Hemocompatibility Testing of Blood-Contacting Implants in a Flow Loop Model Mimicking Human Blood Flow
Summary
This protocol describes a comprehensive hemocompatibility evaluation of blood-contacting devices using laser-cut neurovascular implants. A flow loop model with fresh, heparinized human blood is applied to mimic blood flow. After perfusion, various hematologic markers are analyzed and compared to the values gained directly after blood collection for hemocompatibility evaluation of the tested devices.
Abstract
The growing use of medical devices (e.g., vascular grafts, stents, and cardiac catheters) for temporary or permanent purposes that remain in the body's circulatory system demands a reliable and multiparametric approach that evaluates the possible hematologic complications caused by these devices (i.e., activation and destruction of blood components). Comprehensive in vitro hemocompatibility testing of blood-contacting implants is the first step towards successful in vivo implementation. Therefore, extensive analysis according to the International Organization for Standardization 10993-4 (ISO 10993-4) is mandatory prior to clinical application. The presented flow loop describes a sensitive model to analyze the hemostatic performance of stents (in this case, neurovascular) and reveal adverse effects. The use of fresh human whole blood and gentle blood sampling are essential to avoid the preactivation of blood. The blood is perfused through a heparinized tubing containing the test specimen by using a peristaltic pump at a rate of 150 mL/min at 37 °C for 60 min. Before and after perfusion, hematologic markers (i.e., blood cell count, hemoglobin, hematocrit, and plasmatic markers) indicating the activation of leukocytes (polymorphonuclear [PMN]-elastase), platelets (β-thromboglobulin [β-TG]), the coagulation system (thombin-antithrombin III [TAT]), and the complement cascade (SC5b-9) are analyzed. In conclusion, we present an essential and reliable model for extensive hemocompatibility testing of stents and other blood-contacting devices prior to clinical application.
Introduction
The in vivo application of implants and biomaterials, which interact with human blood, requires intense preclinical testing focusing on the investigation of various markers of the hemostatic system. The International Organization for Standardization 10993-4 (ISO 10993-4) specifies the central principles for the evaluation of blood-contacting devices (i.e., stents and vascular grafts) and considers the device design, clinical utility, and materials needed1.
Human blood is a fluid that contains various plasma proteins and cells, including leukocytes (white blood cells [WBCs]), erythrocytes (red blood cells [RBCs]), and platelets, which carry out complex functions in the human body2. The direct contact of foreign materials with blood can cause adverse effects, such as activation of the immune or coagulation system, which can lead to inflammation or thrombotic complications and serious issues after implantation3,4,5. Therefore, in vitro hemocompatibility validation offers an opportunity prior to implantation to detect and exclude any hematologic complications that may be induced upon contact of the blood with a foreign surface6.
The presented flow loop model was established to assess the hemocompatibility of neurovascular stents and similar devices by applying a flow rate of 150 mL/min in tubing (diameter of 3.2 mm) to mimic cerebral flow conditions and artery diameters2,7. Besides the need for an optimal in vitro model, the source of blood is an important factor in gaining reliable and unaltered results when analyzing hemocompatibility of a biomaterial8. The collected blood should be used immediately after sampling to prevent changes caused by prolonged storage. In general, a gentle collection of blood without stasis using a 21 G needle should be performed to minimize the preactivation of platelets and the coagulation cascade during blood drawing. Furthermore, donor exclusion criteria include those who smoke, are pregnant, are in a poor state of health, or have taken oral contraceptives or painkillers during the previous 14 days.
This study describes an in vitro model for the extensive hemocompatibility testing of stent implants under flow conditions. When comparing uncoated to fibrin-heparin-coated stents, results of the comprehensive hemocompatibility tests reflect improved hemocompatibility of the fibrin-heparin-coated stents9. In contrast, the uncoated stents induce activation of the coagulation cascade, as demonstrated by an increase in thombin-antithrombin III (TAT) concentrations and loss of blood platelet numbers due to the adhesion of platelets to stent surface. Overall, integrating this hemocompatibility model as a preclinical test is recommended to detect any adverse effects on the hemostatic system that are caused by the device.
Protocol
The blood sampling procedure was approved by the Ethics Committee of the medical faculty at the University of Tuebingen (project identification code: 270/2010BO1). All subjects provided written, informed consent for inclusion before participation.
1. Preparation of Heparin-loaded Monovettes
- Mix the undiluted heparin (5,000 IU/mL) with sodium chloride (NaCl, 0.9%) solution and prepare a solution with a resulting concentration of 15 IU/mL of heparin.
- Add 900 µL of the diluted heparin solution to each neutral monovette (9 mL) to obtain a final heparin concentration of 1.5 IU/mL after blood sampling. Prepare three monovettes per donor plus three reserve monovettes and store the heparin-loaded monovettes at 4 °C until blood sampling.
2. Blood Sampling
- Take the heparin-loaded monovettes out of the refrigerator 30 min prior to blood sampling.
- Collect a 27 mL blood sample from each healthy donor (n = 5) by venipuncture for the flow loop. Only apply a smooth tourniquet to avoid premature activation of the platelets and the blood clotting cascade.
- Collect blood samples in three monovettes containing 900 µL of the heparin solution (1.5 IU/mL) and pool all three monovettes in one plastic container to ensure that all components are evenly distributed.
- Directly transfer the pooled heparinized blood into three different monovettes containing either EDTA (1.2 mL), citrate (1.4 mL), or a mixture of citrate, theophylline, adenosine, and dipyridamole (CTAD, 2.7 mL) to collect baseline values. Proceed with the samples as described in sections 5−8.
NOTE: To guarantee uninfluenced clotting behavior, donors should avoid the intake of hemostasis-affecting drugs (e.g., acetylsalicylic acid, naproxen, and carbenicillin) within the last 14 days, as well as oral contraceptives and smoking.
3. Preparation of the Flow Loop
- Cut three heparin-coated polyvinyl chloride tubes with a length of 75 cm and inner diameter of 3.2 mm. Load the tubes with the neurovascular laser-cut implants with or without the fibrin-heparin coating. Remember to leave one tub unloaded as a control.
- Place one end of the tube in a reservoir filled with 0.9% NaCl, connect the tubing to the pump head, and insert the other end into a measuring cylinder.
- Adjust the settings of the peristaltic pump to achieve a flow rate of 150 mL/min by using a timer while checking the fill level in the measuring cylinder.
4. Performance of Hemocompatibility Testing
- Use a 12 mL syringe to fill the tubes with blood. Let 6 mL of blood flow smoothly into each tube containing a sample or unloaded control.
- Form a circuit and close the tubes tightly using a 0.5 cm length of silicone connection tubing. Place the tubes in a water bath of 37 °C and start the perfusion for 60 min.
5. Whole Blood Count Analysis
- Put 1.2 mL of blood after sampling (baseline) or after perfusion into a monovette containing EDTA and carefully invert the tube 5x.
- Insert the monovette into the blood analyzer and perform a blood count analysis for every sample. Then, incubate the monovettes on ice for 15−60 min after the blood count measurement for further analysis, as described in section 7.
6. Collection of Citrate Plasma
- Fill the monovettes containing citrate with 1.4 mL of blood (freshly drawn or after circulation) and carefully invert 5x.
- Centrifuge the tubes for 18 min at 1,800 x g at room temperature (RT). Aliquot three 250 µL samples of the plasma fraction into 1.5 mL reaction tubes and freeze the plasma samples in liquid nitrogen. Store them at -20 °C until analysis.
7. Collection of EDTA Plasma
- Incubate the monovettes on ice for 15−60 min after the blood count measurement. Then, centrifuge the tubes for 20 min at 2,500 x g and 4 °C.
- Aliquot three 250 µL samples of the plasma fraction into 1.5 mL reaction tubes after centrifugation and freeze the tubes in liquid nitrogen. Store them at -80 °C until analysis.
8. Collection of CTAD Plasma
- Fill the monovettes containing the CTAD mixture with 2.7 mL of blood (freshly drawn or after incubation) and carefully invert 5x. Afterwards, incubate the monovettes on ice for 15−60 min. Then, centrifuge the tubes for 20 min at 2,500 x g and 4 °C.
- Transfer 700 µL of the middle plasma fraction into a 1.5 mL reaction tube and centrifuge the filled reaction tubes for 20 min at 2,500 x g and 4 °C.
- Aliquot two 100 µL samples of the middle fraction into 1.5 mL reaction tubes after centrifugation and freeze the tubes in liquid nitrogen. Store them at -20 °C until analysis.
NOTE: The collection of EDTA plasma and CTAD plasma can be performed together because the operating conditions are the same.
9. Measurement of Human TAT from Citrate Plasma
- Thaw the citrate plasma in a water bath of 37 °C.
- Use the TAT enzyme-linked Immunosorbent assay (ELISA) kit according to the manufacturer's instructions. Reconstruct the plasma standards and control and dilute the washing solution, anti-human-TAT peroxidase (POD)-conjugated antibody, and chromogen solution. Leave all reagents and the microtiter plate at RT (15−25 °C) for 30 min before starting the test.
- Pipet 50 µL of the sample buffer into each well of the microtiter plate and add 50 µL of the sample buffer (blank), plasma standard, plasma control, and undiluted plasma sample in duplicates to the well plate. Seal the plate and incubate at 37 °C for 15 min with gentle shaking. Then, wash the plate 3x with 300 μL of washing solution.
- Add 100 µL of the POD-conjugated anti-human-TAT antibody to each well. Seal the plate and incubate at 37 °C for 15 min with gentle shaking. Then, wash the plate 3x with 300 μL of washing solution.
- Add 100 µL of the freshly prepared chromogen solution to each well. Seal the plate and incubate at RT for 30 min.
- Remove the seal film and add 100 µL of stop solution to each well. Read the optical density (OD) with a photometer at 490−500 nm. Fit the standard curve data as a trend line and calculate the concentration of the samples.
10. Measurement of PMN-elastase from Citrate Plasma
- Thaw the citrate plasma in a water bath at 37 °C.
- Use the polymorphonuclear (PMN)-elastase ELISA kit according the to manufacturer's instructions: reconstruct the PMN-elastase control and the PMN-elastase standard to prepare a standard curve using the kit's dilution buffer.
- Dilute the washing solution according to the manufacturer's description. Leave all reagents and the microtiter plate at RT for 30 min before starting the test. Dilute the citrate plasma samples to 1:100 with the dilution buffer.
- Add 100 µL of the sample buffer (blank), PMN-elastase standard curve (15.6−1,000 ng/ mL), PMN-elastase controls (high and low concentrations), and diluted plasma samples in duplicates to the well plate. Seal the plate and incubate at RT for 60 min with gentle shaking. Afterwards, wash the plate 4x with 300 μL of washing solution.
- Add 150 µL of the enzyme-conjugated antibody to each well. Seal the plate and incubate at RT for 60 min with gentle shaking. Afterwards, wash the plate 4x with 300 μL of washing solution.
- Add 200 µL of the 3,3',5,5'-tetramethylbenzidin (TMB)-substrate solution to each well. Seal the plate and incubate at RT for 20 min in the dark. Then, remove the seal film and add 50 µL of the stop solution to each well.
- Read the OD with a photometer at 450 nm with a reference reading at 630 nm. Fit the standard curve data as a trend line and calculate the concentration of the samples.
11. Measurement of Terminal Complement Complex (TCC) from EDTA Plasma
- Thaw the EDTA plasma in a water bath at 37 °C, and store on ice after defrosting.
- Use the complement cascade SC5b-9 ELISA kit according to the manufacturer's instructions: dilute the washing solution as described in the manufacturer's protocol. Leave all reagents and the microtiter plate at RT for 30 min before starting the test. Dilute the EDTA plasma samples to 1:10 with the kit's dilution buffer.
- Add 300 µL of washing solution to each well to rehydrate the surface and aspirate after 2 min. Add 100 µL of the sample buffer (blank), SC5b-9 standards, SC5b-9 controls (high and low concentrations), and the diluted plasma samples in duplicates to the well plate.
- Seal the plate and incubate at RT for 60 min. Next, wash the plate 5x with 300 μL of washing solution.
- Add 50 µL of the enzyme-conjugated antibody to each well. Seal the plate and incubate at RT for 30 min. Then, wash the plate 5x with 300 μL of washing solution.
- Add 100 µL of the TMB-substrate solution to each well. Seal the plate and incubate at RT for 15 min in the dark.
- Remove the seal film and add 100 µL of the stop solution to each well. Read the OD with a photometer at 450 nm. Fit the standard curve data as a trend line and calculate the concentration of the samples.
12. Measurement of β-thromboglobulin from CTAD Plasma
- Thaw the CTAD plasma in a water bath at 37 °C.
- Use the β-thromboglobulin (β-TG) ELISA kit according to the manufacturer's instructions: reconstruct the β-TG control and the β-TG standard and dilute the washing solution using distilled H2O. Reconstruct the POD-conjugated antibody using the provided phosphate buffer. Leave all reagents and the microtiter plate at RT for 30 min before starting the test.
- Prepare the standard curve and the control according to the manufacturer's instructions with the provided phosphate buffer. Dilute the CTAD plasma samples to 1:21.
- Add 200 µL of the phosphate buffer (blank), β-TG standards, β-TG controls (high and low concentrations), and diluted plasma samples in duplicates to the well plate. Seal the plate and incubate at RT for 60 min. Afterwards, wash the plate 5x with 300 μL of washing solution.
- Add 200 µL of the enzyme-conjugated antibody to each well. Seal the plate and incubate at RT for 60 min. Afterwards, wash the plate 5x with 300 μL of washing solution.
- Add 200 µL of the TMB-substrate solution to each well. Seal the plate and incubate at RT for 5 min in the dark. Remove the seal film and stop the reaction by adding 50 µL of 1 M sulfuric acid (H2SO4) to each well.
- Leave the plate for 15−60 min, then read the OD with a photometer at 450 nm. Fit the standard curve data as a trend line and calculate the concentration of the samples.
13. Sample Preparation for Scanning Electron Microscopy
- Remove the implant from the tube using forceps and rinse the implant briefly by dipping it into 0.9% NaCl solution 3x.
- Store in glutaraldehyde solution (2% glutaraldehyde in phosphate-buffered saline [PBS-buffer without Ca2+/Mg2+]) overnight at 4 °C.
- Next, incubate the implants in PBS-buffer for 10 min. Dehydrate the samples by incubating in ethanol with increasing concentration for 10 min each: 40%, 50%, 60%, 70%, 80%, 90%, 96%, and 100%. Store the dehydrated samples in 100% ethanol until further analysis.
- Perform critical point drying according to the instructions of the drying device or literature10 just before scanning electron microscopy (SEM).
14. Scanning Electron Microscopy
- Attach the dried implants to a sample carrier for the scanning microscope and sputter the samples with gold palladium.
- Introduce the sputtered implants into the sample chamber. Take pictures in 100-, 500-, 1,000- and 2,500-fold magnification of the areas with the representative surface and cell adhesion.
Representative Results
Briefly summarized, human whole blood was collected in heparin-loaded monovettes then pooled and used to evaluate the baseline levels of cell counts as well as plasmatic hemocompatibility markers.
Subsequently, the tubing containing the neurovascular implant samples was filled, and the blood was perfused for 60 min at 150 mL/min and 37 °C using a peristaltic pump. Again, the number of cells was analyzed in all groups, and the plasma samples were prepared for ELISA analyses (Figure 1). The quantification of the blood cells and blood parameters, such as hemoglobin and hematocrit, was performed directly after blood collection as well as after perfusion in the flow loop model for all sample types and the control. No changes were detected regarding the number of WBCs (Figure 2A), RBCs (Figure 2B), or the hematocrit values (Figure 2C). However, a decrease in hemoglobin levels was detected after the incubation of blood in the flow loop model when compared to the baseline values, which was due to the perfusion of blood in the flow loop system (Figure 2D). In addition, a decrease in platelet numbers was observed due to blood perfusion. Furthermore, this effect was increased when an uncoated stent was present in the tubing, indicating the adhesion of platelets to the biomaterial. Nonetheless, it was clearly demonstrated that the loss of platelets was significantly higher when the uncoated stent was incubated with blood, as opposed to the fibrin-heparin-coated stent (Figure 2E).
Potential alterations of the hematologic plasma markers were also investigated in the test groups after perfusion and compared to the baseline values of the freshly drawn blood. The TAT complex concentration, which reflects the activation status of the coagulation system, was mildly increased due to blood perfusion (Figure 3A). In the bare metal stent group, however, a significant increase in the TAT was detected, indicating a profound activation of the coagulation system. The fibrin-heparin-coated stent prevented the activation of the coagulation system, since no increase in the TAT was determined.
The perfusion led to an increased activation of the complement cascade, which was determined by measuring SC5b-9 (Figure 3B). However, incubation with uncoated or fibrin-heparin-coated stents did not further increase the SC5b-9 concentration. Similar results were obtained when analyzing the activation of the neutrophil granulocytes through the quantification of PMN-elastase concentrations (Figure 3C).
Visualization of the stent surface was performed using SEM. Clear differences between the two stent groups were detected after blood incubation. While on the surface of the uncoated stent a dense network of blood cells and proteins was present, no adhesion of proteins or cells was detected on the surface of the fibrin-heparin-coated stent (Figure 4).
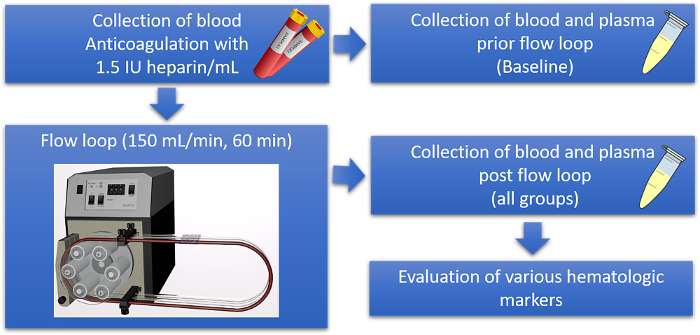
Figure 1: Schematic overview of the hemocompatibility evaluation of stents in a well-established flow loop model. Fresh human whole blood is collected from healthy donors in blood tubes containing heparin for anticoagulation. For each donor, an empty tube as well as tubes preloaded with the sample material are subsequently filled with fresh blood and incubated in the flow loop at a rate of 150 mL/min at 37 °C for 60 min. Additionally, plasma samples are prepared from freshly drawn blood to obtain the baseline values of each donor. After the incubation, the plasma samples from the test tubing, with and without sample materials, are prepared and analyzed using a specific ELISA. Please click here to view a larger version of this figure.
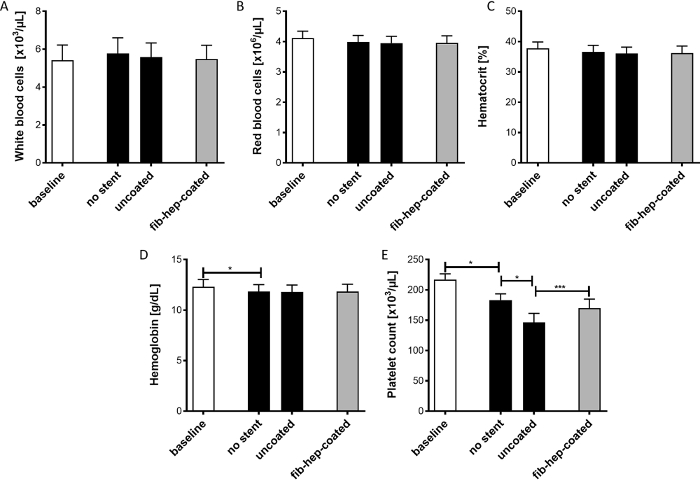
Figure 2: Analysis of different cell types and blood parameters before and after incubation of different stent implants in the flow loop model. The determination of white blood cells (A), red blood cells (B), hematocrit (C), hemoglobin (D), and platelets (E) was performed. The data are displayed as mean ± SEM (n = 5, p* < 0.5, p*** < 0.001). Please click here to view a larger version of this figure.
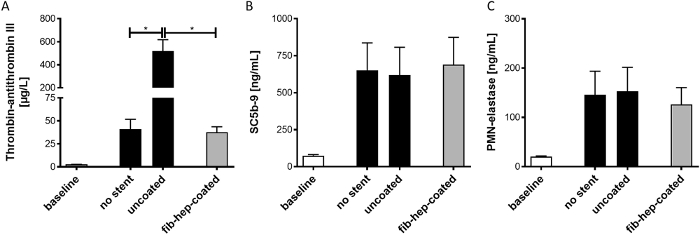
Figure 3: Determination of platelet or immune system activation markers before and after incubation with neurovascular implants. The markers for the (A) activation of blood coagulation (TAT), (B) complement system (SC5b-9), and (C) neutrophils (PMN-elastase) were quantified using ELISA. The analysis was performed on plasma samples gained from freshly drawn blood or blood incubated with different stents in the flow loop model. The data are displayed as mean ± SEM (n = 5, p* < 0.5). Please click here to view a larger version of this figure.
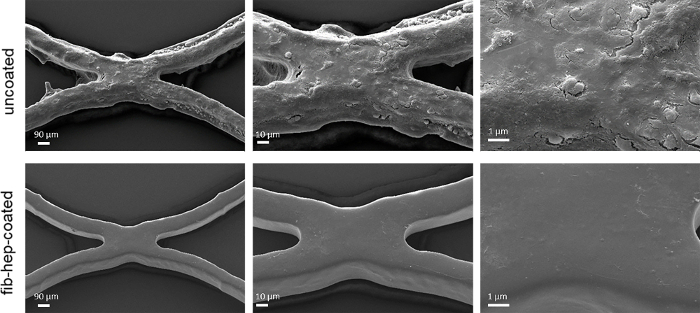
Figure 4: Scanning electron microscopic analysis of stents after incubation with blood. The aggregation of blood plasma proteins and platelets on uncoated stent material was observed. In comparison, stent materials with the fibrin-heparin coating did not demonstrate adhesion of cells or other blood components on the surface (magnification of 500-, 1,000-, and 2,500-fold). Please click here to view a larger version of this figure.
Discussion
The presented protocol describes a comprehensive and reliable method for the hemocompatibility testing of blood-contacting implants in accordance with ISO 10993-4 in a shear flow model imitating human blood flow. This study is based on the testing of laser-cut neurovascular implants but can be performed with a variety of samples. The results demonstrate that this method enables the broad analysis of various parameters such as the blood cell count, prevalence of several hemocompatibility markers, and microscopic visualization of the device surface after blood contact. Using this protocol, potential differences regarding the hemocompatibility of different devices can be detected.
An alternative to in vitro hemocompatibility assessment consists of in vivo animal testing, which is associated with several disadvantages, such as higher variability and distortion of device-related effects due to the overwhelming short-term effects of tissue injury6.
For in vitro hemocompatibility testing, three types of models are available: (1) static blood incubation models, (2) agitated blood incubation models, and (3) shear flow models. The static model provides a simple and rapid method to determine thrombogenicity by incubating the device directly with blood, but it only leads to rudimentary results regarding hemocompatibility11. To overcome the main disadvantages of static models (i.e., sedimentation of blood cells and the large air-contacting surface), the agitating blood incubation model may be used, in which a test chamber containing the implants is filled with blood and incubated on a rocking platform12. However, these model types are still inferior compared to the existing shear flow models, such as the flow loop presented here. The quintessence of these models is that vascular human blood flow can be imitated; thus, a close depiction of the real interaction between the implant and blood cells can be displayed13. In addition to the flow loop model, models such as the Chandler loop or several perfused flow chamber exist14,15,16.
The Chandler loop is a closed tube system that is partly filled with air and clamped into a rotating device, resulting in blood circulation through the tubing17. In the present flow loop system, the tube is completely filled with blood, and the flow is forced by using a peristaltic pump. When using the Chandler loop model, operators face two major disadvantages due to the requirement of including air into the test tubing. First, it is known that the constant interaction of blood and air triggers the aggregation of leukocytes and platelets as well as protein denaturation18,19. Second, the blood circulation rate is limited, because the air always remains at the highest point of the loop20.
These drawbacks can be overcome when using the flow loop system. Since no air-liquid interface is present in the system, no platelet activation occurs. Thus, the model has a low background for thrombotic events so that a low concentration of anticoagulants, typically 1 IU/mL or 1.5 IU/mL of heparin, is sufficient to prevent clotting, even if high flow rates are applied6. The adjustable pump-regulated blood flow rate and the freely selectable tube diameter allow the operator to mimic the physiological conditions of a vein or artery, which correspond to the implant to be tested, and achieve relevant test results21. However, this advantage is at the same time a limitation, due to the mechanical stress applied to the blood through the pump, and the destruction of erythrocytes (i.e., hemolysis) may occur2. This arising intrinsic blood damage reduces the method sensitivity and impedes prolonged exposure to the blood21. Nevertheless, several studies have demonstrated the effective use of the flow loop model for hemocompatibility evaluation22,23,24.
However, the main gap between all in vitro models and the in vivo mechanisms includes the missing endothelium, which expresses cytokines, anti-thrombotic components, and adhesion molecules; therefore, this component plays a crucial role in the interaction of the implant and circulating blood25. In conclusion, the flow loop model is adjustable, efficient, reliable, and cost-effective to assess the hemocompatibility of implants before clinical use.
Disclosures
The authors have nothing to disclose.
Acknowledgements
For the performance of scanning electron microscopy, we thank Ernst Schweizer from the section of Medical Materials Science and Technology of the University Hospital Tuebingen. The research was supported by the Ministry of Education, Youth and Sports of the CR within National Sustainability Program II (Project BIOCEV-FAR LQ1604) and by Czech Science Foundation project No. 18-01163S.
Materials
| aqua ad iniectabilia | Fresenius-Kabi, Bad-Homburg, Germany | 1088813 | |
| beta-TG ELISA | Diagnostica Stago, Duesseldorf, Germany | 00950 | |
| Centrifuge Rotana 460 R | Andreas Hettich, Tuttlingen, Germany | – | |
| Citrat monovettes (1.4 mL) | Sarstedt, Nümbrecht, Germany | 6,16,68,001 | |
| CTAD monovettes (2.7 mL) | BD Biosciences, Heidelberg, Germany | 367562 | |
| EDTA monovettes (1.2 mL) | Sarstedt, Nümbrecht, Germany | 6,16,62,001 | |
| Ethanol p.A. (1000 mL) | AppliChem, Darmstadt, Germany | 1,31,08,61,611 | |
| Glutaraldehyde (25 % in water) | SERVA Electrophoresis, Heidelberg, Germany | 23114.01 | |
| Heparin coating for tubes | Ension, Pittsburgh, USA | – | |
| Heparin-Natrium (25.000 IE/ 5 mL) | LEO Pharma, Neu-Isenburg, Germany | PZN 15261203 | |
| Multiplate Reader Mithras LB 940 | Berthold, Bad Wildbad, Germany | – | |
| NaCl 0,9% | Fresenius-Kabi, Bad-Homburg, Germany | 1312813 | |
| Neutral monovettes (9 mL) | Sarstedt, Nümbrecht, Germany | 2,10,63,001 | |
| PBS buffer (w/o Ca2+/Mg2+) | Thermo Fisher Scientific, Darmstadt, Germany | 70011044 | |
| Peristaltic pump ISM444B | Cole Parmer, Wertheim, Germany | 3475 | |
| Pipette (100 µL) | Eppendorf, Wesseling-Berzdorf, Germany | 3124000075 | |
| Pipette (1000 µL) | Eppendorf, Wesseling-Berzdorf, Germany | 3123000063 | |
| Plastic container (100 mL) | Sarstedt, Nümbrecht, Germany | 7,55,62,300 | |
| PMN-Elastase ELISA | Demeditec Diagnostics, Kiel Germany | DEH3311 | |
| Polyvinyl chloride tube | Saint-Gobain Performance Plastics Inc., Courbevoie France | – | |
| Reaction Tubes (1.5 mL) | Eppendorf, Wesseling-Berzdorf, Germany | 30123328 | |
| neurovascular laser-cut implants | Acandis GmbH, Pforzheim | 01-0011x | |
| SC5b-9 ELISA | TECOmedical, Buende, Germany | A029 | |
| Scanning electron microscope | Cambridge Instruments, Cambridge, UK | – | |
| Sealing tape (96 well plate) | Thermo Fisher Scientific, Darmstadt, Germany | 15036 | |
| Syringe 10/12 mL Norm-Ject | Henke-Sass-Wolf, Tuttlingen, Germany | 10080010 | |
| TAT micro kit | Siemens Healthcare, Marburg, Germany | OWMG15 | |
| Waterbath Type 1083 | Gesellschaft für Labortechnik, Burgwedel, Germany | – |
References
- ISO. . Biological evaluation of medical devices. , (2002).
- Weber, M., et al. Blood-Contacting Biomaterials: In Vitro Evaluation of the Hemocompatibility. Frontiers in Bioengineering and Biotechnology. 6, 99 (2018).
- Li, Y., Boraschi, D. Endotoxin contamination: a key element in the interpretation of nanosafety studies. Nanomedicine (Lond). 11 (3), 269-287 (2016).
- Cattaneo, G., et al. In vitro investigation of chemical properties and biocompatibility of neurovascular braided implants. Journal of Materials Science: Materials in Medicine. 30 (6), 67 (2019).
- Stang, K., et al. Hemocompatibility testing according to ISO 10993-4: discrimination between pyrogen- and device-induced hemostatic activation. Materials Science and Engineering: C Materials for Biological Applications. 42, 422-428 (2014).
- van Oeveren, W. Obstacles in haemocompatibility testing. Scientifica (Cairo). , 392584 (2013).
- Engels, G. E., Blok, S. L., van Oeveren, W. In vitro blood flow model with physiological wall shear stress for hemocompatibility testing-An example of coronary stent testing. Biointerphases. 11 (3), 031004 (2016).
- Blok, S. L., Engels, G. E., van Oeveren, W. In vitro hemocompatibility testing: The importance of fresh blood. Biointerphases. 11 (2), 029802 (2016).
- Kaplan, O., et al. Low-thrombogenic fibrin-heparin coating promotes in vitro endothelialization. Journal of Biomedical Materials Research Part A. 105 (11), 2995-3005 (2017).
- . SEM Imaging of Biological Samples Available from: https://www.jove.com/science-education/10492/sem-imaging-of-biological-samples (2019)
- Mohan, C. C., Chennazhi, K. P., Menon, D. In vitro hemocompatibility and vascular endothelial cell functionality on titania nanostructures under static and dynamic conditions for improved coronary stenting applications. Acta Biomaterialia. 9 (12), 9568-9577 (2013).
- Streller, U., Sperling, C., Hubner, J., Hanke, R., Werner, C. Design and evaluation of novel blood incubation systems for in vitro hemocompatibility assessment of planar solid surfaces. The Journal of Biomedical Materials Research Part B: Applied Biomaterials. 66 (1), 379-390 (2003).
- Sanak, M., Jakieła, B., Węgrzyn, W. Assessment of hemocompatibility of materials with arterial blood flow by platelet functional tests. Bulletin of the Polish Academy of Sciences: Technical Sciences. 58 (2), 317-322 (2010).
- Krajewski, S., et al. Hemocompatibility evaluation of different silver nanoparticle concentrations employing a modified Chandler-loop in vitro assay on human blood. Acta Biomaterialia. 9 (7), 7460-7468 (2013).
- Podias, A., Groth, T., Missirlis, Y. The effect of shear rate on the adhesion/activation of human platelets in flow through a closed-loop polymeric tubular system. Journal of Biomaterials Science, Polymer Edition. 6 (5), 399-410 (1994).
- Van Kruchten, R., Cosemans, J. M., Heemskerk, J. W. Measurement of whole blood thrombus formation using parallel-plate flow chambers-a practical guide. Platelets. 23 (3), 229-242 (2012).
- Müller, M., Krolitzki, B., Glasmacher, B. Dynamic in vitro hemocompatibility testing-improving the signal to noise ratio. Biomedical Engineering/Biomedizinische Technik. 57, 549-552 (2012).
- Ritz-Timme, S., Eckelt, N., Schmidtke, E., Thomsen, H. Genesis and diagnostic value of leukocyte and platelet accumulations around “air bubbles” in blood after venous air embolism. International Journal of Legal Medicine. 111 (1), 22-26 (1998).
- Miller, R., et al. Characterisation of the initial period of protein adsorption by dynamic surface tension measurements using different drop techniques. Colloids and Surfaces A: Physicochemical and Engineering Aspects. 131 (1), 225-230 (1998).
- van Oeveren, W., Tielliu, I. F., de Hart, J. Comparison of modified chandler, roller pump, and ball valve circulation models for in vitro testing in high blood flow conditions: application in thrombogenicity testing of different materials for vascular applications. International Journal of Biomaterials. , 673163 (2012).
- Krajewski, S., et al. Preclinical evaluation of the thrombogenicity and endothelialization of bare metal and surface-coated neurovascular stents. AJNR American Journal of Neuroradiology. 36 (1), 133-139 (2015).
- Monnink, S. H., et al. Silicon-carbide coated coronary stents have low platelet and leukocyte adhesion during platelet activation. Journal of Investigative Medicine. 47 (6), 304-310 (1999).
- Amoroso, G., van Boven, A. J., Volkers, C., Crijns, H. J., van Oeveren, W. Multilink stent promotes less platelet and leukocyte adhesion than a traditional stainless steel stent: an in vitro experimental study. Journal of Investigative Medicine. 49 (3), 265-272 (2001).
- Mulvihill, J., Crost, T., Renaux, J. L., Cazenave, J. P. Evaluation of haemodialysis membrane biocompatibility by parallel assessment in an ex vivo model in healthy volunteers. Nephrology Dialysis Transplantation. 12 (9), 1968-1973 (1997).
- Nordling, S., Nilsson, B., Magnusson, P. U. A novel in vitro model for studying the interactions between human whole blood and endothelium. Journal of Visualized Experiments. (93), e52112 (2014).

