Laparoscopic Radical Antegrade Modular Pancreatosplenectomy via Dorsal-Caudal Artery Approach for Pancreatic Neck-Body Cancer
Summary
With advancements in laparoscopic techniques, laparoscopic radical antegrade modular pancreatosplenectomy (L-RAMPS) has been widely recognized. However, owing to several technical difficulties in this procedure, the artery-first approach in L-RAMPS still remains uncommon. Here, we developed the dorsal-caudal artery approach for L-RAMPS, which might be safe and beneficial for pancreatic neck tumors.
Abstract
Laparoscopic radical resection of the pancreatic neck is one of the most complicated radical operations for pancreatic cancer, especially for patients who have had neoadjuvant chemotherapy. Here, we present a technique to perform laparoscopic radical antegrade modular pancreatosplenectomy (L-RAMPS) using the dorsal-caudal artery approach by making full use of the high-definition vision and operation modes of the laparoscope.
The innovation and optimization of this operation are provided in the protocol. Priority should be given to the dorsal resection plane, including the dorsal side of the superior mesenteric artery (SMA), the dorsal side of the pancreatic head, the root of the celiac artery (CeA), the ventral side of the left renal vessels, and the renal hilum. On the condition that the operation for pancreatic neck-body cancer is feasible and safe, the second step is to perform tumor resection en bloc surrounding the SMA and CeA from the caudal to the cephalic side to increase the rate of R0 (radical zero) resection and further prognosis.
Introduction
Radical antegrade modular pancreatosplenectomy (RAMPS) is an exquisite procedure for malignant tumors located in the pancreatic body or tail, first described by Strasberg in 2003. This operation strategy was designed based on the blood and lymph node drainage of the pancreas, in order to achieve tumor-free dissection planes and radical resection of regional lymph nodes1. RAMPS is becoming increasingly valued by surgeons as it could be conducive to obtain tumor-free margins and relatively favorable survival results2,3,4. With advancements in minimally invasive surgical instruments and techniques, laparoscopic RAMPS (L-RAMPS) has been gradually popularized on account of several advantages, including less intraoperative blood loss, decreased requirement for blood transfusions, and fewer incision events such as pain and infection1. For well-selected patients with distal pancreatic ductal adenocarcinoma (PDAC), recent studies have demonstrated that L-RAMPS could be an effective and safe approach1,5,6.
In pancreaticoduodenectomy (PD) for a malignant tumor around the pancreatic head region, the artery-first approach is a widely accepted strategy with several advantages. The core principle of this method is to explore the superior mesenteric artery (SMA) in the early stages of surgery in order to determine the feasibility of radical resection before pancreatic transection or the ligation of major vessels7,8,9. Recent studies have demonstrated that this artery-first approach may relieve the formation of venous congestion of the distal pancreas and spleen and contribute to the effective bleeding control of the operative regions; besides, it makes lymph node dissection around the SMA more adequate7,10,11. For these reasons, the artery-first strategy is becoming an important component for PD and provides an insight into left-sided pancreatic cancer.
Until now, only some artery-first approaches for L-RAMPS procedures have been reported5,6. The core concept of these approaches is that during pancreatic cancer surgery, tumor infiltration to the SMA should be detected before the performance of irreversible surgical steps, such as transection of the pancreas parenchyma or ligation and resection of the major vessels12,13.
Here, we developed the dorsal-caudal artery approach for L-RAMPS, which might be safe and beneficial for tumors in the pancreatic neck. Our procedure further optimized the routine artery-first approaches for L-RAMPS procedures which were reported by Yamamoto and Kawabata12,14. In other words, we explored and separated the SMA first via the dorsal-caudal approach, a method that has never been reported previously. The goal and advantages of this procedure are to ensure the feasibility and safety of the operation for pancreatic neck-body cancer, which might improve the rate of R0 resection and further prognosis.
Surgeons who intend to use this procedure, however, must have substantial experience in laparoscopic pancreatic surgery. Even if they have passed the learning curve, it is critical to evaluate the patient's condition, including tumor type, vascular condition, and other parameters, because this procedure necessitates sophisticated resection techniques.
In this article, we present a case of a 50-year-old male patient with PDAC, confirmed by endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) biopsy, who underwent L-RAMPS after preoperative neoadjuvant chemotherapy. Our aim is to demonstrate the clinical safety and feasibility of L-RAMPS using the dorsal-caudal artery approach, and its oncologic outcomes in patients with PDAC located in the pancreatic neck, body, or tail.
Protocol
The present protocol follows the guidelines of the Ethics of the Second Affiliated Hospital of Guangzhou University of Chinese Medicine. Informed consent was obtained from the patient for this article and the video.
1. Patient Selection
- Ensure patients have PDAC in the pancreas neck, body, and tail as confirmed by contrast enhanced computed tomography (CT). This procedure is indicated in the following cases: resectable pancreatic cancer and borderline resectable pancreatic cancer after neoadjuvant treatment. The possibility of radical resection increases when the tumor regresses markedly following effective neoadjuvant treatment.
NOTE: In the presented case, CT imaging revealed a tumor in the neck of the patient's pancreas, which was most likely cancerous. - Do not perform this surgery in the following conditions: (1) metastatic PDAC; (2) overall poor patient health, in which the body is unable to withstand major surgery; (3) if a pancreatic tumor is present that infiltrates the splenic vein or the main blood arteries other than the splenic artery.
- At the early age of the learning curve, do not choose challenging cases, for example: obese patients with a body max index (BMI) >35 kg/m2; patients who have suffered from recurrent pancreatitis, as they might have a very poor pancreas parenchyma condition; patients undergoing pancreas targeted radiotherapy; patients with former laparotomy, etc. Once extensive experience with laparoscopic surgery has been obtained, patients with these conditions can also be selected15.
2. Surgical technique
- Operative setting
- Place the anesthetized patient on the operation table in a supine position. Ensure the legs are spread apart.
- During the sterile operation, expose the suprapubic region sufficiently for specimen extraction by the Pfannenstiel incision. Make sure the first surgeon is on the right side of the patient, the first assistant is on the left side, and the second assistant with the laparoscope is between the patient's legs. This procedure employs a five-port technique, as shown in Figure 1.
- After establishing pneumoperitoneum, insert a 12 mm trocar below the navel. Securely install the next four trocars on the abdominal wall in a semicircular arrangement mode with the help of the laparoscopic visual field.
- Exploration phase
- Perform routine diagnostic laparoscopy from the start to examine the liver and the peritoneal surfaces for extra pancreatic metastasis.
- Open the lesser sac using an ultrasound knife by dividing the gastrocolic ligament approximately 2 cm distal from the gastroepiploic artery and vein.
- Dissect and resect the short gastric vessels to facilitate splenectomy.
NOTE: The most superior short gastric vessels can also be detected at this stage for better bleeding control. - Suspend the stomach above the surface of the pancreas with a liver retractor to create enough room for operative procedures around the suprapancreatic area.
- Isolate and excise the splenocolic and perisplenic ligaments with an ultrasound knife, to further expose the splenic flexure of the colon and the tail of the pancreas.
NOTE: Use intraoperative ultrasonography when local structures are not clearly identified. Pancreatic adenocarcinoma appears at intraoperative ultrasonography (US) as a hypoechoic mass with ill-defined margins. - Divide and pass through the gap between the posterior surface of the pancreatic neck and the junction part of the SMV, portal vein (PV), and splenic vein. Then carefully clear this area to prepare for pancreatic transection.
- Dissection phase
- Determine the dorsal resection plane, including the dorsal side of the SMA, the dorsal side of the pancreatic head, the root of the CeA, the ventral side of the left renal artery vessels, and the renal hilum.
- Elevate the transverse colon and its mesentery first on the dorsal side of the pancreatic head. Perform the Kocher maneuver to expose the anterior surface of the left renal vein (LRV).
NOTE: A view of several centimeters of the LRV at this point are helpful for the later procedure steps, when the LRV can be handled on the other side. Explore the group 16 lymph node (para-aortic nodes). - Place the entire small bowel on the right side to provide access to the dorsal side of the SMA. Identify the SMA above the LRV, then dissect it along its periadventitial plane on the anterior-left margin and separate it from the pancreas16.
- Perform the approach on the ventral side of the renal artery vessels and renal hilum as described in step 2.3.5.
- Perform the resection range to the left and posterior plane behind the adrenal gland and onto the surface of the left kidney in the posterior RAMPS.
NOTE: On the left side of the aorta, the left renal artery is followed. - Follow steps 2.3.7-2.3.10 to get to the core of the CeA method.
- Return to the upper colon region. Anatomize the common hepatic artery and gastroduodenal artery (GDA), and then identify the correct hepatic artery.
- Dissect the gastroduodenal lymph nodes, especially the eighth group of lymph nodes17.
- Move the GDA to the right side using a rubber band to expose the anterior surface of the PV.
- Expose the root of the CeA. To do so, dissect the common hepatic artery to its origin, and dissect the CeA free at its origin from the abdominal aorta.
- Dissect around the SMA and CeA from the caudal side to the cephalic side. For this approach, follow steps 2.3.12-2.3.14.
- Dissect the SMA along its periadventitial plane on the anterior-left margin.
NOTE: The anterior surface of the SMA is well displayed under the tunnel which was previously created below the pancreatic neck. The dorsal pancreatic artery is an arterial branching to the pancreas, which will be explored from the root of the SMA. - Divide the distal pancreas from the retroperitoneum and then manage the inferior mesenteric vein (IMV).
NOTE: As a benefit of using the dorsal-caudal artery approach technique, create anatomical space between the SMA and pancreas. Until now, bleeding control of the SMA can be easily achieved, as the most of the space at the left side of the SMA is already exposed. - After excluding tumor infiltration of the anterior surface of the SMA, the SMV is exposed under the pancreatic neck.
NOTE: Exclude tumor infiltration on the anterior surface of the SMA, due to the fact that the vascular plexus and the nerve plexus surrounding the SMA can be completely severed during operation. - According to the pancreatic thickness and texture at the pancreatic neck, transect the pancreas parenchyma using a mechanical stapler device with a corresponding cartridge18,19. Suture the pancreatic duct with a 5-0 polypropylene suture. Perform frozen pathologic examination of the pancreatic neck margin.
- Ligate the root of the splenic vein with a vascular clamp and severe it sharply.
- To skeletonize the common hepatic artery (CHA), left gastric artery, splenic artery (SpA), and CeA, dissect and clear en bloc suprapancreatic lymph nodes.
- Resect fat and fibrous tissues from the caudal to the cephalic side of the SMA and CeA.
- Remove the lymph nodes from the Heidelberg triangle, which is bounded by the PV, CeA, and SMA.
- During this procedure, isolate and divide the origin of the SpA on account of the larger dorsal and caudal spaces created.
- Expose the LRV to determine the dorsal dissection plane.
NOTE: The left adrenal gland and Gerota's fascia should be completely removed for posterior RAMPS20,21. - Finally, using an antegrade method (right-to-left), dissect the distal pancreas and spleen, as well as the surrounding soft tissues.
NOTE: As shown in Figure 2, the cephalic boundary of the resection region is the diaphragmatic crus, the caudal boundary is the LRV, and the interior range is on the left lateral portion of the aorta. Finally, introduce and place two drainage tubes in the pancreatic stump and splenic recess areas.
3. Postoperative follow-up
- Perform a color Doppler ultrasound or CT scan of the abdomen on postoperative day 7 to assess the abdominal condition.
- Check the amylase level of the abdominal drainage fluid on postoperative day 3, day 5, and day 7 to evaluate pancreatic fistula, which is one of the serious complications.
NOTE: Postoperative pancreatic fistula (POPF) can be diagnosed when the amylase level in drainage fluid is more than three times the upper limit of normal serum amylase activity, associated with a clinically relevant development/condition which is related directly to the pancreatic fistula22. - Remove the drain when the amylase level is less than three times the upper limit of normal serum amylase.
- Perform 6-8 cycles of adjuvant chemotherapy using gemcitabine plus nab-paclitaxel program 6-8 weeks after surgery.
- Perform long term follow-up after surgery. Conduct follow-up every 3 months in the first year, every 3-6 months in the second year, and then every 6 months after. Carbohydrate antigen 199 (CA-199), the CeA, the abdominal color Doppler ultrasound, or the CT scan should be rechecked during the follow-up.
Representative Results
A 50-year-old man with upper abdomen discomfort and chronic malnutrition had a 3.2 cm x 2.5 cm tumor in the pancreatic neck identified. He had previously been healthy and had a normal BMI (19.9 kg/m2).
No distant metastasis, major vessels (besides the splenic artery and vein), or lymph node infiltration were detected on the preoperative imaging evaluation. Endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) was performed to confirm the pathological diagnosis of PDAC. The patient underwent four cycles of neoadjuvant chemotherapy using gemcitabine plus nab-paclitaxel program. The medication helped to ease clinical symptoms, and carbohydrate antigen 199 (CA-199) decreased from 4,666 U/mL to 1,350 U/mL, while the tumor's maximal diameter decreased from 3.2 cm to 2.5 cm (see Figure 3).
The outcome of the surgery is presented in Table 1. The total time for the procedure was 240 min, with a blood loss of 50 mL. The patient's recovery was uncomplicated, and he was discharged on the 9th day after the surgery. On postoperative day 3 (POD 3), the amylase level in the drainage fluid was 1,645 U/L. The drain was removed in POD 7 when the amylase level was 54 U/L. This was graded as grade A POPF. No peritoneal fluid was found on the postoperative CT examination on POD 7 (see Figure 4). The patient recovered well and was discharged on POD 9.
Histopathology revealed a moderately poorly differentiated ductal adenocarcinoma with interstitial fibrosis, thus confirming the preoperative diagnosis. The pancreatic interstitial fibrosis may be due to preoperative neoadjuvant chemotherapy (see Table 1). The resection margins of the pancreatic neck and posterior peritoneum were microscopically radical (R0). Only 15 lymph nodes were detected, and none of those were involved. The tumor was staged as T2N0M0 (AJCC 8th edition). The patient underwent six cycles of adjuvant chemotherapy using gemcitabine plus nab-paclitaxel program.
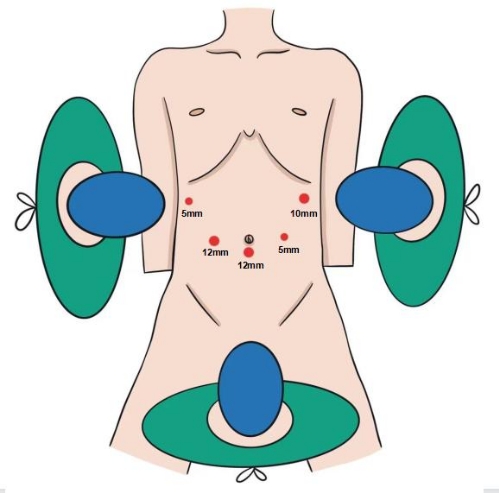
Figure 1: The position of the surgeons. The first surgeon is to the patient's right, the first assistant is to the left, and the second assistant, who is holding the laparoscope, is between the patient's legs. The procedure is performed using a five-port technique. Please click here to view a larger version of this figure.
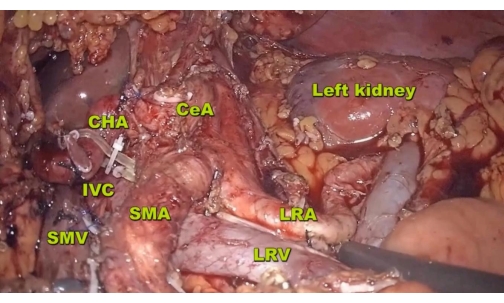
Figure 2: Resection. The resection range extends up to the diaphragmatic crus, down to the LRV, and to the posterior left lateral part of the aorta on the posterior side. Abbreviations: LRA = left renal artery, IVC = inferior vena cava. Please click here to view a larger version of this figure.
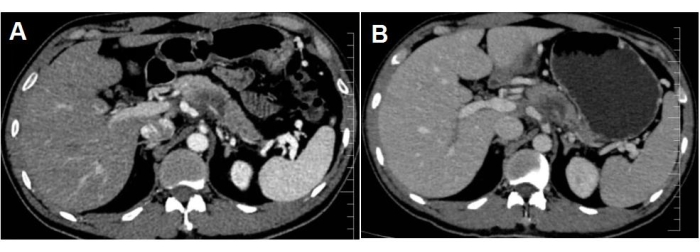
Figure 3: The images show the mass in the pancreatic neck. After neoadjuvant chemotherapy, the tumor maximum diameter was reduced from (A) 3.2 cm to (B) 2.5 cm. Please click here to view a larger version of this figure.
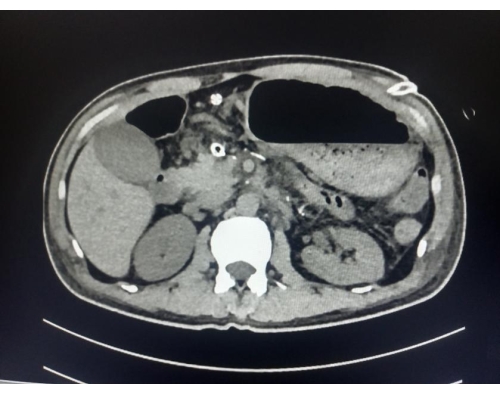
Figure 4: The CT image shows no peritoneal fluid was found on POD 7. Please click here to view a larger version of this figure.
| Variable | Outcome |
| Intraoperative | |
| operative time,minutes | 240 |
| Intraoperative blood loss,mL | 50 |
| Postoperative | |
| Postoperative pancreatic fistula(POPF) | Grade A |
| Drain removal,postoperative day | 7 |
| Postoperative hospital stay,days | 9 |
| Pathological diagnosis | Radically(R0) resected ductal adenocarcinoma with interstitial fibrosis,2cm |
Table 1: Representative outcome of the surgery.
Discussion
Despite the 5 year survival rates of PDAC, this is still unsatisfactory for patients who have the chance for radical resection; surgery has been the only curative therapeutic method till now22. Resectability status is mostly assessed by the conditions of the regional critical vessels, including the SMA, CeA, CHA, and SMV24,25. Although preoperative radiography can provide adequate information about vascular conditions, surgical exploration is the straightforward method for patients who have ambiguity evaluation, especially for the ones who previously had neoadjuvant chemotherapy. Hence, the early assessment of radical resection during surgery is of great importance in order to avoid unnecessary subsequent operation and improve R0 resection rate.
Laparoscopic radical resection of the pancreatic neck is one of the most complicated radical operations for pancreatic cancer. This study provides an L-RAMPS surgical strategy via the dorsal-caudal artery approach, which might develop to be a standardized, reproducible, and oncologically effective procedure in high-volume centers with experienced surgeons. It may offer a new vision of an artery-first aspect by minimally invasive approaches and could determine the feasibility of radical resection at the early stage of RAMPS.
The most important part of this technique is that priority should be given to confirm the dorsal resection plane, which contains the following parts: the dorsal side of the superior mesenteric artery (SMA), the dorsal side of the pancreatic head, the root of the celiac artery (CeA), the ventral side of the left renal vessels, and the renal hilum. On the condition that all the important parts are not infiltrated by the tumor, surgeons could ensure the feasibility and safety of the radical operation, and then resect the tumor en bloc around the SMA and CeA from the caudal side to the cephalic side. This surgical strategy is aimed at improving the rate of R0 resection and further long-term survival.
This complex procedure needs to be performed by highly experienced surgical teams with both open and laparoscopic pancreatic surgical skills. The learning curve is steep as the implementation of this surgical method has high requirements for the cooperation of the surgical team, anatomical cognition of this complicated area, and flexible response capability against unexpected variations. For these reasons, limited cases make the randomized clinical trials difficult to design and accomplish, so high-level evidence for the perioperative and survival results of this technique are difficult to establish.
Disclosures
The authors have nothing to disclose.
Acknowledgements
None.
Materials
| 3D Laparoscope | STORZ | TC200,TC302 | |
| Echelon Flex Powered Plus Articulating Endoscopic Linear Cutter and Endopath Echelon Endoscopic Linear Cutter Reloads with Gripping Surface Technology | Ethicon Endo-Surgery | ECR60G/GST60G | |
| HARMONIC ACE Ultrasonic Surgical Devices | Ethicon Endo-Surgery | HAR36 | |
| Ligating Clips | Teleflex Medical | 5,44,22,05,44,23,05,44,000 | |
| Ultrasonic Surgical & Electrosurgical Generator | Ethicon Endo-Surgery | GEN11CN |
References
- Kim, E. Y., Hong, T. H. Initial experience with laparoscopic radical antegrade modular pancreatosplenectomy for left-sided pancreatic cancer in a single institution: technical aspects and oncological outcomes. BMC Surgery. 17 (1), 2 (2017).
- Strasberg, S. M., Fields, R. Left-sided pancreatic cancer: distal pancreatectomy and its variants: radical antegrade modular pancreatosplenectomy and distal pancreatectomy with celiac axis resection. The Cancer Journal. 18 (6), 562-570 (2012).
- Strasberg, S. M., Linehan, D. C., Hawkins, W. G. Radical antegrade modular pancreatosplenectomy procedure for adenocarcinoma of the body and tail of the pancreas: ability to obtain negative tangential margins. Journal of the American College of Surgeons. 204 (2), 244-249 (2007).
- Strasberg, S. M., Drebin, J. A., Linehan, D. Radical antegrade modular pancreatosplenectomy. Surgery. 133 (5), 521-527 (2003).
- Sunagawa, H., Harumatsu, T., Kinjo, S., Oshiro, N. Ligament of Treitz approach in laparoscopic modified radical antegrade modular pancreatosplenectomy: report of three cases. Asian Journal of Endoscopic Surgery. 7 (2), 172-174 (2014).
- Choi, S. H., Kang, C. M., Lee, W. J., Chi, H. S. Multimedia article. Laparoscopic modified anterior RAMPS in well-selected left-sided pancreatic cancer: technical feasibility and interim results. Surgical Endoscopy. 25 (7), 2360-2361 (2011).
- Weitz, J., Rahbari, N., Koch, M., Büchler, M. W. The "artery first" approach for resection of pancreatic head cancer. Journal of the American College of Surgeons. 210 (2), 1-4 (2010).
- Cho, A., Yamamoto, H., Kainuma, O. Tips of laparoscopic pancreaticoduodenectomy: superior mesenteric artery first approach (with video). Journal of Hepato-Biliary-Pancreatic Sciences. 21 (3), 19-21 (2014).
- Morales, E., et al. Follow "the superior mesenteric artery": laparoscopic approach for total mesopancreas excision during pancreaticoduodenectomy. Surgical Endoscopy. 33 (12), 4186-4191 (2019).
- Nagakawa, Y., et al. Surgical approaches to the superior mesenteric artery during minimally invasive pancreaticoduodenectomy: A systematic review. Journal of Hepato-Biliary-Pancreatic Sciences. 29 (1), 114-123 (2021).
- Pessaux, P., Varma, D., Arnaud, J. P. Pancreaticoduodenectomy: superior mesenteric artery first approach. Journal of Gastrointestinal Surgery. 10 (4), 607-611 (2006).
- Yamamoto, M., et al. New laparoscopic procedure for left-sided pancreatic cancer-artery-first approach laparoscopic RAMPS using 3D technique. World Journal of Surgical Oncology. 15 (1), 213 (2017).
- Rosso, E., et al. Laparoscopic radical antegrade modular pancreatosplenectomy with venous tangential resection: focus on periadventitial dissection of the superior mesenteric artery for obtaining negative margin and a safe vascular resection. Annals of Surgical Oncology. 27 (8), 2902-2903 (2020).
- Kawabata, Y., et al. Laparoscopic versus open radical antegrade modular pancreatosplenectomy with artery-first approach in pancreatic cancer. Langenbeck’s Archives of Surgery. 405 (5), 647-656 (2020).
- Vissers, F. L., et al. Laparoscopic radical left pancreatectomy for pancreatic cancer: surgical strategy and technique video. Journal of Visualized Experiments. (160), e60332 (2020).
- Ome, Y., Seyama, Y., Muto, J. Laparoscopic distal pancreatectomy for left-sided pancreatic cancer using the "Caudo-Dorsal Artery First Approach". Annals of Surgical Oncology. 26 (13), 4464-4465 (2019).
- Tol, J. A., et al. Definition of a standard lymphadenectomy in surgery for pancreatic ductal adenocarcinoma: a consensus statement by the International Study Group on Pancreatic Surgery (ISGPS). Surgery. 156 (3), 591-600 (2014).
- Asbun, H. J., Stauffer, J. A. Laparoscopic approach to distal and subtotal pancreatectomy: a clockwise technique. Surgical Endoscopy. 25 (8), 2643-2649 (2011).
- Asbun, H. J., et al. Technique and audited outcomes of laparoscopic distal pancreatectomy combining the clockwise approach, progressive stepwise compression technique, and staple line reinforcement. Surgical Endoscopy. 34 (1), 231-239 (2020).
- Abu Hilal, M., et al. Laparoscopic radical ‘no-touch’ left pancreatosplenectomy for pancreatic ductal adenocarcinoma: technique and results. Surgical Endoscopy. 30 (9), 3830-3838 (2016).
- Song, K. B., et al. Single-center experience of laparoscopic left pancreatic resection in 359 consecutive patients: changing the surgical paradigm of left pancreatic resection. Surgical Endoscopy. 25 (10), 3364-3372 (2011).
- Bassi, C., et al. The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 Years After. Surgery. 161 (3), 584-591 (2017).
- Mizrahi, J. D., Surana, R., Valle, J. W., Shroff, R. T. Pancreatic cancer. Lancet. 395 (10242), 2008-2020 (2020).
- Varadhachary, G. R., et al. Borderline resectable pancreatic cancer: definitions, management, and role of preoperative therapy. Annals of Surgical Oncology. 13 (8), 1035-1046 (2006).
- Abrams, R. A., et al. Combined modality treatment of resectable and borderline resectable pancreas cancer: expert consensus statement. Ann of Surgical Oncology. 16 (7), 1751-1756 (2009).

