Hyperactive piggyBac Transposase-mediated Germline Transformation in the Fall Armyworm, Spodoptera frugiperda
Summary
Successful germline transformation in the fall armyworm, Spodoptera frugiperda, was achieved using mRNA of hyperactive piggyBac transposase.
Abstract
Stable insertion of genetic cargo into insect genomes using transposable elements is a powerful tool for functional genomic studies and developing genetic pest management strategies. The most used transposable element in insect transformation is piggyBac, and piggyBac-based germline transformation has been successfully conducted in model insects. However, it is still challenging to employ this technology in non-model insects that include agricultural pests. This paper reports on germline transformation of a global agricultural pest, the fall armyworm (FAW), Spodoptera frugiperda, using the hyperactive piggyBac transposase (hyPBase).
In this work, the hyPBase mRNA was produced and used in place of helper plasmid in embryo microinjections. This change led to the successful generation of transgenic FAW. Furthermore, the methods of screening transgenic animals, PCR-based rapid detection of transgene insertion, and thermal asymmetric interlaced PCR (TAIL-PCR)-based determination of the integration site, are also described. Thus, this paper presents a protocol to produce transgenic FAW, which will facilitate piggyBac-based transgenesis in FAW and other lepidopteran insects.
Introduction
The fall armyworm (FAW), Spodoptera frugiperda, is native to tropical and subtropical regions of America. Currently, this is a devastating insect herbivore in more than 100 countries worldwide1. FAW larvae feed on more than 350 host plants, including some important staple food crops2. The strong migration ability of FAW adults contributes to its recent rapid spread from the Americas to other places1,2. As a result, this insect is now threatening food security internationally. Applying new technologies may facilitate advanced studies in FAW and provide novel strategies to manage this pest.
Insect germline transformation has been used to study gene function and generate transgenic insects for use in genetic control3,4. Among the various methods used to achieve genetic transformation in insects, the piggyBac element-based method is the most used method5. However, due to the low rate of transposition, it is still challenging to conduct transgenesis in non-model insects. Recently, a hyperactive version of piggyBac transposase (hyPBase) was developed6,7. Germline transformation was achieved in FAW recently8, which is the first report that used the hyPBase in lepidopteran insects. In this report, detailed information on employing hyPBase mRNA in generating transgenic FAW is described. The method described here could be applied to achieve the transformation of other lepidopteran insects.
Protocol
1. In vitro synthesis of hyPBase mRNA
NOTE: The complete coding sequence of the hyPBase sequence was synthesized and inserted into a pTD1-Cas9 vector (see the Table of Materials) to produce the pTD1-hyPBase construct, which contains a hyPBase-expressing cassette, T7 promoter: polyhedrin-5' UTR: hyPBase: polyhedrin-3' UTR: poly (A). The full sequence of the pTD1-hyPBase construct is provided in the Supplementary Material.
- Preparation of the template for in vitro synthesis
- Perform a PCR reaction to amplify the hyPBase-expressing cassette using a forward primer, 5'- atgcggtgtgaaataccgcacagatgcg-3', and a reverse primer 5'- tagaggccccaaggggttatgctag-3', high-fidelity polymerase, and pTD1-hyPBase plasmid as a template.
NOTE: The PCR reaction (final volume of 50 µL) contains 5.0 µL of 10x Reaction buffer, 4.0 µL of 10 mM deoxynucleoside triphosphate (dNTP), 1.0 µL of plasmid (50 µg/µL), 1.0 µL of high-fidelity polymerase, 1.0 µL each of 10 µM forward and reverse primers, and 33 µL of nuclease-free water. The settings were as follows: an initial incubation of 95 °C for 3 min, followed by 35 cycles of 98 °C for 10 s, 60 °C for 15 s, and 68 °C for 2 min. - Check the PCR product on a 1% agarose gel at 120 V in fresh Tris-acetate-ethylenediamine tetraacetic acid (TAE) buffer for 30 min and purify the product with the gel extraction kit.
NOTE: Do not use the PCR product purification kit. Even a trace amount of the pTD1-hyPBase plasmid significantly decreases the efficiency of in vitro synthesis.
- Perform a PCR reaction to amplify the hyPBase-expressing cassette using a forward primer, 5'- atgcggtgtgaaataccgcacagatgcg-3', and a reverse primer 5'- tagaggccccaaggggttatgctag-3', high-fidelity polymerase, and pTD1-hyPBase plasmid as a template.
- In vitro synthesis of hyPBase mRNA using a commercial kit
- Completely thaw the frozen reagents, keep the enzyme and 2x NTP/CAP solution on ice, and leave the thawed 10x Reaction buffer at room temperature.
- Assemble the reaction at room temperature by adding the following components: 2x NTP/CAP solution: 10 µL; 10x Reaction buffer: 2 µL; Template DNA: 0.1-0.2 µg; Enzyme mix: 2 µL; Nuclease-free water to 20 µL.
- Mix the reagents thoroughly by gently pipetting the mixture up and down and incubate at 37 °C for 2-4 h.
- Recovery of synthesized mRNA by lithium chloride precipitation
- Add 30 µL of LiCl Precipitation Solution, mix thoroughly by flicking the tube, and then keep at -20 °C for ≥ 30 min.
- Centrifuge at 4 °C for 15 min at the maximum speed, and carefully remove the supernatant.
- Wash the pellet once with 1 mL of 70% ethanol, re-centrifuge, and remove the supernatant carefully.
- Dry the pellet at room temperature for 1-2 min, and then resuspend the pellet with 20-40 µL of nuclease-free water.
- Dilute 1 µL of mRNA solution with 9 µL of nuclease-free water. Take 2 µL to determine the mRNA concentration and use 8 µL to check the mRNA quality on a 1% agarose gel at 100 V in fresh TAE buffer for 40 min.
- Aliquot the mRNA solution and store frozen at -70 °C for up to one year.
NOTE: Do not completely dry the mRNA pellet; it may be harder to dissolve it in water.
2. Microinjection
- Egg collection
- Place 12 male pupae and 12 female pupae in a 15 cm x 15 cm x 10 cm box. Cover the box with a paper towel. Place a 10% sucrose solution in the box for feeding adults.
- On day 2 post eclosion, expose the adults to intense light overnight.
- On day 3 post eclosion, transfer the adults from step 2.1.2 to a dark place. Collect the embryos within 2 h after oviposition.
- Add drops of water to the piece of a paper towel with fresh eggs kept in a Petri dish on ice. Transfer the eggs gently to a glass slide with a fine brush and align them one by one on a glass slide. Keep the glass slides with the aligned eggs on ice before microinjection.
- Microinjection setup
- Inject a mixture of a transgenic enhanced green fluorescent protein (EGFP)-expressing plasmid (200 ng/µL), pBac: hr5ie1-EGFP-SV40, hyPBase mRNA (200 ng/µL), and sterile distilled water into the eggs, using the compensation pressure of the automated microinjector (see the Table of Materials).
- Keep the injected eggs at 27 ± 1 °C, 60 ± 10% relative humidity until hatching.
- Feed the hatched larvae on an artificial diet, and transfer each larva to one small cup at the early 3rd instar stage (~6 mm in length, and body-color turns black).
3. Fluorescence screening and genetic crossing
- Collect the pupae and place ~150 female pupae and ~150 male pupae in a 35 cm x 35 cm x 35 cm cage. Hang several pieces of paper towels (~30 cm x 15 cm) in the cage to collect the eggs.
- Collect the paper towels with eggs daily, and keep the eggs at 27 ± 1 °C, 60 ± 10% relative humidity until hatching.
- Keep the neonate larvae at 4 °C for 5 min to immobilize them, and then transfer them onto an ice-cold plate.
- Screen the immobilized neonate larvae under a fluorescence stereomicroscope. Select the EGFP-positive neonates, raise them to the pupal stage in ~2 weeks at 27 ± 1 °C, and separate them by sex according to the phenotype differences at the ventral abdomen segments.
NOTE: The female pupa has a slit-like genital opening on the eighth abdominal segment and the cephalic margins of the ninth and tenth segments curving toward the genital opening. The genital opening of the male pupa has a slight elevation on the ninth abdominal segment. - Place the EGFP-positive pupae and the wild-type pupae of the opposite sex in a male: female ratio of 1:1 in a cage. Sib-cross the EGFP-expressing adults to produce the following generations.
4. PCR detection of transgenic insertion
- Extract genomic DNA from the wild-type and EGFP-positive individual larvae using any commercial genomic DNA extraction kit following the manufacturer's instructions.
- Perform a PCR reaction using the genomic DNA as a template; a forward primer, 5'- acgtacgctcctcgtgttccgttc-3' located in the ie1 promoter region; and a reverse primer, 5'- aagcactgcacgccgtaggtcag-3' located in the EGFP region of the transformation vector.
- Set up each PCR reaction (final volume of 40 µL) to contain 20 µL of the polymerase mixture, 1.0 µL of genomic DNA (40 µg/µL), 1.0 µL each of 10 µM forward and reverse primers, and 17 µL of nuclease-free water. Use the following settings: an initial incubation of 95 °C for 3 min, followed by 35 cycles of 95 °C for 20 s, 56 °C for 20 s, and 68 °C for 40 s settings.
- Check the PCR products on a 1% agarose gel at 120 V for 30 min.
5. Determination of the transgenic insertion site
- Perform high-efficiency TAIL-PCR using genomic DNA as a template to determine the integration site of transgene insertion in the EGFP-positive insects.
- Perform the PCR reactions following the steps described elsewhere9.
- Use three primers, P1: 5'-atcagtgacacttaccgcattgacaagcacgcc-3', P2: 5'-tcacgggagctccaagcggcgactg-3', and P3: 5'-atgtcctaaatgcacagcgacggattcgcgct-3', which are specific to the piggyBac vector, in the 3 rounds of PCR reaction, respectively.
- Perform the PCR reactions following the steps described elsewhere9.
- Sequence the final PCR products to determine the integration site.
Representative Results
A construct for the expression of hyPBase-containing T7 promoter: polyhedrin-5'UTR: hyPBase: polyhedrin-3'UTR: poly (A) signal was generated (Figure 1A) and amplified as a ~2.2 kb PCR fragment to synthesize hyPBase mRNA in vitro (Figure 1B). Then, the hyPBase mRNA was produced and subjected to agarose gel electrophoresis. The mRNA of the expected size (~1.1 kb band) was detected on a 1% agarose gel (Figure 1C).
Then, a piggyBac-based EGFP expression construct was prepared (Figure 2A). A mixture of this plasmid and hyPBase mRNA, or PBS solution, was injected into embryos within 2 h after oviposition. At 24 h after injection, a transient signal of EGFP expression was observed in the injected embryos. The PBS-injected embryos did not show EGFP signal (Figure 2B), whereas the EGFP-expressing plasmid and hyPBase mRNA-injected embryos showed EGFP fluorescence (Figure 2C), indicating that the piggyBac construction was successful. Thus, the hr5ie1 promoter can function during the early embryonic development.
Approximately 2,000 eggs were injected, and ~700 of them developed into pupae. The G0 adults were self-crossed, and eggs were collected. The G1 larvae hatched from these eggs were examined for the EGFP signal under a fluorescence stereomicroscope. As shown in Figure 3A, the transgenic larvae showed a strong EGFP signal. To test the insertion of the EGFP expression cassette into the genomes of EGFP-positive insects, a fragment containing the promoter and EGFP fragment was amplified using the genomic DNA isolated from the EGFP-positive larvae as a template (Figure 3B). This fragment was not detected when genomic DNA was isolated from the wild-type larvae as the template (Figure 3B). By conducting high-efficiency TAIL-PCR and sequencing of the PCR products, the integration site of transgene insertion was determined. As shown in Figure 4, the transgene integration site lies in the 81st intron of the Twinchin-like gene in chromosome 26.
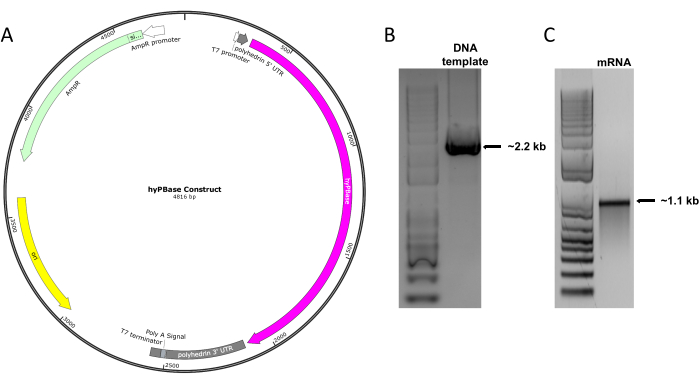
Figure 1: Production of hyPBase mRNA. (A) The hyPBase-expressing cassette containing the T7 promoter: polyhedrin-5'UTR: hyPBase: polyhedrin-3'UTR: poly (A) signal was constructed. (B) The ~2.2 kb DNA template for synthesizing hyPBase mRNA was amplified from the hyPBase construct. The left lane shows the 1-kb ladder run on the same gel. (C) A ~1.1 kb hyPBase mRNA was synthesized from the amplified DNA template. The left lane shows the 1-kb ladder run on the same gel. Please click here to view a larger version of this figure.
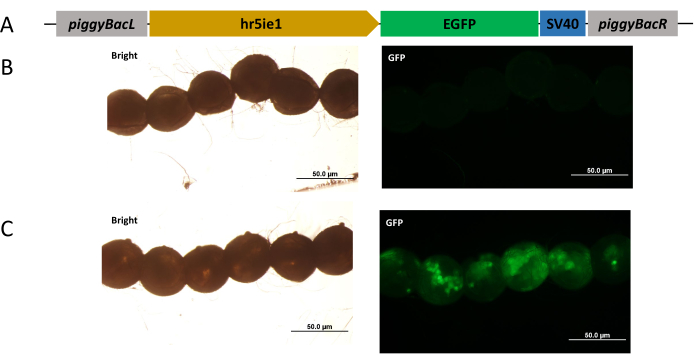
Figure 2: Expression of EGFP in injected embryos. (A) Schematic diagram of the piggyBac vector used in this study. (B) No EGFP signal was detected in embryos injected with 1x PBS. (C) EGFP signal was detected in embryos injected with the piggyBac vector and hyPBase mRNA. Scale bars = 50 µm. Abbreviations: EGFP = enhanced green fluorescent protein and PBS = phosphate-buffered saline. Please click here to view a larger version of this figure.
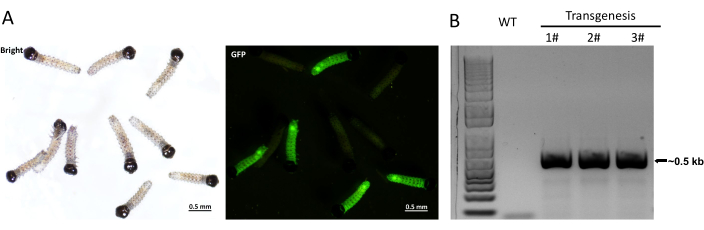
Figure 3: Characterization of transgenic FAW. (A) EGFP expression in transgenic but not in wild-type FAW larvae. Scale bars = 0.5 mm. (B) PCR analysis of genomic insertion of the transgenes. A pair of primers targeting the promoter and EGFP regions were used to amplify a ~0.5 kb fragment using the genomic DNA isolated from transgenic or wild-type FAW larvae as a template. Abbreviations: EGFP = enhanced green fluorescent protein and FAW = Fall armyworm. Please click here to view a larger version of this figure.
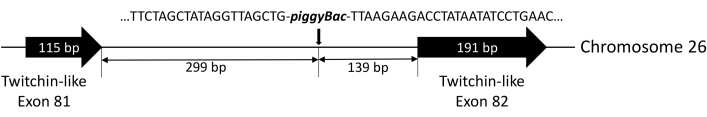
Figure 4: Genomic insertion of piggyBac construct. Genomic insertion of the piggyBac construct in transgenic FAW was determined by TAIL-PCR and sequencing. Chromosome localization and partial genomic DNA sequences flanking the boundaries of the piggyBac construct are shown. Abbreviations: FAW = Fall armyworm and TAIL-PCR = thermal asymmetric interlaced PCR. Please click here to view a larger version of this figure.
Supplementary Material. Please click here to download this File.
Discussion
The low rate of transposition and difficulty of delivering transgenic components into fresh embryos limit the success of germline transformation in many non-model insects, especially those from order, Lepidoptera. To increase the germline transformation rate, a hyperactive version of the most widely used piggyBac transposase (hyPBase) was developed7,10. To date, successful germline transformation in lepidopteran insects is mainly reported in the model insect silkworm, Bombyx mori11. However, conducting transgenesis work in other lepidopteran insects, including some major agriculture pests, is still challenging.
The major limitations and the key steps of establishing a transgenic FAW line using this protocol are synthesizing high-quality hyPBase mRNA and delivering piggyBac transposition components into the early embryos. The two core components of the traditional piggyBac system are a donor plasmid containing the cargo to be inserted into the genome and a helper plasmid providing the transposase12. To increase the transformation efficiency, highly active promoters are used in the helper plasmid to drive the expression of transposase13,14. As these promoters are usually obtained from model insects, their activity in other insects may not be high as in the species from which the promoter was identified. Thus, transposase mRNA is more effective than the helper plasmid, which may improve germline transformation in most non-model insects.
In this protocol, hyPBase mRNA was prepared in vitro by constructing a plasmid containing the hyPBase-expressing cassette (T7 promoter: polyhedrin-5'UTR: hyPBase: polyhedrin-3'UTR: poly (A) signal). The T7 promoter allows in vitro synthesis of mRNA using commercially available mRNA synthesis kits. Both polyhedrin-5'UTR and polyhedrin-3'UTR could help in improving transcription of hyPBase mRNA in vitro and translation of hyPBase protein in vivo. The presence of the poly (A) signal helps enhance the stability of hyPBase mRNA in vivo15. Additionally, injection of the piggyBac components into the very early embryos with a limited number of dividing cells is also critical to improve the chances of delivering piggyBac system component progenitors of germline cells. The transposition events occurring in the germline cells could be inherited. In this protocol, FAW embryos less than 2 hours old were collected and kept on ice to slow down their development before injection. For most insects, collecting embryos as early as possible and finding a way to slow down their development could also help in successful germline transformation.
Using fluorescent proteins in insect transgenesis helps in the identification of transgenic animals. In this protocol, the IE1 promoter, which has been reported to be an efficient and ubiquitous promoter in lepidopteran insects11, derived from the Autographa california multicapsid nuclear polyhedrosis virus (AcNPV) immediate early gene, was fused with the hr5 enhancer to drive the expression of EGFP. In transgenic individuals in which EGFP was inserted into the FAW genome, the EGFP signal could be observed at all stages (data not shown). Molecular confirmation of piggyBac-mediated insertion was usually performed by southern blot or inverse PCR and sequencing. In this protocol, PCR amplification of a fragment in the transgenic cargo was used to confirm the transgenic insertion rapidly. In addition, a PCR-based high-efficiency TAIL-PCR method was employed to identify the integration site of transgenic insertion9, which is easy to manipulate compared with the widely used inverse PCR method. The high-efficiency TAIL-PCR method was developed to identifying the transgenic integration sites in plants. In this study, this method is used for the first time in insects. The presented protocol provides an effective way to generate transgenic FAW and could also be further applied to many other model and non-model insects.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The research reported is supported by the National Science Foundation I/UCRC, the Center for Arthropod Management Technologies, under Grant No IIP-1821936 and by industry partners, Agriculture and Food Research Initiative Competitive Grant No. 2019-67013-29351 and the National Institute of Food and Agriculture, US Department of Agriculture (2019-67013-29351 and 2353057000).
Materials
| 1.5" Dental Cotton Rolls | PlastCare USA | 8542025591 | REARING |
| 1 oz Souffle Cup Lids | DART | PL1N | |
| 1 oz Souffle Cups | DART | P100N | REARING |
| 48 oz Plastic Deli Containers | Genpack | AD48 | REARING |
| Add-on Filter Set (Green) | NightSea LLC | SFA-BLFS-GR | SCREENING |
| Borosilicate Glass | Sutter Instruments | BF100-50-10 | INJECTION |
| Borosilicate Glass | SUTTER INSTRUMENT | BF-100-50-10 | |
| Dissecting Scope | Nikon | SMZ745T | SCREENING |
| Featherweight Forceps | BioQuip | 4750 | REARING |
| Gutter Guard | ThermWell Products | VX620 | REARING |
| Inverted Microscope | Olympus | IX71 | INJECTION |
| Microinjector | Narishige | IM-300 | INJECTION |
| Micropipette Puller | Sutter Instruments | P-1000 | INJECTION |
| Microscope Slides | VWR | 16004-22 | INJECTION |
| NightSea Full System | NightSea LLC | SFA-RB-DIM | SCREENING |
| Nitrogen Gas | AWG/Scott-Gross | NI 225 | INJECTION |
| Paper Towels | Bounty | 43217-45074 | REARING |
| Spodoptera frugiperda Artificial Diet | Southland Products, Inc | N/A [Request Species/Quantity] | REARING |
| Spodoptera frugiperda Eggs | Benzon Research, Inc | N/A [Request Species/Quantity] | REARING |
| Taq MasterMix | polymerase mixture |
References
- Gui, F., et al. Genomic and transcriptomic analysis unveils population evolution and development of pesticide resistance in fall armyworm Spodoptera frugiperda. Protein Cell. , (2020).
- Montezano, D. G., et al. Host plants of Spodoptera frugiperda (Lepidoptera: Noctuidae) in the Americas. African Entomology. 26 (2), 286-300 (2018).
- Li, Z., et al. Ectopic expression of ecdysone oxidase impairs tissue degeneration in Bombyx mori. Proceedings, Biological Sciences. 282 (1809), 20150513 (2015).
- Ogaugwu, C. E., Schetelig, M. F., Wimmer, E. A. Transgenic sexing system for Ceratitis capitata (Diptera: Tephritidae) based on female-specific embryonic lethality. Insect Biochemistry and Molecular Biology. 43 (1), 1-8 (2013).
- Gregory, M., Alphey, L., Morrison, N. I., Shimeld, S. M. Insect transformation with piggyBac: getting the number of injections just right. Insect Molecular Biology. 25 (3), 259-271 (2016).
- Otte, M., et al. Improving genetic transformation rates in honeybees. Scientific Reports. 8 (1), 16534 (2018).
- Eckermann, K. N., et al. Hyperactive piggyBac transposase improves transformation efficiency in diverse insect species. Insect Biochemistry and Molecular Biology. 98, 16-24 (2018).
- Chen, X., Koo, J., Gurusamy, D., Mogilicherla, K., Palli, S. R. Caenorhabditis elegans systemic RNA interference defective protein 1 enhances RNAi efficiency in a lepidopteran insect, the fall armyworm, in a tissue-specific manner. RNA Biology. , 1-9 (2020).
- Liu, Y. G., Chen, Y. High-efficiency thermal asymmetric interlaced PCR for amplification of unknown flanking sequences. Biotechniques. 43 (5), 649-650 (2007).
- Yusa, K., Zhou, L., Li, M. A., Bradley, A., Craig, N. L. A hyperactive piggyBac transposase for mammalian applications. Proceedings of the National Academy of Sciences of the Unites States of America. 108 (4), 1531-1536 (2011).
- Xu, H., O’Brochta, D. A. Advanced technologies for genetically manipulating the silkworm Bombyx mori, a model Lepidopteran insect. Proceedings, Biological Sciences. 282 (1810), 20150487 (2015).
- Wu, S. C. -. Y., et al. piggyBac is a flexible and highly active transposon as compared to sleeping beauty, Tol2, and Mos1 in mammalian cells. Proceedings of the National Academy of Sciences of the Unites States of America. 103 (41), 15008-15013 (2006).
- Tamura, T., et al. Germline transformation of the silkworm Bombyx mori L. using a piggyBac transposon-derived vector. Nature Biotechnology. 18 (1), 81-84 (2000).
- Handler, A. M., Harrell, R. A. Germline transformation of Drosophila melanogaster with the piggyBac transposon vector. Insect Molecular Biology. 8 (4), 449-457 (1999).
- Dreyfus, M., Régnier, P. The poly (A) tail of mRNAs: bodyguard in eukaryotes, scavenger in bacteria. Cell. 111 (5), 611-613 (2002).

