Imaging Spatial Reorganization of a MAPK Signaling Pathway Using the Tobacco Transient Expression System
Summary
At the subcellular level, signaling events are dynamically modulated by developmental and environmental cues. Here we describe a protocol that employs the tobacco transient expression system to monitor dynamic protein-protein interaction and to disclose spatial organization of signal transduction in plant cells.
Abstract
Visualization of dynamic signaling events in live cells has been a challenge. We expanded an established transient expression system, the biomolecular fluorescent complementation (BiFC) assay in tobacco epidermal cells, from testing protein-protein interaction to monitoring spatial distribution of signal transduction in plant cells. In this protocol, we used the BiFC assay to show that the interaction and the signaling between the Arabidopsis MAPKKK YODA and MAPK6 occur at the plasma membrane. When the scaffold protein BASL was co-expressed, the YODA-MAPK interaction redistributed and spatially co-polarized with CFP-BASL. This modified tobacco expression system allows for quick examination of signaling localization and dynamic changes (less than 4 days) and can accommodate multiple of fluorescent protein colors (at least three). We also presented detailed methods to quantify protein distribution (asymmetric spatial localization, or "polarization") in tobacco cells. This advanced tobacco expression system has a potential to be used widely for quick testing of dynamic signaling events in live plant cells.
Introduction
Proteins interact within an intricate hierarchical network and form complexes that play a pivotal role in nearly all biological processes occurring in an unpredictable cellular environment. However, from plant development to growth responses, there has been a lack of convenient tools that can not only quickly but also efficiently identify and monitor these dynamic signaling events at the subcellular level.
The transient protein expression system in tobacco leaf epidermis has appealing advantages for visualizing fluorescent proteins in living cells. This system provides semi-in vivo conditions that allow post-translational protein modification and quick examination of protein localization. By examining the complemented YFP signal, the bimolecular fluorescent complementation (BiFC) assay informs the possibility of protein-protein interaction in plant cells. Compared to other methods, e.g., yeast-two hybrid (Y2H) and co-immunoprecipitation (Co-IP), BiFC also provides a powerful means of visualizing compartments where protein-protein interactions may occur at the subcellular level.
Because asymmetric cell division (ACD) maintains stem cell population while generating new cell types for tissue/organ formation, it is an indispensable mechanism for promoting eukaryotic multicellularity. Arabidopsis stomatal development has been used as a model system for studying ACD in plants. The precursor cell, meristemoid mother cell, divides asymmetrically to produce two different daughter cells, a meristemoid (undergoes stem cell-like divisions before terminating into a pair of guard cells) and a stomatal lineage ground cell (SLGC) (may divide and differentiate into a pavement cell), respectively (Figure 1). In stomatal ACD, the novel protein Breaking of Asymmetry in the Stomatal Lineage (BASL) is polarized premitotically to drive division asymmetries, which include physical asymmetry and cell fate asymmetry1. A MAPK cascade composed of the MAPKKK YODA and the MAPKs, MPK3 and 6 is central for stomatal division patterning and fate adoption2,3, 4,5.
Recently, Zhang et al. linked the polarity protein BASL to the YDA-MAPK signaling pathway in Arabidopsis stomatal ACD 6. The canonical YODA-MAPK pathway, through MPK3/6, phosphorylates BASL and activates its polarization. Phosphorylated BASL functions as a scaffold and recruits YODA (YDA) and MPK3/6 to form a protein complex and concentrate the signaling at the cell cortex6. Polarization of the MAPK components and the positive feedback loop between BASL and the YDA-MAPK pathway represents a novel mechanism for protein polarization in plant cells. Locally enriched MAPK signaling is hypothesized to be closely linked to cell fate differentiation in stomatal ACD6 (Figure 1). One of the key experimental data that supported this model came from the tobacco assays that demonstrated the spatial redistribution of MAPKs induced by the expression of BASL6.
In general, it is not easy to monitor where MAPK signaling occurs because MAPK molecules were often found everywhere inside of a cell. In this study, we utilized the split-YFP system to visualize the interaction between the upstream kinase and downstream ones to suggest where the signaling relay occurs. We further extended the use of the BiFC system by co-expressing a third protein (CFP-tagged) with the split YFP pair (suggestive of protein-protein interaction) to visualize whether and how the complemented YFP could be spatially modulated by the co-expressed CFP protein. By doing so, we demonstrated that co-expression of CFP-BASL induced spatial reorganization of interactions between YDA and MPK6, from even distribution to polarized patterning at the cell cortex of the tobacco epidermal cells. This system therefore has the potential to be developed for monitoring dynamic signaling events in plant cells under conditions when cells are challenged by the internal or external stimuli (e.g., co-expression of other proteins, chemical application, pathogen attack or environmental changes, etc.).
Protocol
1. Plasmid Construction
- Generate the constructs by cloning technology as previously described1,6.
- First use a high fidelity DNA polymerase with appropriate primers to amplify the coding sequences of BASL, YDA and MPK6 and sub-clone them into entry vectors. Find the detailed protocol in 7.
- To generate the dominant negative versions of MPK6 and YDA (DNmpk68 and DNyda4, respectively), use the entry plasmids of MPK6 and YDA as template and introduce the point mutations by a site-directed mutagenesis method9.
- To build the constructs for tobacco transient assays, conduct standard LR (attL x attR recombination) reactions (detailed procedure in 7) to integrate BASL into pH35CG10, DNyda and DNmpk6 into pXNGW and pXCGW vectors11, respectively.
- Confirm the resultant plasmids by enzyme digestion and sequencing. Once successful, transform the binary constructs into the Agrobacterium tumefaciens strain GV310112 and grow them on selection plates at 28 °C for 2 – 3 days.
2. Preparation of the Agrobacterium Infiltration Solution
- Inoculate a few freshly transformed Agrobacterium colonies into 10 ml of Luria-Bertani (LB) medium with appropriate antibiotics (Gentamycin 25 μg/ml, Rifamycin 25 μg/ml, Spectinomycin 100 μg/ml for pH35CG, pXNGW and pXCGW constructs; the same concentrations of Gentamycin and Rifamycin with Kanamycin 50 μg/ml for P19 (expressing proteins against transgenic silencing)13.
- Grow Agrobacterium cultures in a 28 °C shaker at the speed of 200 rpm for about 16 hr and then measure the OD600 (estimated to reach 1.5 – 1.8) (Figure 2A-B). Spin down the cultures at 2,500 x g for 10 min. Re-suspend and wash the cell pellets with 10 mM of MgCl2 (the infiltration solution).
- Spin down the cultures again and re-suspend the pellets with appropriate amount of the infiltration solution to reach the final OD600 = 0.5 (Figure 2C-D). Before infiltrating plants, leave the cell mixtures at room temperature for 1 – 3 hr. This step helps to maintain cell homeostasis.
3. Tobacco Plant and Leaf Injection
- Use tobacco plants (Nicotiana benthamiana), grown 4 – 5 week-old in a standard plant growth chamber (23 °C with cycles of 16 hr-light/8 hr-dark)14 for leaf injection.
Note: At this stage, tobacco plants typically have 6 leaves (2 large, 2 medium, and 2 small). The two medium-sized leaves are the most optimal for leaf injection (the two large ones are too old, while the two small ones are often too young). - Before the infiltration day, water the tobacco plants to saturate the soil.
- On the infiltration day, pre-incubate the plants in the dark for 1 – 3 hr. This step allows stomata in the epidermis to open widely, which greatly facilitates the Agrobacterium when penetrating into the leaf epidermal cells.
- Using color tapes labeled with dates, flag the leaves to be infiltrated. Mark the areas to be infiltrated with a black thick sharpie (optional; the infiltrated areas are visible after being infected.).
- Take equal volumes of the Agrobacterium re-suspension and mix them in a 100 x 15 mm Petri dish by gentle swirling (Figure 2E). Note: In our case, we mixed 1 ml of p19 with 1 ml of CFP-BASL, 1 ml of DNyda-nYFP and 1 ml of DNmpk-cYFP.
- In the process of infiltration, fill a 3 ml needleless syringe with the infiltration mixture (1 – 2 ml) and push into the bottom side (abaxial) of the tobacco leaf (Figure 2F). While infiltrating, it is helpful to use one hand to push and the other hand to gently support the upper side (adaxial, against the pushing force).
Note: Once the infiltration mixture is successfully injected, the infected areas will become easily recognizable in the epidermis. However, the infiltration may fail due to harsh injection (tissue damage) or insufficient push (no liquid penetration). For each test, we suggest at least two injections in two independent leaves (from different plants). - Place the infected plants back into the growth chamber. Generally, protein expressions are detectable after 48 hr following infiltration.
4. Confocal Imaging
- At 48 hr post infiltration, use a hole puncher to excise a leaf disk from the injected area (normally 3 – 4 disks/area can be produced) (Figure 2G). Use tweezers to gently transfer the leaf disks to a slide (the abaxial side upward) and mount them with water drops. Avoid pressing too hard on the cover slip because squeezed/damaged cells will not show well under the confocal microscope (Figure 2H).
- Before confocal imaging, scan the mounted samples on an epi-fluorescent compound microscope to check the expression levels. Note: Based on our experience, the cells that show intermediate expression levels are best represented under the confocal microscope.
- Start with the 40X (NA 1.3) lens on a confocal microscope. Adjust the slide to the desired position so that the cells expressing medium levels of florescent protein remain at the center. The excitation/emission spectrums for CFP and YFP are 458 nm / 480 – 500 nm and 514 nm / 520 – 540 nm, respectively.
- Activate the "Live" button to preview the images then adjust laser intensity and smart gain for the most balanced fluorescence intensity/noise ratio. To improve imaging quality, increase scanning pixels and/or frame/line average. Finally, adjust the focusing knob to reach the median focal plane and click "Capture" to take the image.
Note: The CFP and YFP images can be captured sequentially or simultaneously by checking on or off the option of "sequential" scanning mode, respectively. - When Z-projection is desired for the in-depth view of the cells, select "xyz" imaging mode, define the depth of the scanning range (10 – 15 µm) and collect the images from the top to the bottom of the target cell. Compile the obtained images with the Leica software by right clicking on the image file to select "rename" and export images as .tiff files.
- Image 30 – 40 cells for quantification analysis.
5. Image Processing and Quantification Analysis
- Use Fiji software (http://fiji.sc/Fiji) to process images and conduct quantification.
- Plot the fluorescence intensity graphs.
- To better visualize whether the CFP/YFP expression is uniformly distributed, use Fiji to demonstrate the signal strength of CFP and YFP along the cell periphery. To achieve this, first launch Fiji then select "File" and "Open" to start the image. Click "line" on the tool menu and select "Segmented line" by right clicking. Decide the region of interest and draw a line along the cell periphery to trace the CFP or the YFP signal (Figure 3A-C).
Note: Fiji measures the absolute fluorescence intensity value along the segmented lines. - Select the drop-down menu "Analyze" and "Plot Profile" to generate a graph with position and intensity values that represent the levels of CFP/YFP along the segmented line. If desired, duplicate the intensity values (by selecting the menu "Copy") into EXCEL or other graphic software to generate comparable intensity graphs.
- To better visualize whether the CFP/YFP expression is uniformly distributed, use Fiji to demonstrate the signal strength of CFP and YFP along the cell periphery. To achieve this, first launch Fiji then select "File" and "Open" to start the image. Click "line" on the tool menu and select "Segmented line" by right clicking. Decide the region of interest and draw a line along the cell periphery to trace the CFP or the YFP signal (Figure 3A-C).
- Quantify polarity degree.
- Collect approximately 20 representative cells from 3 replicate experiments to evaluate the polarity degree of CFP and YFP in tobacco cells. Calculate the polarity degree based on the ratio of the high fluorescence intensity (defined as H) over the low fluorescence intensity (defined as L). Note: The quantification method is explained in Figure 3.
- To measure the H and L values, draw a segmented line along the interested region at the cell periphery (described above), click "Analyze" from the drop-down menu, and then select "Measure". When a "Results" window pops up, use "Mean" values to perform quantification.
- From the cells displaying relatively uniform expression of CFP and YFP, e.g., CFP-BASL (Figure 3A) or the YFP complemented from co-expression of DNyda-nYFP and DNmpk6-cYFP (Figure 3B), obtain the H and L values by measuring two randomly selected peripheral segments with equal length.
- From the cells exhibiting obvious polar accumulation of fluorescent proteins, in this case, the co-expression of CFP-BASL, DNyda-nYFP and DNmpk6-cYFP (Figure 3C), collect the H values from the peripheral regions with enriched fluorescence signals and the Ls from the regions with low/no fluorescence accumulation.
- Pool the resultant ratios of H/L from 20 cells together and test for normal distribution. Calculate the p values by the Student's t test (for normal distribution) or the Kolmogorov-Smirnov (K-S) test (for non-normal distribution).
- Collect approximately 20 representative cells from 3 replicate experiments to evaluate the polarity degree of CFP and YFP in tobacco cells. Calculate the polarity degree based on the ratio of the high fluorescence intensity (defined as H) over the low fluorescence intensity (defined as L). Note: The quantification method is explained in Figure 3.
- Plot the fluorescence intensity graphs.
Representative Results
To prevent cell death induced by the hyperactive MAPK signaling, the kinase inactive versions of YDA (DNyda) and MPK6 (DNmpk6) were used in the co-expression assay in tobacco cells. Neither the interaction between DNyda and DNmpk6 revealed by BiFC nor CFP-BASL itself generated uneven distribution pattern (Figure 3A-B). However, when CFP-BASL was introduced into the DNyda-DNmpk6 interaction pair, both CFP and YFP signals were redistributed to a highly polarized manner (Figure 3C). This redistribution suggests that BASL interacts with YDA and MPK6 to spatially re-organize the MAPK signaling pathway in plant cells. The phosphorylation status of BASL has differential impacts on the polarity degree of the MAPK signaling: a phospho-mimicking version of BASL generated very strong polarity, whilst a phospho-deficient version showed weaker activity6. These data supported our working model of a positive feedback regulation between BASL and the YDA-MAPK pathway in generating cell polarity6.
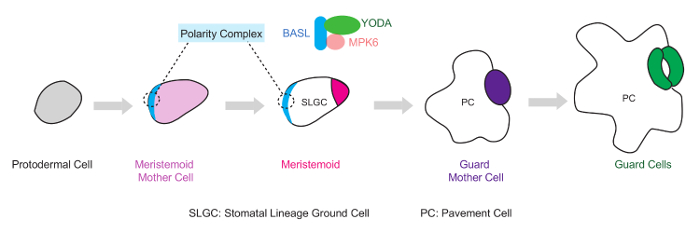
Figure 1. Diagram of the BASL-YDA-MPK6 Polarity Complex During Stomatal Asymmetric Cell Division (ACD) in Arabidopsis. In the leaf epidermis, the stomatal linage initiates from the protodermal cells, which expand to become ACD precursor cells, the Meristemoid Mother Cells (MMCs). A MMC undergoes an ACD to create one Meristemoid and one SLGC (stomatal lineage ground cell), which may divide and eventually differentiate into stomatal guard cells and pavement cells, respectively. At the cell cortex of the ACD precursor cells (MMCs), BASL recruits two components of a MAPK pathway, the MAPKKK YDA and the MAPK MPK6, to form a polarity complex and regulate stomatal ACD. Please click here to view a larger version of this figure.
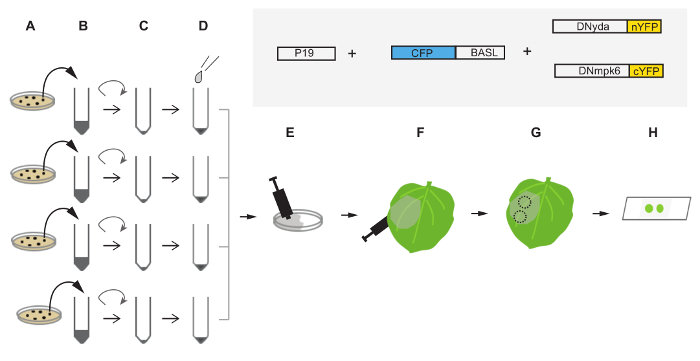
Figure 2. Diagram of the Injection Procedure in Tobacco Leaf Epidermis. (A) Transform the plasmids harboring CFP-BASL, DNyda-nYFP, DNmpk6-cYFP and P19 into Agrobacterium tumefaciens GV3101 and grow them on respective selection plates (from top to bottom in A). (B) Pick up colonies from (A) to inoculate liquid culture for overnight growth. (C) Harvest cells by spinning and decant the supernatant. After briefly pipetting and washing the pellets, spin down the cells again. (D) Re-suspend and dilute the cell pellets with the infiltration solution (see the protocol) to achieve the final OD600 = 0.5. (E) Gently mix the re-suspended cells in a Petri dish. (F) Use a needleless syringe to push the infiltration mixture into the abaxial side of the tobacco leaves. (G) About 48 hr after infiltration, the leaves are ready for confocal imaging. Excise leaf disks containing infected cells with a hole puncher around the infiltration region. Dashed circles indicate excised leave disks. (H) Use water to mount the leaf disks onto a standard slide for confocal imaging. The box diagrams in the shaded area describe the proteins injected into tobacco leaves for polarization assay. Protein sizes are not in scale. Please click here to view a larger version of this figure.
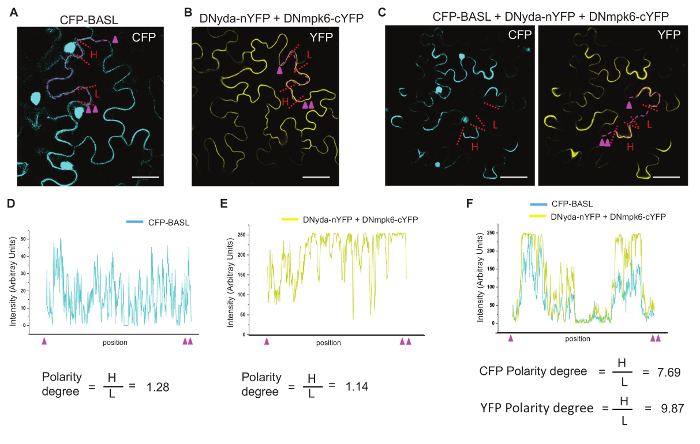
Figure 3. Display and Quantification of Polarity Formation. (A-C) Confocal images to show CFP and YFP florescence in tobacco epidermal cells. Cyan indicates the expression of CFP. Yellow indicates the expression of YFP. (A) Overexpression of CFP-BASL alone. (B) Co-expression of DNyda-nYFP and DNmpk6-cYFP. The YFP expression is complemented when two proteins interact. (C) Co-expression of CFP-BASL, DNyda-nYFP and DNmpk6-cYFP. Uneven distribution (polarity) is evident for both CFP and YFP. Scale bar = 50 µm in (A-C). (D-F) Graphically plotting the CFP/YFP fluorescence intensity along the dashed lines (Magenta) in (A-C). Magenta triangles mark the start and end point of the regions used for intensity plotting. Quantifications of the polarity degree in tobacco cells are presented underneath. The values of florescence intensity (H for high and L for low) are measured and collected by Fiji from the regions traced with red dashed lines in (A-C). For each sample, the values from 20 cells are averaged for significance test. Please click here to view a larger version of this figure.
Discussion
General Use and Possible Modifications of the Tobacco Transient Expression System for Signal Transduction: Overexpression of fluorescent protein (FP)-tagged proteins in the tobacco epidermis has been successfully used for fast examination of protein subcellular localization in plant cells. However, studying dynamic signaling distribution of protein complex in planta is a challenging topic due to complicated subcellular contexts, transient protein-protein interaction and signal transduction events. To address this challenge, we took advantage of the BiFC system to examine MAPK signaling events. By co-expressing YDA-nYFP and MPK6-cYFP, the recovered YFP signal indicated that the activation of the MAPK signaling relay might occur at the plasma membrane.
It would be ideal to co-express fluorescently tagged YDA and MPK6 in Arabidopsis and to examine the dynamic MAPK signaling in vivo. However, generating transgenic plants is time-consuming, particularly when more than one constructs is desired. Protein transient expression in tobacco cells can be a quick preliminary test for the in vivo expression in Arabidopsis or other plants. The other advantage that the tobacco transient expression system offers, but cannot be easily achieved by generating transgenic plants, is that up to 4 or 5 fusion proteins can be co-expressed simultaneously and examined by confocal microscopy when the combination is achievable.
Using tobacco epidermal cells to investigate MAPK signaling in plant cells can be further implemented to other signaling pathways and under different environmental conditions. For example, would the interaction of YDA-MAPK change its intensity or location when the expressing cell is treated with local H2O2 application (a MAPK signaling stimulus)? Such assays can be quickly designed and performed to address specific questions. Multiple members exist in each tier of the MAPK cassettes (3 – 4 tiers in general), but how signaling specificity is achieved in plants or other systems remain largely unknown. By making different combinations of MAPKKKs with MAPKKs, or MAPKKs with MAPKs, the tobacco transient expression system allows a relatively large-scale analysis of not only protein-protein interaction, but also the spatial organization of the signaling flow in plant cells.
Customized Co-expression and BiFC Assay for Investigation of Protein Polarization: One long-standing fundamental question in cell biology is how universal signaling pathways, e.g., MAPKs, achieve their specificity at certain developmental stage or under particular environmental conditions. Co-expressing the polarity protein BASL with the BiFC components of YDA and MPK6 in tobacco cells has allowed us to demonstrate that the YDA-MAPK interaction and signaling is spatially re-distributed by the presence of BASL, a specific polarity proteins during stomatal ACD. Therefore, the YDA-MAPK signaling specificity can be achieved by interaction with BASL to promote cell polarity and asymmetric division.
Although it is not a universal phenomenon, quite a few proteins have been found to be unevenly distributed in plant cells, e.g., small GTPase ROPs15, auxin effluxer PIN proteins16 and their regulators17, boron transporters18 and some unknown proteins (OCTOPUS19, POLAR20, and BRX21). This protocol was not intended to search the genetic or physical partners in a particular protein polarization pathway, but will likely be useful for testing whether the interacting proteins with ROPs, PINs or others may form a polarity complex in plant cells. However, when trying to simulate a polarization event using tobacco cells, one should keep in mind that complications may occur in the assays. For example, the Arabidopsis polarization event may not be recapitulated in tobacco cells, in particular when more components are required than what can be accommodated in the assay. Some unknown molecules derived from the tobacco background, not necessarily the proteins under examination, may be involved in inducing a polarization event. Therefore, it is essential to set up strict control experiments in tobacco and to validate the data in Arabidopsis or other plants.
Key Elements to Succeed in the Experiments: Firstly, greenish and healthy tobacco plants should be used. Since quantification analyses need to pool data from various cells, it is crucial to use thriving tobacco plants with consistently healthy cells. Secondly, the cells expressing medium protein levels should be used for confocal imaging. Low expression levels may not provide sufficient proteins for the assay and saturated expression levels would mask the polarization/translocation effect.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank Yumeng (Helen) Xia (Rutgers University) for manuscript editing. The work on BASL polarity formation is supported by grants from the U.S. National Institute of General Medical Sciences to J.D. (R01GM109080) and Rutgers University.
Materials
| Phusion DNA polymerase | New England Biolabs | M0530S | |
| pENTR/D/TOPO | Life Technologies | K2400-20 | |
| QuickChange II XL Site-directed Mutagenesis Kit | Agilent Technology | 200521 | |
| LB broth | AMRESCO | J106-2KG | |
| Bacto Agar | AMRESCO | J673-1KG | |
| Gentamycin | Sigma-Aldrich | G3632-1G | |
| Rifampicin | Sigma-Aldrich | R3501-1G | |
| Kanamycin | Sigma-Aldrich | K4000-25G | |
| Spectinomycin | Sigma-Aldrich | S4014-5G | |
| MgCl2 | Sigma-Aldrich | M8266-100G | |
| 25x75mm Slide | VWR | 16004-382 | |
| 24×50 mm Cover glass | VWR | 48393-241 | |
| Laser scanning confocal microscope | Leica | TCS SP5 II | LAS AF Lite software |
| 40 x objective lens | Leica | HCX Plan APO | HCX Plan APO , NA 1.30 |
| Centrifuge | Thermo Scientific | SORVALL 6+ | |
| Hole puncher | Staples | ||
| 50 ml falcon tubes | Genesee Scientific | 21-108 | |
| No. 5 Forceps | Canemco & Marivac | 205EQA-Spec |
References
- Dong, J., MacAlister, C. A., Bergmann, D. C. BASL controls asymmetric cell division in Arabidopsis. Cell. 137, 1320-1330 (2009).
- Lukowitz, W., Roeder, A., Parmenter, D., Somerville, C. A MAPKK kinase gene regulates extra-embryonic cell fate in Arabidopsis. Cell. 116, 109-119 (2004).
- Bergmann, D. C., Lukowitz, W., Somerville, C. R. Stomatal development and pattern controlled by a MAPKK kinase. Science. 304, 1494-1497 (2004).
- Lampard, G. R., Lukowitz, W., Ellis, B. E., Bergmann, D. C. Novel and expanded roles for MAPK signaling in Arabidopsis stomatal cell fate revealed by cell type-specific manipulations. Plant Cell. 21, 3506-3517 (2009).
- Wang, H., Ngwenyama, N., Liu, Y., Walker, J. C., Zhang, S. Stomatal development and patterning are regulated by environmentally responsive mitogen-activated protein kinases in Arabidopsis. Plant Cell. 19, 63-73 (2007).
- Zhang, Y., Wang, P., Shao, W., Zhu, J. K., Dong, J. The BASL Polarity Protein Controls a MAPK Signaling Feedback Loop in Asymmetric Cell Division. Dev Cell. 33, 136-149 (2015).
- Xu, R., Li, Q. Q. Protocol: Streamline cloning of genes into binary vectors in Agrobacterium via the Gateway(R) TOPO vector system. Plant methods. 4, 4 (2008).
- Bush, S. M., Krysan, P. J. Mutational evidence that the Arabidopsis MAP kinase MPK6 is involved in anther, inflorescence, and embryo development. J Exp Bot. 58, 2181-2191 (2007).
- Carey, M. F., Peterson, C. L., Smale, S. T. PCR-mediated site-directed mutagenesis. Cold Spring Harbor protocols. , 738-742 (2013).
- Kubo, M., et al. Transcription switches for protoxylem and metaxylem vessel formation. Genes Dev. 19, 1855-1860 (2005).
- Yuan, L., et al. Allosteric regulation of transport activity by heterotrimerization of Arabidopsis ammonium transporter complexes in vivo. Plant Cell. 25, 974-984 (2013).
- Weigel, D., Glazebrook, J. Transformation of agrobacterium using the freeze-thaw method. CSH protocols. 2006 (7), (2006).
- Voinnet, O., Rivas, S., Mestre, P., Baulcombe, D. An enhanced transient expression system in plants based on suppression of gene silencing by the p19 protein of tomato bushy stunt virus. Plant J. 33, 949-956 (2003).
- Rivero, L., et al. Handling Arabidopsis plants: growth, preservation of seeds, transformation, and genetic crosses. Methods in molecular biology. 1062, 3-25 (2014).
- Yang, Z. Cell polarity signaling in Arabidopsis. Annu Rev Cell Dev Biol. 24, 551-575 (2008).
- Zhang, J., Nodzynski, T., Pencik, A., Rolcik, J., Friml, J. PIN phosphorylation is sufficient to mediate PIN polarity and direct auxin transport. Proc Natl Acad Sci U S A. 107, 918-922 (2010).
- Furutani, M., et al. Polar-localized NPH3-like proteins regulate polarity and endocytosis of PIN-FORMED auxin efflux carriers. Development. 138, 2069-2078 (2011).
- Takano, J., et al. Polar localization and degradation of Arabidopsis boron transporters through distinct trafficking pathways. Proc Natl Acad Sci U S A. 107, 5220-5225 (2010).
- Truernit, E., Bauby, H., Belcram, K., Barthelemy, J., Palauqui, J. C. OCTOPUS, a polarly localised membrane-associated protein, regulates phloem differentiation entry in Arabidopsis thaliana. Development. 139, 1306-1315 (2012).
- Pillitteri, L. J., Peterson, K. M., Horst, R. J., Torii, K. U. Molecular profiling of stomatal meristemoids reveals new component of asymmetric cell division and commonalities among stem cell populations in Arabidopsis. Plant Cell. 23, 3260-3275 (2011).
- Mouchel, C. F., Osmont, K. S., Hardtke, C. S. BRX mediates feedback between brassinosteroid levels and auxin signalling in root growth. Nature. 443, 458-461 (2006).

