A Porcine Heterotopic Heart Transplantation Protocol for Delivery of Therapeutics to a Cardiac Allograft
Summary
We present a protocol for utilizing a normothermic ex vivo sanguinous perfusion system for the delivery of therapeutics to an entire cardiac allograft in a porcine heterotopic heart transplant model.
Abstract
Cardiac transplantation is the gold standard treatment for end-stage heart failure. However, it remains limited by the number of available donor hearts and complications such as primary graft dysfunction and graft rejection. The recent clinical use of an ex vivo perfusion device in cardiac transplantation introduces a unique opportunity for treating cardiac allografts with therapeutic interventions to improve function and avoid deleterious recipient responses. Establishing a translational, large-animal model for therapeutic delivery to the entire allograft is essential for testing novel therapeutic approaches in cardiac transplantation. The porcine, heterotopic heart transplantation model in the intraabdominal position serves as an excellent model for assessing the effects of novel interventions and the immunopathology of graft rejection. This model additionally offers long-term survival for the pig, given that the graft is not required to maintain the recipient’s circulation. The aim of this protocol is to provide a reproducible and robust approach for achieving ex vivo delivery of a therapeutic to the entire cardiac allograft prior to transplantation and provide technical details to perform a survival heterotopic transplant of the ex vivo perfused heart.
Introduction
Heart failure is a condition that affects an estimated 6 million adults in the United States and is projected to increase to 8 million adults by the year 20301. Cardiac transplantation is the gold standard treatment for end stage heart failure. However, it is not without its limitations and complications. It remains limited by the number of available donor hearts, primary graft dysfunction, rejection of the heart, and the side effects of long-term immunosuppression2. These limitations are particularly important in young recipients who may experience allograft failure and require subsequent re-transplantation to achieve normal life expectancy.
An ideal intervention to overcome these limitations would treat entire cardiac allografts with therapeutics prior to implantation into the recipient that can improve the viability of the allograft and confer "cardioprotection." Such interventions would be given prophylactically to minimize the incidence of ischemic insults, allograft rejection, cardiac allograft vasculopathy, and even repair marginal allografts. Translational studies for developing these types of interventions require a large-animal model of cardiac transplantation to allow for the long-term surveillance of the cardiac graft. The porcine, heterotopic heart transplantation model in the intraabdominal position has proven ideal for this purpose. Heart transplantation in this position allows for testing the effects of novel therapies and assessing the immunopathology of graft rejection. Additionally, the heterotopic model is advantageous over the orthotopic model due to better overall survival of the recipient, no requirement for cardiopulmonary bypass, and no requirement of the graft to maintain the recipient's circulation3.
Effective delivery of therapeutic interventions to the heart, such as gene, cell, or immuno- therapy, is a significant barrier to clinical application4,5. The technology introduced by ex vivo perfusion devices allows grafts to be continually perfused, maintaining them in a nonworking but metabolically active state6,7,8,9. This offers a unique opportunity to treat a whole heart with advanced therapeutics while minimizing the potential side effects of systemic delivery10,11,12,13. Another advantage of utilizing ex vivo perfusion devices for therapeutic delivery is that they allow the administration of medications to the coronary circulation over extended periods that are not feasible using traditional cold static storage methods. This allows for more global delivery of the therapeutics to the graft14. Using the protocol presented here, we successfully delivered the firefly luciferase gene to a whole porcine cardiac graft using adenoviral vectors15. The aim of this protocol is to provide a reproducible and robust approach for achieving delivery of a therapeutic to the entire cardiac allograft prior to transplantation.
Protocol
NOTE: Two female Yucatan pigs are selected, with one designated to be the cardiac graft donor and the other the recipient. Pigs aged 6-8 months, weighing approximately 30 kg, and having compatible blood types are recommended. The overview of the protocol is demonstrated in Figure 1. Housing and the treatment procedures for the pigs are performed in accordance with the guidelines of the Animal Care and Use Committee of Duke University Medical Center.
1. Preparation of the ex vivo perfusion device
- Prepare the ex vivo perfusion device and a cell saver device for use per the manufacturer's guidelines.
- Have a pacing box and defibrillator available and set them up.
- Have a point-of-care (POC) testing device available to check a complete blood count (CBC), basic metabolic panel (BMP), and arterial blood gas (ABG).
- Add the following medications to the perfusion priming solution provided by the manufacturer, if not already present in the manufacturer's perfusion solution: 100 mL of 25% albumin, 10 mL of 200 mg/100 mL ciprofloxacin, 1 g of cefazolin sodium, two 5 mL vials of multi-vitamin injection, 250 mg of methylprednisolone, 10,000 IU of heparin, and 50 IU of insulin.
- Perform POC testing of the ex vivo device priming solution to ensure that the electrolyte levels are within the normal physiologic range. If not, administer calcium gluconate, dextrose, and/or sodium bicarbonate accordingly to supplement any subtherapeutic electrolyte or glucose levels.
- To add the priming solution with the added medications, spike the solution and de-air the line delivering the solution to the ex vivo perfusion device.
NOTE: Skip to section 6 for instructions on priming the ex vivo perfusion device.
2. Initiation of anesthesia and IV access in the donor pig
- After fasting the pig for 8-12 h, premedicate it with ketamine (5-33 mg/kg, intramuscular) and midazolam (0.2-0.5 mg/kg, intramuscular) and administer isoflurane (1-4%) using a face mask.
- Place the pig in a supine position and intubate with an endotracheal tube (ETT) (5.5-6.5 mm internal diameter) to protect the airway. Secure the ETT by tying it to the pig's snout. Position the extremities using heavy ties attached to the table.
- Apply vet ointment on the eyes to prevent dryness while under anesthesia.
- Place an intravenous (IV) catheter (20-22 G) in an ear vein.
- Initiate maintenance IV fluids (Lactated Ringer's solution at 10 mL·(kg·h)-1).
- Administer intramuscular (IM) Buprenorphine 0.005-0.01 mg/kg for analgesia.
3. Vital signs and central line settings
- Start mechanical ventilation at a tidal volume of 10 mL·(kg·min)-1 and a rate of 10-15 breaths per minute with isoflurane (1-3%) maintained throughout the procedure such that reflexes are absent and the heart rate (>60 bpm, <100 bpm) and blood pressure (systolic blood pressure >90 mmHg, <130 mmHg) remain within the physiologic range.
NOTE: The addition of a paralytic is optional. - Continuously monitor oxygen saturation and heart rates throughout the surgery.
4. Median sternotomy of the donor pig
- Palpate the sternum from the manubrium to the xiphoid. Mark the midline using a sterile surgical marker. Shave any hair from the site with a hair clipper and sterilize the area using 4% chlorhexidine for a total of 3 rounds of sterilization. Apply a sterile surgical drape around the immediate surgical site.
NOTE: Surgeons must wash hands and arms with an alcohol- or iodine-based wash and don sterile gowns and gloves. - Use a no. 10 blade to make an incision from the manubrium down to the xiphoid, measuring 20-30 cm, depending on the size of the pig.
- Use electrocautery to divide the pectoralis major down from the sternum to the xiphoid, being careful to do this along the midline of the sternum. Once down to the sternum, score the midline and begin the sternotomy from the xiphoid by dividing it with heavy scissors.
- Extend the sternotomy cephalad with heavy scissors. After each cut, bluntly separate the heart from the sternum using finger sweeps. In this manner, complete the sternotomy through the manubrium.
- After completing the sternotomy, achieve hemostasis by applying electrocautery to the cut bone edges.
- Place a sternal retractor and open it to optimize the exposure of the surgical field. Identify and remove the thymus with electrocautery. Enter the pericardium longitudinally from the diaphragm to the aorta. Create a pericardial cradle using 5-6 size: 2-0, silk sutures.
5. Cardiac arrest and cardiectomy of the donor pig
- Fully divide the tissue between the aorta and pulmonary artery (PA) and visualize the location of the aortic arch and the brachiocephalic trunk to facilitate proper placement of the aortic cross-clamp.
NOTE: The ascending aorta is much shorter in the pig versus human. - Circumferentially free the superior vena cava (SVC) using scissors and blunt dissection. Pass two, size: 0, silk ties around the SVC.
- Circumferentially free the inferior vena cava (IVC) using scissors and blunt dissection. Similarly, pass two 0 silk ties around the IVC.
- Apply a U-stitch, size: 4-0, polypropylene suture to the ascending aorta.
- Apply a purse-string, size: 4-0, polypropylene suture to the right atrium (RA).
- Administer a bolus of heparin IV using an initial dose of 300 U/kg.
- Insert a pediatric 4-Fr aortic root cannula, secured by the previously placed U-stitch. De-air the cannula and secure it in place with a Rummel tourniquet.
- Connect the aortic root cannula to the cardioplegia tubing after the tubing has been flushed with del Nido cardioplegia. Flush with the necessary amount to remove any air bubbles within the tubing.
NOTE: Communication with the perfusion team is critical at this point to correctly execute the cardiac arrest.- Ensure the perfusionist(s) have installed the cell saver disposables in a sterile fashion, primed the device as recommended by the manufacturer (see section 6), and are ready to process the collected blood.
- Confirm that the cell saver cardiotomy (plastic container attached to the cell saver device where blood is stored after washing) is ready with 10,000 U of heparin and that the cardiotomy is connected to suction, not to exceed -150 mmHg of pressure.
NOTE: This is to avoid hemolysis of red blood cells.
- Create a right atriotomy within the previously placed purse-string, insert a 24 Fr venous cannula into the RA, and secure with a Rummel tourniquet.
- Connect the venous cannula to a sterile suction line connected to the cell saver cardiotomy and collect approximately 1-1.3 L of blood. Then, apply the aortic cross-clamp, carefully ensuring that the clamp completely occludes the ascending aorta. Administer 500 mL of Del Nido cardioplegia into the root at a pressure of 100-150 mmHg using a pressure bag.
NOTE: The heart will blanch and arrest. - Place sterile ice slush on the heart.
- Once the cardioplegia is delivered, remove the aortic root cannula and the RA venous cannula and tie the purse-string sutures down.
- Divide the following: the IVC, the SVC just proximal to the azygos vein, the aorta at the level of the arch just distal to the Innominate artery, the main PA at the bifurcation, and the left azygous vein as it enters the coronary sinus.
NOTE: Pigs have a left azygous vein that drains into the coronary sinus. - Identify the pulmonary veins and ligate them with size: 2-0, silk ties or large-sized clips. Leave one pulmonary vein open for the insertion of the LV vent.
- Remove the heart from the chest and place it in a container with sterile ice slush.
- Move the heart to the backtable to prepare the graft for placement on the ex vivo perfusion device.
6. Washing the donor blood and priming the ex vivo perfusion device
NOTE: This step is necessary to remove any components from the donor serum that might neutralize the delivery of the therapeutic when it is introduced to the perfusate. Perform this step during the explantation of the donor heart to minimize the allograft ischemic time.
- Complete a cell saver prime and wash cycle.
- Install the disposable components into the device per the manufacturer's instructions.
- Prime the cell saver device by spiking Plasmalyte A and selecting the prime function on the device. Add as much Plasmalyte A as the volume of blood collected from the donor pig in a 1:1 fashion.
NOTE: Once the device completes the priming cycle, it is ready for the addition of blood. See sections 5.9-5.11 for how to add the blood from the donor pig. - Once the blood is in the device, select the wash cycle on the cell saver device.
NOTE: During this process, the blood is centrifuged while the Plasmalyte A is introduced to wash the blood. This step concentrates and washes the blood.
- Transfer the washed blood into a blood collection bag for transfer to the ex vivo device.
- Add the washed blood to the ex vivo perfusion device per the manufacturer's guidelines.
- Prepare an epinephrine solution by injecting 0.25 mg epinephrine and 30 IU of insulin into 500 mL of 5% dextrose in water during priming of the ex vivo machine. Spike the solution and de-air the line delivering the solution to the ex vivo device.
- Add 10,000 U of heparin to the ex vivo perfusion device.
- Add 5% albumin to reconstitute the blood.
NOTE: The volume of 5% albumin added to the device equals the amount of plasma removed by the cell saver device. This is done to help achieve a physiologic oncotic pressure and hematocrit. - Turn the pump on to flow at 1-1.5 L/min to prime the circuit with the clear prime, drugs, and blood administered into the reservoir. After turning the pump flow on and circulating the prime through the perfusion module, ensure that the lines of the circuit are air-free.
NOTE: The final maintenance solution volume is 1000 mL in addition to the volume of washed blood. - Obtain a baseline perfusate POC chemistry and lactate using the POC testing device. Replenish electrolytes as needed.
- Add enough dextrose to maintain a minimum glucose level of 100 mg/dL.
- Add enough sodium bicarbonate to maintain a minimum pH goal of 7.4.
NOTE: Importantly, added sodium bicarbonate cannot be removed from the perfusate. Excess sodium levels will contribute to the heart becoming edematous and must be avoided. Caution needs to be taken when treating the base deficit, as the heart will begin to correct the base deficit upon reanimation. - Add enough calcium gluconate to maintain a minimum ionized calcium level of 0.8 mmol/L.
- Set the temperature at 37 °C.
- Set the gas flow rate 세스 150 mL/min and adjust as needed to achieve a physiologic pCO2 level.
- Set the mean arterial pressure (MAP) target to 60-70 mmHg.
- Turn down the pump flow 세스 0.6 L/min.
7. Backtable preparation of the donor heart and reanimating the heart
- Oversew the SVC. Place four pledgeted, size: 4-0, polypropylene sutures in a simple horizontal mattress fashion around the inside of the distal aorta, 5 mm below the cut edge and tie them down.
- While holding up the 4, size: 4-0, pledgeted aortic sutures, insert the aortic connector into the aorta, and tie an umbilical tape around the aorta to secure the connector.
- Place a size: 4-0, polypropylene purse-string around the distal cut edge of the main PA. Insert the PA cannula and tie down the ends of the purse-string to secure the cannula.
- Take the prepared graft from the backtable to the ex vivo perfusion device and connect the aortic connector to the device. Be sure to de-air the aorta/aortic connector before securing the heart to the device.
- Start the perfusion clock, maintain the pump flow around 0.6 L/min, and decrease the temperature set point to 34 °C.
- Start the epinephrine and maintenance drips per the manufacturer's recommendations.
- Connect the PA cannula to the PA connector on the device and secure it with a tie.
- Place the left ventricle (LV) vent drain through the untied pulmonary vein into the left atrium and across the mitral valve into the LV. Secure the vent in place with a single stitch to properly anchor it.
- Place two cardiac pacing leads onto the LV free wall.
- Check lactate, ABG, CBC, and BMP every hour. Administer potassium, 50% dextrose, and calcium as needed to maintain normal physiologic levels.
NOTE: More frequent lactate sampling may be appropriate during early stabilization to establish adequate perfusion based on lactate. - If pacing is required, set the ventricular pace at 80 beats per minute at 10 mA (atrial pacing is typically not utilized).
- If defibrillation is required, start at 10 J after the temperature on the device has reached 34 °C. Do not exceed 50 J.
NOTE: Goal total average flow is 600 mL/min, and average coronary flow is 400 mL/min.
8. Administering the therapeutic
- Draw up the therapeutic into a syringe in a sterile fashion.
- De-air the cardioplegia port by using a sterile 3 mL syringe to draw blood through the port. Administer the therapeutic into the cardioplegia port (or equivalent) such that the therapeutic is introduced directly into the aortic root.
- Flush the port with the volume of collected blood drawn in step 8.2 when de-airing the port; be careful not to flush any air with it.
NOTE: This is to ensure the therapeutic is administered into the heart's aortic root.
NOTE: This section has been previously described in detail in Bishawi et al. to introduce viral vectors for luciferase expression15. - Perfuse the graft on the device for 2 h after introducing the therapeutic.
9. Preparation of the recipient and laparotomy with vascular exposure
- Once the cardiac allograft is secured to the device and the therapeutic is introduced into the circuit, begin the induction of anesthesia and preoperative preparation as described in section 2 for the recipient pig.
- Initiate infusion of the immunosuppression medications: cyclosporine 50 mg/kg total as a slow drip infusion throughout the procedure and methylprednisolone 1 g IV bolus.
- Administer antibiotics: enrofloxacin IM (5 mg/kg) and cefazolin 1 g IV bolus.
- Insert a Foley catheter into the bladder.
NOTE: Decompressing the bladder aids with obtaining an optimal exposure of the infrarenal aorta and IVC. - Mark the abdominal midline from mid-abdomen to the pubis using a sterile surgical marker. Shave any hair from the site with a hair clipper and sterilize the area using 4% chlorhexidine for a total of 3 rounds of sterilization. Apply a sterile surgical drape around the immediate surgical site.
NOTE: Surgeons must wash hands and arms with an alcohol- or iodine-based wash and don sterile gowns and gloves. - Use a 10 blade to incise the skin (20-30 cm incision) and switch to electrocautery to dissect down to the fascia.
- Use two Kocher clamps to lift the fascia and peritoneum and carefully make a small incision (1 cm) into the peritoneal cavity using Metzenbaum scissors.
- Extend the peritoneal opening for the full length of the incision using electrocautery, placing a finger underneath to protect the underlying viscera. Place a Balfour retractor to optimize exposure. Retract the small bowel cranially and with wet towels.
- Open the retroperitoneal space inferior to the kidneys with care directed towards identifying the ureters and avoiding injury.
- Carry the dissection down to the abdominal aorta and IVC. Ligate the lymphatics with medium and large clips.
- Dissect the vessels circumferentially and expose a large enough segment to fit a large Satinsky clamp around each vessel. Take care to avoid disruption of lumbar arterial branches, which come off of the posterior part of the aorta. Place two vessel loops around the aorta and IVC at the proximal and distal ends of the exposure.
10. Final arrest and removing the heart from the ex vivo perfusion device
- At the end of the 2 h of ex vivo perfusion, connect the heater-cooler machine to the ex vivo device. Set the heater cooler temperature to 34 °C.
- In a sterile and air-free fashion, connect the de-aired cardioplegia delivery line to the ex vivo device at the aortic access port.
- Turn the temperature set point on the ex vivo device to off.
- Reduce the heater-cooler temperature to 24 °C and decrease pump flow to maintain MAP between 60 and 70 mmHg (typically a change in pump flow from 1 L/min down to 0.9 L/min).
- Once the temperature reading on the ex vivo perfusion device reaches 24-26 °C, reduce the heater-cooler temperature further to 14 °C and decrease the pump flow further by 100 mL/min.
- Once the temperature reaches 14-16 °C, detach the PA cannula from the PA port, start the delivery of antegrade del Nido (500 mL), close the AO line valve, stop the pump, and quickly clamp the AO vent line.
NOTE: Cardioplegia delivery pressure needs to be titrated to maintain a mean delivery pressure of 45-65 mmHg as displayed on the ex vivo device monitor. - Remove the heart from the ex vivo perfusion device by disconnecting the PA cannula and the aortic connector and cutting the pacing wires.
- Place the heart in a bucket filled with sterile ice slush.
- On the backtable, oversew the pulmonary vein/left atriotomy where the LV vent had been inserted. Trim (1 or 2 mm) of the distal aspect of the aorta and PA where attachment to the cannulas may have crushed the tissue.
NOTE: The heart is now ready for intraabdominal, heterotopic implantation.
11. Heterotopic implantation of the cardiac graft
- Before placing the Satinsky clamps, administer 300 U/kg of IV heparin to the recipient pig.
- Place a Satinsky clamp on the IVC and create a longitudinal venotomy measuring ~1.5 cm using an 11-blade and Pott's scissors.
- Anastomose the graft PA to the recipient's infra-renal IVC in an end-to-side fashion using a running, size: 4-0, polypropylene suture. Perform the inner part of the anastomosis first and reinforce as necessary with interrupted sutures before completing the outer part of the anastomosis.
NOTE: The PA to IVC anastomosis is performed first, and the aorta-to-aorta anastomosis is done last to reduce the duration of aortic occlusion. - Place a Satinsky clamp on the aorta and create a longitudinal aortotomy measuring ~1.5 cm using an 11-blade and Pott's scissors.
NOTE: Obtain an ABG prior to clamp placement. Recheck it immediately after clamp release and again 15-30 min later to assess any changes in hyperkalemia, hyperlactatemia, or acidemia indicative of ischemic injury in the recipient. - Anastomose the graft aorta to the recipient's infra-renal aorta in an end-to-side fashion using a running, size: 4-0, polypropylene suture. Perform the inner part of the anastomosis first and reinforce as necessary with interrupted sutures before completing the outer part of the anastomosis.
- Remove the Satinsky clamps to reperfuse the heart; first, remove the IVC clamp followed by the aortic clamp.
- Place an 18 G angiocath into the LV apex of the graft to de-air. When done, remove the angiocath and close the site with a pledgeted suture.
- Carefully check the anastomoses for any bleeding.
- Carefully place the heart into the right retroperitoneal space, such that there is no tension on the anastomoses and no kinking of the vessels. Replace the small bowel.
12. Closure of the laparotomy
- Close the fascia with looped, size: 0, Maxon suture in a running fashion starting from both ends of the incision and tying in the middle. Take care to avoid any injury to the bowel.
- Close the deep dermal layer with size: 2-0, Vicryl in a running fashion and the skin with size: 4-0, Monocryl in a running fashion.
- Clean the skin incision and apply skin glue.
13. Postsurgical treatment and euthanasia
- After completion of the surgery, turn off the isoflurane flow and monitor the pig for return of muscular tone and neuromuscular reflexes (corneal reflex, withdrawal to painful stimuli, swallowing).
- After confirming the restoration of these functions, turn off mechanical ventilation and observe for spontaneous breathing. If there is spontaneous breathing, remove the endotracheal tube; if there is not, reconnect the endotracheal tube to mechanical ventilation.
- Transfer the pig off the operating table to an isolated enclosure where its vital signs (rectal temperature, blood pressure, heart rate) can be closely monitored. Use a heating lamp to warm the pig as necessary. Provide an IV fluid bolus of 250 mL of Lactated Ringer's solution in the setting of hypotension (systolic blood pressure < 100mmHg). Continue to monitor the pig until it can maintain sternal recumbency and vital signs are fully normalized.
NOTE: The animal is not left unattended until it has regained sufficient consciousness. Additionally, the animal is not returned to the company of other animals until fully recovered. - For pain management, administer a one-time dose of buprenorphine (sustained release) subcutaneous injection 0.12 mg/kg for 72 h of analgesia.
- At the end of the experimental period, euthanize the pig for explantation of the native (thoracic) heart and the allograft (abdominal) heart.
- Prepare the pig as described in sections 2 and 3 for the procedure. Prepare two bags of del Nido and two cardioplegia lines for arresting each heart.
- Expose the thoracic heart as described in section 4. Once complete, proceed to perform a laparotomy as described in section 9.
- Once the aorto-aortic and PA-IVC anastomoses are exposed, place a Satinsky clamp on the recipient aorta and another on the recipient IVC to isolate the allograft from the systemic circulation.
- Insert a pediatric 4-Fr aortic root cannula into the aortic root of the allograft and connect a cardioplegia line to the catheter. Administer 500 mL of del Nido cardioplegia into the root at a pressure of 100-150 mmHg using a pressure bag. After the infusion is started, use Metzenbaum scissors to make a 2 cm incision at the level of the PA-IVC anastomosis to vent the allograft.
- Once the allograft is arrested, proceed to explant the allograft by using Metzenbaum scissors to excise at the level of the aorto-aortic anastomosis and the remainder of the PA-IVC anastomosis. Do not remove any of the Satinsky clamps.
- Proceed with the removal of the thoracic heart as described in section 5.
NOTE: The only significant difference is that the pulmonary veins do not need to be carefully ligated and can instead be grossly dissected using Metzenbaum scissors when performing the cardiectomy.
Representative Results
This group has successfully survived 9 pigs between 5 and 35 days following the protocol as presented here, depending on the study design. Out of 10 pigs that have undergone this protocol, only 1 died prematurely from surgical complications, yielding a 90% survival rate. Demonstrated in Figure 2 is a diagram of the configuration of a heterotopic heart transplanted in the intraabdominal position in a pig. When determining the site for anastomosis of the allograft, select a site that minimizes any tension or kinking on the anastomosis. This ensures that the anastomoses heal properly and that the allograft receives optimal perfusion and drainage of blood.
A representative image of a cardiac allograft being perfused on a normothermic ex vivo perfusion device is shown in Figure 3. Figure 4 outlines representative perfusion parameters acquired during a successful experiment (circulatory flow rate, aortic pressure, heart rate, temperature, mixed venous oxygen saturation, and hematocrit). Inability to achieve the parameter values demonstrated here may lead to compromised allograft function after transplantation. Figure 5 demonstrates an image of an intraabdominal heterotopic heart in situ 35 days after successful transplantation. Representative results of the effectiveness of using the protocol presented here for therapeutic delivery were previously demonstrated by this group15. The cardiac allografts (n = 3) were perfused with perfusate treated with an adenoviral vector carrying the transgene for luciferase. Gene expression proved to be global and robust within the allografts 5 days after the treatment and transplantation. Figure 6 shows an atlas of luciferase protein activity measured and presented as average fold-change in activity from each region of the explanted cardiac allograft in comparison to the thoracic heart of the recipients.

Figure 1: Protocol schematic for therapeutic delivery to an entire cardiac allograft using normothermic ex vivo sanguinous perfusion. (A) The heart and blood are procured from the donor pig. (B) The blood is washed using a cell saver device to remove any therapeutic neutralizing components from the donor serum. (C) The cardiac allograft is mounted onto the normothermic ex vivo perfusion device and perfused for 2 h. (D) Soon after the allograft is mounted, the therapeutic of interest is added to the perfusate. (E) After the allotted ex vivo perfusion period, the allograft is transplanted into the recipient pig in the intraabdominal, heterotopic position. This figure has been modified from15. Please click here to view a larger version of this figure.

Figure 2: Porcine heterotopic heart model in the intraabdominal position. Diagram of the heterotopic heart model where the allograft is transplanted in the intraabdominal position while the recipient's native heart remains in its natural location. The pulmonary artery of the allograft is anastomosed to the infra-renal inferior vena cava, while the aorta of the allograft is anastomosed to the infra-renal aorta of the recipient. Please click here to view a larger version of this figure.
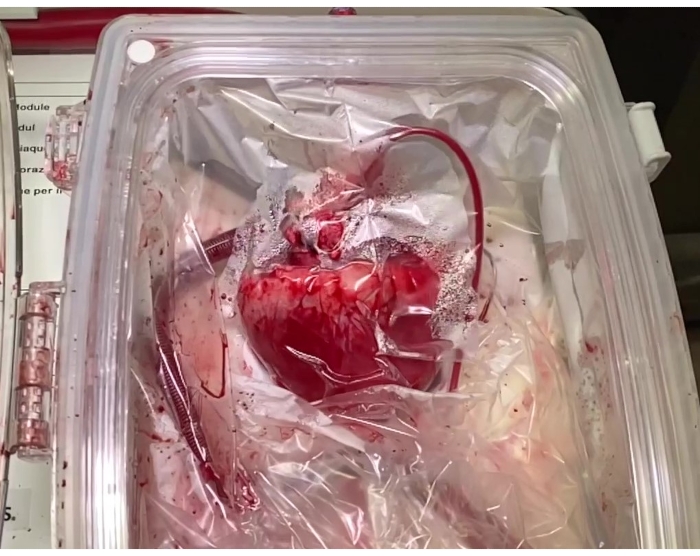
Figure 3: Cardiac allograft on ex vivo perfusion device. The cardiac allograft mounted on a normothermic, ex vivo perfusion device where it is perfused with therapeutic-infused perfusate for 2 h prior to implantation into the recipient. Please click here to view a larger version of this figure.

Figure 4: Representative ex vivo perfusion parameters. (A) Circulatory flow rates measured from the pulmonary artery (blue), the aorta (green), and the coronary arteries (red). (B) Representative aortic pressure measurements: mean pressure (blue), systolic pressure (red), diastolic pressure (green). (C) Heart rate of a cardiac allograft during ex vivo perfusion. (D) Recorded temperature of the cardiac allograft during ex vivo perfusion. (E) demonstrates the values of SvO2 measured from the perfusate during the perfusion period. (F) Hematocrit values measured from the perfusate during the perfusion period. Abbreviations: hct = hematocrit; SvO2 = mixed venous oxygen saturation. Please click here to view a larger version of this figure.

Figure 5: Cardiac allograft transplanted in the recipient. A cardiac allograft on postoperative day 35 treated with therapeutic at the time of implantation. The donor was selected to be a perfect SLA match with the recipient. Abbreviation: SLA = Swine Leukocyte Antigen. Please click here to view a larger version of this figure.

Figure 6: Luciferase activity after transduction of cardiac allografts. Presented are the results of three cardiac allografts that were transduced with adenoviral vectors carrying a luciferase transgene. Demonstrated is the average fold-change in luciferase protein activity in each area of the cardiac allograft. This figure has been modified from Bishawi et al.15. Please click here to view a larger version of this figure.
Discussion
Delivery of therapeutics during ex vivo perfusion in cardiac transplantation offers a strategy to modify the allograft and potentially improve transplant outcomes. The protocol presented here incorporates the state-of-the-art normothermic ex vivo sanguinous perfusion storage and offers promising potential to test isolated delivery of cell, gene, or immunotherapies to the allograft11,12,13. To date, cardiac delivery techniques for these putative therapies for cardiovascular disease and end-stage heart failure have relied on systemic administration, intracoronary perfusion via catheterization, and direct intramyocardial injections, all of which have achieved poor results in terms of myocardial delivery5,16. We had previously demonstrated robust and global expression of a reporter gene to entire cardiac allografts when a viral vector was administered into the perfusate during ex vivo perfusion prior to transplantation15. This is particularly important in the context of cardiac transplantation, where global expression and effect of the therapeutic should reach all areas of the allograft to achieve the desired "cardioprotection" of the whole allograft. This protocol achieves this in a manner that has not been previously achieved using traditionally described routes of administration for therapeutics.
There are several critical steps presented in this protocol to highlight. (1) Every precaution must be taken to minimize blood loss during the procurement of the heart from the donor. At least 1 L of blood needs to be attained from the donor for the perfusion device to achieve adequate flow rates. (2) For therapeutic delivery using normothermic ex vivo sanguinous perfusion, it is necessary to wash the donor blood before adding it to the perfusate to remove any neutralizing components in the donor serum that may negatively affect the delivery of the therapeutic to the heart. (3) Minimize dissection of the heart in the donor until after cardioplegic arrest to avoid fatal arrhythmias. (4) When introducing the therapeutic to the perfusion device, it is important to introduce it through the port closest to the aortic root and always flush the port to ensure complete delivery of the suspension. This is to minimize any potential loss of the therapeutic to the oxygenator or tubing within the circuit and ensure that the graft is receiving as high of a therapeutic concentration as possible. (5) Finally, when selecting the site for graft implantation, it is critical that the location minimizes the potential for tension on the anastomosis and that there be no kinking of the blood vessels/anastomoses.
It is also recommended that the pigs be Swine Leukocyte Antigen (SLA)-typed (i.e., porcine major histocompatibility complex, MHC) beforehand to select for the appropriate degree of matching/mismatching across SLA haplotypes comprising the cell-surface class I (SLA-1, SLA-2, and SLA-3) and/or class II (DR and DQ) antigens based on the investigator's needs (SLA-typing performed by SH as previously described with slight modifications made to the typing primer panels)17,18. For example, ensuring that pigs match across all SLA antigens minimizes the risk of allograft rejection, whereas using pigs with mismatch across all SLA antigens maximizes the incidence of allograft rejection.
A limitation of this model is that while it allows for the study of the immunologic effects on the cardiac graft, it does not allow for a full assessment of the graft's ability to support the cardiovascular system following an intervention. To achieve that, the graft would need to be implanted orthotopically. However, orthotopic transplantation in large-animal models has higher associated mortality and requires cardiopulmonary bypass3. Another limitation of this model is limited access to an ex vivo perfusion device to conduct effective gene delivery to the graft. As these devices become more available in the field of organ transplantation, access is expected to improve. Furthermore, a non-commercial device may be an option for experimental purposes.
Cardiac transplantation offers a unique setting where therapeutics can be introduced to the allograft via ex vivo perfusion prior to implantation into the recipient. The use of an ex vivo perfusion device allows for grafts to be in transit from the donor to the recipient for periods that are much longer than what is safe using traditional cold static storage6. This extended perfusion period enables effective isolated delivery of therapeutics. This model serves as a translational step between preclinical animal testing of therapeutics and transformative clinical therapies.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We would like to thank Duke Large Animal Surgical Core and Duke Perfusion Services for their assistance during these procedures. We would also like to thank Paul Lezberg and TransMedics, Inc. for support.
Materials
| 0 Looped Maxon suture | Covidien | GMM-341L | Used to close fascia of the laparotomy incision |
| 0 Silk ties | Medtronic, Inc | S346 | |
| 18 G Angiocath | BD | 381144 | Used to de-air the left ventricle of the donor heart after implantation |
| 20 Fr LV vent | Medtronic, Inc | 12002 | |
| 2-0 Silk sutures | Ethicon, Inc. | SA11G | |
| 2-0 Silk ties | Ethicon, Inc. | SA65H | |
| 2-0 Vicryl suture | Ethicon, Inc. | J259H | |
| 24 Fr venous cannula | Medtronic, Inc | 68124 | |
| 3-0 Prolene sutures | Ethicon, Inc. | 8522 | |
| 4-0 Monocryl suture | Ethicon, Inc. | Y469G | |
| 4-0 Prolene sutures | Ethicon, Inc. | 8521 | |
| Animal hair cutting clipper | Wahl | 8786-452 | |
| Aortic clamp | V. Mueller | CH6201 | |
| Army Navy retractor | V. Mueller | SU3660 | |
| ATF 40, Cell saver disposable set | Fresenius Kabi | 9108494 | Cell saver device insert |
| Balfour retractor | V. Mueller | SU3042 | Used as an abdominal wall retractor |
| C.A.T.S cell saver | Fresenius Kabi | ES0019 | Cell saver device used to wash donor blood |
| Cardiac defibrillator | Zoll | M Series | Cardiac defibrillator |
| Castro needle holder | V. Mueller | CH8589 | |
| CG4 iStat cartridges | Abbott | 03P85-25 | POC testing |
| CG8 iStat cartridges | Abbott | 03P88-25 | POC testing |
| DeBakey forceps | V. Mueller | CH5902 | |
| Electrocautery disposable pencil | Covidien | E2450H | |
| Gerald forceps | V. Mueller | NL1451 | |
| Hemotherm 400CE Dual Reservoir Cooler/Heater | Cincinnati Sub-Zero | 86022 | Heater cooler used to regulate perfusion temperature on the ex vivo perfusion device |
| iSTAT 1 | Abbott | 04P75-03 | POC testing device |
| Kocher clamp | V. Mueller | SU2790 | |
| Large clip applier | Sklar | 50-4300 | |
| Large clips | Teleflex | 4200 | |
| Large soft pledgets | Covidien | 8886867901 | |
| Medium clip applier | Sklar | 50-4335 | |
| Medium clips | Teleflex | 2200 | |
| Metzenbaum scissor | V. Mueller | CH2006-001 | |
| No. 10 scalpel blade | Swann-Mortan | 301 | Used for skin incision |
| No. 11 scalpel blade | Kiato Plus | 18111 | Used for vascular incision |
| OCS device with base | TransMedics, Inc. | Ex vivo perfusion device | |
| OCS disposable | TransMedics, Inc. | Ex vivo perfusion device insert with perfusion kits | |
| Pacing cable | Remington Medical | FL-601-97 | |
| Pediatric cardioplegia catheter (4Fr) | Medtronic, Inc | 10218 | Used to deliver cardioplegia to the donor aortic root |
| Pediatric Foley catheter | Teleflex | RSH170003080 | Placed pre-op to decompress the recipient's bladder |
| Potts scissors | V. Mueller | CH13038 | |
| Pressure bag x2 (1,000 mL) | Novaplus | V4010H | Used to deliver cardioplegia at a set pressure |
| Satinsky clamp | V. Mueller | CH7305 | Vascular clamp used for creating anastomoses between donor heart and recipient vessels |
| Scissors | Felco | FELCO 200A-50 | Used to perform sternotomy |
| Small hard pledgets | Covidien | 8886867701 | |
| Sternal retractor | V. Mueller | CH6950-007 | |
| Temporary cardiac pacing wires | Ethicon, Inc. | TPW32 | |
| Temporary dual chamber pacemaker | Medtronic, Inc | 5388 | Cardiac pacing device |
| Tourniquet kit | Medtronic, Inc | 79005 | Rummel tourniquets |
| Umbilical tape | Covidien | 8886861903 | |
| Vessel loops | Covidien | 31145686 |
References
- Virani, S. S., et al. Heart disease and stroke statistics-2021 update: A report from the American Heart Association. Circulation. 143 (8), 254 (2021).
- Stehlik, J., Kobashigawa, J., Hunt, S. A., Reichenspurner, H., Kirklin, J. K. Honoring 50 years of clinical heart transplantation in circulation: in-depth state-of-the-art review. Circulation. 137 (1), 71-87 (2018).
- Kadner, A., Chen, R. H., Adams, D. H. Heterotopic heart transplantation: experimental development and clinical experience. European Journal of Cardiothorac Surgery. 17 (4), 474-481 (2000).
- Hastings, C. L., et al. Drug and cell delivery for cardiac regeneration. Advanced Drug Delivery Reviews. 84, 85-106 (2015).
- Sahoo, S., Kariya, T., Ishikawa, K. Targeted delivery of therapeutic agents to the heart. Nature Reviews. Cardiology. 18 (6), 389-399 (2021).
- Stamp, N. L., et al. Successful heart transplant after ten hours out-of-body time using the TransMedics Organ Care System. Heart, Lung & Circulation. 24 (6), 611-613 (2015).
- Ragalie, W. S., Ardehali, A. Current status of normothermic ex-vivo perfusion of cardiac allografts. Current Opinion in Organ Transplantation. 25 (3), 237-240 (2020).
- Koerner, M. M., et al. Normothermic ex vivo allograft blood perfusion in clinical heart transplantation. Heart Surgery Forum. 17 (3), 141-145 (2014).
- Rosenbaum, D. H., et al. Perfusion preservation versus static preservation for cardiac transplantation: effects on myocardial function and metabolism. Journal of Heart and Lung Transplantation. 27 (1), 93-99 (2008).
- Cullen, P. P., Tsui, S. S., Caplice, N. M., Hinchion, J. A. A state-of-the-art review of the current role of cardioprotective techniques in cardiac transplantation. Interactive Cardiovascular and Thoracic Surgery. 32 (5), 683-694 (2021).
- Rurik, J. G., Aghajanian, H., Epstein, J. A. Immune cells and immunotherapy for cardiac injury and repair. Circulation Research. 128 (11), 1766-1779 (2021).
- Rincon, M. Y., VandenDriessche, T., Chuah, M. K. Gene therapy for cardiovascular disease: advances in vector development, targeting, and delivery for clinical translation. Cardiovascular Research. 108 (1), 4-20 (2015).
- Kieserman, J. M., Myers, V. D., Dubey, P., Cheung, J. Y., Feldman, A. M. Current landscape of heart failure gene therapy. Journal of the American Heart Association. 8 (10), 012239 (2019).
- Perin, E. C., Perin, E. C., Miller, L. W., Taylor, D. A., Wilkerson, J. T. . Stem cell and gene therapy for cardiovascular disease. , 279-287 (2016).
- Bishawi, M., et al. A normothermic ex vivo organ perfusion delivery method for cardiac transplantation gene therapy. Scientific Reports. 9 (1), 8029 (2019).
- Hulot, J. S., Ishikawa, K., Hajjar, R. J. Gene therapy for the treatment of heart failure: promise postponed. European Heart Journal. 37 (21), 1651-1658 (2016).
- Ho, C. S., et al. Molecular characterization of swine leucocyte antigen class I genes in outbred pig populations. Animal Genetics. 40 (4), 468-478 (2009).
- Ho, C. S., et al. Molecular characterization of swine leucocyte antigen class II genes in outbred pig populations. Animal Genetics. 41 (4), 428-432 (2010).

