Proximal Cadaveric Femur Preparation for Fracture Strength Testing and Quantitative CT-based Finite Element Analysis
Summary
We present a robust protocol on how to carefully preserve and prepare cadaveric femora for fracture testing and quantitative computed tomography imaging. The method provides precise control over input conditions for the purpose of determining relationships between bone mineral density, fracture strength, and defining finite element model geometry and properties.
Abstract
Cadaveric fracture testing is routinely used to understand factors that affect proximal femur strength. Because ex vivo biological tissues are prone to lose their mechanical properties over time, specimen preparation for experimental testing must be performed carefully to obtain reliable results that represent in vivo conditions. For that reason, we designed a protocol and a set of fixtures to prepare the femoral specimens such that their mechanical properties experienced minimal changes. The femora were kept in a frozen state except during preparation steps and mechanical testing. The relevant clinical measures of total hip and femoral neck bone mineral density (BMD) were obtained with a clinical dual X-ray absorptiometry (DXA) bone densitometer, and the 3D geometry and distribution of bone mineral were obtained using CT with a calibration phantom for quantitative estimations based on the greyscale values. Any possible bone disease, fracture, or the presence of implants or artifacts affecting the bone structure, was ruled out with X-ray scans. For preparation, all bones were carefully cleaned of excess soft tissue, and were cut and potted at the internal rotation angle of interest. A cutting fixture allowed the distal end of the bone to be cut off leaving the proximal femur at a desired length. To allow positioning of the femoral neck at prescribed angles during later CT scanning and mechanical testing, the proximal femoral shafts were potted in polymethylmethacrylate (PMMA) using a fixture designed specifically for desired orientations. The data collected from our experiments were then used for validation of quantitative computed tomography (QCT)-based finite element analysis (FEA), as described in a different protocol. In this manuscript, we present the protocol for the precise bone preparation for mechanical testing and subsequent QCT/FEA modeling. The current protocol was successfully applied to prepare about 200 cadaveric femora over a 6-year time period.
Introduction
Determining the true cadaveric proximal femoral fracture strength with mechanical testing is a destructive method that requires a rigorous testing approach for accurate measurements. In particular, proper bone preparation methods are necessary to maintain near in vivo integrity of the bones prior to mechanical fracture testing1. This is achieved by proper bone storage and minimizing handling at room temperature. This test data is extensively used to validate QCT/FEA models of femoral fracture which have the potential to be used clinically to understand the fracture risk, especially in osteoporotic patients. Unfortunately, there is no current standard procedure to prepare proximal femur samples for mechanical testing. A good testing procedure should ensure repeatability and reproducibility of the preparation process. Therefore, fixtures required for sample preparation need to be carefully designed and fabricated to minimize the likelihood of various testing errors. We also need to minimize the preparation time for which bone tissue is at room temperature and thus in danger of degradation with irreversible changes in mechanical properties.
To this end, we have developed a procedure that preserves bone tissue across multiple preparation steps. This is important to ensure minimal exposure time at room temperature while also minimizing the number of freeze/thaw cycles which can affect tissue physical properties2. The entire procedure is long and nontrivial as the steps occurred over multiple weeks and required scheduling for scanning procedures and personnel availability. The steps included thawing bone samples, screening the samples using DXA scanning to obtain bone mineral density (BMD) values, X-ray to rule out any diseased specimens, and finally CT scanning to estimate distribution of bone mineral and femoral geometry. All the specimens were prepared for testing by removing extraneous soft tissues from the bone surface, cutting the femur to a length required for testing, and potting the femur in a desired orientation for simulating a sideways fall on the hip during subsequent testing. It is essential to keep the time period for all these operations as short as possible. A robust protocol is thus mandatory for consistent specimen preparation, tissue preservation between steps, and for reducing the overall preparation time.
The aim of this paper is to present in detail the procedures involved in the preparation of femoral samples for subsequent mechanical testing under various conditions. Preservation of the bone tissue is crucial in this process and we achieved it by keeping specimens frozen between steps and keeping them carefully wrapped in saline saturated towels at all times except when scanning and mechanically testing the bones. Femora were also kept wrapped in saline wet towels during the steps involving PMMA curing to prevent dryness of the bone tissue.
Protocol
NOTE: All studies presented in this protocol were approved by the Institutional Review Board (IRB) at Mayo Clinic. The bones were obtained over a period of 6 years from various organizations. All specimens were collected within 72 hours of death, wrapped in saline saturated towels, and stored at -20 °C until preparation.
1. Measuring Bone Mineral Density Using DXA
- Remove specimens kept at -20 °C freezer to thaw at room temperature for about 24 h; specimens do not need to be removed from original packing if most soft tissue has been removed.
- Use two 5 lb bags of rice to account for soft tissue. Cover the two rice bags on the DXA table with plastic bags to prevent contamination. The rice bags will simulate surrounding soft tissue (in vivo) during scanning as shown in Figure 1.
- Protect the surface of the DXA scanner with plastic lined paper and place 2 plastic wrapped rice bags on the scanner table (Figure 1A).
- Lay 2 femora (right and left) on top of rice bags such that the proximal end (including the femoral head) is centered on bags and the posterior side is down (Figure 1B). This mimics a patient lying on their back.
- Cover anterior/exposed proximal femur end with additional two 5 lb rice bags (Figure 1C).
- Position machine head over proximal femur and scan the femur according to the standard institutional procedure for patient BMD measurement (Figure 1C). Follow the specific DXA manufacturer instructions.
- From the DXA machine software interface perform a normal femur scan. Select the femur exam, position the DXA arm on top of the cadaveric femur by pressing on the left or right arrow accordingly on the DXA arm, and start the exam by clicking on the "start" button. Perform the BMD analysis by clicking on "Analyze".
NOTE: The resulting automatic T-score from the scan classifies the bone as normal, osteopenic or osteoporotic (Figure 1D). Follow the specific DXA manufacturer instructions.
- From the DXA machine software interface perform a normal femur scan. Select the femur exam, position the DXA arm on top of the cadaveric femur by pressing on the left or right arrow accordingly on the DXA arm, and start the exam by clicking on the "start" button. Perform the BMD analysis by clicking on "Analyze".
2. Cleaning, Cutting and Drilling the Distal End of the Bone
- Clean the most proximal 300 mm of the femur by carefully removing any remaining soft tissue from the bone. This step is needed to allow PMMA to contact the bone during the potting process in preparation for mechanical testing. The bones need not be thawed to room temperature for this process.
- Sanitize the workspace with 70% isopropyl alcohol and cover the table with absorbent paper pads with plastic film on one side (Figure 2A). Set the whole femur on the table (Figure 2B) for the entire process starting with cleaning to cutting (Figure 2A 세스 2H). Wear personal protective equipment (PPE) including gloves and eye protection.
- Scrape off excess periosteum and cut away excess tissue using a scraper and scalpel (Figure 2C, 2H).
- Place the bone in the custom-made cutting fixture as shown in Figure 2D with the head of the femur against the acrylic plate of the fixture.
- Align and hold the diaphysis against the two pins on the cutting fixture (Figure 2D).
- Secure the bone to the fixture by tightening down the slotted plate on the diaphysis; if bone does not lie flat on fixture, hang distal end off table top and also optionally zip tie the femoral neck as needed to hold the specimen in place (Figure 2D).
- Cut the distal shaft of the femur using the cast cutter (Figure 2E) through slotted plate as a guide; hold bone with a dry rag/towel for better grip.
- Remove the bone from the fixture; normal bone length after cut is 255 mm (Figure 2F).
- Clean the medullary cavity of marrow using a curette approximately 25 mm deep. Then, insert a gauze sponge to help dry the interior surface. Remove the gauze just prior to placing the distal end of the femur in the mold. Grip the bone with a dry towel/rag and drill a 10 mm hole through the distal end at approximately 25 mm from the proximal cut end of the specimen. Note: This is to allow the PMMA to penetrate the canal and secure the bone firmly.
3. Potting the Bone
- Design and fabricate potting container. The potting containers are made of 5 mm thick acrylic sheets and have the following outer dimensions: 50 mm by 50 mm square cross section and 100 mm tall (Figure 3A).
- Label potting container with appropriate bone identification (Figure 3A and 3B – see label on acrylic box).
- Adjust the embedding fixture to the proper orientation (left leg or right leg; e.g. 15° or 30° internal rotation).
- Place potting container in base of embedding fixture, place bone in the potting container (Figure 3B) and align neck with pointer in fixture (Figure 3C) to adjust the internal rotation angle of the bone to the desired value.
- Measure 60 g of PMMA powder and mix with 30 g of liquid resin under fume hood until the powder has dissolved. The mixture should be pourable. Use a disposable paper cup for this process.
- Pour mixture into potting container with bone under a fume hood (Figure 3D), allow to cure for about 10 – 15 min until PMMA is clear and hard. This should only fill ~ 1/2 of the potting container with PMMA. Carefully wrap the bone in saline saturated towels to prevent tissue dryness due to heat generation during PMMA polymerization (Figure 3E).
- Periodically check the femur to ensure it remains aligned in the container during curing.
- Remove femur from potting fixture and wrap with a saline soaked paper towel (Figure 3E).
- Prepare 90 g of PMMA under a fume hood as explained in step 3.5 and fill the potting container completely. Cure the PMMA for about 10 – 15 min until it becomes hard.
- After resin has cured, tightly wrap/rewrap bone in saline soaked paper towels, cover with plastic bag and store specimens at -20 °C in freezer.
4. Imaging the Bone with X-ray
(CAUTION! Operate with proper care for X-ray radiation when using the machine)
- If using films for X-rays, turn X-ray developer on at least 20 min (per manufacturer instructions) prior to scanning by turning the knob clockwise (in the developer room).
- Ensure there is unexposed film in the cassette before X-raying; cassette should only be opened in a dark room.
- Turn machine on to unlock and extend the machine head.
- Place a cart under the path of the beam and place a cassette on the cart under the beam (Figure 4A).
- Place and position the femur on the cassette (Figure 4B); two orientations will be captured: medial-lateral view and anterior-posterior view. Label the specimen images accordingly.
- After first exposure, swap the lead and bone locations.
- Cover the already exposed half with lead and expose the bone in the second orientation on the unexposed side. This allows the user to use one X-ray film for a single bone in two orientations (Figure 4C-D).
- Change the cassette and rotate each femur into the second orientation.
- In the case of a single femur, cover half of the cassette with a lead cover to avoid initial exposure of the entire film to X-rays.
- Move behind the lead-lined portable wall for personal protection and use the trigger to expose the bones.
- On completion, return X-ray head to lock and store position, turn key to OFF on X-ray machine and remove exposed X-ray film.
- Develop the film to obtain X-ray images (Figure 4E) using a regular film developer. Turn on the white light in the room and locate the film developer. Turn on the red light and turn off the white light before opening the cassette and proceed with the film developing process. Open the cassette and put the film through the developer. Turn on the white light and turn off the red light once the film has been developed.
5. CT Scanning of Bones
- Remove bones from freezer about 24 h prior to scanning. Bones must be completely thawed prior to scanning.
- Ensure the bones are wrapped in plastic bags for scanning to minimize clean-up at the end.
- Place and secure the femur and the calibration phantom in the CT scanning fixture (Figure 5A-B). The fixture holds the calibration phantom (Figure 5C) and also holds the femur in an orientation (Figure 5D-E) identical with the orientation desired for subsequent mechanical testing. This cross registration is required to use the data from CT scanning (Figure 5F) in the QCT/FEA modeling process (described in a different protocol).
NOTE: The fixture is designed such that it exposes the femur for CT scanning without obstructing the proximal femur (femoral head, neck, greater trochanter, and proximal shaft). - Make sure the fixture along with the femur in the CT scanner is properly aligned using the system lasers as guides (Figure 5D-E). Re-check the alignment of the fixture with the CT long axis laser (by pressing laser on/off button). The phantom does NOT need to be aligned with the laser as it is secured in the fixture. Zero the table position from the control panel of the machine by pressing the zero button (→ 0 ←) on the control panel.
- Following CT standard operating procedure, operate the CT machine at 120 kVp, 216 mAs, 1 s rotation time, and pitch of 1 using ultra high resolution mode (zUHR). This gives a slice thickness of 0.4 mm, and a pixel size of 0.30 – 0.45 mm depending on the size of the field of view (FOV).
- Check CT-scan data prior to mechanical testing to ensure that images of interest are captured and saved. Refreeze the bone at -20 °C until the day of the experiment.
Representative Results
The cadaveric femora were shipped frozen and maintained at -20 °C until preparation began. BMD scanning was performed using a DXA scanner to measure total hip and neck BMD as well as T-score for each specimen (Figure 1). A T-score is the number of standard deviations of the measured BMD compared to average values for young healthy subjects. It can range from -2.5 or lower for osteoporotic bones, between -1 and -2.5 for osteopenic bones and higher than -1 for normal bones. Once finished, bones were cleaned of excess tissue, and cut to remove the distal end using an in-house designed and fabricated cutting fixture (Figure 2). The specimens were then potted distally using a fixture designed for holding the bones in the desired internal rotation orientation; after placing the distal end into the potting container, the PMMA in liquid form was poured to fill the container (Figure 3). The X-ray images were obtained for paired bones together and for single bones separately to discern the presence of fracture or diseases, such as cancer, that might affect femoral strength (Figure 4). In the presence of such abnormalities, the condition of the bone should be documented when considered for future analyses. Finally, the femora were CT-scanned, in order to obtain CT images, using an acrylic CT scanning fixture designed to hold the bone in appropriate predetermined orientations (adduction and internal rotation angles) (Figure 5). The CT images are used to obtain 3-dimensional bone geometry and volumetric bone mineral distribution to be used in quantitative CT-based finite element analysis. Prior to subsequent fracture testing, all relevant data characterizing each femur such as the BMD values, X-ray images, and CT images were checked to ensure that data of interest were recorded and saved.
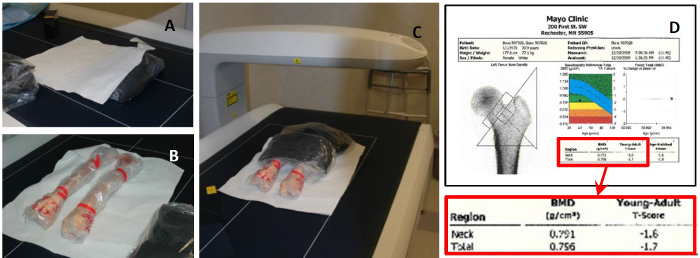
Figure 1: BMD Measurement Using DXA Scanning. (A) Rice bags and plastic lined papers; (B) Two bone specimens in desired orientations in scanner bed; (C) proximal femur ends covered with 2 rice bags during scanning; (D) Neck and total hip BMD measurements with associated T-scores. DXA scanning is performed using a clinical scanner to measure bone mineral density and estimate T-score. Please click here to view a larger version of this figure.

Figure 2: Cleaning and Cutting Bones. (A) Cleaning and cutting desk; (B) bone sample tools for cleaning; (C) cleaning the shaft of a femur; (D) securing a sample in the cutting fixture; (E) cast cutter; (F) completed sample after cutting. A special fixture and bone cleaning and cutting tools are used to prepare the most proximal 255 mm length for testing; (G) curette used for cleaning intramedullary canal of the femur; (H) tool for cleaning superficial tissue in samples. Please click here to view a larger version of this figure.

Figure 3: Femur Cleaning and Potting Process. (A) Potting fixture; (B) potting a femur in the fixture; (C) adjusting internal rotation angle to desired value; (D) pouring PMMA in the container; (E) potted bone wrapped in a saline saturated towels. A special fixture is used to set the internal rotation angle to a specified value. Please click here to view a larger version of this figure.
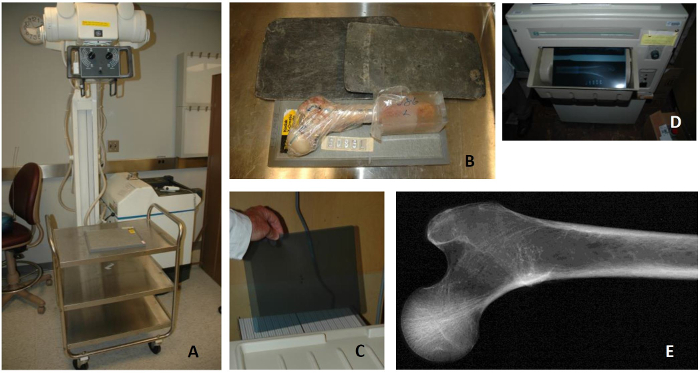
Figure 4: Bone X-ray Process. (A) X-ray machine; (B) bone sample on a cassette with unexposed film, a second half of the cassette is covered by lead to avoid exposure of the entire film; (C) placing unexposed film in loading tray of developer in dark room; (D) developed film; (E) resulting X-ray image of a healthy femur. X-ray equipment is used to scan the bones in two positions to rule out prior fractures, implants, bone metastasis, or any structural abnormalities. Please click here to view a larger version of this figure.

Figure 5: CT Scanning Using an Acrylic Fixture to Hold Bones in a Desired Orientation. (A) CT scanner; (B) fiberglass femur mounted in an acrylic fixture designed to hold bones in a desired orientation; (C) mounting a cadaveric femur in the fixture; (D) vertical alignment of the fixture using the CT long axis laser; (E) Alignment of the femur. An in-house designed fixture is used to hold the bone in a position identical to the subsequent testing position; bone alignment is obtained with the aid of the CT built-in lasers; (F) CT imaging of the femur. Trabecular and cortical bone can be visualized in the CT image. Cortical bone is represented by bright voxels (large Hounsfield Unit (HU) values) surrounding the trabecular bone which is represented by smaller HU values. Note: Care has to be taken to include all five rods and the entire proximal femur in the CT images. Please click here to view a larger version of this figure.
Discussion
We presented a robust bone preparation protocol for ensuring mechanical testing and QCT/FEA modeling of femoral strength in a sideways fall on the hip configuration. This method became our standard in-house protocol. Over the course of 6 years, with varying personnel, about 200 femora were successfully prepared following this protocol. The outcomes of the protocol includes classifying bone conditions using DXA, ruling out metastatic diseases, previous fractures, or implants using X-ray, and obtaining mineral distribution and 3D geometry using CT for subsequent QCT/FEA modeling. Cutting, potting, and scanning fixtures were designed to accommodate left and right femora as well as for different bone orientations required for future testing, modeling, and analysis. The in-house fixtures assured repeatability and reproducibility of test samples.
Due to the complexity of bone experiments and the need for the combination of BMD, X-ray, and CT scanning before mechanical testing, femora must undergo multiple freeze/thaw cycles. With a proper protocol that minimizes the exposure to room temperature, freezing bone specimens preserves the tissue for mechanical testing, even long term3,4. Previous studies showed that freezing bones at -20 °C does not alter their mechanical properties and that a few freeze/thaw cycles before testing is considered a safe and feasible process5,6. In our study, all femora experienced three freeze/thaw sequences at -20 °C and room temperature, respectively, for DXA scanning, CT scanning, and mechanical testing.
In line with several previous studies, standardized rice bags were used while measuring BMD values of specimens using DXA to mimic in vivo soft tissue around bone7. We compared BMD values of our cadaveric cohort with BMD values of different patient populations and found their distributions to be very similar, suggesting the robustness of our protocol for BMD measurements8.
Femoral samples lack flat surfaces to be easily and properly aligned to a desired orientation for testing. If not done properly, this may impact the repeatability of the procedure and limit the accuracy of the experimental results9. To address this issue, several fixtures were designed and fabricated and standard operating procedures were implemented to make tissue handling independent of the users' skill throughout the sample preparation process. While the femora were acquired and tested over several years, the protocol and the hardware remained the same reducing the potential preparation errors.
One important step of our bone preparation process was to perform CT scanning for 3D modeling of bone fracture using QCT/FEA. Thus, registration between CT scanning and future fracture testing was a necessary step in our femoral sample preparation protocol10.
The current method for bone preparation has some limitations. Although careful planning was implemented during the acquisition of the cadavers, dissection, potting and CT scanning, scheduling the various phases of femur preparation could be challenging due to personnel and equipment availability. Our process requires the specimens to be frozen and thawed at multiple time points. Nevertheless, the freezing time never exceeded more than two weeks, and the bones were subjected to a total of three freeze/thaw cycles. Also the bone preparation process was designed to minimize the operator errors. We observed only one error in potting the distal end of the proximal femur. One right leg femur was potted at the wrong internal rotation angle which, was discovered only after CT imaging. Subsequently, this femur was discarded from further data analysis. Therefore, a second operator might be needed for this step to check on the orientation of the femur before pouring the PMMA for potting. No other errors were observed in any of the other steps. Thus, it is significant to note that our process was very robust allowing for only one error, with multiple operators, during the preparation of about 200 proximal femora in the course of several years.
To provide a good quality control system that will minimize the likelihood of operator errors, certain parts of the protocol need to be repeated or re-checked by a second operator. For example, care should be taken during potting to assure that the femoral shaft is drilled to allow bone cement to enter the femur cavity, guaranteeing that the femur is rigidly fixed and will not loosen during testing. Additionally, potting the femur at the internal rotation angle of interest is usually performed by one operator. Before the PMMA is poured for potting the distal end, a second operator might be required to check that the internal rotation angle was set at the required value. Finally, during CT scanning of the femur, the alignment of the fixture holding the bone on the CT scanner bed is critical. One operator should precisely align the fixture with the CT-laser beams and a second operator should confirm that the fixture is properly aligned.
While the current protocol was designed specifically for fracture testing and modeling of femoral specimens in a sideways fall on the hip configuration, it can be easily extended to other loading scenarios including non-destructive testing, or adopted to test other bone types with appropriate fixture redesign.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We would like to thank the Materials and Structural Testing Core Facility at Mayo Clinic for technical support. In addition we would like to thank Lawrence J. Berglund, Brant Newman, Jorn op den Buijs, Ph.D., for their help during the study. This study was financially supported by the Grainger Innovation Fund from the Grainger Foundation.
Materials
| CT potting container and scanning fixture | Internally manufactured | N/A | Custom designed and manufactured |
| CT scanner | Siemens | Somatom Definition scanner (Siemens, Malvern, PA) | CT scanning equipment |
| Quantitative CT Phantom | Midways Inc, San Francisco, CA | Model 3 CT calibration Phantom | Used for obtaining BMD values from Hounsfield units in the CT image |
| Dual Energy X-ray Absorptiometry scanner | General Electric | N/A | GE Lunar iDXA scanner for bone health or any similar BMD scanners |
| Hygenic Orhodontic Resin (PMMA) | Patterson Dental Supply | H02252 | Controlled substance and can be purchased with proper approval |
| Freezer | Kenmore | N/A | This is a -20oC storage for bones |
| X-ray scanner | General Electric | 46-270615P1 | X-ray imaging equipment. |
| X-ray films | Kodak | N/A | Used to display x-ray images |
| X-ray developer | Kodak X-Omatic | M35A X-OMAT | Used for developing X-ray images |
| X-ray Cassette | Kodak X-Omatic | N/A | Used for holding x-ray films |
| 5-pound Rice Bags | Great Value | N/A | Used for mimicking soft tissue during the DXA scanning process |
| Physiologic Saline (0.9% Sodium Chloride) | Baxter | NDC 0338-0048-04 | Used for keeping samples hydrated |
| Scalpels and scrapers | Bard-Parker | N/A | Used to clean the bone from soft tissue |
| Cast cutter | Stryker | 810-BD001 | Used to cut femoral shaft |
| Drilling machine | Bosch | N/A | Used to drill the femoral shaft |
| Fume Hood | Hamilton | 70532 | Used for ventilation when using making PMMA |
References
- Cristofolini, L., Schileo, E., Juszczyk, M., Taddei, F., Martelli, S., Viceconti, M. Mechanical testing of bones: the positive synergy of finite-element models and in vitro experiments. Philos Trans A Math Phys Eng Sci. 368, 2725-2763 (2010).
- Cartner, J. L., Hartsell, Z. M., Ricci, W. M., Tornetta, P. Can we trust ex vivo mechanical testing of fresh-frozen cadaveric specimens? The effect of postfreezing delays. J Orthop Trauma. 25 (8), 459-461 (2011).
- An, Y. H., Draughn, R. A. . Mechanical testing of bone and the bone-implant interface. , (1999).
- van Haaren, E. H., van der Zwaard, B. C., van der Veen, A. J., Heyligers, I. C., Wuisman, P. I., Smit, T. H. Effect of long-term preservation on the mechanical properties of cortical bone in goats. Acta Orthop. 79, 708-716 (2008).
- Shaw, J. M., Hunter, S. A., Gayton, J. C., Boivin, G. P., Prayson, M. J. Repeated freeze-thaw cycles do not alter the biomechanical properties of fibular allograft bone. Clin Orthop Relat Res. 470 (3), 937-943 (2012).
- Topp, T., et al. Embalmed and fresh frozen human bones in orthopedic cadaveric studies: which bone is authentic and feasible?. Acta Orthop. 83 (5), 543-547 (2012).
- Manske, S., et al. Cortical and trabecular bone in the femoral neck both contribute to proximal femur failure load prediction. Osteoporos Int. 20 (3), 445-453 (2009).
- Rezaei, A., Dragomir-Daescu, D. Femoral Strength Changes Faster With Age Than BMD in Both Women and Men: A Biomechanical Study. J Bone Miner Res. 30, 2200-2206 (2015).
- Cristofolini, L., McNamara, B., Freddi, A., Viceconti, M. In vitro measured strains in the loaded femur: quantification of experimental error. J Strain Anal Eng Des. 32, 193-200 (1997).
- Dragomir-Daescu, D., et al. Robust QCT/FEA models of proximal femur stiffness and fracture load during a sideways fall on the hip. Ann Biomed Eng. 39, 742-755 (2011).

