm activity, Gzm cleavage assays using known protein substrates are indicated. These cleavage assays can be performed with purified recombinant or native protein (if available), with cell lysates or most physiological with intact cells. If a protein substrate is available, proteolytic activity can be analyzed in simple co-incubation experiments with the Gzm preparations as this is demonstrated with the known bacterial GzmA and GzmB substrates nuoCD and nuoF9, as well as with the novel human mitochondrial GzmM substrate NDUFAF3 (Figure 4A).
Cell lysates represent another obvious source of multiple Gzm substrates. Commercial antibodies are available against many of the substrates. A fast and simple method to generate cell lysates is by applying multiple freeze/thaw cycles as previously demonstrated33. The use of detergents is not recommended as they can interfere with Gzm activity. In Figure 4B, GzmA mediated cleavage of hnRNPA1 in a HeLa cell lysate is demonstrated in an immunoblot using an anti-hnRNPA1 antibody, as well as an anti-Β-actin antibody as loading control.
For cleavage assays (or cytotoxicity assays) in intact cells, the availability of an additional delivery molecule is necessary (PFN or Streptolysin O, SLO, for mammalian cells; GNLY or other antimicrobial peptides for prokaryotic cells). Cleavage assays in intact cells are most challenging as the concentration of the delivery molecules is critical and needs extensive fine tuning. Introducing these complex protocols of apoptosis assays is beyond the scope of this paper. Here, we may only refer to the vast body of literature, as examples12,13.
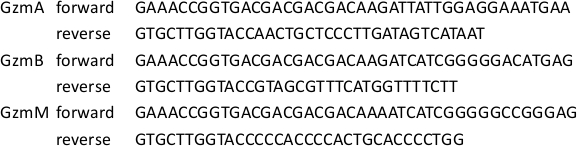
Table 1: Gzm Cloning Primer Sequences.
The primer sequences that were used to clone GzmA, GzmB and GzmM are indicated. Forward primers were designed to introduce an EK site before the N-terminus of the active protease (see Figure 1)

Table 2: Stock Solutions and Buffer Compositions.
The recipes of the most important stock solutions and buffers are indicated.
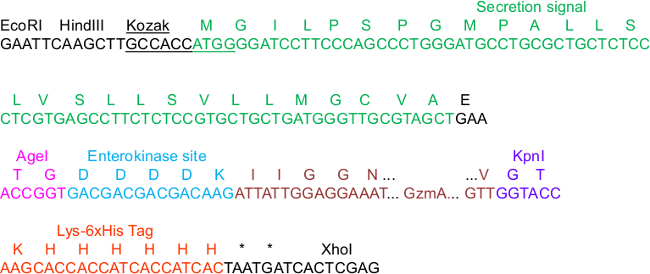
Figure 1. pHLsec Sequence Critical for Construct Cloning.
In the sequence, a few elements in the vector are highlighted that are important for cloning, secretion, IMAC purification and activation of the proteases: The Kozak consensus and secretion signal sequences, the AgeI and KpnI restriction sites, as well as the C-terminal Lys_6xHis tag. We exemplified GzmA as the insert that is N-terminally fused to an enterokinase (EK) site. For the full map of the complete vector backbone, we refer to the supplementary information in23.
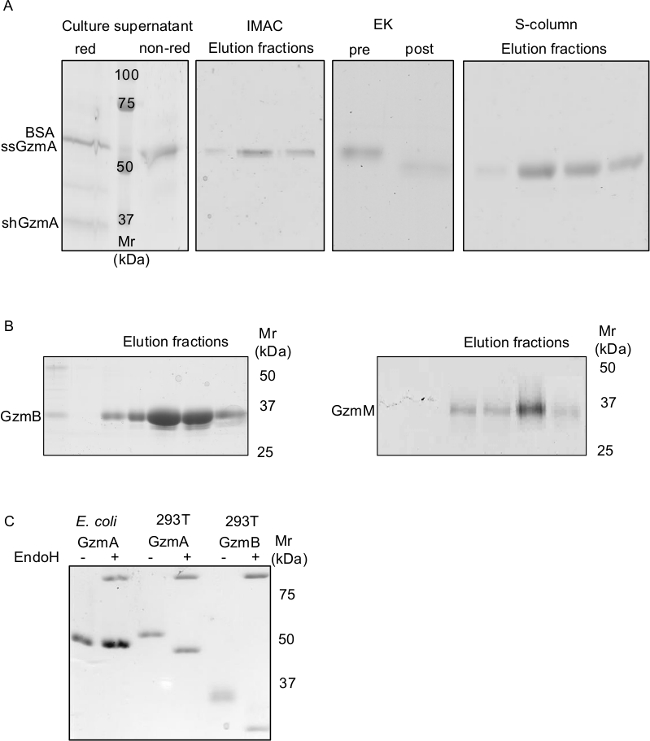
Figure 2. Expression, Purification and Activation of Fully Glycosylated Human Gzms from HEK293T Cells.
A) shows a series of Coomassie-stained, non-reducing SDS-PAGE gels demonstrating the whole GzmA production process from its secretion in the supernatant to first Nickel-IMAC purification, EK treatment and final polishing via cation exchange chromatography. The culture supernatant was run on SDS-PAGE under reducing (red) and non-reducing (non-red) conditions. GzmA is a homodimer (ssGzmA) that is stabilized by a disulfide bond. The GzmA homodimer (~60 kDa) runs close to contaminating BSA (~66 kDa). Under reducing conditions the GzmA monomer (shGzmA) appears as a ~34 kDa band. To demonstrate the purity of the final GzmB and GzmM preparations, representative Coomassie-stained gels under non reducing conditions are shown in B. Bacterially (E. coli) generated GzmA as well as GzmA and GzmB produced in HEK293T cells were treated with EndoH and analyzed by non-reducing SDS-PAGE and Coomassie staining. Glycosylation of the Gzms produced in HEK293T cells is demonstrated by a mobility shift after EndoH treatment as compared to non-glycosylated GzmA produced in bacteria (C).
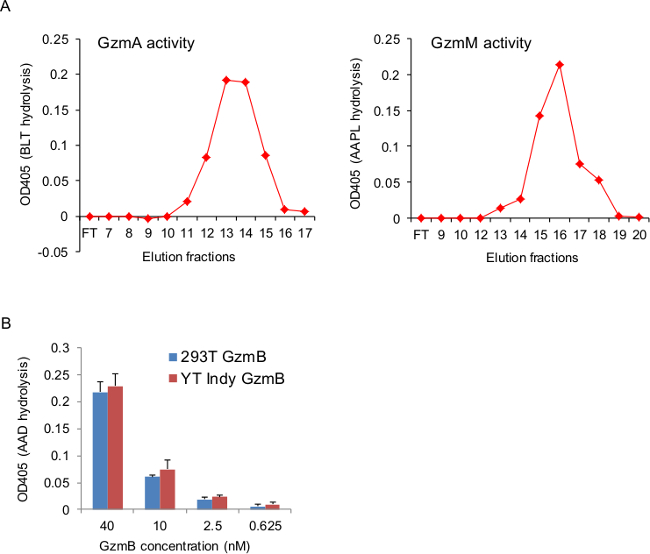
Figure 3. Recombinant Gzms Hydrolyze Synthetic Peptides.
A), small samples of the elution fractions from the final S column were tested for GzmA and GzmM activity in chromogenic assays using BLT and AAPL, respectively, as substrates. Graphs show peptide cleavage that was indicated as an OD increase at 405 nm. The activity peak fractions correlated with the protein peak in the elution as indicated in UV absorbance and SDS-PAGE analysis (Figure 2A and B). B), recombinant 293T and native GzmB (prepared as described12) at indicated concentrations were incubated in presence of the chromogenic substrate AAD. GzmB activity was indicated as an OD change at 405 nm. Graph shows the mean ± SEM of our most recent Gzm preparations that were tested in triplicates.
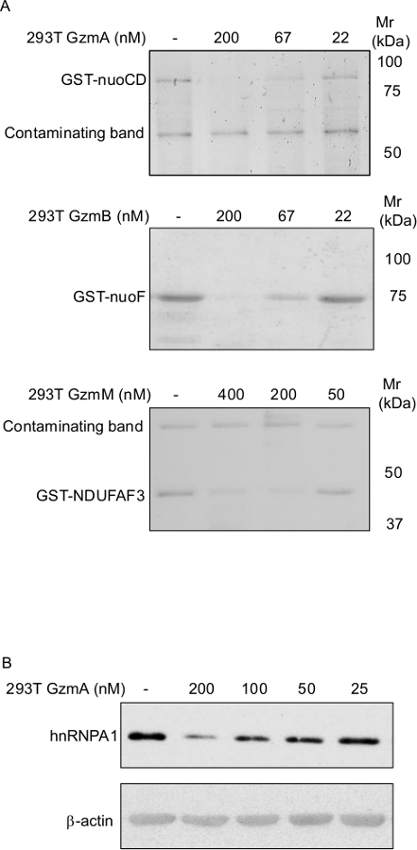
Figure 4. Recombinant Gzms Cleave Protein Substrates.
A) Recombinant GST-tagged, bacterial respiratory chain proteins nuoCD and nuoF, as well as the mammalian respiratory chain protein NDUFAF3, were treated with indicated concentration of indicated Gzms for 15 min at 37 °C. 1 μg of purified proteins in 20 μl reactions were used. Cleavage was analyzed by SDS-PAGE and Coomassie staining. B) HeLa cell lysate (at a protein concentration of ~1 mg/ml) was incubated with indicated concentrations of recombinant GzmA for 30 min and analyzed by immunoblotting using anti hnRNPA1 and Β-actin antibodies.
Discussion
The classical and extensively studied role of GzmA and GzmB is the induction of apoptosis in mammalian cells after their cytosolic delivery by the pore-forming protein PFN1. Recently, the cytotoxic spectrum of the Gzms was broadened significantly from mammalian cells to bacteria9,10, as well as to certain parasites11. Furthermore, the non-classical, extracellular functions of GzmA and GzmB as well as the biological significance of the various orphan Gzms are still obscure. Therefore, a robust time- and cost-efficient expression and purification system of the Gzms, as provided by this protocol, will be of great help for these future studies.
The strength of this method is particularly based on the expression vector, pHLsec23, that is used in this system. This plasmid has a high copy number in E. coli, thus enabling efficient DNA production. Its enhancer/promoter elements provide the strongest activity in various cell lines24. It contains an intron within the transcription unit. Introns have been found to enhance gene expression in mammalian cells up to 100-fold37. In addition, the plasmid provides a Kozak consensus and an optimized secretion signal, a Lys-6xHis-tag and a poly-A signal (Figure 1). Inserts can be cloned by using the AgeI and KpnI sites between the secretion signal and the Lys-6xHis tag. Using AgeI and XhoI will avoid the polyHis tag. For the Gzms, it was necessary to insert an enterokinase site at the N-terminus of the proteases enabling activation of the proteases after expression and IMAC purification. For the expression of other enzymes this might not be necessary. The expression plasmid was successfully tested for multiple constructs of various lengths, glycosylation states, and numbers of disulfide bonds, as well as for proteins that contained multiple domains with different folds23.
HEK293T cells grow rapidly in low cost medium and are very well suited for cost-efficient calcium-phosphate transfection. For this protocol, we recommend not to over-passage the HEK293T cells. Frozen stocks of low-passage-cells should be prepared. Expansion takeoff the cells usually took about 8 to 9 days (3 splits) to obtain the desired amount of cells. The cell density at the day of transfection (60 to 70% confluency) was critical for efficient transfection and consequently expression. Also, avoiding antibiotics but adding chloroquine (that inhibits lysosomal DNA degradation) to the medium significantly enhanced efficiency38. We found that transfection efficiency was linearly dependent of the DNA amount saturating only at about 80 μg for a 15-cm dish (unpublished observation). Therefore, we recommend using up to 80 μ g of plasmid DNA/dish in this protocol for maximal efficiency. This will amount to about 2 mg of DNA per preparation (corresponding to 2 MaxiPrep columns). The transfection efficiency was most easily monitored by running a sample of culture supernatant (about 20 μ l) on SDS-PAGE after the 72 hr incubation period. A protein band of the secreted protease should be visible by Coomassie staining. If the Gzm band was weak and the cells did not detach and appeared viable, the cultures were incubated for another 24 hr before IMAC purification.
Another most critical step was the enterokinase (EK) treatment that activates the enzyme. EK activity is calcium dependent and is inhibited by high concentrations of NaCl39or imidazole (unpublished observation). Therefore, it was crucial to dialyze the eluate from the IMAC against a sufficient volume (20 ml eluate per 2 L of dialysis buffer) of EK buffer containing enough calcium and not more than 50 mM of NaCl at neutral pH. EK treatment is recommended for 16 hr at RT and should be monitored by SDS-PAGE that revealed a mobility shift of the treated protease if the cleavage was complete. Fresh EK was added and the incubation in fresh EK Dialysis buffer was continued if the shift was incomplete. Only if the cleavage was complete, dialysis against S buffer A was started and purification continued. The catalytic light chain of EK has a pI value of 5.2, allowing the complete separation of EK from the Gzms in the final cation exchange chromatography.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by grants from the Novartis Foundation for Medical-Biological Research and from the Research Pool of the University of Fribourg (to MW). We thank Li Zhao, Zhan Xu, and Solange Kharoubi Hess for technical support, as well as Radu Aricescu and Yvonne Jones (Oxford University, UK) for providing the pHLsec plasmid, and Thomas Schürpf (Harvard Medical School) for helpful discussions.
Materials
| TRIzol Reagent | Invitrogen | 15596-026 | Total RNA isolation kit |
| SuperScript II Reverse Transcriptase | Invitrogen | 18064-014 | |
| Phusion High-Fidelity DNA Polymerase | NEB | M0530L | |
| EndoFree Plasmid Maxi Kit | QIAGEN | 12362 | |
| EX-CELL 293 Serum-Free Medium for HEK 293 Cells | Sigma | 14571C | |
| DMEM, high glucose, GlutaMAX Supplement, pyruvate | Gibco | 31966-021 | |
| SnakeSkin Dialysis Tubing, 10K MWCO | Thermo Scientific | 68100 | |
| Enterokinase from bovine intestine | Sigma | E4906 | recombinant, ≥20 units/mg protein |
| HisTrap Excel 5ml-column | GE Healthcare | 17-3712-06 | Nickel IMAC |
| HiTrap SP HP 5 ml-column | GE Healthcare | 17-1152-01 | S column |
| N-α-Cbz-L-lysine thiobenzylester (BLT) | Sigma | C3647 | GzmA substrate |
| Boc-Ala-Ala-Asp-S-Bzl (AAD) | MP Biomedicals | 2193608 _10mg | GzmB substrate |
| Suc-Ala-Ala-Pro-Leu-p-nitroanilide (AAPL) | Bachem | GzmM substrate | |
| 5,5′-dithio-bis(2-nitrobenzoic acid) (DTNB) | Sigma | D8130 | Ellman`s reagent |
References
- Chowdhury, D., Lieberman, J. Death by a thousand cuts: granzyme pathways of programmed cell death. Annu Rev Immunol. 26, 389-420 (2008).
- Thiery, J., Lieberman, J. Perforin: a key pore-forming protein for immune control of viruses and cancer. Subcell Biochem. 80, 197-220 (2014).
- Krensky, A. M., Clayberger, C. Biology and clinical relevance of granulysin. Tissue Antigens. 73, 193-198 (2009).
- Bovenschen, N., Kummer, J. A. Orphan granzymes find a home. Immunol Rev. 235, 117-127 (2010).
- Lieberman, J. Granzyme A activates another way to die. Immunol Rev. 235, 93-104 (2010).
- Ewen, C. L., Kane, K. P., Bleackley, R. C. A quarter century of granzymes. Cell death and differentiation. 19, 28-35 (2012).
- Hiebert, P. R., Granville, D. J. Granzyme B in injury, inflammation, and repair. Trends Mol Med. 18, 732-741 (2012).
- Afonina, I. S., Cullen, S. P., Martin, S. J. Cytotoxic and non-cytotoxic roles of the CTL/NK protease granzyme. B. Immunol Rev. 235, 105-116 (2010).
- Walch, M., et al. Cytotoxic cells kill intracellular bacteria through granulysin-mediated delivery of granzymes. Cell. 157, 1309-1323 (2014).
- Lee, W. Y., et al. Invariant natural killer T cells act as an extravascular cytotoxic barrier for joint-invading Lyme Borrelia. Proc Natl Acad Sci U S A. , (2014).
- Kapelski, S., de Almeida, M., Fischer, R., Barth, S., Fendel, R. Antimalarial activity of granzyme B and its targeted delivery by a granzyme B-scFv fusion protein. Antimicrob Agents Chemother. , (2014).
- Thiery, J., Walch, M., Jensen, D. K., Martinvalet, D., Lieberman, J. Isolation of cytotoxic T cell and NK granules and purification of their effector proteins. Curr Protoc Cell Biol. Chapter 3 (Unit3 37), (2010).
- Shi, L., Yang, X., Froelich, C. J., Greenberg, A. H. Purification and use of granzyme B. Methods Enzymol. 322, 125-143 (2000).
- Masson, D., Tschopp, J. A family of serine esterases in lytic granules of cytolytic T lymphocytes. Cell. 49, 679-685 (1987).
- Lorentsen, R. H., Fynbo, C. H., Thogersen, H. C., Etzerodt, M., Holtet, T. L. Expression, refolding, and purification of recombinant human granzyme B. Protein Expr Purif. 39, 18-26 (2005).
- Sun, J., et al. Expression and purification of recombinant human granzyme B from Pichia pastoris. Biochem Biophys Res Commun. 261, 251-255 (1999).
- Xia, Z., et al. Expression and purification of enzymatically active recombinant granzyme B in a baculovirus system. Biochem Biophys Res Commun. 243, 384-389 (1998).
- Stahnke, B., et al. Granzyme B-H22(scFv), a human immunotoxin targeting CD64 in acute myeloid leukemia of monocytic subtypes. Mol Cancer Ther. 7, 2924-2932 (2008).
- Gehrmann, M., et al. A novel expression and purification system for the production of enzymatic and biologically active human granzyme. B. J Immunol Methods. 371, 8-17 (2011).
- Metkar, S. S., et al. Cytotoxic cell granule-mediated apoptosis: perforin delivers granzyme B-serglycin complexes into target cells without plasma membrane pore formation. Immunity. 16, 417-428 (2002).
- Motyka, B., et al. Mannose 6-phosphate/insulin-like growth factor II receptor is a death receptor for granzyme B during cytotoxic T cell-induced apoptosis. Cell. 103, 491-500 (2000).
- Pinkoski, M. J., et al. Entry and trafficking of granzyme B in target cells during granzyme B-perforin-mediated apoptosis. Blood. 92, 1044-1054 (1998).
- Aricescu, A. R., Lu, W., Jones, E. Y. A time- and cost-efficient system for high-level protein production in mammalian cells. Acta Crystallogr D Biol Crystallogr. 62, 1243-1250 (2006).
- Fukuchi, K., et al. Activity assays of nine heterogeneous promoters in neural and other cultured cells. In Vitro Cell Dev Biol Anim. 30 A, 300-305 (1994).
- Tran, T. V., Ellis, K. A., Kam, C. M., Hudig, D., Powers, J. C. Dipeptidyl peptidase I: importance of progranzyme activation sequences, other dipeptide sequences, and the N-terminal amino group of synthetic substrates for enzyme activity. Arch Biochem Biophys. 403, 160-170 (2002).
- Somanchi, S. S., Senyukov, V. V., Denman, C. J., Lee, D. A. Expansion, purification, and functional assessment of human peripheral blood NK cells. Journal of visualized experiments : JoVE. , (2011).
- Barry, M., et al. Granzyme B short-circuits the need for caspase 8 activity during granule-mediated cytotoxic T-lymphocyte killing by directly cleaving Bid. Mol Cell Biol. 20, 3781-3794 (2000).
- Darmon, A. J., Nicholson, D. W., Bleackley, R. C. Activation of the apoptotic protease CPP32 by cytotoxic T-cell-derived granzyme B. . Nature. 377, 446-448 (1995).
- Rajani, D. K., Walch, M., Martinvalet, D., Thomas, M. P., Lieberman, J. Alterations in RNA processing during immune-mediated programmed cell death. Proc Natl Acad Sci U S A. 109, 8688-8693 (2012).
- Domselaar, R., et al. Noncytotoxic inhibition of cytomegalovirus replication through NK cell protease granzyme M-mediated cleavage of viral phosphoprotein 71. J Immunol. 185, 7605-7613 (2010).
- Thiery, J., et al. Perforin activates clathrin- and dynamin-dependent endocytosis, which is required for plasma membrane repair and delivery of granzyme B for granzyme-mediated apoptosis. Blood. 115, 1582-1593 (2010).
- Thiery, J., et al. Perforin pores in the endosomal membrane trigger the release of endocytosed granzyme B into the cytosol of target cells. Nat Immunol. 12, 770-777 (2011).
- Thomas, M. P., et al. Leukocyte protease binding to nucleic acids promotes nuclear localization and cleavage of nucleic acid binding proteins. J Immunol. 192, 5390-5397 (2014).
- Jacquemin, G., et al. Granzyme B-induced mitochondrial ROS are required for apoptosis. Cell death and differentiation. , (2014).
- Kummer, J. A., Kamp, A. M., Citarella, F., Horrevoets, A. J., Hack, C. E. Expression of human recombinant granzyme A zymogen and its activation by the cysteine proteinase cathepsin C. The Journal of biological chemistry. 271, 9281-9286 (1996).
- Hagn, M., Sutton, V. R., Trapani, J. A. A colorimetric assay that specifically measures Granzyme B proteolytic activity: hydrolysis of Boc-Ala-Ala-Asp-S-Bzl. Journal of visualized experiments : JoVE. , e52419 (2014).
- Huang, M. T., Gorman, C. M. Intervening sequences increase efficiency of RNA 3′ processing and accumulation of cytoplasmic RNA. Nucleic Acids Res. 18, 937-947 (1990).
- Luthman, H., Magnusson, G. High efficiency polyoma DNA transfection of chloroquine treated cells. Nucleic Acids Res. 11, 1295-1308 (1983).
- Magee, A. I., Grant, D. A., Hermon-Taylor, J., Offord, R. E. Specific one-stage method for assay of enterokinase activity by release of radiolabelled activation peptides from alpha-N-[3H]acetyl-trypsinogen and the effect of calcium ions on the enzyme activity. Biochem J. 197, 239-244 (1981).

