A Modified Two Kidney One Clip Mouse Model of Renin Regulation in Renal Artery Stenosis
Summary
A modified 2 kidney 1 clip (2K1C) Goldblatt mouse model was developed using polyurethane tubing to initiate renal artery stenosis, inducing an increase in renin expression and kidney injury. Here, we describe a detailed procedure of preparing and placing the cuff onto the renal artery to generate a reproducible and consistent 2K1C mouse model.
Abstract
Renal artery stenosis is a common condition in patients with coronary or peripheral vascular disease where the renin angiotensin aldosterone system (RAAS) is overactivated. In this context, there is a narrowing of the renal arteries that stimulate an increase in the expression and release of renin, the rate-limiting protease in RAAS. The resulting rise in renin expression is a known driver of renovascular hypertension, frequently associated with kidney injury and end organ damage. Thus, there is a great interest in developing novel treatments for this condition. The molecular and cellular mechanism of renin control in renal artery stenosis is not fully understood and warrants further investigation. To induce renal artery stenosis in mice, a modified 2 kidney 1 clip (2K1C) Goldblatt mouse model was developed. The right kidney was stenosed in wild type mice and sham operated mice were used as control. After renal artery stenosis, we determined renin expression and kidney injury. Kidneys were harvested, and fresh cortices were used to determine protein and mRNA expression of renin. This animal model is reproducible and can be used to study pathophysiological responses, molecular and cellular pathways involved in renovascular hypertension and kidney injury.
Introduction
Renal artery stenosis (RAStenosis) is an intractable problem affecting about 6% of people over 65 and in up to 40% of people with coronary or peripheral vascular disease1,2. Current treatments for the disease are limited; therefore, there is a critical need to develop new therapies to treat renovascular hypertension or resistant hypertension induced by RAStenosis. Renin angiotensin aldosterone system (RAAS) is the key pathway involved in the pathogenesis of RAStenosis induced hypertension or renovascular hypertension3,4. Known therapies targeting RAAS, such as ACE inhibitors or angiotensin receptor blockers, alleviate hypertension, but need close examining for kidney failure and hyperkalemia5,6,7. Renin catalyzes the rate-limiting step in RAAS; it converts angiotensinogen to angiotensin I. In atherosclerosis, plaque formation causes the narrowing of renal artery that drives renin secretion, resulting in renovascular hypertension and kidney damage8. A number of studies have reported increased levels of oxidative stress during renovascular hypertension in humans, which were corroborated with the two kidney one clip (2K1C) mice model as well as other hypertensive animal models2,9,10,11,12,13,14,15,16. The molecular mechanism of renin expression control during RAStenosis induced renovascular hypertension is not well understood and warrants further investigation.
Experimental animal models that reliably and reproducibly recapitulate RAStenosis are important in elucidating the cellular and molecular mechanisms of renin expression control for the development of novel therapies. The 2K1C mouse model is a well-established experimental model to study the pathogenesis of renovascular hypertension17,18,19,20. This model is generated by the constriction of the renal artery using a clip17,20,21, therefore producing renal artery occlusion that results in an increase in renin expression and hypertension17,19,20,21. However, there are no technical reports available, which describe a step by step procedure to generate renal artery stenosis in animal models.
Conventional U-shaped silver clips, polyurethane tubes and other clips have been used to constrict the renal artery to induce renal artery stenosis. Some studies have shown that the design and material of the clip are critical to obtaining reliable and reproducible data with the 2K1C animal model. According to Lorenz et al., the use of conventional U-designed silver clips induces a low success rate of hypertension (40-60%)21. Due to the clip design, the renal artery is press laterally, triggering a few constrictions and greater probability to be dislodged from the renal artery. Silver malleability and ductility may allow changes in clip widths; therefore, causing different hypertension levels among mice. Silver dioxides on the clip can cause perivascular inflammation, intimal proliferation, and tissue granulation, altering the renal artery diameter22. Due to the variability in the levels of hypertension obtained with the conventional U-design silver clip, Warner et al. and Lorenz et al. have successfully used a rounder-design polyurethane tubing to initiate renal artery stenosis in mice, generating a more reliable and consistent induction of the two kidney one clip animal model20,21.
In this report, we describe a surgical protocol to generate experimental RAStenosis in mice, using the polyurethane tubing to constrict the renal artery. The polyurethane round-design cuff is a more reproducible, reliable and low-cost clip to generate stenosis in mouse. The goal of this experimental model is to study and define the molecular and cellular mechanism of renin expression control during renal artery stenosis. We confirmed the success of RAStenosis mice model by measuring renin expression and kidney injury marker neutrophil gelatinase-associated lipocalin (N-GAL).
Protocol
Mice were housed and cared at the Vanderbilt University Medical Center (VUMC) Division of Animal Care following the National Institutes of Health (NIH) guidelines and the Guide for the Care and Use of Laboratory Animals, US Department of Health and Human Services. All animal procedures were approved by the VUMC Institutional Animal Care and Use Committee prior to starting the experiments.
1. Animal preparation and dissection
- Turn on the germinator and water pump of the heating pad about 30 min before starting surgery.
- Cut 0.5 mm length polyurethane tubing with a sharp scalpel. Remove 0.2 mm of the circumference by making a cut lengthwise to make a cuff.
NOTE: This is a critical part of the renal artery stenosis procedure that requires extreme precision, attention to details and patience. Try to cut several pieces of polyurethane tubing at a time. Perform all of this procedure under microscope. - Before proceeding any further, put on surgical sterile gloves and a surgical mask.
- Use C57BL/6 wild-type (WT) mice of 6-8 weeks. Use an equal number of male and female mice to avoid any sex bias.
- Record the weight of the mice before performing surgery. The ideal weight to perform renal artery stenosis with polyurethane tube is 18-22 g. Handle mice gently and do not agitate while injecting the anesthesia. Administer pre-operative analgesia (Ketofgen, 5 mg/kg body wieght).
NOTE: While transferring the mice from their housing room to the surgery room, carry them with great gentleness and care to avoid agitation. Carrying mice cages in hands instead of a trolley is highly recommended. - Anesthetize the mice with a mixture of ketamine (100 mg/kg BW) and xylazine (10 mg/kg BW) via intraperitoneal (I.P.) injections.
- Place the mouse back inside the cage until fully anesthetized. It takes about 4-5 minutes before the mouse is fully unconscious. Pinch the tail or toe with forceps to check if the mouse is fully unconscious and ready for the surgery.
- Lay the mouse on its back on a paper towel. Remove the hair of the lateral abdomen using an electric hair clipper following the opposite direction of hair growth. After shaving the surgical site, clean the area with a surgical scrub containing iodine (or chlorhexidine) followed by a rinse with 70% ethanol (or sterile saline). Repeat 3 times.
NOTE: The hair removal procedure must be performed at some distance or preferably at a different bench than the surgery procedure bench to avoid any hair interference and hair contamination during the surgery procedure. - Cover the heating pad with a sterile sheet. Bring the mouse to the surgical bench and place on to the sterile sheet, facing the mouse’s right lateral side towards the microscope. Maintain a constant pad temperature of 37 °C with circulating water.
- Open the sterilized bag containing all the surgical equipment. Using a dissecting microscope and sterile sharps scissors, make a small flank incision (close to 13th thoracic rib, the last rib in mouse) and about 0.5 cm away from vertebrae. Proceed along the lumbar vertebrae and make a 1 inch incision.
- Pull back the skin and muscle to expose the kidney.
- Clean and remove the surrounding fat using sterile cotton swabs to isolate the renal artery. Isolate the renal nerve from the renal artery using curved forceps.
- When performing the sham surgery, apply sutures to close the skin and apply antibiotic to the closed wound, then proceed to Post-operative care. If not, proceed with the following section to stenose the artery.
NOTE: Every experiment should have sham animals as controls of the surgical procedure. Sham animals consist of mice that have gone through the surgical procedure of exposing the renal artery without placing a cuff on it.
2. Right renal artery stenosis
- Place two nylon sutures under the right renal artery, make loose knots, and then place the cuff around the main renal artery approximately equidistant between the kidney and aorta bifurcation
- Close the cuff using the nylon sutures. Make four knots for each suture to avoid the probability of losing the sutures after the surgery.
- Close the incision in the muscle by applying a simple continuous suture.
- Make simple interrupted sutures to close the skin.
- Administer IACUC approved analgesics.
NOTE: Autoclave surgical tools before every use. If more than one surgery is being performed at once, wipe all used tools with a sterile alcohol gauge and place them in hot germinator for 15-30 s after every surgery. Change sterile gloves also for each mouse.
3. Post-operative care
- Return mice to their cage and leave the cage half on, half off, a circulating water heating pad for 2 – 3 hours. Add gel diet recovery food inside the cage.
- Administer painkiller (ketoprofen) intraperitoneally (dose: 5 mg/kg BW) the next day.
- Weigh the mice for the next two days; if some mice lose more than 20% of their weight consult with the veterinarian and decide if the animal needs to be euthanized following the appropriate IACUC authorized procedure.
- Monitor the mice daily to assess for redness, swelling, pain or infection.
- Seek veterinary consultation for postoperative complications in accordance with the institutional IACUC policies..
4. Tissue harvest
- Harvest tissue 3 weeks after surgical procedure. Record the weight of each mouse.
- Euthanize the mouse with an IACUC approved procedure.
- Place the mouse on a sterile platform in supine position to dissect.
- Secure and extend the limbs to limit movement.
- Thoroughly spray the mice with 70% ethanol.
- Make a midline incision to open the abdomen and chest area using sharp scissors.
- Pull back the skin and peritoneal wall.
- Carefully expose the heart and puncture the right ventricle and exsanguinate the mouse.
- Remove both kidneys using forceps. Kidneys are located on the back of the mice.
NOTE: Do not mix both kidneys. Be aware of stenosed, sham, and contralateral kidneys. - Remove the kidney capsule, clean them from any fat and record the weight of each kidney separately.
- Cut a longitudinal section of both kidneys and fixed in 4% PFA overnight at 4 °C to be later processed for paraffin embedding, to perform in situ hybridization (ISH) and immunohistochemistry (IHC). Follow ISH and IHC protocols as reported23,24.
- Isolate the cortex of the remaining kidney and flash freeze in liquid nitrogen to perform western blot. Store samples at -80 °C until analysis.
- Quantify renin, and N-GAL expressions with Western blot as described in literature23,24.
5. Statistics
- Use one-way or two-way ANOVA for experiments with three or more conditions followed by Bonferroni post-hoc tests for comparisons between individual groups. Consider a p-value equal or less than 0.05 significant. Use software (e.g., GraphPad Prism 8.2) to perform all statistical analysis.
Representative Results
Renal artery constriction increases renin expression in the stenosed kidney while repressing expression in the contralateral kidney. The two kidney one clip (2K1C) or Goldblatt model of stenosis induces increased renin expression and kidney injury. This is recognized as the best representative model of unilateral renal artery stenosis in humans.
Expression of renin and prorenin (precursor of renin) were measured using immunoblotting. The data show that renin and prorenin expression increased in the stenosed kidney comparing to contralateral and sham kidneys, suggesting that the cuff was constricting the renal artery causing changes in renal perfusion (Figure 1). To visualize the localization of renin expression, IHC was performed. IHC corroborated immunoblotting data showing increased expression of renin in the clipped kidney (Figure 2). Moreover, juxtaglomerular (JG) cells recruitment along the afferent arteriole was seen in the stenosed kidney (Figure 2). To investigate the effect on renin mRNA expression levels, ISH was performed. The ISH data suggest increased renin mRNA and JG cells recruitment in the stenosed kidney when compared to contralateral and sham kidneys (Figure 3).
Another characteristic of renal artery stenosis is the upregulation of kidney injury markers due to changes in kidney perfusion, superoxide production and hypertension2,25,26. Neutrophil gelatinase-associated lipocalin (NGAL) is a well characterized acute injury marker and is overexpressed during kidney injury27,28. Therefore, acute kidney injury marker NGAL was measured using immunoblotting. Immunoblotting data showed that N-GAL was highly upregulated in the stenosed kidney when compared to the contralateral and sham kidneys (Figure 4).
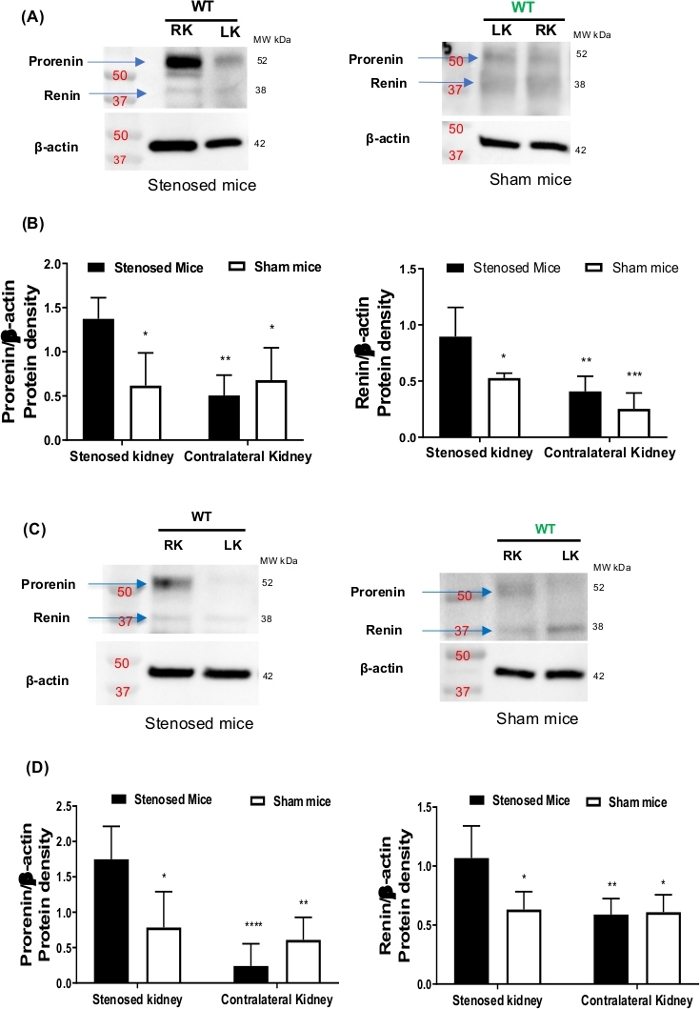
Figure 1: Renin expression. After 15- and 3-days of renal artery stenosis, mice were euthanized. Kidneys were harvested, and renin expression was determined by western blot. (A) is showing representative western blots images from 15-days stenosed (left panel) and sham mice (right panel). (B) is showing the densitometric analysis of prorenin (left panel) and renin (right panel) protein bands. Beta-actin was used as loading control. (C) is showing the representative western blots images from 3-day stenosed (left panel) and sham mice (right panel). (D) is showing densitometric analysis of prorenin (left panel) and renin (right panel) protein bands. Beta-actin was used as loading control. Data are presented as the mean ± SD. P value calculated with two-way ANOVA followed by Tukey post-hoc test. *P<0.05, **P< 0.01, ***P< 0.001, N=3-6. Please click here to view a larger version of this figure.
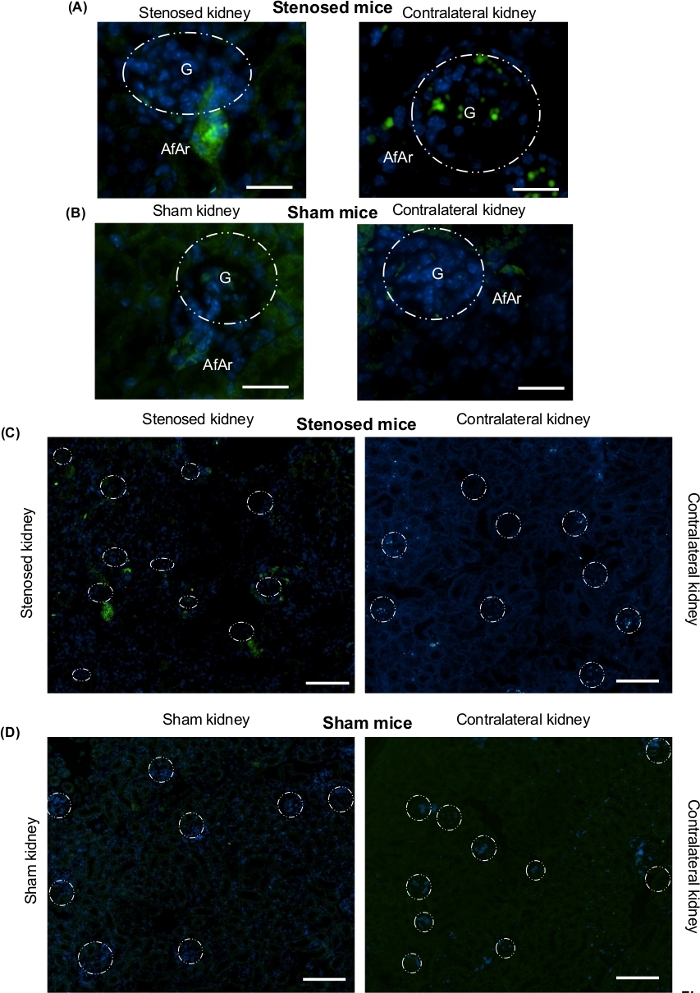
Figure 2: Immunohistochemistry analysis for visualization and localization of renin expression after renal artery stenosis. Kidneys were isolated after euthanizing mice from 3-day renal artery stenosis. One piece from longitudinal cut of the whole kidney was fixed with 4% neutral buffered formalin solution, after that dehydrated in a graduated ethanol series, and embedded in paraffin. Green staining represents renin protein expression; blue, nuclei. (A) Representative microscopy images of stenosed kidney (left side), contralateral kidney (right side) from stenosed mice. (B) Representative microscopy images of sham kidney (left side), and contralateral kidney (right side) from sham mice. Scale bar 30 microns. 90X magnification. (C) Representative microscopy images of stenosed kidney (left side), contralateral kidney (right side) from stenosed mice. (D) Representative microscopy images of sham kidney (left side), and contralateral kidney (right side) from sham mice. These images were mainly taken from cortex region. Scale bar 50 µm. 15x magnification. White dotted circles denote the location of glomeruli. G: Glomeruli, AfAr: Afferent arteriole, N=4. Please click here to view a larger version of this figure.
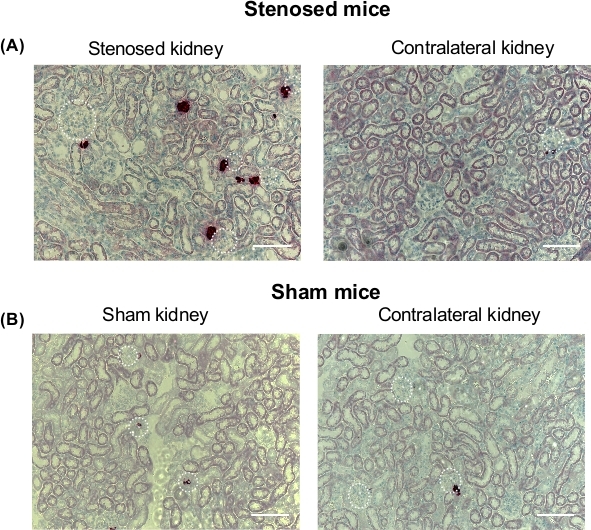
Figure 3: In situ hybridization analysis of renin mRNA expression after renal artery stenosis. After 3-days of renal artery stenosis, mice were euthanized and kidneys were isolated and perfusion-fixed with 4% neutral buffered formalin solution, dehydrated in a graduated ethanol series, and embedded in paraffin. In situ hybridization was performed following the manufacturer’s instructions. Dark red staining represents mRNA renin expression; blue, nuclei. (A). Representative microscopy image of stenosed kidneys (left side), and contralateral kidney (right side) from stenosed mice. (B). Representative microscopy image of sham kidney (left side), and contralateral kidney (right side) from sham mice. Scale bar 50 µm. White dotted circles denote glomeruli expressing renin. G: Glomeruli, AfAr: Afferent arteriole, N=4. Please click here to view a larger version of this figure.
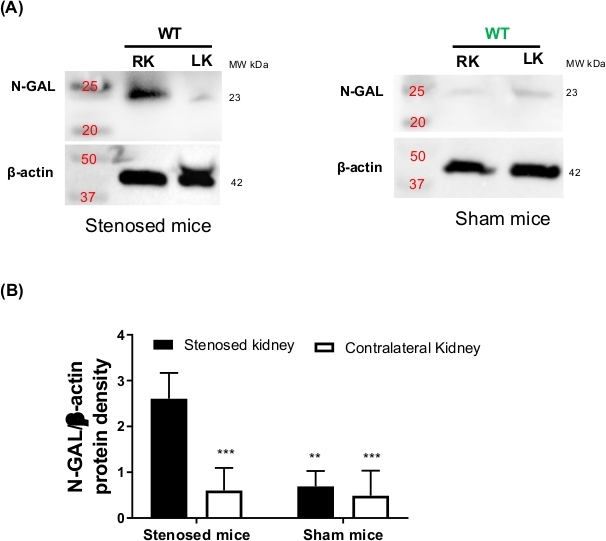
Figure 4: Neutrophil gelatinase-associated lipocalin (N-GAL) expression after renal artery stenosis. After 3-day renal artery stenosis, mice were euthanized and kidneys were harvested, and N-GAL expression measure by Western blot. (A) Representative Western blots images from 3-day stenosed (left panel) and sham mice (right panel). Beta-actin was used as loading control. (B) Densitometric analysis of N-GAL bands. Protein density values of N-GAL were normalized to β-actin. Data are presented as the mean ± SD. P value calculated with two-way ANOVA followed by Tukey post-hoc test. **P<0.01, ***P< 0.001, N=3-5. Please click here to view a larger version of this figure.
Discussion
Renal artery stenosis is an important cause of secondary or resistant hypertension, and kidney injury1,29. The two kidney one clip (2K1C) Goldblatt model has been employed to study RAStenosis induced renovascular hypertension1,17,18,19. A number of previous studies using various animals models have shown that stenosis in the renal artery is a strong stimulator of renin overexpression and release, and kidney injury18,30,31,32,33,34,35. Moreover, this model is used to study immune cell infiltration, fibrosis, inflammation, and acute and chronic kidney injury markers29.
Here, we described a detailed and step by step procedure to generate reproducible, reliable and consistent renal artery stenosis model in mice. Earlier, metal clips have been employed to initiate renal artery stenosis36,37,38. As an alternative, we used polyurethane round tubing (MRE 025; internal diameter (ID) = 0.30 mm; outside diameter (OD) = 0.63 mm; wall thickness, (WT) = 0.16 mm). We used tubing, since placement of a polyurethane cuff would result constriction in two dimensions (constriction) rather than one (flattening), as with a metal clip. Also, using polyurethane round tubing provides an advantage of uniform constriction in the renal artery. The critical step and challenges are to cut the right size of polyurethane tubing, which requires extreme attention to details that must be performed using a microscope. Another critical criterion is to keep mice between 18-22 g to fit the tubing onto the renal artery. Mice within this weight range normally have a renal artery outer diameter (OD) that is consistently within the range of the tubing cuff diameter. A limitation of the method is that heavy (above 25 g) or small (below 16 g) mice are difficult to perform surgery on because of the size of the tube and cuff made in it. However, when required, changes in the polyurethane tubing can be made to accommodate younger or older mice.
We have conducted 3-day and 15-day studies to initiate renal artery stenosis in mice with about 95% success rate. In our experience, induction of the renal artery stenosis using this method produced reliable, reproducible, and consistent results among the mice regardless of the sex. To confirm the constriction of renal artery, we measured renin expression and kidney injury. Our data suggest that renin expression significantly increased in the stenosed kidney.
Disclosures
The authors have nothing to disclose.
Acknowledgements
Research was supported by NHLBI Research Scientist Development Grant (1K01HL135461-01) to JAG. Thank you to David Carmona-Berrio, and Isabel Adarve-Rengifo for their technical assistance.
Materials
| Diet Gel | Clear H2O | Diet-Gel 76A | Surgery recovery diet |
| EMC Heated Hard pad | Hallowell | 000A2788B | Heating pads were used to keep mice warm |
| Ethilon Nylon Suture | Ethicon | 662G | 4-0 (1.5 metric), This suture was used to close the peritoneum, and skin |
| Ethilon Nylon Suture | Ethicon | 2815 G | 8-0 (0.4 metric), This suture was used to close cuff to tie and constrict the artery |
| Germinator 500 | Braintree Scientific Inc. | GER 5287 | Sterilize surgical tools between surgeries |
| Ketoprofen | Zoetis | Ketofen | Painkiller |
| Polyurethane | Braintree Scientific Inc. | MRE-025 | This tube was used to initiate stenosis |
| Povidone-iodine antiseptic swabsticks | Medline | MDS093901 | It was applied after hair removal and surgery on the skin |
| Reflex 7 Clip Applier | Roboz Surgical Instrument Co | 204-1000 | This clip applier was used to apply clip in case one or more sutures went off |
| Sterile towel drapes | Dynarex | 4410 | It was used as a bedsheet for mice during surgery |
| Triple antibiotic ointment | Medi-First | 22312 | |
| Water pump | Stryker | T/pump Professionals | Used to warm and circulate water in the heating hard pad to keep mice warm during and post-surgery |
References
- Kashyap, S., et al. Blockade of CCR2 reduces macrophage influx and development of chronic renal damage in murine renovascular hypertension. American Journal of Physiology-Renal Physiology. 310 (5), 372-384 (2016).
- Wang, W., et al. Changes in inflammatory biomarkers after renal revascularization in atherosclerotic renal artery stenosis. Nephrology Dialysis Transplantation. 31 (9), 1437-1443 (2016).
- Yerram, P., Karuparthi, P. R., Chaudhary, K. Pathogenesis and management of renovascular hypertension and ischemic nephropathy. Minerva Urologica e Nefrologica. 64 (1), 63-72 (2012).
- Covic, A., Gusbeth-Tatomir, P. The role of the renin-angiotensin-aldosterone system in renal artery stenosis, renovascular hypertension, and ischemic nephropathy: diagnostic implications. Progress in Cardiovascular Diseases. 52 (3), 204-208 (2009).
- Barreras, A., Gurk-Turner, C. Angiotensin II receptor blockers. Proceedings. 16 (1), 123-126 (2003).
- Sica, D. A. Angiotensin-converting enzyme inhibitors side effects–physiologic and non-physiologic considerations. Journal of Clinical Hypertension. 6 (7), 410-416 (2004).
- Hill, R. D., Vaidya, P. N. Angiotensin II Receptor Blockers (ARB, ARb). StatPearls. , (2019).
- Durante, A., et al. Role of the renin-angiotensin-aldosterone system in the pathogenesis of atherosclerosis. Current Pharmaceutical Design. 18 (7), 981-1004 (2012).
- Chen, K., et al. Plasma reactive carbonyl species: Potential risk factor for hypertension. Free Radical Research. 45 (5), 568-574 (2011).
- Zhang, X., et al. Angiotensin receptor blockade has protective effects on the poststenotic porcine kidney. Kidney International. 84 (4), 767-775 (2013).
- Zou, X., et al. Renal scattered tubular-like cells confer protective effects in the stenotic murine kidney mediated by release of extracellular vesicles. Scientific Reports. 8 (1), 1263 (2018).
- Kinra, M., Mudgal, J., Arora, D., Nampoothiri, M. An insight into the role of cyclooxygenase and lipooxygenase pathway in renal ischemia. European Review for Medical and Pharmacological Sciences. 21 (21), 5017-5020 (2017).
- Cavalcanti, C. O., et al. Inhibition of PDE5 Restores Depressed Baroreflex Sensitivity in Renovascular Hypertensive Rats. Frontiers in Physiology. 7, 15 (2016).
- Dias, A. T., et al. Sildenafil ameliorates oxidative stress and DNA damage in the stenotic kidneys in mice with renovascular hypertension. Journal of Translational Medicine. 12, 35 (2014).
- Lerman, L. O., Chade, A. R., Sica, V., Napoli, C. Animal models of hypertension: an overview. Journal of Laboratory and Clinical Medicine. 146 (3), 160-173 (2005).
- Reckelhoff, J. F., Romero, D. G., Yanes Cardozo, L. L. Sex, Oxidative Stress, and Hypertension: Insights From Animal Models. Physiology (Bethesda). 34 (3), 178-188 (2019).
- Goldblatt, H., Lynch, J., Hanzal, R. F., Summerville, W. W. Studies on Experimental Hypertension : I. The Production of Persistent Elevation of Systolic Blood Pressure by Means of Renal Ischemia. Journal of Experimental Medicine. 59 (3), 347-379 (1934).
- Gollan, F., Richardson, E., Goldblatt, H. Hypertension in the systemic blood of animals with experimental renal hypertension. Journal of Experimental Medicine. 88 (4), 389-400 (1948).
- Lewis, H. A., Goldblatt, H. Studies on Experimental Hypertension: XVIII. Experimental Observations on the Humoral Mechanism of Hypertension. Bulletin of the New York Academy of Medicine. 18 (7), 459-487 (1942).
- Warner, G. M., et al. Genetic deficiency of Smad3 protects the kidneys from atrophy and interstitial fibrosis in 2K1C hypertension. American Journal of Physiology-Renal Physiology. 302 (11), 1455-1464 (2012).
- Lorenz, J. N., et al. Renovascular hypertension using a modified two-kidney, one-clip approach in mice is not dependent on the alpha1 or alpha2 Na-K-ATPase ouabain-binding site. American Journal of Physiology-Renal Physiology. 301 (3), 615-621 (2011).
- Ebina, K., Iwabuchi, T., Suzuki, S. Histological change in permanently clipped or ligated cerebral arterial wall. Part II: Autopsy cases of aneurysmal neck clipping. Acta Neurochirurgica. 66 (1-2), 23-42 (1982).
- Saleem, M., et al. Sox6: A new modulator of renin expression during physiological conditions. bioRxiv. , (2019).
- Saleem, M., et al. Sox6 as a new modulator of renin expression in the kidney. American Journal of Physiology-Renal Physiology. , (2019).
- Chade, A. R., Williams, M. L., Engel, J., Guise, E., Harvey, T. W. A translational model of chronic kidney disease in swine. American Journal of Physiology-Renal Physiology. 315 (2), 364-373 (2018).
- Xue, Y., Xu, Z., Chen, H., Gan, W., Chong, T. Low-energy shock wave preconditioning reduces renal ischemic reperfusion injury caused by renal artery occlusion. Acta Cirúrgica Brasileira. 32 (7), 550-558 (2017).
- Lalanne, A., Beaudeux, J. L., Bernard, M. A. NGAL: a biomarker of acute and chronic renal dysfunction. Annales de Biologie Clinique. 69 (6), 629-636 (2011).
- Bolignano, D., et al. Neutrophil gelatinase-associated lipocalin (NGAL) as a marker of kidney damage. American Journal of Kidney Diseases. 52 (3), 595-605 (2008).
- Kashyap, S., et al. Development of renal atrophy in murine 2 kidney 1 clip hypertension is strain independent. Research in Veterinary Science. 107, 171-177 (2016).
- Anderson, W. P., Woods, R. L., Kline, R. L., Korner, P. I. Acute haemodynamic responses to unilateral renal artery stenosis in conscious dogs. Clinical and Experimental Pharmacology and Physiology. 12 (3), 305-309 (1985).
- Imanishi, M., et al. Critical degree of renal arterial stenosis that causes hypertension in dogs. Angiology. 43 (10), 833-842 (1992).
- Ziecina, R., Abramczyk, P., Lisiecka, A., Papierski, K., Przybylski, J. Adrenal-renal portal circulation contributes to decrease in renal blood flow after renal artery stenosis in rats. Journal of Physiology and Pharmacology. 49 (4), 553-560 (1998).
- Johnson, J. A., Ichikawa, S., Kurz, K. D., Fowler, W. L., Payne, C. G. Pressor responses to vasopressin in rabbits with 3-day renal artery stenosis. American Journal of Physiology. 240 (6), 862-867 (1981).
- Eirin, A., et al. Changes in glomerular filtration rate after renal revascularization correlate with microvascular hemodynamics and inflammation in Swine renal artery stenosis. Circulation: Cardiovascular Interventions. 5 (5), 720-728 (2012).
- Ma, Z., Jin, X., He, L., Wang, Y. CXCL16 regulates renal injury and fibrosis in experimental renal artery stenosis. American Journal of Physiology-Heart and Circulatory. 311 (3), 815-821 (2016).
- Cheng, J., et al. Temporal analysis of signaling pathways activated in a murine model of two-kidney, one-clip hypertension. American Journal of Physiology-Renal Physiology. 297 (4), 1055-1068 (2009).
- Wiesel, P., Mazzolai, L., Nussberger, J., Pedrazzini, T. Two-kidney, one clip and one-kidney, one clip hypertension in mice. Hypertension. 29 (4), 1025-1030 (1997).
- Johns, C., Gavras, I., Handy, D. E., Salomao, A., Gavras, H. Models of experimental hypertension in mice. Hypertension. 28 (6), 1064-1069 (1996).

