Halogenated Agent Delivery in Porcine Model of Acute Respiratory Distress Syndrome via an Intensive Care Unit Type Device
Summary
We describe a model of hydrochloric acid-induced acute respiratory distress syndrome (ARDS) in piglets receiving sedation with halogenated agents, isoflurane and sevoflurane, through a device used for inhaled intensive care sedation. This model can be used to investigate the biological mechanisms of halogenated agents on lung injury and repair.
Abstract
Acute respiratory distress syndrome (ARDS) is a common cause of hypoxemic respiratory failure and death in critically ill patients, and there is an urgent need to find effective therapies. Preclinical studies have shown that inhaled halogenated agents may have beneficial effects in animal models of ARDS. The development of new devices to administer halogenated agents using modern intensive care unit (ICU) ventilators has significantly simplified the dispensing of halogenated agents to ICU patients. Because previous experimental and clinical research suggested potential benefits of halogenated volatiles, such as sevoflurane or isoflurane, for lung alveolar epithelial injury and inflammation, two pathophysiologic landmarks of diffuse alveolar damage during ARDS, we designed an animal model to understand the mechanisms of the effects of halogenated agents on lung injury and repair. After general anesthesia, tracheal intubation, and the initiation of mechanical ventilation, ARDS was induced in piglets via the intratracheal instillation of hydrochloric acid. Then, the piglets were sedated with inhaled sevoflurane or isoflurane using an ICU-type device, and the animals were ventilated with lung-protective mechanical ventilation during a 4 h period. During the study period, blood and alveolar samples were collected to evaluate arterial oxygenation, the permeability of the alveolar-capillary membrane, alveolar fluid clearance, and lung inflammation. Mechanical ventilation parameters were also collected throughout the experiment. Although this model induced a marked decrease in arterial oxygenation with altered alveolar-capillary permeability, it is reproducible and is characterized by a rapid onset, good stability over time, and no fatal complications.
We have developed a piglet model of acid aspiration that reproduces most of the physiological, biological, and pathological features of clinical ARDS, and it will be helpful to further our understanding of the potential lung-protective effects of halogenated agents delivered through devices used for inhaled ICU sedation.
Introduction
Acute respiratory distress syndrome (ARDS) is a common cause of hypoxemic respiratory failure and death in critically ill patients1. It is characterized by both diffuse alveolar epithelial and endothelial injuries, leading to increased permeability and pulmonary edema, altered alveolar fluid clearance (AFC), and worsened respiratory distress2. The resorption of alveolar edema and recovery from ARDS require epithelial fluid transport through the alveoli to remain intact, which suggests that a therapy improving AFC could be useful3,4. Although lung-protective ventilation and a restrictive strategy for intravenous fluid therapy have proven beneficial in improving outcomes2,5, they are still associated with high mortality and morbidity6. Therefore, there is an urgent need to develop effective therapies for the syndrome and to better understand the precise mechanisms through which such therapies might work.
Halogenated anesthetics, such as isoflurane or sevoflurane, have been widely used for general anesthesia in the operating room. Sevoflurane is associated with decreased inflammation in the lungs of patients undergoing thoracic surgery and with a decrease in postoperative pulmonary complications, such as ARDS7. Similar results have been found in a meta-analysis of patients after cardiac surgery8. Halogenated volatiles also have a bronchodilatory effect9,10 and perhaps some properties that protect several organs, such as the heart8,11 and the kidneys12,13,14. Recently, there has been growing interest in the clinical use of inhaled anesthetics as sedatives in the intensive care unit (ICU). Both animal and human studies support the protective effects of pretreatment with halogenated agents before prolonged ischemia of the liver15, the brain16, or the heart11. Halogenated agents also have potential pharmacokinetic and pharmacodynamic advantages over other intravenous agents for the sedation of critically ill patients, including a rapid onset of action and fast offset due to little accumulation in tissues. Inhaled halogenated agents decrease intubation times in comparison with intravenous sedation in patients undergoing cardiac surgery17. Several studies support the safety and efficacy of halogenated agents in the sedation of ICU patients18,19,20. In experimental models of ARDS, inhaled sevoflurane improves gas exchange21,22, reduces alveolar edema21,22, and attenuates both pulmonary and systemic inflammation23. Isoflurane also ameliorates lung repair after injury by maintaining the integrity of the alveolar-capillary barrier, possibly by modulating the expression of a key tight junction protein24,25,26. In addition, mouse macrophages that were cultured and treated with isoflurane had better phagocytic effects on neutrophils than macrophages that were not treated with isoflurane27.
However, the precise biological pathways and mechanisms accounting for the lung-protective properties of volatile anesthetics remain largely unknown to date, requiring further investigation18. Additional studies are also warranted to investigate the precise effects of sevoflurane on lung injury and to verify whether experimental evidence can be translated to patients. The first randomized control trial from our team found that the administration of inhaled sevoflurane in patients with ARDS was associated with oxygenation improvement and decreased levels of both pro-inflammatory cytokines and lung epithelial injury markers, as assessed by plasma and alveolar soluble receptors for advanced glycation end products (sRAGE)28. As sRAGE is now considered as a marker of alveolar type 1 cell injury and a key mediator of alveolar inflammation, these results could suggest some beneficial effects of sevoflurane on the lung alveolar epithelial injury21,29,30.
The use of halogenated agents for inhaled ICU sedation has long required operating room anesthesia ventilators and gas vaporizers to be deployed in the ICU. Since then, anesthetic reflectors suitable for the use with modern critical care ventilators have been developed for specific use in the ICU31. These devices feature modified heat and moisture exchanging filters inserted between the Y-piece of the respiratory circuit and the endotracheal tube. They allow the administration of halogenated agents, with isoflurane and sevoflurane being the most frequently used, and they consist of a porous polypropylene evaporator rod, into which a liquid agent, delivered by a specific syringe pump, is released. The halogenated agent is absorbed during expiration by a reflecting medium contained in the device and it is released during the next inspiration, allowing recirculation of approximately 90% of the expired halogenated agent31,32. Recently, a miniaturized version of the device was developed with an instrumental dead space of 50 mL, making it even more suitable for use during ultra-protective ventilation in ARDS patients, with tidal volumes that could be as low as 200 mL31. Such a miniaturized device has never been studied in an experimental piglet model of ARDS.
Because previous research supports the promising roles of halogenated volatiles in lung alveolar inflammation and injury during ARDS, we designed an experimental animal model to achieve a translational understanding of the mechanisms of the effects of halogenated agents on lung injury and repair33,34,35. In this study, we developed a model of hydrochloric acid (HCl)-induced ARDS in piglets in whom inhaled sedation can be delivered using the miniaturized version of the anesthetic conserving device, an ICU-type device. This large animal model of ARDS could be used to further our understanding of the potential lung-protective effects of inhaled halogenated agents.
Protocol
The study protocol was approved by the Animal Ethics Committee of the French Ministère de l’Education Nationale, de l’Enseignement Supérieur et de la Recherche (approval number 01505.03) before being registered at preclinicaltrials.eu (Pre-clinical registry identifier PCTE0000129). All procedures were performed in the Centre International de Chirurgie Endoscopique, Université Clermont Auvergne, Clermont-Ferrand, France, in accordance with the Animal Research: Reporting In Vivo Experiments (ARRIVE) guidelines36.
1. Animal preparation and anesthesia
- Piglet mode
- Ensure that the experimental protocol is consistent with guidelines for animal experiments, including the 3R principles (replacement, reduction, and refinement) and national/international regulations.
- Obtain approvals from the ethics committee for care and use of experimental animals at the relevant institution before starting the protocol.
- Use a male white Landrace piglet (2–4 months old; weighing 10–15 kg).
- Place the piglet in the supine position after premedication using intramuscular azaperone (described in 1.2.2).
- Anesthesia induction
- Restrict animals from having food for overnight while allowing free access to water.
- Administer anxiolytic premedication to the piglet using intramuscular azaperone (2 mg.kg-1) behind the ear.
- Apply a finger pressure on the soft tissues of the auricular base of the piglet to identify the medial and lateral auricular vein.
- Insert a peripheral intravenous 22 G catheter in the medial or lateral auricular vein of the piglet. Follow with the catheter at a shallow angle of 45˚ through the skin and advance until blood appears through the catheter.
- Induce general anesthesia with intravenous propofol (3 mg.kg-1) and sufentanil (0.3 µg.kg-1)37. Check the depth of the anesthesia by lack of response to pedal reflex.
- Tracheal intubation38,39
- Prepare the laryngoscope using a size 4 straight Miller laryngoscope blade.
- Pass the laryngoscope into the pharyngeal cavity and depress the tongue with the laryngoscope blade, making the epiglottis visible.
- Visualize the larynx opening of the piglet prior to orotracheal intubation.
- Insert a 6 mm internal diameter cuffed endotracheal tube.
- Inflate the endotracheal tube cuff to reach a cuff pressure around 20–30 cmH2O.
- Fix the endotracheal tube to the piglet’s nose with micropore surgical tape.
- Connect to the ventilator and initiate mechanical ventilation following the settings described in section 3.
- Sedation maintenance
- Maintain anesthesia with continuous intravenous infusion of propofol (5 mg.kg-1.h-1) before acid-induced lung injury. The infusion of propofol will be stopped when halogenated agents are started.
- Add a continuous intravenous infusion of remifentanil (10–20 μg.kg−1.h−1 = 0.15–0.33 μg.kg−1.min−1) for pain management.
- Add continuous intravenous infusion of cisatracurium (0.2 mg.kg-1.h-1) for a neuromuscular blockade.
- Keep the body temperature of the piglet at approximately 38 °C using warm blankets.
- Monitor electrocardiogram activity, the peripheral oxygen saturation (SpO2), and arterial pressure continuously using an external monitor.
- Surgery
- Insert central venous access using a surgical exposure of the right internal jugular vein and the Seldinger method to insert a 3-lumen catheter (7 French, 16 cm).
- Make a cutaneous midline incision on the ventral aspect of the neck, 2 cm lateral from the trachea. Use surgical forceps to dissect the tissues.
- Localize the internal jugular vein (approximately 1–2 cm deep, lateral to the internal carotid artery) and, using the needle (18 G, 6.35 cm), make a puncture with a craniocaudal direction orientation.
- With the hand, insert the “J” guidewire (0.81 mm diameter, 60 cm) through the needle. Gently remove the needle and quickly insert a venous catheter with three lines into the internal jugular vein along the “J” guidewire. Remove the “J” guidewire while maintaining the venous catheter in place.
- Aspirate blood through each line of the venous catheter to remove the air from the different lines and flush with 5 mL of saline solution (0.9% NaCl) to rinse the three lines.
- Suture the skin with a 3.0 non-absorbable suture thread following the continuous Lembert pattern and fix the catheter to the skin with a single stitch and triple knots on each lateral perforation of the central venous catheter.
- Insert an arterial line via surgical exposure of the right femoral artery and use the Seldinger method to insert the thermodilution catheter (3–5 French, 20 cm).
- Place the right forelimb of the piglet in extension.
- Make a cutaneous incision on the right groin area of the piglet. Use surgical forceps to dissect the subcutaneous and muscular tissues.
- Localize the right femoral artery by palpating the femoral pulse (approximately 3–4 cm deep) and, using the needle (19 G, 54 mm), make a puncture with a caudocranial direction orientation.
- Insert the “J” guidewire through the needle. Gently remove the needle and quickly insert an arterial catheter into the femoral artery up along the guidewire. Remove the guidewire while maintaining the catheter in place.
- Remove the air from the arterial catheter and flush with saline solution to rinse the line.
- Suture the skin with a 3.0 non-absorbable suture thread following the continuous Lembert pattern and fix the catheter to the skin with a single stitch and triple knots on each lateral perforation of the arterial catheter.
- Plug the catheter on an arterial line tubing to allow retrieval of serial blood samples and continuous hemodynamic monitoring (arterial pressure, cardiac index, and extravascular lung water, as indexed to body weight) with a pulse contour cardiac output monitor device.
- Insert central venous access using a surgical exposure of the right internal jugular vein and the Seldinger method to insert a 3-lumen catheter (7 French, 16 cm).
2. Acid-induced acute lung injury
CAUTION: Use gloves and glasses during this step to avoid any risk of contact of the acid with the skin or the eyes)
- Make 100 mL of HCl at 0.05 M and pH 1.4.
- Using the anatomical landmark of the last segment of the sternum, measure the distance between the tip of the endotracheal tube and the carina of the piglet.
- Mark this distance with a black pen on a Ch14 suction catheter.
- Insert the suction catheter through the endotracheal tube up to the black landmark.
- Gently instill 4 mL.kg-1 (bodyweight) of acid through the suction catheter for over 3 min.
- Remove the suction catheter.
3. Mechanical ventilation
- Use volume-controlled ventilation on an intensive care ventilator.
- Use a tidal volume of 6 mL.kg-1, a positive end-expiratory pressure (PEEP) of 5 cmH2O, and an inspired oxygen fraction (FiO2) of 40%.
- Adjust the respiratory rate to maintain the end-tidal carbon dioxide between 35 and 45 mmHg.
NOTE: Based on previous studies37,40,41, lung injury is considered established when the arterial oxygen tension (PaO2)-to-FiO2 ratio decreases to 25% from the baseline, approximately 1 h after airway HCl instillation.
4. Halogenated anesthetics
NOTE: Start sedation using halogenated anesthetics (sevoflurane or isoflurane) once acid-induced lung injury is achieved. The intravenous sedation using propofol should then be interrupted.
- Filling the syringe (Figure 1A): Attach the filling adapter provided by the manufacturer to the 250 mL bottle of the halogenated agent and a 60 mL syringe to the filling adapter. Turn the bottle upside down and fill the syringe by pushing and pulling the plunger. Turn the bottle upright and remove the syringe.
- Scavenging (Figure 1B)
- Place the charcoal filter, used to remove halogenated hydrocarbon anesthetic gases, close to the ventilator.
- Remove the protective cap from the charcoal filter.
- Connect the charcoal filter to the expiratory valve of the ventilator with a flex tube.
- Use the anesthetic conserving device (device used for inhaled ICU sedation) (Figure 1C) as described below.
- Connect the ionomer membrane dryer line to the gas sampling port of the anesthetic conserving device.
- Connect one side of the gas sampling line to the ionomer membrane dryer line.
- Connect the other side of the gas sampling line to the gas analyzer.
- Insert the anesthetic conserving device between the Y-piece of the respiratory circuit and the endotracheal tube.
- Ensure that the anesthetic conserving device has the black side up and is sloped down toward the piglet.
- Deliver inhaled sedation through the anesthetic conserving device (Figure 2).
- Place the specific syringe in the syringe pump.
- Connect the anesthetic agent line to the syringe.
- Prime the agent line with a bolus of 1.5 mL of the halogenated agent.
- Adapt the initial pump rate in mL.h-1 (initial syringe pump rate settings of isoflurane and sevoflurane are 3 and 5 mL/h, respectively) to the targeted expired sevoflurane fraction (FEsevo) or the expired isoflurane fraction (FEiso) value, as displayed on the gas analyzer.
- Ensure that the gas analyzer displays a FEsevo %–FEiso % or equivalent minimal alveolar concentration value greater than zero. If necessary, give an additional bolus of 0.3 mL of the halogenated agent.
- Adapt the syringe pump rate necessary to reach a certain concentration depending on the minute volume and the targeted concentration, with rates of 2–7 mL.h-1 and 4–10 mL.h-1 being, in general, associated with expired fractions of 0.2%–0.7% and 0.5%–1.4% for isoflurane42 and sevoflurane28,43, respectively.
- During the experiment, continue administration of the halogenated agents with FEsevo and FEiso targets of 0.8–1.1 and 0.5–0.8, respectively.
5. Measurements
- Monitoring
- Collect different parameters as measured by the external monitor: heart rate, blood pressure, and peripheral oxygen saturation.
- Record parameters as measured by the ventilator: tidal volume, respiratory rate, set PEEP, auto-PEEP (by applying an expiratory hold maneuver of 5 s on the ventilator), compliance of the respiratory system, airway resistance, inspiratory plateau pressure (by applying an inspiratory hold maneuver of 2 s on the ventilator), peak inspiratory pressure, and driving pressure.
- Calculate the lung functional residual capacity using the Nitrogen Wash In/Wash Out method if integrated in the ventilator.
- Use the thermal indicator previously inserted in the femoral artery to measure the extravascular water volume of the lungs, cardiac index, and systemic vascular resistance.
- Undiluted pulmonary edema fluid sampling to measure the net AFC rate.
- Insert a soft 14 Fr suction catheter into a wedged position in the distal bronchus through the endotracheal tube.
- Sample edema fluid into a suction trap by applying gentle suction.
- Centrifuge all samples at 240 x g at 4 °C for 10 min in a refrigerated centrifuge.
- Collect the supernatants.
NOTE: Total protein concentration in undiluted pulmonary edema fluid is measured with a colorimetric method. Because the rate of clearance of edema fluid from the alveolar space is much faster than the rate of protein removal, the net AFC rate was calculated as Percent AFC = 100 × [1 – (initial edema protein/final edema total protein)] and thereafter was reported as %/h37. Undiluted pulmonary edema fluid samples are collected from the animals at baseline and 4 h later, as previously described34,44,45,46,47,48,49.
- Mini bronchoalveolar lavage sampling.
- Insert a soft 14 Fr suction catheter into a wedged position in a distal bronchus through the endotracheal tube.
- Instill 50 mL of a 0.9% sodium chloride solution into the suction catheter.
- Promptly sample the fluid into a suction trap.
- Collect the mini bronchoalveolar lavage.
NOTE: Total protein concentration in mini BAL is measured with a colorimetric method and, for example, the levels of proinflammatory cytokines, such as TNF-α, IL-6, IL-1β, and IL-18, are measured using a multiplex immunoassay method. Samples are collected 4 h after the acid-induced lung injury.
- Blood gas analysis
- Collect arterial blood gases through the arterial line in a 3 mL BD Preset syringe with BD Luer-Lok tip at baseline. Immediately measure PaO2/FiO2, PaCO2, pH, serum lactate, and serum creatinine using a point-of-care blood gas analyzer.
- Repeat this step every hour for 4 h after acid instillation.
- Lung sampling
- Sacrifice the piglet with an intravenous injection of pentobarbital (150 mg.kg-1) at the end of the experiment (4 h after acid-induced lung injury).
- Dissect and remove the whole lungs. Fix with alcohol acetified formalin.
- Embed in paraffin and slice at a 10 μm thickness.
- Stain with hematoxylin and eosin.
NOTE: Histological evidence of lung injury can be assessed using a standardized histology injury score50.
Representative Results
For this experiment, 25 piglets were anesthetized and divided in two groups: 12 piglets in the untreated group (SHAM group) and 13 piglets in the acid-injured group (HCl group). No piglet died before the end of the experiment. A two-way repeated-measures analysis of variance (RM-ANOVA) indicated a significant time by group interaction (P < 10−4) with a detrimental effect of HCl-induced ARDS on PaO2/FiO2, compared to sham animals without ARDS (Figure 3). A significant between-group difference was noted in the undiluted pulmonary edema fluid levels of the total protein measured after 4 h of mechanical ventilation (P < 10−4). HCl-induced ARDS was associated with increased BAL protein when compared to the sham animals (Figure 4). A two-way RM-ANOVA indicated a significant time by group interaction (P < 10−4) with HCl-induced ARDS being associated with increased extravascular lung water, compared to sham animals without ARDS (Figure 5A). Cardiac output and systemic vascular resistance values are reported in Figure 5B and Figure 5C, respectively. Inspiratory and expiratory fractions of sevoflurane measured in all animals are reported in Figure 6, and macroscopic evidence of histologic lung injury is shown in Figure 7.
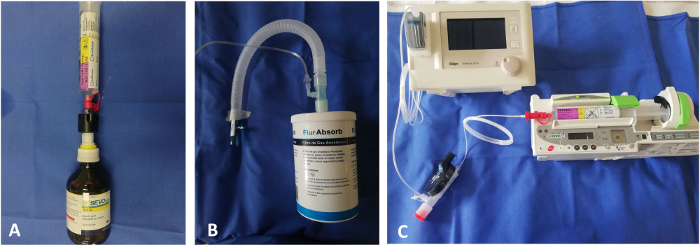
Figure 1: Illustration of the set up needed to administer sedation with halogenated volatile agents using the anesthetic conserving device. (A) Filling the specific syringe with the bottle adapter. (B) Scavenging the halogenated agents using the scavenging charcoal filter. (C) Assembling both the syringe pump and the gas analyzer with the anesthetic conserving device to use along with the ventilator. Please click here to view a larger version of this figure.
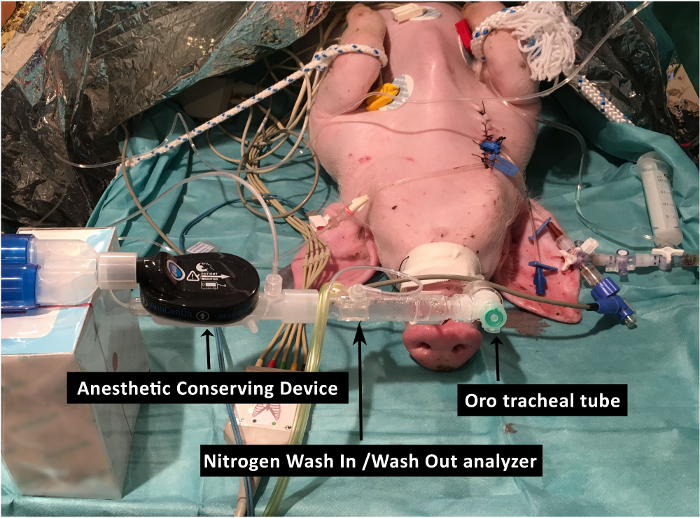
Figure 2: Schematic representation of the connection of the anesthetic conserving device to the respiratory circuit of the ventilator. This includes the integration of the module to measure lung functional residual capacity. Please click here to view a larger version of this figure.
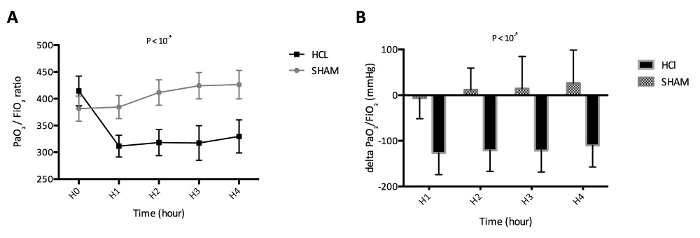
Figure 3: Measurement of the alteration in arterial oxygenation. (A) Evolution of arterial oxygen tension (PaO2) to inspired oxygen fraction (FiO2) ratio in untreated piglets (SHAM group, N = 12) and acid-injured piglets (HCl group, N = 13) during a 4 h period. (B) Evolution of the delta of PaO2/FiO2 at a specific time point and of PaO2/FiO2 at H0 in untreated piglets (SHAM group, N = 12) and acid-injured piglets (HCl group, N = 13). Values are expressed in mmHg and represented as means, with error bars representing standard errors of the means. Please click here to view a larger version of this figure.
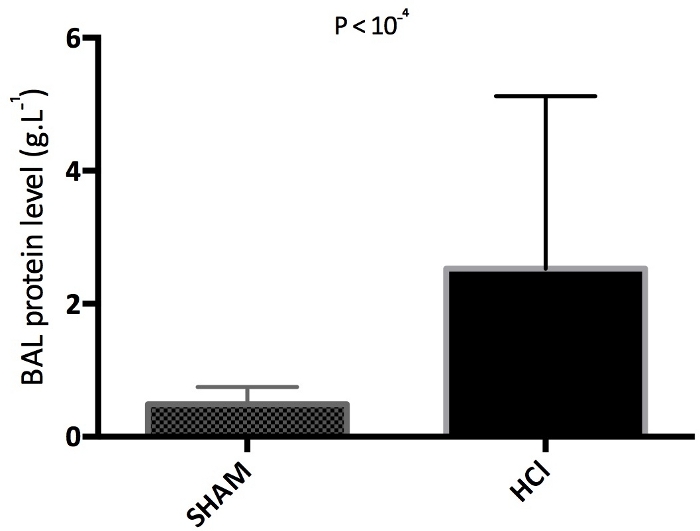
Figure 4: Measurements of the alteration in alveolar-capillary membrane permeability. Mini bronchoalveolar levels (BAL) of total protein at 4 h in untreated piglets (SHAM group, N = 12) and acid-injured piglets (HCl group, N = 13). Values are expressed in g.L-1 and represented as means, with error bars representing standard errors of the means. Please click here to view a larger version of this figure.
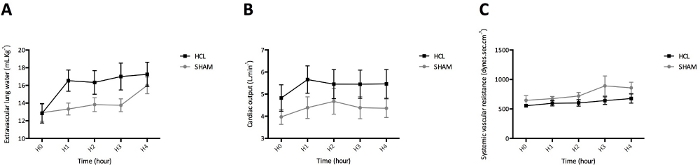
Figure 5: Measurements provided by transpulmonary thermodilution. (A) Lung edema, as assessed by extravascular lung water. (B) Cardiac output. (C) Systemic vascular resistance. Transpulmonary thermodilution was performed in untreated piglets (SHAM group, N = 12) and acid-injured piglets (HCl group, N = 13) using a pulse contour cardiac output monitor. Values are expressed in mL.kg-1, L.min-1, dynes.s.cm-5, respectively, and are reported as means, with error bars representing standard errors of the means. Please click here to view a larger version of this figure.
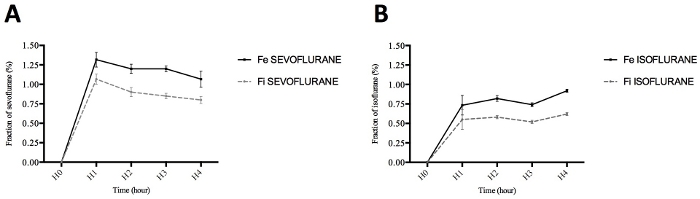
Figure 6: Measurements of the expired fractions of halogenated agents, sevoflurane and isoflurane. (A) Expired (FEsevoflurane) and inspired (FIsevoflurane) sevoflurane fractions during the 4-h study period. (B) Expired (FEisoflurane) and inspired (FIisoflurane) isoflurane fractions during the 4-h study period. Values are expressed in % and represented as means, with error bars representing standard errors of the means. Please click here to view a larger version of this figure.
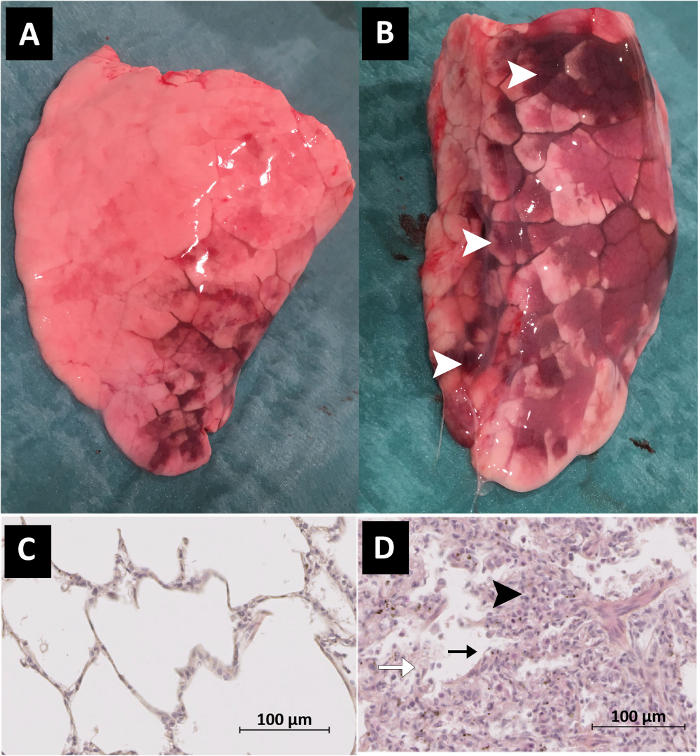
Figure 7: Macroscopic evaluation of the whole lung after the 4 h study period. (A) Whole lung of an untreated piglet (SHAM group). (B) Whole lung of an acid-injured piglet (HCl group). Macroscopic lung injury, with visible hemorrhage and congestion, is noticeable in the red parts of the lung (white arrows). Histologic evaluation of the lung after the 4 h study period. (C) Histological slice of the lung of an untreated piglet (SHAM group). (D) Histological slice of the lung of an acid-injured piglet (HCl group). Histologic evidence of lung injury was a greater cellularity consisting mainly of neutrophils (black arrowheads), with more areas of atelectasis and increased alveolar disruption, hyaline membranes, protein debris, hemorrhage (white arrow), and the thickening of the alveolar wall (black arrows). Scale bars equal 100 µm. Please click here to view a larger version of this figure.
Discussion
This article describes a reproducible experimental model of ARDS induced by the intratracheal instillation of HCl in piglets to investigate the lung-protective effects of halogenated volatiles, such as sevoflurane or isoflurane, delivered using an anesthetic conserving device.
The primary goal of this study was to develop an experimental model of ARDS in which volatile agents could be delivered by an anesthetic conserving device, such as those used in ICU patients. Although some effects of halogenated agents have been previously studied in animal models, the strength of our model is that it is a clinically relevant, translational model to further our understanding of such effects. Another advantage of this model is that substantial lung injury can be induced in animals larger than mice with low mortality over time. Indeed, an important consideration in the choice of an animal model of ARDS should be the experimental question to be addressed51. In a mouse model, experimental techniques to induce lung injuries, such as intravenous oleic acid52, lavage-induced surfactant depletion40, and high-stretch mechanical ventilation53, can induce intensive injury over a timescale from hours to days, but they do not allow the investigation of lung injury repair/resolution. Furthermore, some challenges of the animal model (e.g., extremely large tidal volumes in certain models of ventilator-induced lung injury) may be extreme, such that they are unrepresentative of the range of conditions present in humans with ARDS. Conversely, such models as intratracheal endotoxin54 may allow the investigation of certain aspects of the resolution of inflammation and fibrotic processes that can occur following clinical ARDS, but they do not produce the substantial hypoxemia that is a prerequisite for a diagnosis of the syndrome51. To characterize their specific effects better, therapeutics should likely be tested in multiple models, as none sufficiently reproduces the heterogeneity of ARDS55.
Our model has some inherent limitations. First, because we euthanized the animals 4 h after experimental ARDS onset, we only collected parameters during the early phase of ARDS. More elaborate facilities, such as “animal ICUS,” are needed to investigate later phases of ARDS in piglets. Second, during the current experiments, we only assessed the degree of lung injury using an index of arterial oxygenation, such as PaO2/FiO2. However, most features of experimental ARDS were present when we previously reported use of this acid-induced ARDS model37. To improve our model, it could be interesting to add non-invasive measures of the degree of lung injury, determined, for example, using electrical impedance tomography or lung ultrasound56. Third, we only report the use of a “one-hit model” to induce lung injury in piglets, while models in which more than one inciting stimulus for lung injury is induced are likely more reflective of the pathological human situation, in which a single inciting stimulus is rarely present (“two-hit hypothesis”)51. From this perspective, ventilator-induced lung injury could, for example, be added to our model to produce an additional hit, and this model can be combined with other injurious “hits” if needed to investigate more complex clinical scenarios involving multiple features of the ARDS pathophysiology, such as lung endothelial injury, alveolar macrophage activation, and the effects of cell-free hemoglobin57, among others58. Fourth, we did not test other halogenated agents such as desflurane. Indeed, we only used isoflurane and sevoflurane for inhaled sedation in piglets because they are most frequently used in clinical ICU practice, at least in Europe and in some other areas of the world.
The porcine model has contributed to significant advances in experimental research over the last decades, and it has become an increasingly important translational bridge between traditional small laboratory animal models and human medicine. A major advantage of using experimental models in large animals is to allow investigations that involve the ventilation of animals over time. Nevertheless, such models can be extremely expensive, and they may sometimes require the availability of an animal ICU. In addition, the limited availability of some molecular reagents in pigs is an important limitation. Studies in smaller animals, such as mice, rats, or rabbits, have been highly useful in studying individual pathways, but the generalizability of the results to humans appears limited59. Larger animal studies can provide focused evaluations of key physiological and molecular pathways and can be used to test new therapies in humans, such as sedation with halogenated agents. Furthermore, the size of the animals supports the use of clinically used catheters, endotracheal tubes, ventilators, and monitors that are not fully available for use in smaller mammals. Indeed, the important advantages of using experimental models with large animals include the ability to take multiple longitudinal blood samples and perform blood gas analyses over time. In addition, invasive hemodynamic monitoring could be used as transpulmonary thermodilution with a pulse contour cardiac output monitor device, allowing the study of the degree of alveolar edema by measuring extravascular lung water, a highly relevant parameter in the alteration of the alveolar–capillary barrier during ARDS51. Nevertheless, caution is necessary when data aside from size are interpreted from animal models, because important anatomical, physiological, and immunological differences exist among animal species. Animal models could have anatomical differences that will impact research and the translation to humans. Indeed, many animals, such as mice or rabbits, have an incomplete mediastinum and thin visceral membranes, prohibiting, for example, the use of the contralateral pleura as a control. However, larger animals (e.g., sheep or pigs) have one pleural cavity around each lung and a thick visceral pleura resembling that of humans60.
The administration of volatile anesthetics to ICU patients has been increasingly studied in the last decade, mostly because of the development of dedicated devices based either on reflection or on a circle system. Such devices can be inserted into any mechanical ventilation circuit to administer the two agents most frequently used in the ICU setting, sevoflurane and isoflurane61. Due to their hypnotic, bronchodilator, and anticonvulsant properties, halogenated agents have been used for a long time in the ICU to manage patients with refractory status asthmaticus, status epilepticus, and complex sedation scenarios with high sedation requirements, such as burns, chronic pain, high-risk surgeries, or a history of drug abuse. Although recent international guidelines do not recommend using volatile agents for procedural pain management62, halogenated anesthetics are increasingly popular in Europe, and they are considered a feasible option for sedation in the 2015 German Guidelines63, especially if short wake-up times are needed. Potential therapeutic end-organ-protective properties via the cytoprotective and anti-inflammatory mechanisms64 of volatile anesthetics have attracted the attention of researchers and physicians. In fact, their emerging use in ICUs has paved the way to the study of their potential benefits in patients with ARDS. ARDS represents the ultimate and most severe form of lung organ dysfunction, as well as a major challenge for patients, their families, healthcare providers from various disciplines, and healthcare systems when caring for critically ill patients, especially under some exceptional circumstances, such as during the current COVID-19 pandemic65,66,67. Beyond the current efforts to find specific antiviral therapies, improving supportive care and treatment options for patients with COVID-19-related ARDS is, therefore, of major importance65,68,69. From this perspective, the rationale supporting inhaled sedation with sevoflurane or isoflurane as a way to improve lung epithelial permeability, to decrease the inflammatory response, and, potentially, to improve patient outcomes is strong. In addition, several non-human models have shown that a volatile anesthetic agent, such as inhaled sevoflurane, improves gas exchange21,70,71, reduces alveolar edema22, and decreases levels of pro-inflammatory cytokines72,73. These effects could be explained by restored lung epithelial function and by the immunomodulatory effects of the halogenated agent. In a previous pilot randomized control trial, the use of a halogenated agent, such as sevoflurane, to sedate ARDS patients in the ICU improved oxygenation and decreased levels of a marker of lung epithelial injury and of some pro-inflammatory cytokines (interleukin [IL]-1β, IL-6, and IL-8 and tumor necrosis factor-α) compared to intravenous sedation28. These results reinforce the protective effect of sevoflurane on inflammation and on reduced epithelial injury or improved AFC, as assessed by plasma sRAGE34.
Understanding the biological mechanisms and pathophysiological pathways involved in acute lung injury and its resolution under inhaled sedation with halogenated agents requires the use of experimental and preclinical models. Although in vitro studies represent an important step in describing these mechanisms74, in vivo experiments are fundamental before results can be extrapolated to the clinical setting. Furthermore, in this large animal model, halogenated agents could be administered using the same anesthetic conserving device as in humans. In fact, devices based either on reflection or on a circle system, which are both available for patients in some countries, do not have specific equivalents available for small animals, such as for mice, rats, or rabbits. Consequently, when researchers want to administer halogenated agents to animals, they must choose between either pre- or post-exposure to halogenated agents, usually via anesthesia chamber induction over a more or less long time with no specific mechanical ventilation during this period75. This piglet model allows the specific reproduction of the same treatment conditions as in ICU patients with ARDS, that is, the administration of halogenated agents, such as sevoflurane, in addition to delivering lung-protective mechanical ventilation with low tidal volumes and PEEP. Interestingly, our model reported the use of the recent, miniaturized version of the anesthetic conserving device to administer sevoflurane for the first time in piglets, thereby allowing smaller tidal volumes and further instrumental dead space to be set compared to the previous version of the device. Furthermore, in addition to administering halogenated volatiles, this model of acid-induced ARDS could be useful in studying specific pathways, such as those involved in lung epithelial injury and its repair37.
In conclusion, this experimental model of ARDS in piglets has significant advantages compared to the existing ones. These include rapid onset (within 1 h in general), good reproducibility and stability over time, a low mortality rate, and, more importantly, the use of a clinically relevant device to deliver inhaled ICU sedation, thus allowing novel translational approaches to the study of the effects of halogenated agents in ARDS.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors would like to thank the staff from the GreD, the Université Clermont Auvergne, and the Centre International de Chirurgie Endoscopique (all in Clermont-Ferrand, France).
Materials
| Tracheal intubation | |||
| Endotracheal tube 6-mm | Covidien | 18860 | |
| Animal preparation | |||
| Central venous catheter 3-lumens catheter (7 French – 16 cm) | Arrow | CV-12703 | |
| Pulse contour cardiac output monitor PiCCO catheter (3-5 French – 20 cm) | Getinge Pulsion Medical System | catheter | |
| Warm blankets WarmTouch5300 | MedTronic | 5300 | |
| Monitoring | |||
| External monitor IntelliVue MP40 | Phillips | MNT 142 | |
| Point-of-care blood gas analyzer Epoc® Blood Analysis System | Siemens | 20093 | |
| Pulse contour cardiac output monitor PiCCO Device PulsioFlex Monitor | Getinge Pulsion Medical System | PulsioFlex | |
| Mechanical ventilation | |||
| Ventilator Engström Carestation | General Electrics | Engström | |
| Halogenated anesthetics | |||
| Anaconda Syringe | SedanaMedical | 26022 | |
| Anesthetic conserving device AnaConDa-S | SedanaMedical | 26050 | |
| Charcoal filter FlurAbsorb | SedanaMedical | 26096 | |
| Filling Adaptaters | SedanaMedical | 26042 | |
| Ionomer membrane dryer line Nafion | SedanaMedical | 26053 | |
| Products | |||
| Propofol | Mylan | 66617123 | |
| Isoflurane | Virbac | QN01AB06 | |
| Pentobarbital | PanPharma | 68942457 | |
| Sevoflurane | Abbvie | N01AB08 | |
| Sufentanil | Mylan | 62404996 |
References
- ARDS Definition Task Force et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA: The Journal of the American Medical Association. 307 (23), 2526-2533 (2012).
- Thompson, B. T., Chambers, R. C., Liu, K. D. Acute Respiratory Distress Syndrome. The New England Journal of Medicine. 377 (6), 562-572 (2017).
- Ware, L. B., Matthay, M. A. Alveolar fluid clearance is impaired in the majority of patients with acute lung injury and the acute respiratory distress syndrome. American Journal of Respiratory and Critical Care Medicine. 163 (6), 1376-1383 (2001).
- McAuley, D. F., Frank, J. A., Fang, X., Matthay, M. A. Clinically relevant concentrations of beta2-adrenergic agonists stimulate maximal cyclic adenosine monophosphate-dependent airspace fluid clearance and decrease pulmonary edema in experimental acid-induced lung injury. Critical Care Medicine. 32 (7), 1470-1476 (2004).
- Fan, E., et al. An official American thoracic society/European society of intensive care medicine/society of critical care medicine clinical practice guideline: Mechanical ventilation in adult patients with acute respiratory distress syndrome. American Journal of Respiratory and Critical Care Medicine. 195 (9), 1253-1263 (2017).
- Bellani, G., et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA: The Journal of the American Medical Association. 315 (8), 788-800 (2016).
- De Conno, E., et al. Anesthetic-induced improvement of the inflammatory response to one-lung ventilation. Anesthesiology. 110 (6), 1316-1326 (2009).
- Uhlig, C., et al. Effects of volatile anesthetics on mortality and postoperative pulmonary and other complications in patients undergoing surgery: A systematic review and meta-analysis. Anesthesiology: The Journal of the American Society of Anesthesiologists. 124 (6), 1230-1245 (2016).
- Campagna, J. A., Miller, K. W., Forman, S. A. Mechanisms of actions of inhaled anesthetics. The New England Journal of Medicine. 348 (21), 2110-2124 (2003).
- Dikmen, Y., Eminoglu, E., Salihoglu, Z., Demiroluk, S. Pulmonary mechanics during isoflurane, sevoflurane and desflurane anaesthesia. Anaesthesia. 58 (8), 745-748 (2003).
- Hert, S. G. D., et al. Choice of primary anesthetic regimen can influence intensive care unit length of stay after coronary surgery with cardiopulmonary bypass. Anesthesiology. 101 (1), 9-20 (2004).
- Hashiguchi, H., et al. Isoflurane protects renal function against ischemia and reperfusion through inhibition of protein kinases, JNK and ERK. Anesthesia and Analgesia. , 1584-1589 (2005).
- Fukazawa, K., Lee, H. T. Volatile anesthetics and AKI: risks, mechanisms, and a potential therapeutic window. Journal of the American Society of Nephrology: JASN. 25 (5), 884-892 (2014).
- Obal, D., Rascher, K., Favoccia, C., Dettwiler, S., Schlack, W. Post-conditioning by a short administration of desflurane reduced renal reperfusion injury after differing of ischaemia times in rats. British Journal of Anaesthesia. 97 (6), 783-791 (2006).
- Lv, X., et al. Isoflurane preconditioning at clinically relevant doses induce protective effects of heme oxygenase-1 on hepatic ischemia reperfusion in rats. BMC Gastroenterology. 11, 31 (2011).
- Sakai, H., et al. Isoflurane provides long-term protection against focal cerebral ischemia in the rat. Anesthesiology. 106 (1), 92-99 (2007).
- Jerath, A., et al. Volatile-based short-term sedation in cardiac surgical patients: a prospective randomized controlled trial. Critical Care Medicine. 43 (5), 1062-1069 (2015).
- Jerath, A., Parotto, M., Wasowicz, M., Ferguson, N. D. Volatile Anesthetics. Is a New Player Emerging in Critical Care Sedation. American Journal of Respiratory and Critical Care Medicine. 193 (11), 1202-1212 (2016).
- Perbet, S., et al. A pharmacokinetic study of 48-hour sevoflurane inhalation using a disposable delivery system (AnaConDa®) in ICU patients. Minerva Anestesiologica. 80 (6), 655-665 (2014).
- Mesnil, M., et al. Long-term sedation in intensive care unit: a randomized comparison between inhaled sevoflurane and intravenous propofol or midazolam. Intensive Care Medicine. 37 (6), 933-941 (2011).
- Schläpfer, M., et al. Sevoflurane reduces severity of acute lung injury possibly by impairing formation of alveolar oedema. Clinical and Experimental Immunology. 168 (1), 125-134 (2012).
- Voigtsberger, S., et al. Sevoflurane ameliorates gas exchange and attenuates lung damage in experimental lipopolysaccharide-induced lung Injury. Anesthesiology. 111 (6), 1238-1248 (2009).
- Steurer, M., et al. The volatile anaesthetic sevoflurane attenuates lipopolysaccharide-induced injury in alveolar macrophages. Clinical and Experimental Immunology. 155 (2), 224-230 (2009).
- Englert, J. A., et al. Isoflurane Ameliorates Acute Lung Injury by Preserving Epithelial Tight Junction Integrity. Anesthesiology. 123 (2), 377-388 (2015).
- Li, Q. F., Zhu, Y. S., Jiang, H., Xu, H., Sun, Y. Isoflurane preconditioning ameliorates endotoxin-induced acute lung injury and mortality in rats. Anesthesia and Analgesia. 109 (5), 1591-1597 (2009).
- Reutershan, J., Chang, D., Hayes, J. K., Ley, K. Role of a reduction of cytokine levels in isoflurane-mediated protection from endotoxin-induced lung Injury. Anesthesiology. 105 (6), 1280-1281 (2006).
- Du, X., et al. Isoflurane promotes phagocytosis of apoptotic neutrophils through AMPK-mediated ADAM17/Mer signaling. PloS One. 12 (7), 0180213 (2017).
- Jabaudon, M., et al. Sevoflurane for Sedation in Acute Respiratory Distress Syndrome. A Randomized Controlled Pilot Study. American Journal of Respiratory and Critical Care Medicine. 195 (6), 792-800 (2017).
- Yue, T., et al. Postconditioning with a volatile anaesthetic in alveolar epithelial cells in vitro. The European Respiratory Journal: Official Journal of the European Society for Clinical Respiratory Physiology. 31 (1), 118-125 (2008).
- Blondonnet, R., Constantin, J. M., Sapin, V., Jabaudon, M. A pathophysiologic approach to biomarkers in acute respiratory distress syndrome. Disease Markers. 2016, 3501373 (2016).
- Farrell, R., Oomen, G., Carey, P. A technical review of the history, development and performance of the anaesthetic conserving device “AnaConDa” for delivering volatile anaesthetic in intensive and post-operative critical care. Journal of Clinical Monitoring and Computing. 32 (4), 595-604 (2018).
- Sturesson, L. W., Bodelsson, M., Jonson, B., Malmkvist, G. Anaesthetic conserving device AnaConDa: dead space effect and significance for lung protective ventilation. British Journal of Anaesthesia. 113 (3), 508-514 (2014).
- Blondonnet, R., et al. RAGE inhibition reduces acute lung injury in mice. Scientific Reports. 7 (1), 7208 (2017).
- Jabaudon, M., et al. Soluble receptor for advanced glycation end-products predicts impaired alveolar fluid clearance in acute respiratory distress syndrome. American Journal of Respiratory and Critical Care Medicine. 192 (2), 191-199 (2015).
- Jabaudon, M., et al. Soluble forms and ligands of the receptor for advanced glycation end-products in patients with acute respiratory distress syndrome: An observational prospective study. PloS One. 10 (8), 0135857 (2015).
- Kilkenny, C., Browne, W. J., Cuthill, I. C., Emerson, M., Altman, D. G. Improving bioscience research reporting: The ARRIVE guidelines for reporting animal research. PLoS Biology. 8, 1000412 (2010).
- Audard, J., et al. Inhibition of the receptor for advanced glycation end-products in acute respiratory distress syndrome: A randomised laboratory trial in piglets. Scientific Reports. 9 (1), 9227 (2019).
- Wu, C. W., et al. Intra-operative neural monitoring of thyroid surgery in a porcine model. Journal of Visualized Experiments. (144), e57919 (2019).
- Russ, M., et al. Lavage-induced surfactant depletion in pigs as a model of the acute respiratory distress syndrome (ARDS). Journal of visualized experiments. (115), e53610 (2016).
- Marumo, C. K., et al. Hemodynamic effects of PEEP in a porcine model of HCl-induced mild acute lung injury. Acta Anaesthesiologica Scandinavica. 53 (2), 190-202 (2009).
- Ambrosio, A. M., et al. Effects of positive end-expiratory pressure titration and recruitment maneuver on lung inflammation and hyperinflation in experimental acid aspiration-induced lung injury. Anesthesiology. 117 (6), 1322-1334 (2012).
- Sackey, P. V., Martling, C. R., Granath, F., Radell, P. J. Prolonged isoflurane sedation of intensive care unit patients with the Anesthetic Conserving Device. Critical Care Medicine. 32 (11), 2241-2246 (2004).
- Blanchard, F., et al. Minimal alveolar concentration for deep sedation (MAC-DS) in intensive care unit patients sedated with sevoflurane: A physiological study. Anaesthesia, Critical Care & Pain. , (2020).
- Verghese, G. M., Ware, L. B., Matthay, B. A., Matthay, M. A. Alveolar epithelial fluid transport and the resolution of clinically severe hydrostatic pulmonary edema. Journal of Applied Physiology. 87 (4), 1301-1312 (1999).
- Sakuma, T., et al. Alveolar fluid clearance in the resected human lung. American Journal of Respiratory and Critical Care Medicine. 150 (2), 305-310 (1994).
- Matthay, M. A., Wiener-Kronish, J. P. Intact epithelial barrier function is critical for the resolution of alveolar edema in humans. The American Review of Respiratory Disease. 142 (6), 1250-1257 (1990).
- Ware, L. B., Matthay, M. A. Alveolar fluid clearance is impaired in the majority of patients with acute lung injury and the acute respiratory distress syndrome. American Journal of Respiratory and Critical Care Medicine. 163 (6), 1376-1383 (2001).
- Ware, L. B., Golden, J. A., Finkbeiner, W. E., Matthay, M. A. Alveolar epithelial fluid transport capacity in reperfusion lung injury after lung transplantation. American Journal of Respiratory and Critical Care Medicine. 159 (3), 980-988 (1999).
- Constantin, J. M., et al. Response to recruitment maneuver influences net alveolar fluid clearance in acute respiratory distress syndrome. Anesthesiology. 106 (5), 944-951 (2007).
- Kemming, G. I., et al. Effects of perfluorohexan vapor on gas exchange, respiratory mechanics, and lung histology in pigs with lung injury after endotoxin infusion. Anesthesiology. 103 (3), 585-594 (2005).
- Matute-Bello, G., et al. An official American Thoracic Society workshop report: features and measurements of experimental acute lung injury in animals. American Journal of Respiratory Cell and Molecular Biology. 44 (5), 725-738 (2011).
- Chiew, Y. S., et al. Physiological relevance and performance of a minimal lung model: an experimental study in healthy and acute respiratory distress syndrome model piglets. BMC Pulmonary Medicine. 12, 59 (2012).
- Hochhausen, N., et al. Optimizing PEEP by Electrical Impedance Tomography in a Porcine Animal Model of ARDS. Respiratory Care. 62 (3), 340-349 (2017).
- Fu, H., Sun, M., Miao, C. Effects of different concentrations of isoflurane pretreatment on respiratory mechanics, oxygenation and hemodynamics in LPS-induced acute respiratory distress syndrome model of juvenile piglets. Experimental Lung Research. 41 (8), 415-421 (2015).
- Yehya, N. Lessons learned in acute respiratory distress syndrome from the animal laboratory. Annals of Translational Medicine. 7 (19), 503 (2019).
- Hochhausen, N., et al. Comparison of two experimental ARDS models in pigs using electrical impedance tomography. PloS One. 14 (11), 0225218 (2019).
- Shaver, C. M., et al. Cell-free hemoglobin: a novel mediator of acute lung injury. American Journal of Physiology. Lung Cellular and Molecular Physiology. 310 (6), 532-541 (2016).
- Matthay, M. A., et al. Acute respiratory distress syndrome. Nature Reviews. Disease Primers. 5 (1), 18 (2019).
- Martin, T. R., Matute-Bello, G. Experimental models and emerging hypotheses for acute lung injury. Critical Care Clinics. 27 (3), 735-752 (2011).
- Light, R. W., Gary Lee, Y. C. . Textbook of Pleural Diseases Second Edition. , (2008).
- Laferriere-Langlois, P., d’Arogon, F., Manzanares, W. Halogenated volatile anesthetics in the intensive care unit: current knowledge on an upcoming practice. Minerva Anestesiologica. 83 (7), 737-748 (2017).
- Devlin, J. W., et al. Clinical Practice Guidelines for the Prevention and Management of Pain, Agitation/Sedation, Delirium, Immobility, and Sleep Disruption in Adult Patients in the ICU. Critical Care Medicine. 46 (9), 825-873 (2018).
- DAS-Taskforce 2015 et al. Evidence and consensus based guideline for the management of delirium, analgesia, and sedation in intensive care medicine. Revision 2015 (DAS-Guildeline 2015) – short version. German Medical Science: GMS e-journal. 13, (2015).
- O’Gara, B., Talmor, D. Lung protective properties of the volatile anesthetics. Intensive Care Medicine. 42 (9), 1487-1489 (2016).
- Murthy, S., Gomersall, C. D., Fowler, R. A. Care for critically ill patients with COVID-19. JAMA: The Journal of the American Medical Association. , (2020).
- Liao, X., Wang, B., Kang, Y. Novel coronavirus infection during the 2019-2020 epidemic: preparing intensive care units-the experience in Sichuan Province, China. Intensive Care Medicine. 46 (2), 357-360 (2020).
- Grasselli, G., Pesenti, A., Cecconi, M. Critical care utilization for the COVID-19 outbreak in Lombardy, Italy: Early experience and forecast during an emergency response. JAMA: The Journal of the American Medical Association. , (2020).
- Arentz, M., et al. Characteristics and outcomes of 21 critically ill patients with COVID-19 in Washington State. JAMA: The Journal of the American Medical Association. , (2020).
- Xu, Z., et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. The Lancet. Respiratory Medicine. 8 (4), 420-422 (2020).
- Ferrando, C., et al. but not propofol, reduces the lung inflammatory response and improves oxygenation in an acute respiratory distress syndrome model: a randomised laboratory study. European Journal of Anaesthesiology. 30 (8), 455-463 (2013).
- Voigtsberger, S., et al. Sevoflurane ameliorates gas exchange and attenuates lung damage in experimental lipopolysaccharide-induced lung injury. Anesthesiology. 111 (6), 1238-1248 (2009).
- Suter, D., et al. The immunomodulatory effect of sevoflurane in endotoxin-injured alveolar epithelial cells. Anesthesia and Analgesia. 104 (3), 638-645 (2007).
- Steurer, M., et al. The volatile anaesthetic sevoflurane attenuates lipopolysaccharide-induced injury in alveolar macrophages. Clinical and Experimental Immunology. 155 (2), 224-230 (2009).
- Blondonnet, R., et al. In vitro method to control concentrations of halogenated gases in cultured alveolar epithelial cells. Journal of Visualized Experiments. (144), e58554 (2018).
- Gargiulo, S., et al. Mice anesthesia, analgesia, and care, Part I: Anesthetic considerations in preclinical research. ILAR Journal / National Research Council, Institute of Laboratory Animal Resources. 53 (1), 55-69 (2012).

