A Simplified Stepwise Approach to Echo Guidance during Percutaneous Mitral Valve Repair
Summary
This protocol presents in detail how to perform real-time echocardiographic guidance during transcatheter mitral valve repair. The fundamental views and the necessary measurements are described for each stage of the procedure.
Abstract
Percutaneous transcatheter edge-to-edge reconstruction of the mitral valve is a safe and well-established therapy for severe symptomatic mitral regurgitation in patients with high surgical risk. Echocardiographic guidance in addition to fluoroscopy is the gold standard and should be performed using a standardized technique.
This article lays out our reproducible step by step echocardiographic guide including views, measurements as well as highlighting possible difficulties that may arise during the procedure.
This article provides detailed and chronological echocardiographic views for each step of the procedure, especially preferences between 2D and 3D imaging. If needed, pulse wave, continuous wave and color doppler measurements are described. Furthermore, as there are no official recommendations for the quantification of mitral regurgitation during the percutaneous edge-to-edge-repair procedure, advice is also included for echocardiographic quantification after grasping the mitral leaflets and after device deployment. In addition, the article deals with important echocardiographic views to prevent and deal with possible complications during the procedure.
Echocardiographic guidance during transcatheter mitral valve repair is mandatory. A structured approach improves the collaboration between interventionist and imager and is indispensable for a safe and effective procedure.
Introduction
Mitral regurgitation (MR) is the second-most frequent indication for valve surgery in Europe1. Untreated, it may lead to severe heart failure and reduced quality of life2,3,4. The percutaneous mitral valve repair (PMVR) is a catheter-based technique, which mimics the Alfieri stitch surgical method to mitral repair by connecting the A2 and P2 scallops5. For patients with high surgical risk, this technique offers a minimally invasive approach for the treatment of severe MR. Data from several registries and trials has shown that the MitraClip procedure, a transcatheter mitral valve repair therapy, is an effective and safe method6,7,8,9. In 2019, a similar device, the PASCAL transcatheter valve repair system, was introduced to the market. It has shown feasibility and acceptable safety in the treatment of patients with severe MR10. The duration and success of the PMVR are dependent on the individual operator's skill and experience11. In contrast to other percutaneous techniques, like the percutaneous transvalvular replacement (TAVR), which can be done with fluoroscopy only, PMVR requires echocardiographic guidance12,13.
This article describes step by step the echocardiographic approach during PMVR, including measurements, suggestions for intraprocedural quantification of the MR and important views to prevent periprocedural complications.
Protocol
The protocol follows the guidelines of our's institution's human research ethics committee.
1. Evaluation before intervention
- Exclude pericardial effusion before transseptal puncture. If a small pericardial effusion is present, measure the maximal end-diastolic echolucent space in a four-chamber (4Ch) view with a focus on the right ventricle (RV), a mid-oesophageal right ventricular inflow-outflow view, and a long-axis (LAX) view.
- Evaluate the pulmonary venous flow pattern with pulse wave doppler (PW) in the left upper pulmonary vein (LUPV) and exclude thrombus formation in the left atrial appendage (LAA). Show the short axis (SAX) view with focus on the LAA, sweep then at 40-60° and rotate the probe anti-clockwise to show the LUPV. Assess the flow in the right upper pulmonary vein (RUPV) by sweeping at 90-110° (Figure 1 and Supplementary Figure 1).
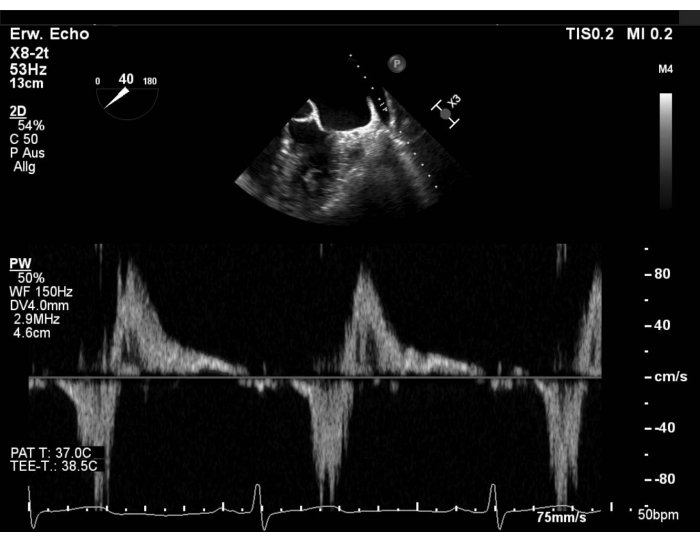
Figure 1: Modified SAX view: PW flow in the left upper pulmonary vein Please click here to view a larger version of this figure.
- Ensure hemodynamic state is the same during pre- and post-procedural evaluation.
NOTE: AS MR is a dynamic valve disease, regurgitation may seem less severe under general anaesthesia. In this case, consult the operator and increase the afterload and/or the preload. - Find the best intercommissural view (50-70°). Take a perpendicular view (X-plane) in the three segments with and without color doppler and measure the length of the posterior mitral leaflet (PML). Then, check the leaflet morphology again (Figure 2 and Supplementary Figure 2).
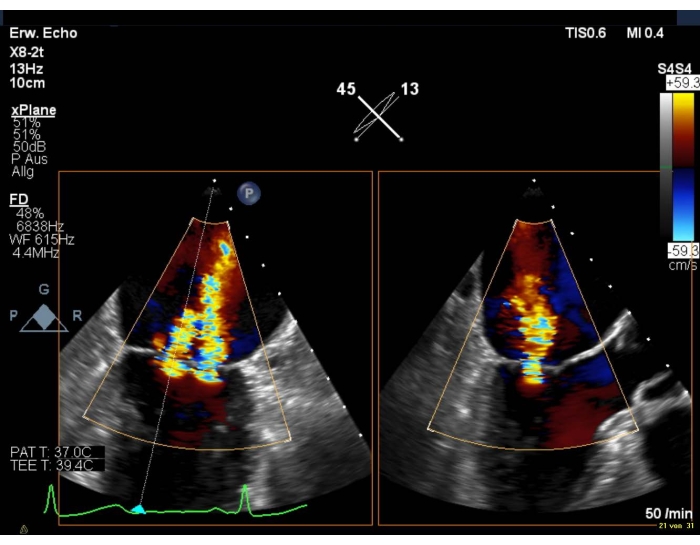
Figure 2: 2D biplanar view of the MV with color doppler: medial insufficiency jet Please click here to view a larger version of this figure.
- Assess the transmitral pressure gradient with continuous wave doppler (CW) in the long axis view (120-140°).
NOTE: A mean pressure gradient (MPG) > 5 mmHg is a relative contraindication for PMVR. - Take a 3D dataset with color doppler or a wide-sector zoom image with color and measure the 3D-vena contracta (3D-VCA) (Figure 3).
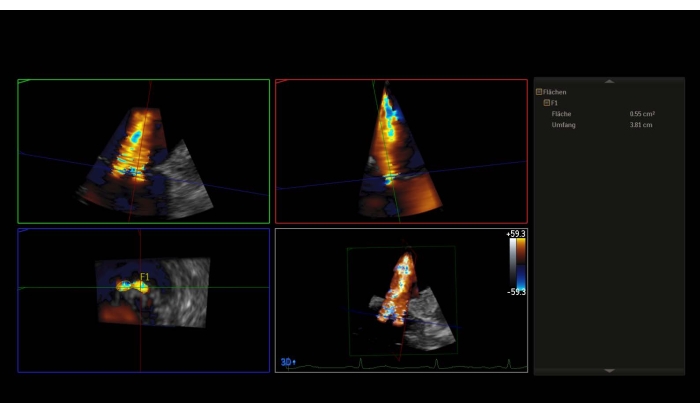
Figure 3: Multiplanar reconstruction of the 3D dataset with color doppler: 3D-Vena contracta Please click here to view a larger version of this figure.
- Without color, use the 3D volume to measure the mitral valve area (MVA) (Supplementary Figure 3).
NOTE: An area < 4 cm2 is a relative contraindication, an area < 3 cm2 an absolute contraindication to perform the procedure. Otherwise assess the MVA in the transgastric basal SAX view. - Show the 3D en-face surgical atrial view (aortic valve at 12 o'clock) of the mitral valve.
NOTE: The segments of the valve are named "lateral" for the segments 1 and "medial" for the segments 3. The sequence of the segments in the en-face surgical view, is inverse to the sequence in the commissural view. Perform a 180° clockwise rotation into the 3D en-face surgical view (aortic valve at 6 o'clock), that will result in an equal sequence of segments in both views (Figure 4 and Figure 5).
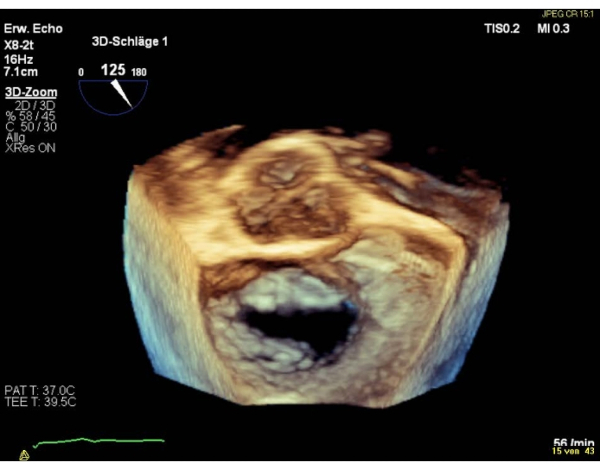
Figure 4: Wide sector zoom image: 3D en-face surgical atrial view (aortic valve at 12 o'clock) Please click here to view a larger version of this figure.
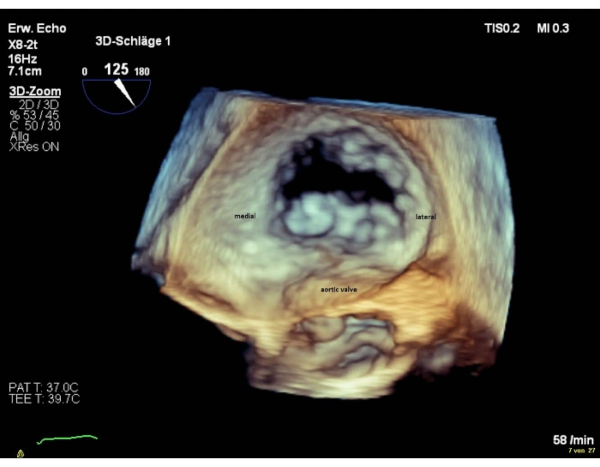
Figure 5: Wide sector zoom image: 3D en-face atrial view (aortic valve at 6 o'clock) Please click here to view a larger version of this figure.
- Finally, take a bicaval view (90-110°) with X-plane, to show the aortic valve (AV), for the transseptal puncture.
2. Strategy
- Discuss the strategy with the operator before inserting the steerable guide catheter (SGC) and the clip delivery system (CDS) into the left atrium.
- Evaluate a one device strategy if the orifice is < 1 cm wide and position the clip directly above the regurgitation jet if the orifice is circular.
- Evaluate the implantation of ≥ 2 clips in case of large elliptic or multiple jets. Implant the device starting medially of the regurgitant orifice, as the positioning of a second device is often easier when the first has been implanted in this way, rather than after starting laterally (Supplementary Figure 4).
3. Transseptal puncture
- Show a bicaval view combined with a SAX view. Ensure the AV is visible, to avoid aortic injury.
- Make sure the puncture site is slightly superior and posterior (Figure 6).
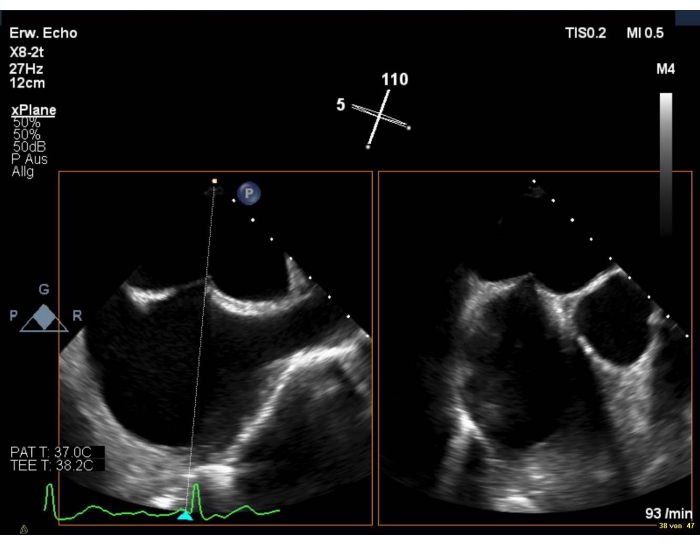
Figure 6: 2D biplanar view: transseptal puncture Please click here to view a larger version of this figure.
- Choose a puncture height of 4-5 cm in case of degenerative MR (e.g., prolapse) and of > 3.5 in functional MR. Avoid a patent foramen ovale, as the entry is far too anterior.
- Once the transseptal needle leads to tenting of the interatrial septum, measure the puncture height in the 4Ch view in mid-systole (Supplementary Figure 5).
NOTE: In patients with large atria, if the puncture site is too posterior the tenting cannot be visualized in the 4Ch view. In this case, retroflex and insert the probe deeper into the oesophagus. - After transseptal puncture, always exclude pericardial effusion in the 4Ch view.
- Show a SAX view with a focus on the LAA and pulmonary vein to visualize the entering of the stiff guidewire into the LUPV.
4. Introduction of the SGC into the LA
- Visualize the tenting and advancement of the SGC with the dilator in a SAX view with continuous 2D-echocardiography and fluoroscopic guidance to avoid injuries to the left atrial wall.
NOTE: The tip of the SGC is defined by a radiopaque and echo bright double ring (Supplementary Figure 6). - Show the operator the SAX view and the bicaval view (90-120°) to position the SGC in the direction of the left ventricle (LV).
5. Advancement of the CDS into the LA
- Take a 3D-volume including the interatrial septum, the left lateral ridge and the MV and ensure that the left lateral ridge is visible because protrusion of the CDS is common (Figure 7).
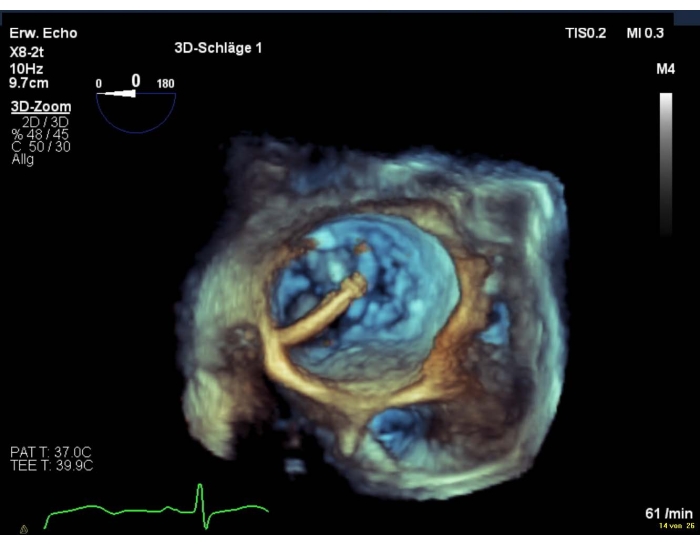
Figure 7: Wide sector zoom image: SGC in the LA including the interatrial septum, the left lateral ridge and the MV Please click here to view a larger version of this figure.
- Otherwise choose the SAX view and the LAX view (X-plane) to ensure, the CDS does not have contact with the ridge and the LA-wall.
NOTE: The operator may ask the imager to show the interatrial septum and pull back the SGC a few millimetres to bypass the ridge. If the double ring in 3D cannot be visualized, switch to 2D and show the SGC in the SAX view.
- Check that the CDS is positioned perpendicularly to the coaptation line to guarantee a correct trajectory.
- Show the intercommissural view in 2D at ca. 60° to display the medial – lateral plane and the LAX view at 120-140° to identify the anterior – posterior plane of the MV (Supplementary Figure 7).
- Alternatively optimize the medial, lateral, anterior, and posterior positioning of the CDS in the 3D en-face view (Supplementary Figure 8).
6. Orientation of the device above and below the MV
- Take the 3D en-face view to show a perpendicular positioning of the arms to the coaptation line.
- In the event of poor image quality, show an intercommissural view combined with a LAX view (Figure 8 and Figure 9).
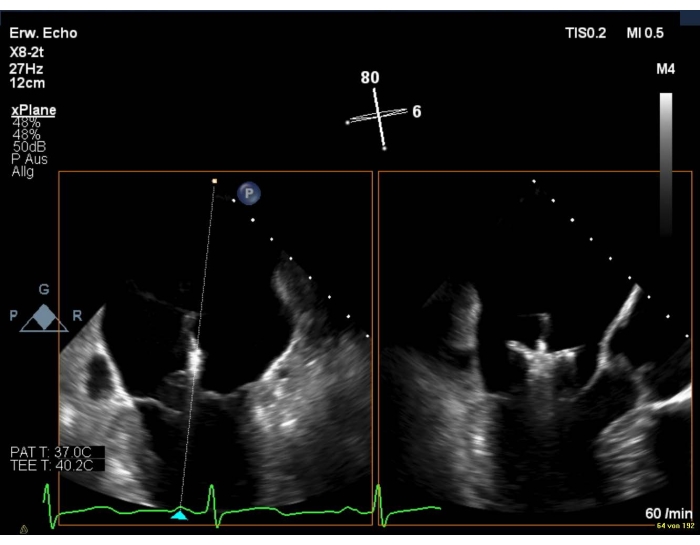
Figure 8: 2D biplanar view of the MV: positioning of the device over the mitral valve Please click here to view a larger version of this figure.
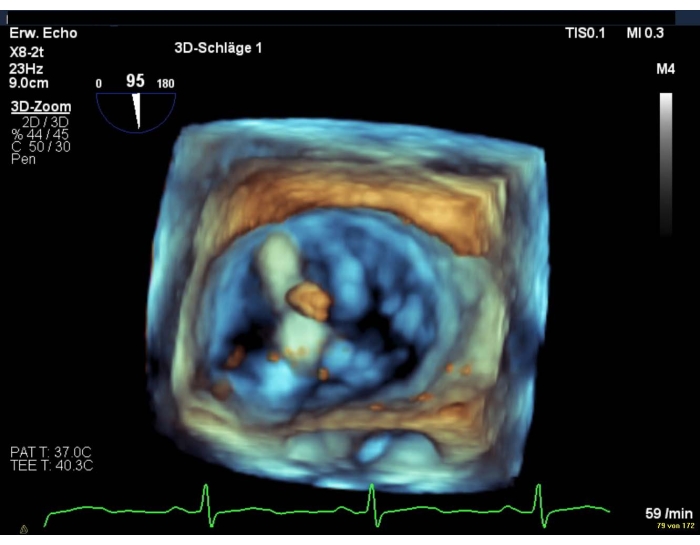
Figure 9: Wide sector zoom image: positioning of the device over the mitral valve Please click here to view a larger version of this figure.
NOTE: The clip arms are visible only in the LAX view.
- Adjust the intercommissural view angle for medially and laterally positioned devices to visualise the complete length of both arms. Sweep at ca. 30-45° for medially positioned devices and ca. 70-90° for laterally positioned devices.
- Choose the intercommissural view combined with a LAX view to visualize the advancing of the CDS into the LV.
- Ensure the CDS is placed just a few millimetres below the MV.
- Verify in the 3D en-face view that the clip arms are still in the planned position, as a rotation of the clip while crossing the valve is frequent.
NOTE: If the position of the clip arms has changed, clockwise or anticlockwise rotation will be performed to obtain symmetric grasping. Be careful during this manoeuvre to minimize chordal and subchordal entanglement. - If gross reorientation of the device is necessary, show the intercommissural view with X-plane to visualize the inversion of the clip that will be retrieved into the LA.
7. Grasping of the mitral leaflets and assessment of MR before and after clip deployment
- Record the grasping of the leaflets in the intercommissural view combined with the LAX view (X-plane) or in the LAX view only (Supplementary Figure 9).
- Ask the anaesthesiologist to perform a breath-hold manoeuvre to reduce shifting during ventilation and to facilitate the grasping of the leaflets.
- Ensure continuous visualisation of leaflet insertion to avoid rolling of the leaflets or the chordae.
NOTE: Grasping a rolled leaflet or a chorda may result in a partial leaflet detachment and/or an aggravation of the MR. - Carefully evaluate the regurgitation reduction before clip deployment. Ensure both, operator and imager analyse this crucial step.
- Rotate the TEE probe medially and laterally to the clip or use X-plane with color doppler to find eccentric jets close to the clip (Supplementary Figure 10).
NOTE: Due to shadowing artifacts caused by the CDS underestimation of the MR may occur. Insert the probe deeper into the esophagus or show the transgastric view to visualize the residual insufficiency jets without shadowing artifacts. - Evaluate the PW flow in the pulmonary veins.
NOTE: if a previous systolic flow reversal changes into a systolic dominant pattern, a relevant reduction has probably occurred. - Measure the MPG across the mitral valve.
NOTE: A gradient > 5 mmHg is a relative contraindication for clip deployment (Supplementary Figure 11 and Supplementary Figure 12). - Use the 3D en-face view of the MV or a transgastric SAX view of the MV to show the double orifice (Figure 10).
- Rotate the TEE probe medially and laterally to the clip or use X-plane with color doppler to find eccentric jets close to the clip (Supplementary Figure 10).
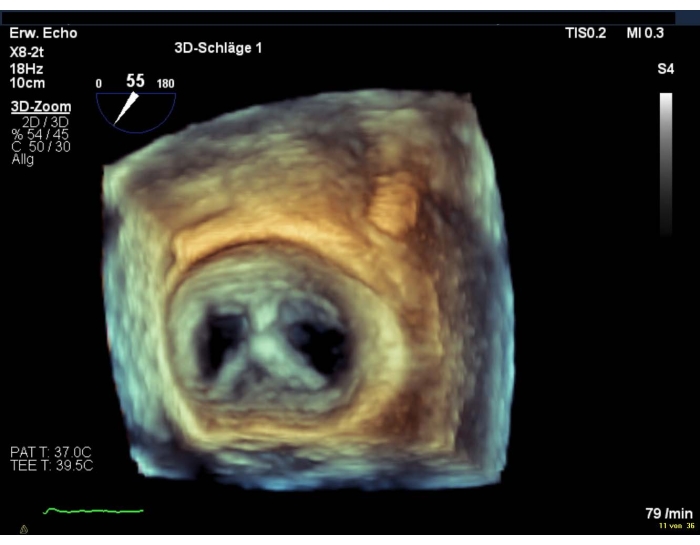
Figure 10: Wide sector zoom image: double orifice of the MV after device deployment Please click here to view a larger version of this figure.
- Finally, if the result is satisfactory, check leaflet insertion in 2D.
- After releasing the clip from the CDS, repeat the last five steps.
NOTE: Due to the tension of the system on the MV, the residual insufficiency jets after releasing the device may be aggravated. - Show when the delivery catheter tip is retrieved from the SGC in the LAX view with X-plane and ensure that the spike avoids contact with the LA (Supplementary Figure 13).
8. Final MR assessment
- Show the intercommissural view with color doppler in combination with perpendicular X-planes in the residual insufficiency jets if present.
- Calculate the 3D-VCA in a 3D-volume. NOTE: usually the orifices are not in the same plane. In this case measure separated planimetries of each orifice in the appropriate planes (Supplementary Figure 14).
- Evaluate once again the pulmonary vein flow and the mean gradient across the mitral valve.
NOTE: continuous LA pressure monitoring may be a useful tool during transcatheter mitral valve repair. - Finally, show the 3D en-face view of the mitral valve.
9. Implantation of additional devices
- Ensure MR reduction is sufficient.
NOTE: If the result is not satisfactory, evaluate the implantation of additional devices. - Ensure that the additional device does not contact the implanted device diving into the left ventricle.
NOTE: fluoroscopy is essential to show the real distance between the clips. - Show the 3D en-face view to visualize the commissural line, as it might be deferred after implantation of the first clip.
- Repeat the five steps as explained in point 7.4 to evaluate MR after grasping the leaflets with the additional clip.
Representative Results
Percutaneous edge-to-edge valve repair is an alternative to surgical valve repair or replacement in patients not eligible for surgery with symptomatic severe MR. The first clinical application of the MitraClip was investigated in the Endovascular Valve Edge-to-Edge Repair Study I (EVEREST I)14 trial. Many other trials have proven the effectiveness of the procedure with an improvement of symptoms as well as low rates of hospital mortality and adverse events (ACCESS EU, TRAMI, EVEREST II)15,16,17. Percutaneous edge-to-edge procedures have been incorporated into the European guidelines for the intervention of primary and secondary MR18. Two randomized trials MITRA-FR and COAPT compared the MitraClip procedure with optimal medical therapy and verified the feasibility of this intervention in patients with secondary MR. Although MITRA-FR did not show any significant benefit for the MitraClip group concerning the composite endpoint (all-cause mortality and rehospitalization for heart failure) at 12 months19 (Supplementary Figure 15), COAPT showed significant superiority of the MitraClip in terms of mortality and rehospitalization rates, compared to the conservative treatment alone at 24 months20 (Supplementary Figure 16). Compared with patients in the COAPT trial, those enrolled in the MITRA-FR trial had substantially more left ventricular damage and less severe MR due to the use of different classifications of MR (Supplementary Figure 17). To classify severe MR MITRA-FR used the 2012 European guidelines21, whereas COAPT used the 2006/2008 American guidelines22, which may explain the different outcomes observed in these two trials.
Since 2019, another device for transcatheter mitral valve repair has become available. Compared to the MitraClip device, the PASCAL system has wider paddles. It can independently grasp leaflets, as the fourth generation MitraClip, in the presence of a central spacer. After a multi-centre, prospective, observational, first-in-man study23 and the multi-centre early-feasibility trial of the PASCAL transcatheter mitral repair device (CLASP study), the PASCAL repair system received CE mark approval for the treatment of both primary and secondary MR. The early feasibility study of the PASCAL repair system showed high survival, low complication rates as well as improvements in functional status and quality of life (Supplementary Figure 18). Furthermore, the CLASP study showed a significant reduction of the MR with this device. As there are no head-to-head studies available yet, the preferred device related to the valve anatomy (e.g., larger flail segments, large prolapse, cleft, short posterior leaflet, mitral annular calcification) is still not defined. A head-to-head comparison trial of the MitraClip with the Pascal System (CLASP IID trial) started in 2018 and the estimated primary completion date is expected to be in 2023.
A critical appraisal of echocardiographic assessment of MR, especially a functional one, is still to be done.
Because of its complex structure, the MV is altered by dynamic changes24 and quantification of MR requires an understanding of the normal valve anatomy. The echocardiographic guidelines for the quantification of native valvular regurgitation have been updated in 2017.25 Official recommendations for the quantification of valve regurgitation after percutaneous valve repair have been available since 2019.26 Due to missing validation of 2D vena contracta and EROA in multiple jets, neither 2D vena contracta nor EROA and regurgitant volume/fraction by PISA is recommended after PMVR (Supplementary Figure 19). Thresholds for 3D-VCA were first introduced and 3D-VCA gained a relevant role for the quantification of valve regurgitations which is superior to the 2D PISA method27,28. 3D-VCA is highly dependent on good image quality. Therefore, semi-quantitative parameters such as the signal intensity of the CW doppler of the MR-jet, the pulmonary vein flow pattern and the mitral inflow pattern are still necessary29.
Although the utility of 3D-VCA has not yet been fully validated to assess MR, further development of 3D technologies will probably improve its value.
Continuous hemodynamic monitoring may complement transesophageal echocardiography to assess and optimize percutaneous edge-to-edge mitral valve repair. Previous studies demonstrate the value of real-time monitoring of LA pressure during PMVR30,31,32,33. An increase in left atrial mean pressure is predictive of worse clinical outcomes at short-term follow-up, independent from echocardiographic findings.
Supplementary Figure 1: Modified bicaval view: PW flow in the right upper pulmonary vein Please click here to download this File.
Supplementary Figure 2: 2D biplanar view of the MV: length of the PML Please click here to download this File.
Supplementary Figure 3: Multiplanar reconstruction of the 3D dataset: mitral valve area in 3D Please click here to download this File.
Supplementary Figure 4: 2D biplanar view of the MV with color doppler: medially implanted device and residual lateral insufficiency jet Please click here to download this File.
Supplementary Figure 5: 4Ch view: puncture height Please click here to download this File.
Supplementary Figure 6 SAX view in 2D: SGC with echo bright double ring Please click here to download this File.
Supplementary Figure 7: 2D biplanar view of the MV: advancement of the CDS into the LA Please click here to download this File.
Supplementary Figure 8: Wide sector zoom image: advancement of the CDS into the LA Please click here to download this File.
Supplementary Figure 9: 2D biplanar view of the MV: grasping of the leaflets Please click here to download this File.
Supplementary Figure 10: 2D biplanar view of the MV with color doppler: residual insufficiency jets Please click here to download this File.
Supplementary Figure 11: LAX in 2D: transmitral pressure gradient before device deployment Please click here to download this File.
Supplementary Figure 12: Intercommissural view in 2D: transmitral pressure gradient after device deployment Please click here to download this File.
Supplementary Figure 13: 2D biplanar view of the MV: delivery catheter tip Please click here to download this File.
Supplementary Figure 14: Multiplanar reconstruction of the 3D dataset with color doppler: 3D-VCA after device deployment Please click here to download this File.
Supplementary Figure 15: MITRA-FR Trial: Kaplan-Meier Estimates of Survival without a Primary Outcome Event Please click here to download this File.
Supplementary Figure 16: COAPT Trial: Primary Effectiveness and Safety End Points and Death Please click here to download this File.
Supplementary Figure 17: MITRA-FR vs. COAPT: Similarities and differences between MITRAFR and COAPT with respect to baseline characteristics of the study populations Please click here to download this File.
Supplementary Figure 18: CLASP Study: Functional and Clinical Paired Analyses at Baseline and 30 Days for the PASCAL Transcatheter Valve Repair System Please click here to download this File.
Supplementary Figure 19: Guidelines for the Evaluation of Valvular Regurgitation After Supplementary Percutaneous Valve Repair or Replacement: Echocardiographic parameters and related comments in the assessment of MR severity with TTE after transcatheter MV interventions Please click here to download this File.
Discussion
Echo-guidance for the PMVR is a safe method. Complications due to echocardiography may occur but rarely lead to significant damage. Nevertheless, esophageal lesions are possible after performing transesophageal echocardiography. This incidence is reduced with a shorter duration of the intervention34. On the contrary, several complications associated with the transcatheter edge-to-edge mitral valve repair are described35,36. Major bleeding is the most frequent major complication and blood transfusion after transcatheter valve procedures have been strongly associated with mortality37. Pericardial tamponade is rare and may occur during several steps of the procedure. Caution during transseptal puncture, insertion of the SGC into the left atrium and retrieving of the SGC-tip after clip deployment reduces the probability of this life-threatening complication. Rates of clip-specific complications, as embolization and partial clip detachment, are low and can be reduced by an accurate echocardiographic assessment during and after grasping of the leaflets.
Despite the carefulness during echocardiography, aggravation of the MR, may occur. Additional clip deployment in the medial and lateral position may increase the risk of entangling and rupturing of the chordae38. Furthermore, repeated grasping of the leaflets can lead to leaflet laceration and perforation. Residual MR is not only a determinant of procedural success but is also associated with poorer prognosis39,40, especially when there is a concomitant transmitral pressure gradient >5 mm Hg41,42. Clear communication with the interventionist and a meticulous echocardiographic assessment are necessary to reduce peri-procedural clip-related complications.
The percutaneous edge-to-edge repair is not eligible for every pathomechanism of MR. For some determinants of MR, such as annulus dilatation, rupture of the chordae tendinae or severe calcifications of the mitral leaflets, the device reaches its limits. In this case, mitral ring dilation can be sutured surgically or percutaneously in place to provide annular stabilization. Polytetrafluoroethylene (PTFE) neochordae can be surgically sewed as new chordae tendinee. A MV with severe calcified leaflets may be replaced with a prosthetic valve. New percutaneous devices designed to address these aspects are under development.
A structured echocardiographic approach for the PMVR is mandatory. Several techniques of how to perform echoguidance for the PMVR procedure have been described43,44,45,46,47,48. In our opinion, it is important to define a detailed sequence of steps together with the interventionist, which should be performed during every intervention. Although the 3D en-face view is defined showing the aortic valve at 12 o'clock, many centres prefer the "cardiologist" 3D en-face view, which shows the aortic valve at 6 o'clock. There is no literature comparing both views during the PMVR. Several options for the assessment of specific views in 2D or 3D as well as for the quantification of MR are available. 3D transesophageal echocardiography allows the measurement of each orifice after edge-to-edge repair. The guidelines propose thresholds for 3D-VCA as <0,2cm2 for mild, 0,2-0,39 cm2 for moderate and ≥ 0,4 cm2 for severe regurgitation26. Previous studies have shown that improvement of clinical symptoms correlates to a significant reduction of the 3D-VCA, independently from an absolute value49. Furthermore, reduction of left atrial and ventricular volume is significantly greater in patients who show a reduction in 3D-VCA of more than a 50% after MV repair50. Our experience confirms this data. Nevertheless, the percentual reduction of the 3D-VCA required to significantly improve a severe MR has not been defined. Further validation of this method is needed and the assessment of other parameters for the adequate evaluation of residual MR remains indispensable. Due to possible mild mitral stenosis caused after clip implantation, the MV inflow pattern rarely gives additional information to the MR reduction. On the contrary, the pulmonary flow pattern is a reliable parameter and predicts recurrent MR and worse long-term outcomes.
In conclusion, percutaneous transcatheter mitral valve repair is fully dependent on transesophageal echocardiography. Both, the imager and the interventionist should have a fundamental understanding of echocardiographic assessment to achieve a successful and safe procedure.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank Ms. Dorothea Scheurlen for technical video support.
Materials
| EPIQ 7 Ultrasound System | Philips | US218B0542 | Cardiac Ultrasound Machine |
| X8-2t xMATRIX 3D-TEE probe | Philips | B34YYK | TEE-probe |
| Sheath 6F 25 cm | Merit Medical | B60N25AQ | Sheath |
| Dilator 16 F | Abbott | 405544 | Dilator |
| BRK-1 transseptal needle 71 cm | St. Jude Medical | ABVA407201 | Transseptal Needle |
| Swartz Lamp 90° | St. Jude Medical | 407356 | Transseptal Guiding Introducer Sheath |
| Amplatz super stiff | Kook Medical | 46509 | Wire |
| Steerable Guide Catheter | Abbott | SGC0302 | Steerable Guide Catheter |
| MitraClip NTR Delivery System | Abbott | CDS0602-NTR | Clip Delivery System |
| MitraClip NTR Bundle | Abbott | MSK0602-NTR | Device |
References
- Iung, B., et al. A prospective survey of patients with valvular heart disease in Europe: The Euro Heart Survey on Valvular Heart Disease. European Heart Journal. 24 (13), 1231-1243 (2003).
- Hauptman, P. J., Rector, T. S., Wentworth, D., Kubo, S. Quality of life in advanced heart failure: role of mitral regurgitation. American Heart Journal. 151 (1), 213-218 (2006).
- Lewis, E. F. Assessing the impact of heart failure therapeutics on quality of life and functional capacity. Current Treatment Options in Cardiovascular Medicine. 15 (4), 425-436 (2013).
- Vaishnava, P., Lewis, E. F. Assessment of quality of life in severe heart failure. Current Heart Failure Reports. 4 (3), 170-177 (2007).
- Feldman, T., et al. Percutaneous repair or surgery for mitral regurgitation. New England Journal of Medicine. 364 (15), 1395-1406 (2011).
- Feldman, T., et al. Percutaneous mitral repair with the MitraClip system: safety and midterm durability in the initial EVEREST (Endovascular Valve Edge-to-Edge REpair Study) cohort. Journal of the American College of Cardiology. 54 (8), 686-694 (2009).
- Whitlow, P. L., et al. Acute and 12-month results with catheter-based mitral valve leaflet repair: the EVEREST II (Endovascular Valve Edge-to-Edge Repair) High Risk Study. Journal of the American College of Cardiology. 59 (2), 130-139 (2012).
- Baldus, S., et al. MitraClip therapy in daily clinical practice: initial results from the German transcatheter mitral valve interventions (TRAMI) registry. European Journal of Heart Failure. 14 (9), 1050-1055 (2012).
- Stone, G. W., et al. Transcatheter Mitral-Valve Repair in Patients with Heart Failure. New England Journal of Medicine. 379 (24), 2307-2318 (2018).
- Lim, D. S., et al. Transcatheter Valve Repair for Patients With Mitral Regurgitation: 30-Day Results of the CLASP Study. JACC: Cardiovascular Interventions. 12 (14), 1369-1378 (2019).
- Schillinger, W., et al. Impact of the learning curve on outcomes after percutaneous mitral valve repair with MitraClip and lessons learned after the first 75 consecutive patients. European Journal of Heart Failure. 13 (12), 1331-1339 (2011).
- Katz, W. E., Conrad Smith, A. J., Crock, F. W., Cavalcante, J. L. Echocardiographic evaluation and guidance for MitraClip procedure. Cardiovascular Diagnosis and Therapy. 7 (6), 616-632 (2017).
- Altiok, E., et al. Optimized guidance of percutaneous edge-to edge repair of the mitral valve using real-time 3-D transesophageal echocardiography. Clinical Research in Cardiology. 100 (8), 675-681 (2011).
- Feldman, T., et al. Percutaneous mitral valve repair using the edge-to-edge technique: six-month results of the EVEREST Phase I Clinical Trial. Journal of the American College of Cardiology. 46 (11), 2134-2140 (2005).
- Maisano, F., et al. Percutaneous mitral valve interventions in the real world: early and 1-year results from the ACCESS-EU, a prospective, multicenter, nonrandomized post-approval study of the MitraClip therapy in Europe. Journal of the American College of Cardiology. 62 (12), 1052-1061 (2013).
- Kalbacher, D., et al. Long-term outcome, survival and predictors of mortality after MitraClip therapy: Results from the German Transcatheter Mitral Valve Interventions (TRAMI) registry. International Journal of Cardiology. 277, 35-41 (2019).
- Glower, D. D., et al. Percutaneous mitral valve repair for mitral regurgitation in high-risk patients: results of the EVEREST II study. Journal of the American College of Cardiology. 64 (2), 172-181 (2014).
- Baumgartner, H., et al. 2017 ESC/EACTS Guidelines for the management of valvular heart disease. European Heart Journal. 38 (36), 2739-2791 (2017).
- Obadia, J. F., et al. MITRA-FR Investigators. Percutaneous Repair or Medical Treatment for Secondary Mitral Regurgitation. New England Journal of Medicine. 379 (24), 2297-2306 (2018).
- Stone, G. W., et al. COAPT Investigators. Transcatheter Mitral-Valve Repair in Patients with Heart Failure. New England Journal of Medicine. 379 (24), 2307-2318 (2018).
- Vahanian, A., et al. Joint Task Force on the Management of Valvular Heart Disease of the European Society of Cardiology (ESC); European Association for Cardio-Thoracic Surgery (EACTS), Guidelines on the management of valvular heart disease (version 2012). European Heart Journal. 33 (19), 2451-2496 (2012).
- American College of Cardiology/American Heart Association Task Force on Practice Guidelines; Society of Cardiovascular Anesthesiologists; Society for Cardiovascular Angiography and Interventions. ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 114 (5), 84 (2006).
- Praz, F., et al. Compassionate use of the PASCAL transcatheter mitral valve repair system for patients with severe mitral regurgitation: a multicentre, prospective, observational, first-in-man study. Lancet. 390 (10096), 773-780 (2017).
- McCarthy, K. P., Ring, L., Rana, B. S. Anatomy of the mitral valve: understanding the mitral valve complex in mitral regurgitation. European Journal of Echocardiography. 11 (10), 3-9 (2010).
- Zoghbi, W. A., et al. Recommendations for Noninvasive Evaluation of Native Valvular Regurgitation: A Report from the American Society of Echocardiography Developed in Collaboration with the Society for Cardiovascular Magnetic Resonance. Journal of the American Society of Echocardiography. 30 (4), 303-371 (2017).
- Zoghbi, W. A., et al. Guidelines for the Evaluation of Valvular Regurgitation Percutaneous Valve Repair or Replacement: A Report from the American Society of Echocardiography Developed in Collaboration with the Society for Cardiovascular Angiography and Interventions, Japanese Society of Echocardiography, and Society for Cardiovascular Magnetic Resonance. Journal of the American Society of Echocardiography. 32 (4), 431-475 (2019).
- Abudiab, M. M., Chao, C. J., Liu, S., Naqvi, T. Z. Quantitation of valve regurgitation severity by three-dimensional vena contracta area is superior to flow convergence method of quantitation on transesophageal echocardiography. Echocardiography. 34 (7), 992-1001 (2017).
- Dietl, A., et al. 3D vena contracta area after MitraClip procedure: precise quantification of residual mitral regurgitation and identification of prognostic information. Cardiovasc Ultrasound. 16 (1), 1 (2018).
- Ikenaga, H., et al. Usefulness of Intraprocedural Pulmonary Venous Flow for Predicting Recurrent Mitral Regurgitation and Clinical Outcomes After Percutaneous Mitral Valve Repair With the MitraClip. JACC: Cardiovascular Interventions. 12 (2), 140-150 (2019).
- Tang, G. H. L., et al. Continuous invasive hemodynamic monitoring using steerable guide catheter to optimize mitraclip transcatheter mitral valve repair: A multicenter, proof-of-concept study. Journal of Interventional Cardiology. 31 (6), 907-915 (2018).
- Maor, E., et al. Acute Changes in Left Atrial Pressure After MitraClip Are Associated With Improvement in 6-Minute Walk Distance. Circulation: Cardiovascular Interventions. 10 (4), 004856 (2017).
- Kuwata, S., et al. Continuous Direct Left Atrial Pressure: Intraprocedural Measurement Predicts Clinical Response Following MitraClip Therapy. JACC: Cardiovascular Interventions. 12 (2), 127-136 (2019).
- Corrigan, F. E., et al. Pulmonary Venous Waveforms Predict Rehospitalization and Mortality After Percutaneous Mitral Valve Repair. JACC Cardiovasc Imaging. 12 (10), 1905-1913 (2019).
- Ruf, T. F., et al. ELMSTREET (Esophageal Lesions during MitraClip uSing TRansEsophageal Echocardiography Trial. EuroIntervention. 13 (12), 1444-1451 (2017).
- Eggebrecht, H., et al. Risk and outcomes of complications during and after MitraClip implantation: Experience in 828 patients from the German TRAnscatheter mitral valve interventions (TRAMI) registry. Catheterization and Cardiovascular Interventions. 86 (4), 728-735 (2015).
- von Bardeleben, R. S., et al. Incidence and in-hospital safety outcomes of patients undergoing percutaneous mitral valve edge-to-edge repair using MitraClip: five-year German national patient sample including 13,575 implants. EuroIntervention. 14 (17), 1725-1732 (2019).
- Généreux, P., et al. Incidence, predictors, and prognostic impact of late bleeding complications after transcatheter aortic valve replacement. Journal of the American College of Cardiology. 64 (24), 2605-2615 (2014).
- Benito-González, T., Estévez-Loureiro, R., Gualis, J. Chordal Rupture Following MitraClip Implantation Resulting in Massive Mitral Regurgitation. Journal of Invasive Cardiology. 27 (10), 224-225 (2015).
- Kaneko, H., Neuss, M., Weissenborn, J., Butter, C. Impact of residual mitral regurgitation after MitraClip implantation. International Journal of Cardiology. 227, 813-819 (2017).
- Buzzatti, N., et al. What is a “good” result after transcatheter mitral repair? Impact of 2+ residual mitral regurgitation. Journal of Thoracic and Cardiovascular Surgery. 151 (1), 88-96 (2016).
- Feldman, T., Guerrero, M. Assessing the Balance Between Less Mitral Regurgitation and More Residual Transmitral Pressure Gradient After MitraClip. JACC: Cardiovascular Interventions. 10 (9), 940-941 (2017).
- Neuss, M., et al. Elevated Mitral Valve Pressure Gradient After MitraClip Implantation Deteriorates Long-Term Outcome in Patients With Severe Mitral Regurgitation and Severe Failure. JACC: Cardiovascular Interventions. 10 (9), 931-939 (2017).
- Katz, W. E., Conrad Smith, A. J., Crock, F. W., Cavalcante, J. L. Echocardiographic evaluation and guidance for MitraClip procedure. Cardiovascular Diagnosis and Therapy. 7 (6), 616-632 (2017).
- Labrousse, L., et al. Guidance of the MitraClip procedure by 2D and 3D imaging. Archives of Cardiovascular Diseases. 111 (6-7), 432-440 (2018).
- Khalique, O. K., Hahn, R. T. Percutaneous Mitral Valve Repair: Multi-Modality Cardiac Imaging for Patient Selection and Intra-Procedural Guidance. Frontiers in Cardiovascular Medicine. 6, 142 (2019).
- Bushari, L. I., et al. Percutaneous Transcatheter Edge-to-Edge MitraClip Technique: A Practical “Step-by-Step” 3-Dimensional Transesophageal Echocardiography Guide. Mayo Clinic Proceedings. 94 (1), 89-102 (2019).
- Sherif, M. A., et al. MitraClip step by step; how to simplify the procedure. Netherlands Heart Journal. 25 (2), 125 (2017).
- Guarracino, F., et al. Transesophageal echocardiography during MitraClip procedure. Anesthesia & Analgesia. 118 (6), 1188-1196 (2014).
- Avenatti, E., et al. Diagnostic Value of 3-Dimensional Vena Contracta Area for the Quantification of Residual Mitral Regurgitation After MitraClip Procedure. JACC: Cardiovascular Interventions. 12 (6), 582-591 (2019).
- Altiok, E., et al. Analysis of procedural effects of percutaneous edge-to-edge mitral valve repair by 2D and 3D echocardiography. Circulation: Cardiovascular Imaging. 5 (6), 748-755 (2012).

