Quantifying Social Motivation in Mice Using Operant Conditioning
Summary
This article describes a new protocol for the quantitative measurement of social motivation in mice using operant conditioning for a social reward. The protocol is useful for the measurement of social motivation in mouse models of autism and other disorders of social behavior.
Abstract
In this protocol, social motivation is measured in mice through a pair of operant conditioning paradigms. To conduct the experiments, two-chambered shuttle boxes were equipped with two operant levers (left and right) and a food receptacle in one chamber, which was then divided from the second chamber by an automated guillotine door covered by a wire grid. Different stimulus mice, rotated across testing days, served as a social stimulus behind the wire grid, and were only visible following the opening of the guillotine door. Test mice were trained to lever press in order to open the door and gain access to the stimulus partner for 15 sec. The number of lever presses required to obtain the social reward progressively increased on a fixed schedule of 3. Testing sessions ended after test mice stopped lever pressing for 5 consecutive minutes. The last reinforced ratio or breakpoint can be used as a quantitative measure of social motivation. For the second paradigm, test mice were trained to discriminate between left and right lever presses in order to obtain either a food reward or the social reward. Mice were rewarded for every 3 presses of each respective lever. The number of food and social rewards can be compared as a measurement of the value placed upon each reward. The ratio of each reward type can also be compared between mouse strains and the change in this ratio can be monitored within testing sessions to measure satiation with a given reward type. Both of these operant conditioning paradigms are highly useful for the quantification of social motivation in mouse models of autism and other disorders of social behavior.
Introduction
In recent years, the linkage of genetic and environmental factors to disorders of social behavior including autism spectrum disorder, has led to the development of mouse models designed to explore the role of these factors in social behavior. This has created a demand for new techniques for the characterization of social behavior in mice including a quantitative measure of social motivation1.
There are several established paradigms that can be used to demonstrate social motivation in mice, however; they all have severe limitations on their ability to quantify the strength of the motivated behavior. For example, some of the earliest behavioral assessments included simple observations of social interactions between two rodents that have never met. Several behaviors including approaching, following, sniffing, and grooming are often measured, and may provide an index of social motivation. These studies were initially conducted in rats2-5, and then later adapted for use in mice6,7. Habituation/dishabituation social recognition paradigms in which the test animal is exposed repeatedly to the same stimulus animal (habituation) and then exposed to a novel animal (dishabituation) also provide a measurement of social motivation8. As the labels suggest, typical animals will display a habituated behavioral response to the repeated presence of the same stimulus animals but will demonstrate dishabituation, or a return to baseline, when exposed to the novel animal. The habituation response likely demonstrates a decline in the motivation of the test mouse to interact with the stimulus mouse. Social choice paradigms allowing a test mouse to choose between an empty chamber and a stimulus mouse restricted to a different chamber have also been used to assess social motivation. A trial typically begins by placing the test mouse in a neutral, center chamber. Some versions of this task involve tethering the stimulus mouse to one chamber9. A modification of this task employed the use of a wire enclosure over the stimulus mice avoiding tethering of stimulus animals10. More recently, a social conditioned place preference task was developed in which mice were conditioned to associate visually distinct chambers with either the presence or absence of a social partner, and then allowed to explore the empty chambers following conditioning11. More time spent in the chamber where social pairings previously occurred is considered to be an index of social motivation.
All of the above paradigms provide valuable data related to the social phenotyping of mice. However, while one can observe whether a mouse demonstrates a “desire” to seek social interaction in the above paradigms, the strength of this “desire”, i.e., social motivation, is difficult to measure. The strength of motivated behavior has traditionally been measured in rodents by creating a scenario in which they have to work in order to gain access to the source of their motivation. This usually involves training rodents in an operant paradigm involving food as the motivating factor. However, this method has been employed previously to investigate social motivation in rats, but either involving access to a sexual partner for male rats or, for females, access to their own pups (reviewed in Matthews et al., 2005) 12,13.
Prior to the development of the social motivation paradigms described below, there was only one other study that investigated social motivation outside of sexual motivation using an operant paradigm with mice12. In this series of experiments, outbred Swiss Webster mice were trained to press a lever in order to gain access to a same-sex conspecific. In one experiment, a significantly reduced latency to begin responding for access to the target mouse was demonstrated in individually-housed mice compared to group-housed mice suggesting increased motivation to seek social access in these socially isolated mice. In another experiment of that same study, a comparison was made between saline and oxytocin antagonist (atosiban) infused mice. Interestingly, the drug-infused mice demonstrated a significantly increased latency to begin responding for access to the target mouse suggesting reduced motivation for a social reward in these mice.
While these experiments represent a noble attempt to quantify social motivation in mice using operant conditioning, they were also highly limited and are greatly improved upon using the methods described below. For example, an apparent fundamental flaw in the first experiment described above (experiment 2 of the actual study) was the researchers’ use of only 2 target mice alternated across testing days. Previous research has shown that social recognition in group-housed mice can last for at least 7 days leading to reduced social investigation with repeated exposure to the same target14. However, social recognition in individually-housed rats and mice dissipates within 1 – 2 hr so that social investigation durations remain at baseline levels if a repeated exposure to the same target is at least 2 hr after the initial exposure15-17. The reduced latency of individually-housed mice to begin responding for social access reported in the Matthews et al. experiment may have been a consequence of the lack of social recognition of the target mice across testing days12. This potential confound is controlled for in the below methods by using a novel target mouse on each test day. Also, in both experiments from the Matthews et al. study described above, there were no reported differences in the total number of lever presses made between groups. It is possible that a difference in total lever presses could have been detected if the researchers utilized a progressively increasing ratio of responses to reward as used in our research design outlined below. In addition, access to the target mouse was controlled manually by the experimenter, by lifting up a box that covered the target. The use of an automatically-controlled guillotine door for access to the target mouse eliminates the possibility of artifact due to the presence of the experimenter. In fact, in order to fully automate the social motivation operant paradigms, the research equipment was customized as detailed below.
Protocol
This research was approved by the Azusa Pacific University Institutional Animal Care and Use Committee. All procedures involving animals were carried out following the recommendations of the Guide for the Care and Use of Laboratory Animals from the National Institutes of Health.
1. Modifying Apparatuses for Social Motivation and Valence Comparison Testing Paradigms
- In order to test mice in operant tasks of social behavior, modify center channel modular shuttle boxes. These shuttle boxes measure 44 cm x 17.3 cm and are made of Plexiglas and stainless steel.
- Modify a programmable guillotine door by mounting a wire grid to the face of the doorframe. The wire grid should allow limited social contact but not allow the mice to cross from one chamber to the other.
- Using the programmable guillotine door, divide each box into two chambers: each measuring approximately 22 cm x 17.3 cm. Designate one chamber as the test chamber and the other as the target chamber.
- Place mouse levers in the test chamber on the wall opposite of the guillotine door. Place a liquid dipper food receptacle between the two mouse levers.
- Program the mouse levers to either open the guillotine door or present a liquid food reward, depending upon the testing paradigm.
- Connect a handheld button to the apparatus and program it to open the guillotine door (for manual shaping of test mice).
- Fit each shuttle box with an eight-channel I/R controller to monitor activity across the metal grid floor of each chamber. Mount a USB digital video camera above the test chamber and connect it to a computer. Enclose the entire apparatus within a melamine sound-attenuating cubicle.
- Run the operant programs using customized programs written in the laboratory.
2. Shaping the Mice for the Social Motivation Paradigm
- Place a test mouse in the test chamber and an age and sex-matched stimulus mouse in the target chamber.
- Train the test mouse to press the levers to open the guillotine door using the method of shaping or reinforcement of successive approximations to the desired operant response. During this shaping procedure, deliver reinforcement manually by pressing the handheld button programmed to control the guillotine door.
Note: Reinforcement consists of opening the guillotine door for 15 sec, thereby allowing access to the stimulus mouse through the wire grid. - Observe the mouse in the test chamber using the camera mounted above the test chamber.
- Train each mouse in a series of 30 min daily sessions until they demonstrate at least 10 operant responses (lever presses) in 3 out of 5 consecutive testing sessions. Rotate mice through shuttle boxes using random assignment.
- If mice do not reach this criterion after 30 daily training sessions, remove the mice from the experiment and designate as non-learners.
Note: The number of daily training sessions may be extended beyond 30 depending on researcher preferences. - Alternate the stimulus mice used during shaping so that a different mouse is used every other day.
- Clean shuttle boxes between each training session using 70% ethyl alcohol. Additionally, following the completion of testing each day, clean all equipment using a disinfecting detergent.
3. Testing the Mice in the Social Motivation Paradigm
- Place a trained test mouse in the test chamber and an age and sex-matched stimulus mouse in the target chamber. During testing, use a pool of at least 10 stimulus mice to assign to each test mouse so that stimulus mice are only repeated after 10 daily test sessions. Do not use stimulus mice used during shaping for the testing.
- Program the guillotine door to open on a progressive ratio schedule of reinforcement so that the number of operant responses (lever presses) necessary to obtain the social reward of 15 sec access to the target mouse arithmetically increases by a fixed rate of 3 each trial.
- Program the test sessions to end when the test mouse stops lever pressing for five consecutive minutes.
- Record the last completed (reinforced) ratio as the breakpoint. Use this dependent measure as an index of social motivation. Test each mouse for 20 consecutive daily sessions or for at least 10 test sessions after asymptotic performance levels are observed.
- Wipe down shuttle boxes between each testing session using 70% ethyl alcohol and following the completion of testing each day using a disinfecting detergent.
4. Valence Comparison of Social Versus Food Reward
- After all mice complete the social motivation paradigm, test these same mice in a task designed to compare the valence of the social reward with that of a food reward.
- Program one lever to open the guillotine door and present the social reward for 15 sec and program the opposite lever to present a liquid food reward for 15 sec.
Note: The liquid food reward used in this protocol consisted of evaporated milk sweetened with 0.2% sucrose. Prepare sufficient amounts (typically 500 ml) for repeatedly presenting rewards of 0.02 ml to each mouse. - Assign test mice alternating lever/reward contingencies to control for potential lever biases among mice. Thus, assign half of the mice in each testing group to a left lever/food reward and right lever/social reward contingency and the other half to a left lever/social reward and right lever/food reward contingency.
- Train the mice for valence comparison of social versus food reward.
- Place a test mouse in the test chamber and place an age and sex-matched stimulus mouse in the target chamber.
- Make only one lever/reward contingency active during each training session. Training consists of six 1 hr sessions that alternate between contingencies each day. Alternate stimulus mice assigned to each test mouse every other social reward session.
- Maintain the mice on their ad libitum chow diet during training and testing with the food reward. Food deprivation is unnecessary for operant conditioning in mice with the use of the evaporated milk and sucrose solution18.
- Testing the mice for the valence comparison of social versus food reward.
- Program the schedule of reinforcement at a fixed ratio of 3:1, so that every third lever press is reinforced, but only by its respective associated reward.
- Program test sessions to run for 60 min.
- Place a test mouse in the test chamber and place an age and sex-matched stimulus mouse in the target chamber. Note: The same pool of 10 stimulus mice used in the social motivation task can be used again but assigned to the test mice using a 10-day rotation.
- Test the mice in 20 daily sessions following the 6 days of training or for at least 10 daily sessions following the observation of asymptotic performance levels. The primary dependent measures for this paradigm is the total number of lever presses for each reward type.
- Once again, wipe down shuttle boxes between testing sessions using 70% ethyl alcohol and following each test day using a disinfecting detergent.
Representative Results
This protocol has been used to compare the prosocial C57BL/6J inbred mouse strain with the BTBR inbred mouse strain which has been previously reported to show decreases in social behavior10,18. It has also been used to characterize social motivation in different knockout mouse strains hypothesized to have either increased or decreased social motivation. Previously published results comparing the C57BL/6J and BTBR inbred mouse strains are shown in Figure 1. These results demonstrated decreased social motivation for the BTBR mice compared to the C57BL/6J mice. The Valence Comparison paradigm also demonstrated decreased food motivation for the BTBR mice suggesting an overall decrease in motivated behavior for this strain (Figure 1B).
Figure 2A presents some unpublished preliminary data comparing wild-type siblings on the C57BL/6 background strain with heterozygous and homozygous knockout siblings with reduced or absent protein product from a gene (gene X) hypothesized to promote social motivation. Figure 2B presents some unpublished preliminary data comparing wild-type siblings on the C57BL/6 background strain to heterozygous knockout siblings with reduced protein product from a gene (gene Y) hypothesized to reduce social motivation. The preliminary data from each of these studies support each respective hypothesis and further demonstrate the value of the described operant social motivation paradigms.
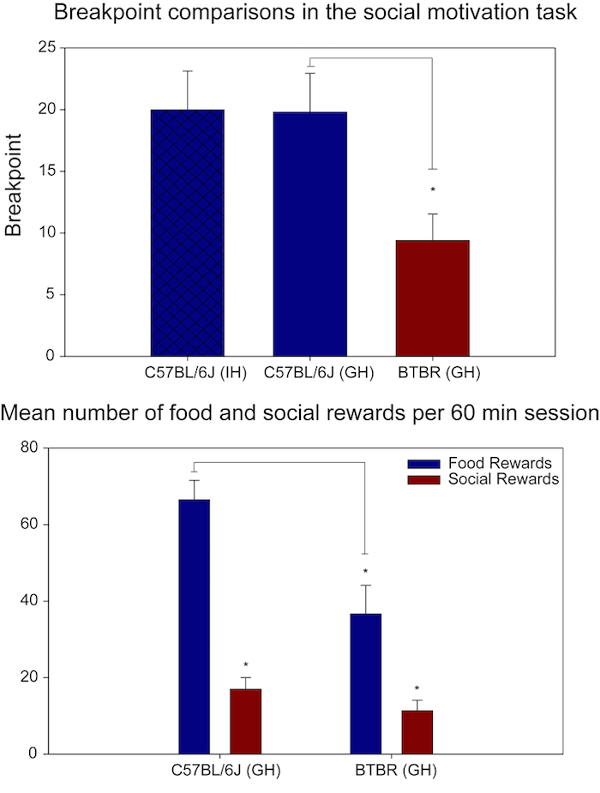
Figure 1. Previously published results from the social motivation and valence comparison paradigms. (A) The mean breakpoint of individually-housed (IH) B6 mice, group-housed (GH) B6 mice, and group-housed BTBR mice across the last 10 days of testing in the social motivation task. There were a total of 9 mice in each group. Individually and group-housed B6 mice performed similarly but there was a significant difference between group-housed B6 and BTBR mice (t = 2.741, df = 16, p = 0.015). (B) The mean number of food rewards and social rewards for the group-housed B6 and BTBR mice across the last 10 sessions of the valence comparison paradigm. Both mouse groups obtained significantly more food rewards than social rewards (BTBR: t = 3.551, df = 8, p = 0.007; B6: t = 9.478, df = 8, p < 0.001). However, the BTBR mice acquired significantly fewer food rewards than the B6 mice and also the fewest number of social rewards (t = 3.321, df = 16, p = 0.004). Figure modified from Reference 18.
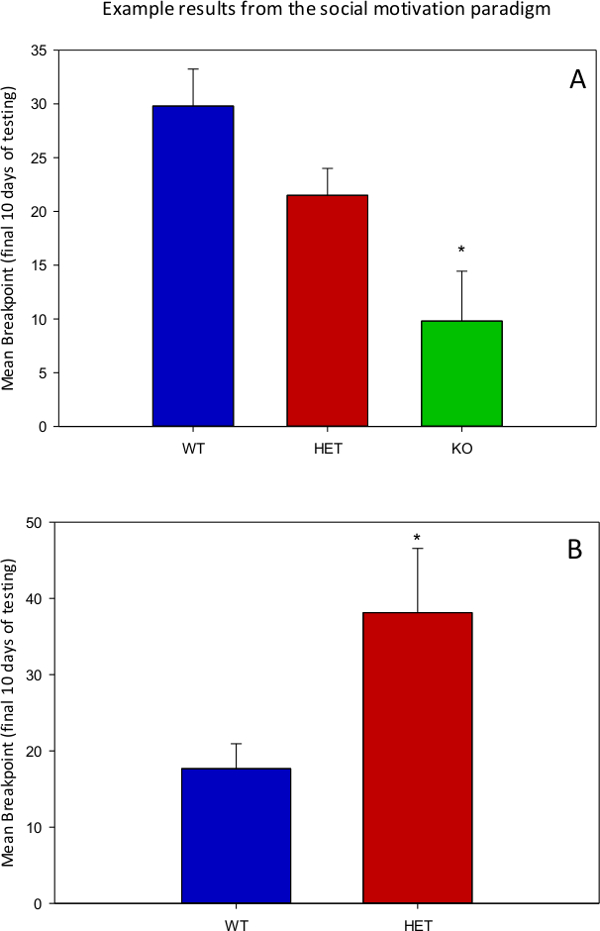
Figure 2. Representative results from unpublished research utilizing the social motivation paradigm. (A) Mean breakpoint (last reinforced ratio) achieved before the mice stopped lever pressing for 5 min. For gene X, a total of 12 WT, 7 HET, and 6 KO mice were tested in the social motivation paradigm. ANOVA revealed a significant difference between genotypes (F = 7.18, df = 2, 22, p = 0.004). Post hoc Tukey HSD tests showed that KO mice performed significantly below WT controls. (B) For gene Y, a total of 8 HET and 6 WT mice were tested. An independent samples t-test revealed a significant difference between genotypes (t = 2.26, df = 8.97, p = 0.05). * indicates significant difference from WT mice at the .05 alpha level.
Discussion
The operant social motivation paradigms described in this article are powerful tools for the characterization of social behavior in mouse models of human disease. Rather than allow a test mouse to passively choose a social encounter, these paradigms require effort from the test mouse in order to obtain a social reward. The amount of effort that a test mouse is willing to produce to obtain the social reward provides a quantitative measure of social motivation. Furthermore, through the valence comparison paradigm, motivated behavior can be compared across different reward types. Both of these operant paradigms can be used to assess sexual and parental motivations as well by presenting the appropriate stimulus on the opposite side of the guillotine door.
The development of these social motivation paradigms involved the troubleshooting of several key variables. For example, through pilot studies the amount of time that the social reward is presented was lowered from 30 sec to 15 sec. While this time is sufficient for social interactions between test and stimulus mice, future research may optimize the amount of time for the social reward. Similarly, the progressively increasing ratio of lever presses to social rewards was lowered from a fixed rate of 5 to 3 based upon pilot research.
The number of days necessary to reach asymptotic lever pressing behavior was initially assessed through 30 days of daily test sessions. It was determined that most mice reach asymptote in 7 days so the number of test sessions was lowered to 20. Daily performance does fluctuate however, so averaging the data over the last 10 days ensures asymptotic performance while also providing a better index of true mean performance than fewer test days. Researchers should monitor their data to ensure that asymptotic performance is reached within 10 test days or modify the protocol as necessary.
Training sessions were also lowered from 1 hr to 30 min. The number of training sessions required does depend on the strain of mouse and some mice may be arbitrarily designated as nonlearners after a fixed number of training sessions18. The number of training days necessary to reach the criterion for advancement to the testing paradigm provides another index of social motivation and is thus worthy of tracking. Researchers may also want to experiment with different food reward options for the valence comparison paradigm. However, it is important that the test mice are initially trained using a purely social reward and then tested in the social motivation paradigm before beginning the valence comparison paradigm.
These operant social motivation paradigms can be modified and improved upon by measuring additional variables than those indicated in the above protocol. For example, the location of each mouse in his respective chamber can be tracked using currently available commercial software. In addition, specific behaviors including nose-to-nose contact or ultrasonic vocalizations can be scored during social interactions. Researchers may further experiment with adjustments to the ratios used in the social motivation paradigm or with trials involving the absence of a social partner or even comparisons involving other stimuli behind the guillotine door such as a video of a conspecific or even a puppet. For the valence comparison paradigm, analyses of choice dynamics across trials can provide further information regarding shifts in motivation due to satiation for a particular reward type18.
While these operant social motivation paradigms provide a much-needed tool for quantifying social motivation in mice, they do have some limitations. First, these paradigms require mice to have the cognitive ability to make learned associations between lever pressing and the social reward. Certain mouse models may have deficits in both cognitive and social abilities making it difficult to assess social motivation using these paradigms. Second, the nature of the social encounter is limited as the mice are only allowed to interact across a wire grid. However, mice are still able to successfully engage in many typical social behaviors, most notably, nose-to-nose investigations. One final limitation of these methods is that they are labor-intensive and require much time to be completed. The training and testing through both paradigms requires a minimum of two months of daily sessions. Some mice may take much longer to learn the lever-pressing association stretching the required number of sessions. While the paradigms certainly do not qualify as high-throughput techniques, the data that they generate is highly valuable and worth the required labor investment. However, to provide a fuller characterization of the social behavior of a particular mouse model, it is recommended that these paradigms be used in conjunction with other established methods of measuring social behavior in mice.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank the Faculty Research Council at Azusa Pacific University for grant support. We also thank Ashley Schneider, Michael Gregg, and Hannah Sample for technical support.
Materials
| Modular Shuttle Box | Med Associates Inc. | ENV-010MC | |
| Auto-Guillotine Door | Med Associates Inc. | ENV-010B | |
| Mouse Levers | Med Associates Inc. | ENV-3010M | |
| Liquid Dipper with Food Receptacle | Med Associates Inc. | ENV-202M-S | |
| Eight-Channel I/R Controller | Med Associates Inc. | ENV-253C | |
| Melamine Sound-Attenuating Cubicle | Med Associates Inc. | ENV-016MD | |
| MED-PC IV Software Program | Med Associates Inc. | MED-PC IV | |
| Microsoft Lifecam Camera | Microsoft | ||
| Wire Grid | Wire grids can be purchases from a hardward store. We cut wire grids from stainless steel mouse cage tops. | ||
| 70% Ethyl Alcohol | Used to clean shuttle boxes between mice. Specific brand is unnecessary. | ||
| Evaportated Milk | Carnation | 0.02 mL combined with 0.2% sucrose. We always purchase the same brand for better consistency. | |
| Sucrose | Any brand of pure granular sugar will due as it is dissolved in the evaporated milk. |
References
- Silverman, J. L., Yang, M., Lord, C., Crawley, J. N. Behavioural phenotyping assays for mouse models of autism. Nat Rev Neurosci. 11, 490-502 (2010).
- File, S. E. The use of social interaction as a method for detecting anxiolytic activity of chlordiazepoxide-like drugs. J Neurosci Methods. 2, 219-238 (1980).
- File, S. E. Animal models for predicting clinical efficacy of anxiolytic drugs: social behaviour. Neuropsychobiology. 13, 55-62 (1985).
- File, S. E. Anxiolytic action of a neurokinin1 receptor antagonist in the social interaction test. Pharmacol Biochem Behav. 58, 747-752 (1997).
- File, S. E., Hyde, J. R. Can social interaction be used to measure anxiety. Br J Pharmacol. 62, 19-24 (1978).
- Winslow, J. T. Mouse social recognition and preference. Curr Protoc Neurosci. 8, Unit 8 16 (2003).
- Angelis, L., File, S. E. Acute and chronic effects of three benzodiazepines in the social interaction anxiety test in mice. Psychopharmacology (Berl). 64, 127-129 (1979).
- Gheusi, G., Bluthe, R. M., Goodall, G., Dantzer, R. Ethological study of the effects of tetrahydroaminoacridine (THA) on social recognition in rats. Psychopharmacology (Berl). 114, 644-650 (1994).
- Carter, C. S., DeVries, A. C., Getz, L. L. Physiological substrates of mammalian monogamy: the prairie vole model). Neurosci Biobehav Rev. 19, 303-314 (1995).
- Moy, S. S., et al. Sociability and preference for social novelty in five inbred strains: an approach to assess autistic-like behavior in mice. Genes Brain Behav. 3, 287-302 (2004).
- Pearson, B. L., et al. Absence of social conditioned place preference in BTBR T+tf/J mice: relevance for social motivation testing in rodent models of autism. Behav Brain Res. 233, 99-104 (2012).
- Matthews, T. J., Abdelbaky, P., Pfaff, D. W. Social and sexual motivation in the mouse. Behav Neurosci. 119, 1628-1639 (2005).
- Lee, A., Clancy, S., Fleming, A. S. Mother rats bar-press for pups: effects of lesions of the mpoa and limbic sites on maternal behavior and operant responding for pup-reinforcement. Behav Brain Res. 108, 215-231 (2000).
- Kogan, J. H., Frankland, P. W., Silva, A. J. Long-term memory underlying hippocampus-dependent social recognition in mice. Hippocampus. 10, 47-56 (2000).
- Sekiguchi, R., Wolterink, G., van Ree, J. M. Short duration of retroactive facilitation of social recognition in rats. Physiol Behav. 50, 1253-1256 (1991).
- Bluthe, R. M., Gheusi, G., Dantzer, R. Gonadal steroids influence the involvement of arginine vasopressin in social recognition in mice. Psychoneuroendocrinology. 18, 323-335 (1993).
- Thor, D. H., Holloway, W. R. Anosmia and play fighting behavior in prepubescent male and female rats. Physiol Behav. 29, 281-285 (1982).
- Martin, L., Sample, H., Gregg, M., Wood, C. Validation of operant social motivation paradigms using BTBR T+tf/J and C57BL/6J inbred mouse strains. Brain Behav. 4, 754-764 (2014).

