Snap Chip for Cross-reactivity-free and Spotter-free Multiplexed Sandwich Immunoassays
Summary
We demonstrate a snap chip technology for performing cross-reactivity-free multiplexed sandwich immunoassays by simply snapping two slides. A snap apparatus is used for reliably transferring reagents from microarray-to-microarray. The snap chip can be used for any biochemical reactions requiring colocalization of different reagents without cross-contamination.
Abstract
Multiplexed protein analysis has shown superior diagnostic sensitivity and accuracy compared to single proteins. Antibody microarrays allow for thousands of micro-scale immunoassays performed simultaneously on a single chip. Sandwich assay format improves assay specificity by detecting each target with two antibodies, but suffers from cross-reactivity between reagents thus limiting their multiplexing capabilities. Antibody colocalization microarray (ACM) has been developed for cross-reactivity-free multiplexed protein detection, but requires an expensive spotter on-site for microarray fabrication during assays. In this work, we demonstrate a snap chip technology that transfers reagent from microarray-to-microarray by simply snapping two chips together, thus no spotter is needed during the sample incubation and subsequent application of detection antibodies (dAbs) upon storage of pre-spotted slides, dissociating the slide preparation from assay execution. Both single and double transfer methods are presented to achieve accurate alignment between the two microarrays and the slide fabrication for both methods are described. Results show that <40 μm alignment has been achieved with double transfer, reaching an array density of 625 spots/cm2. A 50-plexed immunoassay has been conducted to demonstrate the usability of the snap chip in multiplexed protein analysis. Limits of detection of 35 proteins are in the range of pg/mL.
Introduction
A panel of biomarkers comprising multiple proteins may provide higher sensitivity and specificity than a single biomarker in the diagnosis of complex diseases such as cancers1,2. The enzyme-linked immunosorbent assay (ELISA) has been the gold standard technology used in clinical laboratories achieving a limit of detection at low pg/mL in plasma, but limits to one target per assay3,4,5. Antibody microarrays have been developed for accommodating thousands of miniaturized assays conducted in parallel on a single microscope slide6,7,8. However, the multiplexing capability of this method is limited by reagent-driven cross-reactivity, arising from the application of a mixture of dAbs, and it becomes more problematic with an increasing number of targets9,10,11. Pla et al. have stated that the resulting vulnerability of a multiplex sandwich assay scales as 4N(N-1) where N is the number of the targets12.
To mitigate cross-reactivity in antibody microarrays, antibody colocalization microarray (ACM) has been developed in our laboratory for multiplex sandwich assay12. Capture antibodies (cAbs) are spotted on a substrate with a microarray spotter. After blocking samples are applied on the surface, and then individual dAbs are spotted on the same spots with the cAb-antigen complex. All cross-reactivity scenarios between antibodies and antigens can be mitigated with ACM, and limits of detection at pg/mL have been achieved. However, the assay protocol requires preparing and spotting the dAbs during the experiments using an on-site microarray spotter with high precision for alignment purpose, which is expensive and time consuming, limiting the wide application of this technology in other laboratories. A handheld ACM, named snap chip has been developed for cross-reactivity-free and spotter-free multiplex sandwich immunoassays13,14,15. cAbs and dAbs are pre-spotted onto an assay slide and a transfer slide respectively in microarray format and stored. During the assay, the slides are retrieved and a microarray of dAbs are transferred collectively onto the assay slide by simply snapping the two chips together. A snap apparatus is used for reliable reagent transfer. Nitrocellulose coated slides with a relatively large antibody binding capacity have been used as the assay slides to absorb the liquid droplets and thus facilitating reagent transfer, however, the slides are more expensive than regular glass slides and microarray scanners compatible with non-transparent slides are needed for signal acquisition.
In this work, we demonstrate the protocol of performing a multiplex sandwich immunoassay with a snap chip. A novel snap apparatus has been developed for more convenient and reliable reagent transfer from microarray-to-microarray. Importantly, here we have established the reagent transfer method onto regular glass slides with the snap chip. 1024 spots were successfully transferred and aligned onto a glass slide, significantly expanding the use of this technology in most laboratories.
Protocol
1. Fabrication and storage of snap chips
- Single transfer method (Figure 1a)
- Spot cAb solutions containing 400 µg/mL antibodies and 20% glycerol in phosphate-buffered saline (PBS) onto a nitrocellulose (or a functionalized glass) assay slide with an inkjet microarray spotter13 at a relative humidity of 60% (1.2 nL for each spot) with 800 µm center-to-center spacing. Make sure the slide is fixed on the spotter deck according to one corner (here, the bottom left corner was used).
- Incubate the spotted assay slide at room temperature for 1 h with a relative humidity of 60%.
- Clamp a slide module gasket with 16 compartments on the assay slide to divide it into 16 wells. Rinse the slide three times by adding 80 µL of PBS containing 0.1% Tween-20 (PBST) into each well, and shaking at 450 rpm on a shaker, 5 min each time.
- Add 80 µL of blocking solutions to each well and shake for 1 h at 450 rpm.
- Remove the gasket and dry the assay slide with a stream of nitrogen.
- Fix a transfer slide on the spotter deck and push the bottom left corner of the slide against bottom left corner of the spotter deck. Inkjet spot an array of alignment marks (a solution of polystyrene micro-beads) with the same layout as that for the cAbs.
- Let the alignment marks dry. Flip the transfer slide and put it back onto the spotter deck, fixing against the bottom left corner.
- Prepare the dAb spotting solutions containing 20 µg/mL antibodies, 20% glycerol, and 1% BSA.
- Using the spotter's camera, take a picture of an alignment mark. Implement this picture into the spotting program as a fiducial and get the image recognition system of the inkjet spotter to identify the most top left mark. Use its coordinate as the first spot of the dAb array. Spot 8 nL per droplet with a center-to-center spacing of 800 µm.
- Double transfer method (Figure 1b)
- Place a transfer slide 1 on the inkjet spotter deck against the bottom left corner. Spot cAbs solutions containing 400 µg/mL antibodies, 20% glycerol, and 1% BSA in PBS onto the slide with an inkjet microarray spotter at a relative humidity of 60% (0.4 nL for each spot and ~ 200 µm in diameter). The center-to-center spacing is 400 µm.
- Snap the transfer slide with a nitrocellulose coated (or a functionalized glass) assay slide using a snap apparatus (Figure 2) to transfer the cAb droplets onto the assay slide.
- Turn all six buttons on the side of the snap apparatus by 90 degrees to pull and lock all the plungers in the open position. Insert the transfer slide into the slide holder in its receptacle with the clipped corner facing the fixed pogo pin. Turn the first set of three buttons to release the plungers with the golden pogo pin and make sure the slides are pushed against the alignment pins.
- Insert a nitrocellulose-coated slide in the snap apparatus upside-down sitting on the 4 pogo pins. Turn the second set of three buttons to release plungers with the silver pin and make sure the slides are pushed against the alignment pins.
- Close the snap apparatus by placing the top shell on the slide holder using the alignment pillars and holes for a precise positioning.
- Insert the closed snap apparatus into its cage and push the closure tab completely down to bring the microarrays face-to-face while applying the appropriate pressure. Keep closed for 1 min.
- Separate the slides. Incubate the assay slide at room temperature for 1 h with a relative humidity of 60%. Wash, block, and dry the assay slide as described in steps 1.1.3 – 1.1.5.
- Place another transfer slide on the inkjet spotter deck and push against the bottom left corner. Spot dAb solutions containing 50 or 100 µg/mL of antibodies (see Table 1), 20% glycerol, and 1% BSA in PBS. Ensure that each spot is 0.8 nL and the center-to-center spacing between spots is 400 µm.
- Store the assay and the transfer slide. Seal both assay and transfer slides in an airtight bag containing desiccant and put in a -20 °C freezer.
2. Multiplexed immunoassays with snap chips
- Retrieve the assay slide from the freezer. Leave the bag sealed for 30 min until the slide comes to room temperature.
- Prepare 7-point serial diluted sample solutions by spiking proteins in PBS containing 0.05% Tween-20.
NOTE: Here, a 5-fold dilution factor is used. The starting concentrations for each protein are listed in Table 1. - Prepare sample solutions as appropriate. For example, dilute human serum 4 times in PBST buffer.
- Clamp a 16-compartment gasket on the assay slide.
- Fill one column of 8 wells with the 7 protein dilution solutions and a protein-free PBST buffer solution by pipetting. Pipette the sample solutions in the other 8 wells on the same slide.
NOTE: 80 µL solutions can be fit in each well. More slides can be used to measure additional samples when necessary. - Incubate the samples on a shaker at 450 rpm either for 1 h at room temperature or for overnight at 4 °C. Wash the slide three times with PBST on the shaker at 450 rpm, 5 min each time. Remove the gasket and dry the slide under a stream of nitrogen gas.
- Retrieve the transfer slide with dAbs from the freezer. Keep the bag sealed for 30 min at room temperature. Then, incubate the slide in a closed chamber (e.g. empty tips box) containing 60% humidity stabilization beads for 20 min for rehydration.
- Snap the transfer slide with the assay slide using a snap apparatus (Figure 2) to transfer the dAb droplets. See section 1.2.2 for the operation of the snap apparatus.
- Separate the slides. Incubate the assay slide in a closed chamber containing 60% humidity stabilization beads for 1 h. Clamp the assay slide with a 16-compartment gasket and rinse the slide 4 times using PBST on a shaker at 450 rpm, 5 min each time.
- Pipette 80 µL of solutions containing 2.5 µg/mL streptavidin fluorophore in PBS into each well. Incubate for 20 min on a shaker at 450 rpm.
- Rinse the slide 3 times with PBST and once with distilled water on the shaker at 450 rpm. Remove the gasket and dry the slide using nitrogen gas.
3. Slide scanning and data analysis
- Scan the assay slide with a fluorescence microarray scanner using the 635-nm-laser.
- Extract the net intensity of each spot using an analysis software (e.g. array-pro analyzer)16.
- Calculate the limit of detection (LOD) of each protein using a statistics software and determine the protein concentrations in the samples.
NOTE: The LOD is defined as the Y-intercept of the standard curve incremented by three times the standard deviation of three independent assays.
Representative Results
The assay procedure for both single and double transfer methods is shown in Figure 1. In single transfer, the cAbs are spotted directly on the assay slide and the dAbs are transferred onto the assay slide upon use in a mirror pattern of the cAbs (Figure 1a). Only one transfer procedure is required, but this method suffers from misalignment between the two microarrays, mainly caused by the angular misalignment between the slide and the inkjet gantry (Figure 2). One approach to tackle this challenge is to transfer both cAbs and dAbs sequentially onto the assay slide after spotting, and fixing the slides in the snap apparatus properly (Figure 1b). Using this method, both microarrays are transferred to the exact same position. No image recognition system or alignment marker is required for the double transfer method. The misalignment for 98% of the spots was within 41 µm, 6-fold improvement compared to the single transfer method14.
The snap apparatus has been designed to be easy to use without any training and minimize the risk of array misalignment. The slides are brought together with accurate positioning thanks to alignment pins common to both assay and transfer slides. The transfer slide is slightly smaller to fit below the assay slide when suspended by the pogo pins, until the snap apparatus is closed. A spacer is included on the transfer slide maintaining a micrometer-sized gap that permits reliable droplet transfer to the glass slide. Finally, the cage and its closure tab are designed to apply pressure needed for effective transfer at every snap for reproducible, high quality transfer. A photo of the snap apparatus is shown in Figure 3.
Representative images illustrating the results of reagent transfer onto a nitrocellulose slide and a glass slide are shown in Figure 4. Alexa 532 labeled IgGs were used as the first reagent, and Alexa 647 labeled IgGs were used as the second reagent. After transfer, the slide was scanned with 532 nm and 635 nm lasers. The result shows that 3136 (for the nitrocellulose slide) or 1024 (for the glass slide) microspots were transferred with zero failure onto the assay slides and no cross-contamination was observed upon transferring the second reagent. When using glass slides, it is possible to create more hydrophobic surface by functionalization, thus reducing the size of each spot and decreasing the spot-to-spot distance for transferring a larger number of spots. This work expands the application of the snap chip technology to commonly used glass substrates.
Standard curves of a 50-plex immunoassay targeting breast cancer related proteins are illustrated in Figure 5, corresponding to the largest multiplex sandwich antibody microarray to date without cross-reactivity. Double-transfer method was used in this assay, the slide was scanned, and the fluorescence intensity was quantified to generate the standard curves. 35 out of 50 proteins reached LODs at pg/mL (see Table 1) and may be further improved by optimizing the assay conditions. Colocalization of reagents facilitates the use of multiple antibody pairs on a single slide without cross-reactivity and allows the optimization of each pair independently with no interference on other pairs12. These results indicate that snap chip is a scalable technology that can be used for multiplexed protein quantification.
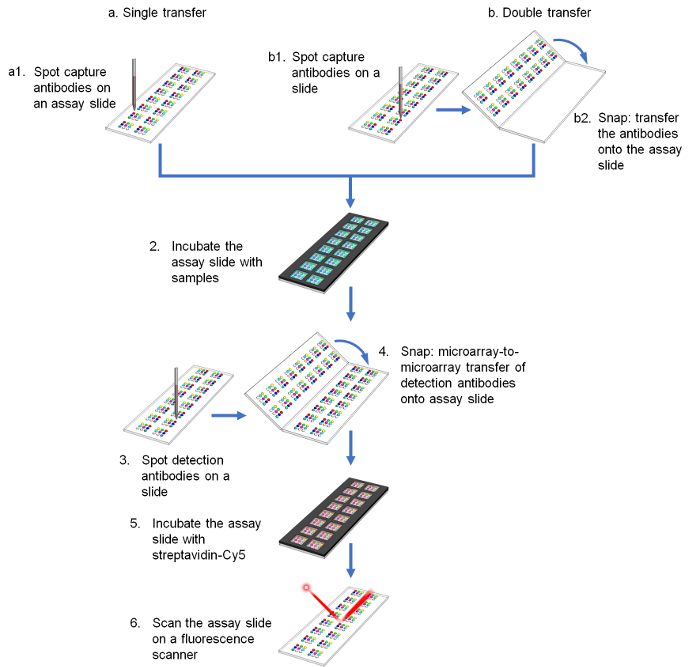
Figure 1. Schematic of assay procedure comparing (a) single and (b) double transfer methods. Please click here to view a larger version of this figure.
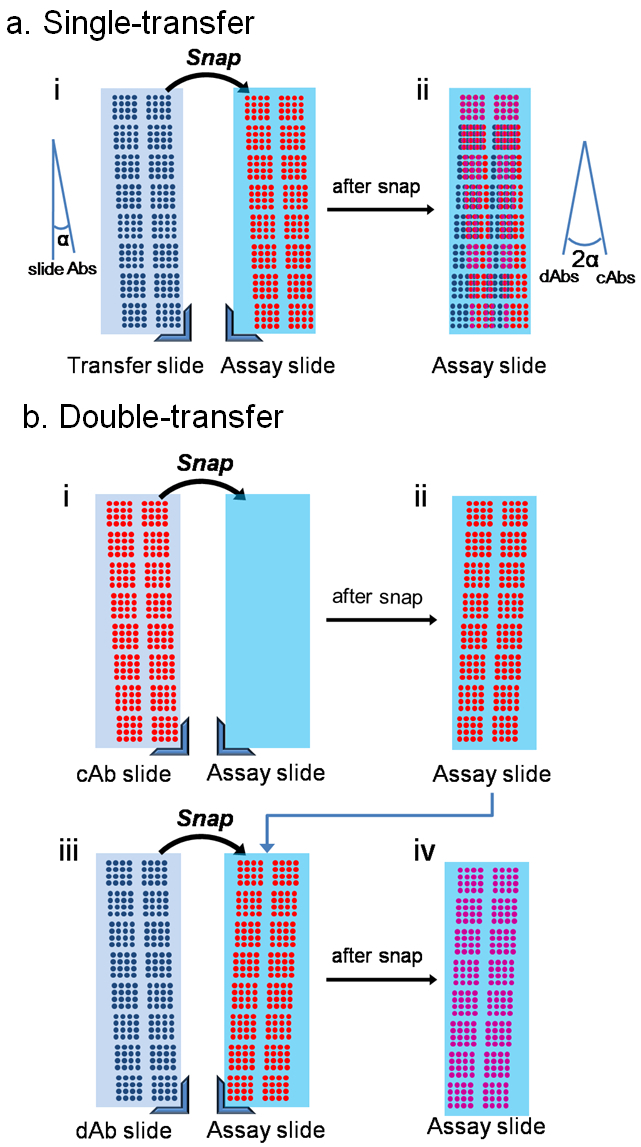
Figure 2. Schematic showing differences between single- and double-transfer. (a) mirroring of the transfer reagents amplifies the angular misalignment between the slide and the inkjet XY stage. (b) double-transfer method overcomes the angular misalignment. Adapted from reference 14 with permission. Please click here to view a larger version of this figure.
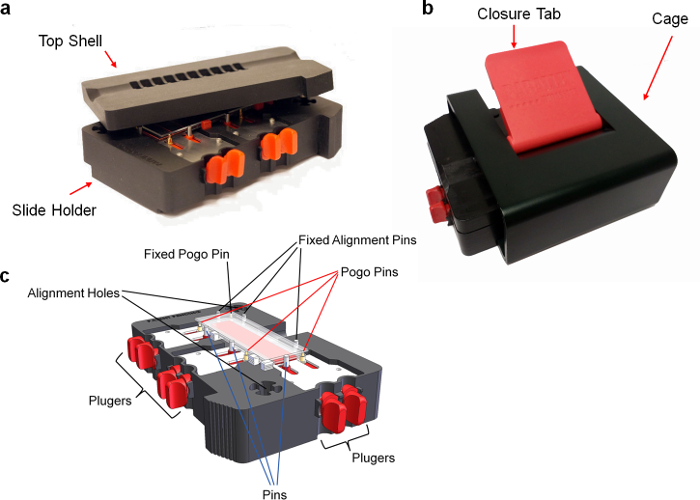
Figure 3. Photo of the snap apparatus. (a) Opened device. (b) Closed device. (c) Perspective view of the device. Briefly, (i) transfer slides are positioned into the slide holder and pushed against alignment pins using the pogopin mounted on plungers. (ii) The assay slides are placed on top of the pogopins and pushed against the alignment pins using a second set of plungers. (iii) The top shell is positioned on top of the slide holder and inserted into the cage. A pressure is applied using the closure tab to compress the pogopin and bring the slides together. Please click here to view a larger version of this figure.
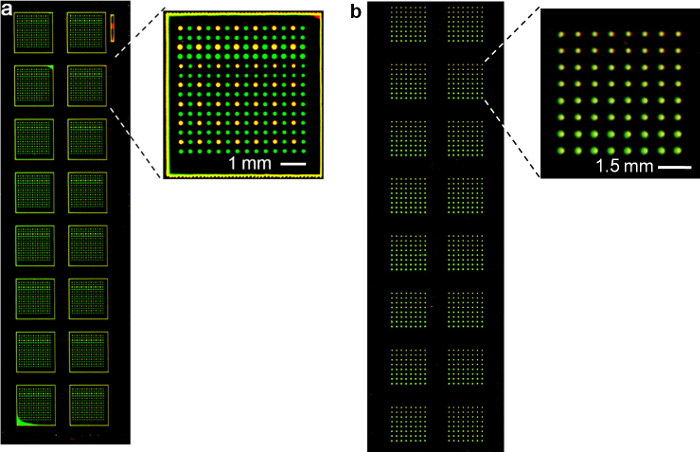
Figure 4. Collective transfer of spots using a snap chip. Fluorescence scan using 532 nm and 633 nm lasers to demonstrate transfer of antibodies onto a (a) nitrocellulose slide (adapted from reference 14 with permission) and (b) glass slide. Please click here to view a larger version of this figure.
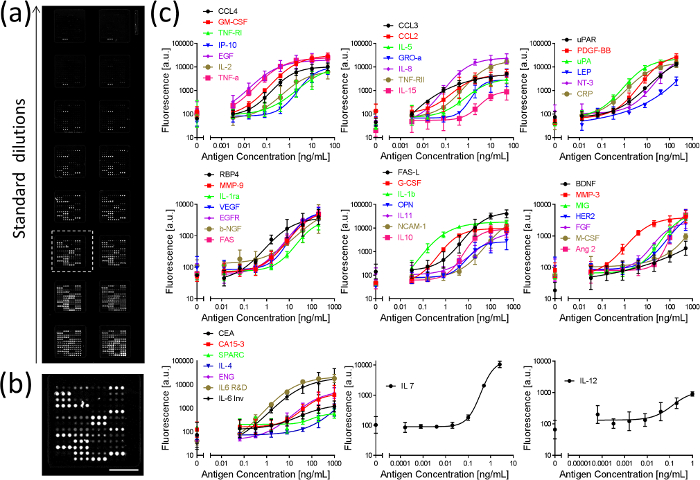
Figure 5. Fluorescence image of an assay slide and standard curves of 50 proteins measured in parallel using double transfer method. (a) Fluorescence image of an assay slide for generating a standard curve. (b) Close-up of an array. Scale bar = 2 mm. (c) Standard curves for 50 proteins. Error bars were calculated as standard deviations from three independent experiments. Adapted from reference 14 with permission. Please click here to view a larger version of this figure.
| Protein name | Starting concentration (ng/ml) | LOD (pg/ml) | Protein name | Starting concentration (ng/ml) | LOD (pg/ml) |
| ANG2 | 500 | 1.3 × 104 | IL-6b | 1 | 2.1 × 102 |
| BDNF | 500 | 1.0 × 102 | IL-5 | 50 | 77 |
| CA 15-3* | 1 | 4.9 × 103 | IL-4 | 1000 | 1.5 × 104 |
| CEA | 1000 | 5.4 × 103 | IL-2 | 50 | 76 |
| CXCL10/IP-10 | 50 | 1.7 × 102 | LEP | 200 | 4.0 × 102 |
| CRP | 200 | 44 | MIG | 500 | 11 × 102 |
| ENG | 1000 | 6.2 × 102 | CCL3/MIP-1α | 50 | 3.3 |
| EGF | 50 | 1.8 × 102 | CCL4/MIP-1β | 50 | 12 |
| EGFR | 200 | 1.9 × 102 | MMP-3 | 500 | 1.0 × 102 |
| FAS-L | 500 | 4.5 × 102 | M-CSF | 500 | 8.2 × 103 |
| FGF | 500 | 1.0 × 103 | MMP-9 | 200 | 3.1 × 102 |
| G-CSF | 500 | 39 | CCL2/MCP-1 | 50 | 55 |
| GM-CSF | 50 | 3.8 | NCAM-1 | 500 | 1.7 × 103 |
| GRO-α | 50 | 3.0 × 102 | β-NGF | 200 | 1.4 × 103 |
| HER2 | 500 | 3.5 × 103 | NT-3 | 200 | 5.8 × 102 |
| PDGF-BB | 200 | 73 | OPN | 500 | 1.9 × 103 |
| IL-1β | 500 | 7.9 × 102 | RBP4 | 200 | 1.2 ×102 |
| IL-1ra | 200 | 1.1 × 103 | SPARC | 1000 | 4.6 ×104 |
| IL-15 | 50 | 8.2 × 102 | TNF-α | 50 | 4.4 |
| IL-12 | 1 | 30 | TNF-RI | 50 | 1.3 × 102 |
| IL-11 | 500 | 1.2 × 103 | TNF-RII | 50 | 12 |
| IL-10 | 500 | 6.1 ×102 | FAS/TNFRSF6 | 200 | 2.8 ×102 |
| IL-8 | 50 | 6.6 | uPA | 200 | 24 |
| IL-7 | 2.5 | 26 | uPAR(CD87) | 200 | 76 |
| IL-6a | 1000 | 84 | VEGF | 200 | 6.7 × 102 |
Table 1. Protein concentrations and LODs in buffer from the 50-plex assay. The LOD for CA 15-3 is in U/ml (*). Adapted from Reference 14 with permission.
Discussion
In this work, we have presented a snap chip technology that makes the cross-reactivity-free multiplex immunoassays widely available for the researchers with basic experimental setup. Different from existing antibody microarrays, no microarray spotter is needed for end-users. Both single and double transfer methods are demonstrated, and double transfer affords superior alignment accuracy down to ~ 40 μm for 98% spots, with the largest misalignment of 63 µm14. A novel snap apparatus was developed to conveniently snap the two slides with consistent pressure, making this technology accessible to researchers. Reliable reagent transfer is realized not only on nitrocellulose-coated slides, but also on regular glass slides, significantly expanding the application of this technology without the need of extensive training. To demonstrate the usability of the snap chip, a 50-plex immunoassay was conducted using pre-spotted and stored slides, and 70% of the proteins achieved LODs in the range of pg/mL14. Compared to existing multiplex immunoassays that apply dAbs as a mixture, an important benefit of the ACM and the snap chip technology is that the assays on a chip are independent to each other, thus allowing for adding or removing any antibody pairs without interfering with any other pairs. Tens to hundreds of proteins can be measured simultaneously with a snap chip, thus shortening assay time compared to other methods such as the automated serial measurement by sequential incubation which requires serial application of each antibody 17. Only sub-nanoliter of each antibody is needed for the snap chip fabrication, making it more economic than conventional immunoassays.
The antibody concentrations used and the antigen incubation steps are critical to achieve high assay sensitivity. In the fabrication of the snap chips, antibody concentration can be optimized depending on the affinity of the antibody pairs and the types of the assay slides (nitrocellulose or non-nitrocellulose). The volumes of each antibody spot can be adjusted based on the spotter capacity and alignment accuracy, and the center-to-center spacing between the spots can be changed accordingly. After transferring cAbs to the assay slide, we incubated the assay slide at room temperature for 1 h. Overnight incubation at 4 °C might give better antibody binding capacity. BSA free stabilizer solutions (see the Table of materials) were used here to block the assay slide after incubating cAbs. Other blocking reagents such as animal serum or milk may also work for this application.
In the multiplexed immunoassays, protocols for non-nitrocellulose slides with different surface chemistry can be developed and optimized. A different dilution factor can be used to make standard curves depending on the target proteins. Different fluorophores can be used to label dAbs, and the slide can be scanned using a laser at a different wavelength for signal collection.
Currently, a density of 625 spots/cm2 has been achieved with double transfer method14. To further improve the multiplexing capability of the snap chip, several strategies are available. Firstly, smaller volumes can be used to reduce the size of the spots, thus allowing for shorter center-to-center distance between spots and increased array density. Secondly, one can choose a transfer slide with a more hydrophobic surface to reduce the spot size. Thirdly, better alignment between the two microarrays might be achieved by performing fine adjustment of the inkjet spotter, and further optimizing the spotting parameters such as the distance between nozzles and the slide surface.
As for all antibody-based immunoassays, the performance of the snap chip immunoassay is subject to the availability and quality of antibodies. The snap chip operates using a sandwich assay format, which is the most widely used assay format in ELISA owing to the high sensitivity and specificity afforded by the dual, sequential binding of a pair of antibodies targeting different epitopes, but it is dependent on the availability of a pair of compatible antibodies needed for capture and detection of the target protein.
The value of the snap chip technology is established for immunoassays, and it is expected that it may be used for any chemical and biochemical reactions that require applying different reagents without cross-contaminations18,19,20,21,22. Actually, the snap chip provides a general solution to delivering various pre-spotted reagents from microarray-to-microarray, thus disassociating the chip fabrication with the assay procedure, and therefore helps addressing the "macro-to-micro" interfacing challenge23. As an example, the snap chip has been used for a 4-plex homogeneous enzyme inhibition assay to analyze 128 conditions with precise timing, and comparable results to large volume experiments14. It has also been applied for multiplexed tissue staining. In addition, it can contribute to drug screening applications to test thousands of chemicals on different bacteria or cells, for single cell analysis21,24, or to test different conditions in single or multi-step chemical reactions.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank Dr. Rob Sladek for the use of the inkjet spotter. We acknowledge the final support from the Canadian Institutes for Health Research (CIHR), the Natural Science and Engineering Research Council of Canada (NSERC), the Canadian Cancers Society Research Institute and the Canada Foundation for Innovation (CFI). D.J. thanks support from a Canada Research Chair.
Materials
| Phosphate buffered saline tablet | Fisher Scientific | 5246501EA | |
| Streptavidin-conjugated Cy5 | Rockland | s000-06 | |
| Tween-20 | Sigma-Aldrich | p1379 | |
| Bovine serum albumin | Jackson ImmunoResearch Laboratories, Inc | 001-000-162 | |
| Glycerol | Sigma-Aldrich | G5516 | |
| Blocking solution: BSA-free StabilGuard Choice Microarray Stabilizer | SurModics, Inc | SG02 | |
| Nitrocellulose coated slides | Grace Bio-Laboratories, Inc | 305116 | |
| Aminosilane coated slides | Schott North America | 1064875 | |
| Snap Device | Parallex BioAssays Inc. | PBA-SD01 | |
| Inkjet microarray spotter | GeSiM | Nanoplotter 2.0 | |
| Slide module gasket | Grace Bio-Laboratories, Inc | 204862 | |
| Humidity Stabilization Beads | Parallex BioAssays Inc. | PBA-HU60 | |
| Array-Pro Analyzer software | Media Cybernetics | Version 4.5 | |
| Fluorescence microarray scanner | Agilent | SureScan Microarray Scanner | |
| Biostatistics software | GraphPad Software | GraphPad Prism 6 | |
| Endoglin capture antibody | R&D Systems | MAB10972 | |
| Endoglin protein | R&D Systems | 1097-EN | |
| Endoglin detection antibody | R&D Systems | BAF1097 | |
| IL-6a (see Table 1) | R&D Systems | ||
| IL-6b (see Table 1) | Invitrogen |
References
- Mor, G., et al. Serum protein markers for early detection of ovarian cancer. Proc. Natl. Acad. Sci. U.S.A. 102 (21), 7677-7682 (2005).
- Nicolini, A., et al. Intensive post-operative follow-up of breast cancer patients with tumour markers: CEA, TPA or CA15.3 vs MCA and MCA-CA15.3 vs CEA-TPA-CA15.3 panel in the early detection of distant metastases. BMC Cancer. 6 (1), 1-9 (2006).
- Hnasko, R., Lin, A., McGarvey, J. A., Stanker, L. H. A rapid method to improve protein detection by indirect ELISA. Biochem. Biophys. Res. Commun. 410 (4), 726-731 (2011).
- Percy, A. J., Chambers, A. G., Yang, J., Hardie, D. B., Borchers, C. H. Advances in multiplexed MRM-based protein biomarker quantitation toward clinical utility. Biochim. Biophys. Acta – Proteins and Proteomics. 1844 (5), 917-926 (2014).
- Ekins, R. P. Multi-analyte immunoassay. J Pharm Biomed Anal. 7 (2), 155-168 (1989).
- Mahlknecht, P., et al. An antibody microarray analysis of serum cytokines in neurodegenerative Parkinsonian syndromes. Proteome Sci. 10 (1), 71-80 (2012).
- Miller, J. C., et al. Antibody microarray profiling of human prostate cancer sera: Antibody screening and identification of potential biomarkers. Proteomics. 3 (1), 56-63 (2003).
- Li, H., Leulmi, R. F., Juncker, D. Hydrogel droplet microarrays with trapped antibody-functionalized beads for multiplexed protein analysis. Lab Chip. 11 (3), 528-534 (2011).
- Juncker, D., Bergeron, S., Laforte, V., Li, H. Cross-reactivity in antibody microarrays and multiplexed sandwich assays: shedding light on the dark side of multiplexing. Curr. Opin. Chem. Biol. 18, 29-37 (2014).
- Blank, K., et al. Double-chip protein arrays: force-based multiplex sandwich immunoassays with increased specificity. Anal. Bioanal. Chem. 379 (7), 974-981 (2004).
- Albrecht, C., et al. DNA: A Programmable Force Sensor. Science. 301 (5631), 367-370 (2003).
- Pla-Roca, M., et al. Antibody Colocalization Microarray: A Scalable Technology for Multiplex Protein Analysis in Complex Samples. Mol. Cell. proteomics. 11 (4), (2012).
- Li, H., Bergeron, S., Juncker, D. Microarray-to-Microarray Transfer of Reagents by Snapping of Two Chips for Cross-Reactivity-Free Multiplex Immunoassays. Anal. Chem. 84 (11), 4776-4783 (2012).
- Li, H., Munzar, J. D., Ng, A., Juncker, D. A versatile snap chip for high-density sub-nanoliter chip-to-chip reagent transfer. Sci. Rep. 5, 11688 (2015).
- Li, H., Bergeron, S., Annis, M. G., Siegel, P. M., Juncker, D. Serial analysis of 38 proteins during the progression of human breast tumor in mice using an antibody colocalization microarray. Mol. Cell. Proteomics. 14, 1024-1037 (2015).
- Bergeron, S., Laforte, V., Lo, P. S., Li, H., Juncker, D. Evaluating mixtures of 14 hygroscopic additives to improve antibody microarray performance. Anal. Bioanal. Chem. 407 (28), 8451-8462 (2015).
- Whiteaker, J. R., et al. Sequential Multiplexed Analyte Quantification Using Peptide Immunoaffinity Enrichment Coupled to Mass Spectrometry. Mol. Cell. Proteomics. 11 (6), (2012).
- Xu, K., Wang, X., Ford, R. M., Landers, J. P. Self-Partitioned Droplet Array on Laser-Patterned Superhydrophilic Glass Surface for Wall-less Cell Arrays. Anal. Chem. 88 (5), 2652-2658 (2016).
- Lee, M. -. Y., Park, C. B., Dordick, J. S., Clark, D. S. Metabolizing enzyme toxicology assay chip (MetaChip) for high-throughput microscale toxicity analyses. Proc. Natl. Acad. Sci. U.S.A. 102 (4), 983-987 (2005).
- Fernandes, T. G., et al. Three-dimensional cell culture microarray for high-throughput studies of stem cell fate. Biotechnol. Bioeng. 106 (1), 106-118 (2010).
- Kwon, C. H., et al. Drug-Eluting Microarrays for Cell-Based Screening of Chemical-Induced Apoptosis. Anal. Chem. 83 (11), 4118-4125 (2011).
- Jogia, G. E., Tronser, T., Popova, A. A., Levkin, P. A. Droplet Microarray Based on Superhydrophobic-Superhydrophilic Patterns for Single Cell Analysis. Microarrays. 5 (4), 28 (2016).
- Schena, M., Shalon, D., Davis, R. W., Brown, P. O. Quantitative Monitoring of Gene Expression Patterns with a Complementary DNA Microarray. Science. 270 (5235), 467-470 (1995).
- Wu, J., et al. A sandwiched microarray platform for benchtop cell-based high throughput screening. Biomaterials. 32 (3), 841-848 (2011).

