Isolation and Functional Analysis of Arteriolar Endothelium of Mouse Brain Parenchyma
Summary
Intensive preparation of intact mouse cerebral endothelial "tubes" from cerebral parenchymal arterioles is illustrated for studying cerebral blood flow regulation. Further, we demonstrate the experimental strengths of this endothelial study model for fluorescence imaging and electrophysiology measurement of key cellular signaling pathways, including changes in intracellular [Ca2+] and membrane potential.
Abstract
Cerebral blood flow is conveyed by vascular resistance arteries and downstream parenchymal arterioles. Steady-state vascular resistance to blood flow increases with decreasing diameter from arteries to arterioles that ultimately feed into capillaries. Due to their smaller size and location in the parenchyma, arterioles have been relatively understudied and with less reproducibility in findings than surface pial arteries. Regardless, arteriolar endothelial cell structure and function—integral to the physiology and etiology of chronic degenerative diseases—requires extensive investigation. In particular, emerging evidence demonstrates that compromised endothelial function precedes and exacerbates cognitive impairment and dementia.
In the parenchymal microcirculation, endothelial K+ channel function is the most robust stimulus to finely control the spread of vasodilation to promote increases in blood flow to areas of neuronal activity. This paper illustrates a refined method for freshly isolating intact and electrically coupled endothelial "tubes" (diameter, ~25 µm) from mouse brain parenchymal arterioles. Arteriolar endothelial tubes are secured during physiological conditions (37 °C, pH 7.4) to resolve experimental variables that encompass K+ channel function and their regulation, including intracellular Ca2+ dynamics, changes in membrane potential, and membrane lipid regulation. A distinct technical advantage versus arterial endothelium is the enhanced morphological resolution of cell and organelle (e.g., mitochondria) dimensions, which expands the usefulness of this technique. Healthy cerebral perfusion throughout life entails robust endothelial function in parenchymal arterioles, directly linking blood flow to the fueling of neuronal and glial activity throughout precise anatomical regions of the brain. Thus, it is expected that this method will significantly advance the general knowledge of vascular physiology and neuroscience concerning the healthy and diseased brain.
Introduction
Parenchymal arterioles directly deliver essential oxygen and nutrients throughout the brain1. While interfacing with capillaries, highly vasoactive arterioles respond to retrograde signaling initiated by capillary ion channels that sense metabolic signals from specific neuronal regions2. With brain parenchyma having historically received the bulk of investigation, a role for endothelial dysfunction has now emerged for clarifying pathological mechanisms associated with various cerebrovascular disorders that underlie dementia (e.g., ischemic stroke, Alzheimer's disease)3,4,5,6. The endothelium is integral to perfusion of the brain in accord with the heterogeneity of genetics, structure, and function throughout vascular segments7. Pial arteries have been extensively studied due to their relatively large size, high segmental vascular resistance, and role in blood flow distribution to the underlying cerebrum8,9. Thus, a better understanding of arteriolar endothelial mechanisms will likely enhance the understanding of brain blood flow regulation in health and disease towards the development of novel therapeutic regimens.
Emerging evidence highlights the importance of studying parenchymal arterioles in relation to different signaling pathways and diseases8,10. However, this approach has been limited to using intact pressurized arteriole11 and/or capillary-parenchymal arteriole (CaPA) preparations12. Freshly isolated, native cerebral arteriolar endothelial cells devoid of other cell types and confounding factors have not been examined, likely due to technical difficulties in their isolation. This paper advances a previous technique highlighting the isolation of pial arterial endothelium13 to now reliably and reproducibly isolate the endothelium of brain parenchymal arterioles (width: ~25 µm, length: ~250 µm). This technique helps achieve optimal resolution of electrically and chemically coupled cells in their individual orientation and cellular networks.
Key pathways of interest have included the interaction of intracellular Ca2+ ([Ca2+]i) signaling and hyperpolarization of membrane potential (Vm)14,15—integral to vasodilation16—to allow blood to enter the capillaries and deliver oxygen and nutrients to active parenchyma17. These preparations allow for real-time electrophysiological recordings of ion channels, including Ca2+-permeant, transient receptor potential (TRP) and K+ channels and/or fluorescent imaging of intracellular organelles within endothelial cell tubes in near-physiological conditions. This is a suitable technique for researchers interested in physiological cellular mechanisms that govern endothelial cell control of cerebral blood flow delivery to the brain parenchyma. Altogether, this technique will help researchers better understand fundamental endothelial signaling pathways and network communication of arterioles embedded in brain parenchyma while addressing questions related to cerebrovascular physiology and pathology.
Protocol
Experimenters should ensure that designated use of animals and associated protocols are approved by their Institutional Animal Care and Use Committee (IACUC) and performed in accordance with the National Research Council's "Guide for the Care and Use of Laboratory Animals" (8th Edition, 2011) and the ARRIVE guidelines. The IACUC of Loma Linda University and the University of Arizona has approved all protocols used for this manuscript for C57BL/6N and 3xTg-AD mice (males and females; age range: 2-30 months). See Figure 1 as an overview of the isolation and examination of arteriolar endothelial tubes freshly isolated from mouse parenchymal arterioles of the brain.
1. Materials and equipment
NOTE: See the Table of Materials for all reagents and materials required for this protocol. In addition, manuals and websites associated with the respective vendors can also be consulted as needed. Illustrations of dissection stations and experimental apparatuses have been previously provided13.
- Flow chamber
- Fasten a superfusion chamber with a glass coverslip onto a platform composed of anodized aluminum. Secure the platform with the chamber onto an aluminum microscope stage.
- Set a micromanipulator holding a pinning pipette at each end of the platform on the aluminum microscope stage.
NOTE: If necessary, use a transferable stage apparatus with a flow chamber unit to move a secured, isolated endothelial tube from one microscope apparatus to another for experimentation.
- Microscopes
- Use stereomicroscopes (5x to 50x magnification range) and fiber optic light sources for dissection procedures.
- For isolation of endothelial tubes, use an inverted microscope rig equipped with phase contrast- or differential interference contrast (DIC)-compatible objectives (10x, 20x, and 40x) and an aluminum stage.
- Have a microsyringe pump controller ready next to the apparatus dedicated to isolating endothelial tubes from partially digested blood vessels.
- Set up the experimental apparatus by arranging an inverted microscope (objectives: 4x, 10x, 20x, 40x, and 60x) and a manual aluminum stage on a vibration isolation table.
- Intracellular Vm recording equipment
- Connect the electrometer to a compatible headstage. Use accessories, such as a function generator and stimulator, for protocols requiring current injection.
- Connect amplifier outputs to a data digitizer system, oscilloscope and audible baseline monitors. Secure the reference bath electrode (Ag/AgCl pellet) near the flow chamber exit.
- Assemble a photometric system with integrated components of a fluorescence system interface, high-intensity arc lamp and power supply, hyperswitch, photomultiplier tube (or PMT), and camera to measure [Ca2+]i in endothelial cells.
- Assemble a temperature controller equipped with an inline heater to raise and maintain a physiological temperature (37 °C) throughout the experiment.
- Assemble a multichannel platform connected to a valve controller with an inline flow control valve to control delivery of solutions to endothelial tubes secured in the chamber.
- Micropipettes and sharp electrodes
NOTE: The experimenter will need an electronic glass puller and a microforge for preparing trituration and pinning pipettes.- To separate the endothelial tube from the partially digested arteriolar segment, prepare heat-polished trituration pipettes (internal tip diameter: 30-50 µm) from borosilicate glass capillaries.
- To secure the endothelial tube in the superfusion chamber, prepare heat-polished pinning pipettes with a blunted, spherical end (outer diameter: 50-70 µm) prepared from thin-wall borosilicate glass capillaries.
- To record Vm of an endothelial cell, prepare sharp electrodes with a tip resistance of ~150 ± 30 MΩ from glass capillaries using the glass puller only.
2. Solutions and drugs
- Physiological salt solution (PSS)
- Prepare a minimum of 1 L of PSS using 140 mM NaCl, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, 10 mM N-2-hydroxyethylpiperazine-N'-2-ethanesulfonic acid (HEPES), and 10 mM glucose.
- Prepare necessary solutions lacking CaCl2 (zero Ca2+ PSS) for dissection of cerebral arterioles and isolation of endothelial tube.
NOTE: Prepare all solutions in ultrapure deionized H2O, followed by filtration (0.22 µm). Ensure that the final product contains a pH 7.4 with osmolality between 290 and 300 mOsm.
- Bovine serum albumin (BSA, 10%)
- Dissolve 1 g of the lyophilized BSA powder in a beaker of 10 mL of zero Ca2+ PSS. Cover the beaker and allow at least 2 h with slow stirring for the BSA to dissolve.
- Filter solution using a 10 mL syringe plus a 0.22 µm filter and prepare 1 mL aliquots to be stored at -20 °C.
- Dissection solution
- Add 500 µL of BSA (10%) to 49.5 mL of zero Ca2+ PSS for a total volume of 50 mL.
- Transfer solution to a Petri dish for arteriolar dissections.
- Dissociation solution
- Add 5 µL of CaCl2 solution (1 M) and 500 µL of BSA (10%) to 49.5 mL of zero Ca2+ PSS for a total volume of 50 mL. Incubate solution at room temperature for at least 1 h.
- Enzymes
- Prepare 1 mL of digestion solution with 100 µg/mL papain, 170 µg/mL dithioerythritol, 250 µg/mL collagenase (Type H blend), and 40 µg/mL elastase.
NOTE: Enzyme activities can vary, although successful protocols will likely require the following enzymatic activities: ≥ 10 units/mg for papain, ≥ 1 unit/mg for collagenase, and ≥ 5 units/mg for elastase.
- Prepare 1 mL of digestion solution with 100 µg/mL papain, 170 µg/mL dithioerythritol, 250 µg/mL collagenase (Type H blend), and 40 µg/mL elastase.
- Fura-2 and pharmacological agents
- Prepare Fura-2 AM stock in dimethyl sulfoxide (DMSO; 1 mM). Prepare 500 µL of working concentration (10 µM) by adding 5 µL of the stock to 495 µL of PSS for loading.
- Prepare at least 50 mL of working concentrations of pharmacological agents in PSS or DMSO as appropriate.
- Conducting solution
- Prepare 2 M KCl by dissolving KCl in deionized H2O (7.455 g of KCl in 50 mL of H2O). Pass the solution through a syringe with a 0.22 µm filter prior to backfilling the sharp electrodes.
- Fluorescent organelle trackers and antibodies
- Prepare plasma membrane or organellar (e.g., nucleus, endoplasmic reticulum) trackers in the appropriate physiological saline solution according to the manufacturer's instructions.
- Prepare primary and secondary antibodies according to the manufacturer's instructions in the appropriate physiological salt solution.
3. Dissection and isolation of mouse cerebral arterioles
NOTE: Stereomicroscopes and sharpened microdissection tools (e.g., fine-tipped forceps, Vannas-style dissection scissors) must be used for specimen magnification (up to 50x) in all these dissection procedures.
- Isolation of mouse brain
- Anesthetize a standard laboratory mouse (e.g., C57BL/6, 3xTg-AD; 2-30 months old, male or female) using isoflurane inhalation (3% for 2-3 min), followed by immediate decapitation using sharp scissors or guillotine per IACUC approval. Place the mouse head in a Petri dish (diameter 10 cm, depth 1.5 cm) containing cold (4 °C) dissection solution.
- While viewing under a stereomicroscope, remove the skin and hair over the skull and wash away excessive blood with cold dissection solution. Using the tips of standard dissection scissors (e.g., 24 mm blades), make an incision starting with the occipital bone and extending up through the nasal bone of the skull. Use coarse-tipped forceps to carefully open the skull along the incision, and separate connective tissue (meningeal membrane) to isolate the brain containing an intact Circle of Willis.
- Wash the isolated brain with cold dissection solution in a beaker or Petri dish to remove remaining blood from the surface of the brain. Place the brain ventral side facing up in a chamber containing cold dissection solution for isolation of parenchymal arterioles (Figure 2A).
- Isolation of parenchymal arterioles
NOTE: Arterioles arising from the middle cerebral arteries (MCAs) and embedded in brain parenchyma are chosen for this protocol. The arterioles are isolated as previously described11, with modification.- Use steel pins (diameter: 0.2 mm, length: 11 to 12 mm) to secure the isolated brain in cold dissection solution in a Petri dish containing a charcoal-infused silicon polymer coating (depth ≥ 50 cm).
- Using sharp and aligned dissection scissors, cut a rectangle of brain tissue (length: 5 mm, width: 3 mm) (Figure 2B) around the MCA while ensuring that the upper part of the tissue segment is past the branching point from the Circle of Willis. Cut another rectangle of brain tissue from the other hemisphere of the brain to access more arterioles if necessary.
- Secure the separated brain tissue segment into the dish with the MCA facing upwards (distal from the Circle of Willis) using steel pins (diameter: 0.1 mm, length: 13 to 14 mm). Carefully make a shallow incision near the pins to remove the pia with small forceps, gently peeling from one end towards the other (Figure 2C). Remove the pia from the other tissue segment to access more arterioles if needed. Carefully secure the isolated pia with parenchymal arterioles branched from MCA in the dish with the pins, and dissect the parenchymal arterioles (Figure 2D), ensuring that the arterioles are not damaged.
NOTE: If the arterioles are not easily pulled out with the pia from the parenchyma, use fine forceps and dig into the brain, starting at the MCA branch point from the Circle of Willis. Locate the arteriole, carefully loosen the tissue around arterioles (Figure 2E), and gently pull the arteriole out of the parenchyma, holding the upper end of the arteriole (Figure 2F). Multiple arterioles (length: ~1.5-2 mm) can be isolated from one rectangle of brain tissue. - Ensure the arteriole is clean with no tissue attached to it, and cut off any remaining distal branches (Figure 3A). Use this clean, intact arteriole for enzymatic digestion. Alternatively, cut each arteriole into two pieces (length: 0.75-1 mm) for enzymatic digestion for the preparation of endothelial tubes if desired.
4. Digestion of parenchymal arterioles and preparation of endothelial tubes
- Arrangement of equipment and pipettes
NOTE: Isolation of arterial endothelial tubes from the brain has been described previously13,18. The current protocol incorporates modifications for the isolation of endothelium from parenchymal (intracerebral) arterioles. Multiple arterioles or/and pieces of arterioles can be used together for enzymatic digestion.- Assemble the trituration apparatus13 for preparing endothelial tubes using an aluminum stage supporting a chamber and micromanipulators. Assemble a microscope equipped with objectives (5x-60x) and a camera connected to a computer monitor. Secure a microsyringe with a pump controller next to the stage and specimen.
- Secure a trituration pipette backfilled with mineral oil over the microsyringe piston. Use the microsyringe with the pump controller to withdraw the dissociation solution into the trituration pipette (~130 nL) on top of the mineral oil while taking care to avoid air bubbles in the pipette.
- Partial digestion of arteriolar segments
- Place intact arteriolar segments into 1 mL of dissociation solution in a 10 mL glass tube containing 100 µg/mL papain, 170 µg/mL dithioerythritol, 250 µg/mL collagenase, and 40 µg/mL elastase. Incubate arteriolar segments at 34 °C for 10-12 min.
- After digestion, replace the enzyme solution with 3-4 mL of fresh dissociation solution at room temperature.
- Isolation of arteriolar endothelial tube
NOTE: Following digestion and replacement of the enzyme solution, transfer one or multiple partially digested segments into the dissociation solution in a superfusion chamber attached to a mobile platform. While viewing at 100x to 200x magnification, select one partially digested but unbroken vessel segment and focus on it under the microscope.- Place the trituration pipette attached with the microsyringe injector in the dissociation solution in the chamber and position it close to one end of the digested vessel segment. Set a rate within the range of 1-3 nL/s on the pump controller for gentle trituration.
- While maintaining 100x to 200x magnification, withdraw the arteriolar segment into the pipette and then eject to dissociate the adventitia and smooth muscle cells (Figure 3B). Triturate the vessel segment until all smooth muscle cells are dissociated, and only endothelial cells remain as an intact "tube."
NOTE: If necessary during trituration, carefully use fine-tipped forceps to remove the dissociated adventitia and internal elastic lamina from the endothelial tube. Typically, only a few cycles of trituration yield an intact endothelial tube. - Use micromanipulators to secure each end of the endothelial tube on the glass coverslip in the chamber with borosilicate glass pinning pipettes (Figure 3C,D).
- Securing the arteriolar endothelial tube
- Replace the dissociation solution with superfusion solution (PSS containing 2 mM CaCl2) while ensuring that nonendothelial material is washed out of the chamber.
- Transfer secured endothelial tube in the mobile platform to the microscope rig designed for continuous superfusion and intracellular or fluorescent recordings/imaging.
- Apply continuous delivery of PSS during the experiment. Manually set the flow rate (5-7 mL/min) throughout the experiment using the inline flow control valve, consistent with the laminar flow, while matching the feed of solution flow to the extent of vacuum suction. Allow ≥5 min of PSS flow to the chamber for superfusion of the endothelial tube before acquiring background data and/or dye loading.
5. Utilization of arteriolar endothelial tubes for the examination of cellular physiology
NOTE: Isolated and secured arteriolar endothelial tubes can be used for intracellular recordings of [Ca2+]i dynamics and Vm using photometry and sharp electrode electrophysiology, respectively, as previously illustrated13 (Figure 4). [Ca2+]i and Vm can be measured as separate or combined experimental variables as needed13 (Figure 4). However, arteriolar endothelial tubes are more delicate than arterial endothelium, and experimentation time should not exceed 1 h.
- Measurement of [Ca2+]i
- Turn on the equipment and software for [Ca2+]i recordings while maintaining continuous superfusion at a flow rate of 5-7 mL/min.
- Load the endothelial tube with the Ca2+ dye Fura-2 AM for 30 min at room temperature. Wash the cells with superfusion solution for another 20-30 min while gradually raising the bath temperature to 37 °C. Maintain the temperature at 37 °C throughout the experiment.
- Manually adjust the imaging window using photometry software to focus on ~20 endothelial cells (Figure 4A). In the absence of light, turn the PMT on the fluorescence interface and begin acquisition of [Ca2+]i by exciting Fura-2 alternately (≥10 Hz) at 340 nm and 380 nm while collecting fluorescence emission at 510 nm. Once a stable baseline recording of [Ca2+]i is established, apply pharmacological agents (e.g., purinergic receptor agonists) per experimental objective (Figure 4B).
- Measurement of Vm
- Turn on the equipment and software for Vm recordings and set the data acquisition rate (≥10 Hz) while maintaining continuous superfusion at a flow rate of 5-7 mL/min. Gradually raise the bath temperature to 37 °C and maintain it until the end of the experiment.
- Pull a sharp electrode using a borosilicate glass capillary, backfill with 2 M KCl, and secure it over a silver wire coated with chloride in the pipette holder attached to an electrometer, that in turn, is held by a micromanipulator.
- While viewing through the 4x objective, use a micromanipulator to carefully position the sharp electrode tip just over a cell of the arteriolar endothelial tube into the flowing PSS in the chamber.
- Gradually increase magnification to 400x and reposition the electrode tip as needed.
- Using the micromanipulator, gently insert the tip of a sharp electrode into one of the cells of the endothelial tube and start recording Vm using an electrometer (Figure 4A).
- Once the endothelial resting Vm is stable (−30 to −40 mV), apply the desired pharmacological agents per experimental objective (Figure 4C).
6. Cellular imaging
NOTE: Endothelial tubes secured in the chamber of the mobile platform can also be used for microscopic imaging in both live and fixed conditions19 using standard fluorescence or confocal microscopy. Immunohistochemistry with different antibodies for receptors and ion channels can also be applied as previously described20.
- Load the endothelial tube with the fluorescence tracker for the plasma membrane or desired organelle (e.g., nucleus, endoplasmic reticulum) at 37 °C for 15-30 min.
- Wash the cells with fresh superfusion PSS and image live cells under the microscope at the excitation wavelength of respective dyes (Figure 4D,E).
NOTE: Immunohistochemistry can be performed on this tube model using antibodies of interest.
Representative Results
A demonstration of the protocol is shown in Figure 1 with arteriolar dissection and endothelial tube isolation steps as Figure 2 and Figure 3, respectively. Here, endothelial function was assessed by measuring [Ca2+]i and Vm using Fura-2 photometry and sharp electrode electrophysiology (Figure 4A) in response to a pharmacological agent [2-methylthioadenosine diphosphate (MTA), a potent purinergic receptor (P2YR) agonist] at 37 °C. Upon application of MTA (1 µM), [Ca2+]i rapidly increases (Figure 4B) with a concomitant hyperpolarization (Figure 4C). These measurements are a reflection of the activation of Gq-protein-coupled receptors (P2YRs) followed by the activation of small- and intermediate-conductance Ca2+-activated K+ channels (SKCa/IKCa), which initiate an endothelium-dependent hyperpolarization (EDH) vasodilatory pathway. Thus, endothelium isolated from a parenchymal arteriole is functional in isolation and can be used to study key components of pathways that involve mobilization of [Ca2+]i and/or regulation of Vm via K+ channels.
Furthermore, the intact endothelium was incubated at 37 °C with fluorescent trackers for visualization to examine cellular morphology. Live endothelial cell imaging shows co-staining for plasma membrane (green) and nuclei (red) (Figure 4D). In Figure 4E, the plasma membrane (green) was stained together with an endoplasmic reticulum (ER; red) fluorescent stain, whereby areas of apparent overlap of ER within proximity of the plasma membrane appear orange (Figure 4E). These fluorescent images further reveal the cellular integrity of arteriolar endothelium and their possible use to study signaling microdomains located at junctional membrane complexes.

Figure 1: Examination of arteriolar endothelial tube freshly isolated from mouse parenchymal arteriole. As depicted, the brain is isolated from a mouse, and a piece of rectangular brain tissue containing the MCA was removed. Parenchymal arterioles branching from the MCA and entering the brain parenchyma are isolated. Arterioles are enzymatically digested and triturated, producing an intact endothelial tube for study. The prepared endothelial tube enables measurement of intracellular Ca2+ dynamics and Vm while also enabling cellular imaging for fluorescence imaging and immunohistochemistry. Abbreviations: MCA = middle cerebral artery; Vm = membrane potential. Please click here to view a larger version of this figure.

Figure 2: Dissection and isolation of parenchymal arterioles. (A) Isolated mouse brain with intact Circle of Willis. (B) A dissected rectangular piece of brain tissue containing the MCA (white arrow). (C) Isolation of pia from the upper part of the tissue (arteriole, blue arrow). (D) Arterioles (outlined with blue arrowheads) connected to middle cerebral arteries (yellow arrows). (E) Identified arteriole (blue arrow) in the brain tissue. (F) Pulling out the arteriole (blue arrow) from the loosened tissue. Note that panels E and F indicate an alternative method for isolation of the arteriole (see protocol step 3.2.3). Scale bars = 2 mm (A,B,C), 0.2 mm (D), and 1 mm (E,F). Please click here to view a larger version of this figure.
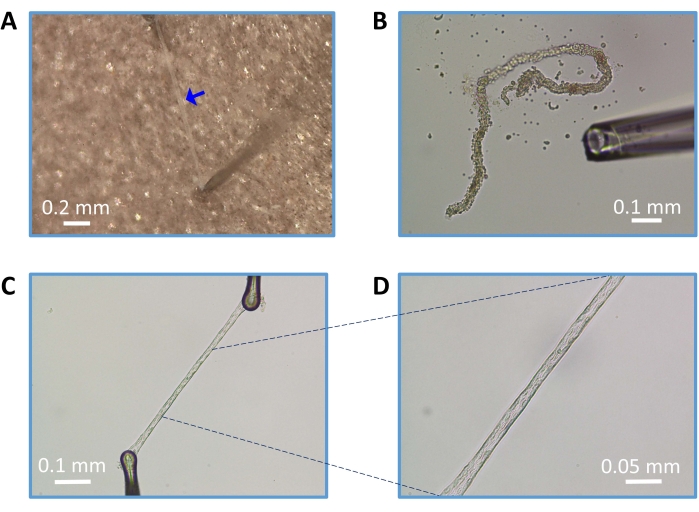
Figure 3: Preparation of endothelial tubes from parenchymal arterioles. (A) Isolated and clean arteriole (blue arrow). (B) Trituration of partially enzyme-digested arteriole. (C) Intact endothelial tube (magnification: 100x) prepared and secured on the glass coverslip using pinning pipettes. (D) Endothelial tube at 200x magnification. Scale bars = 0.2 mm (A), 0.1 mm (B, C), and 0.05 mm (D). Please click here to view a larger version of this figure.

Figure 4: Application of endothelial tube for physiological examination of pathways related to blood flow regulation. (A) A sharp electrode (purple arrow) is positioned in a cell of an endothelial tube focused in the data acquisition window for photometry. (B) Representative recording of intracellular Ca2+ using Fura-2 photometry in response to MTA (1 µM). (C) Representative recording of Vm simultaneous with intracellular Ca2+ trace in panel B. (D) Representative image of plasma membrane (green) and nuclei (red) of live endothelial cells stained with fluorescent dyes. (E) Representative image of plasma membrane (green) and endoplasmic reticulum (ER, red) of live endothelial cells stained with fluorescent dyes. Areas of proximity for ER and plasma membrane are indicated in orange. Images were acquired using a monochrome camera and were pseudo-colored. Scale bars = 20 µm (D), 10 µm (E). Abbreviations: MTA = 2-methylthioadenosine diphosphate; F340/F380 = Fura-2 dye ratio signal; Vm = membrane potential; PM = plasma membrane; ER = endoplasmic reticulum. Please click here to view a larger version of this figure.
Discussion
Growing evidence suggests that cerebrovascular disease (CVD), aging, and Alzheimer's disease are strongly correlated and are a current topic of dementia research4,8,14,21. Thus, it is obvious that studies of the cerebrovascular network would have a broad impact on health while requiring continued extensive investigation during conditions of disease. As a significant point of vascular resistance for cerebral perfusion, the general importance of parenchymal arterioles in the etiology and development of CVD has been highlighted for over 50 years22.
However, scientific examination tools have not been adequately developed to study intact parenchymal arterioles and their composite cell types until the recent onset of ex vivo approaches such as the isolated, pressurized parenchymal arteriole11 and CaPA12 preparations. The cerebrovascular physiology field would also benefit from complementary techniques in homocellular study models (smooth muscle, pericytes, and endothelium) to focus on specific cell types and their genetic and pharmacological characteristics underlying cerebral blood flow regulation and blood-brain barrier (BBB) permeability.
The endothelium is a central regulatory organ that "feeds" all other organs throughout the body, including the brain23. Although all vascular segments (arteries, arterioles, capillaries, venules, veins) are essential for cerebral perfusion, the endothelium of arterioles requires dedicated investigation as a central component to cerebrovascular autoregulation and neurovascular coupling24. Further, Ca2+ signaling25 and K+ channel activity17,26 appear to be enhanced during the regulation of myogenic tone in parenchymal arterioles relative to pial arteries. Thus, we have now extended the previously established method for the pial cerebral arteries (posterior)13 to parenchymal arterioles.
Relative to these previously illustrated approaches11,13, this protocol was refined by empirically adjusting various steps (e.g., enzymatic treatment, trituration) to reproducibly yield intact endothelial tubes from parenchymal arterioles. In addition, this protocol demonstrates the use of this study model to optimize the application of cellular photometry, electrophysiology, and fluorescent imaging (Figure 4). With general strengths and limitations of Fura-2 photometry and sharp electrode electrophysiology previously described in detail13, the experimenter is encouraged to try an array of other Ca2+ signaling and ion channel assays to test specific hypotheses of interest. Further, the advancement and generation of novel genetic models with endothelial cell-specific genetically encoded indicators (e.g. [Ca2+]i27 and voltage28 indicators) can expand the usefulness of this preparation.
For investigative context, this isolated endothelial model is useful for research questions that require studying the role of the arteriolar endothelium in regulation of vascular dynamics, whereby en face measurements are not experimentally feasible due to their small size. Endothelial regulation of cerebral blood flow begins with the activation of various G-protein coupled receptors (e.g., purinergic, muscarinic)18 and ion channels (TRPV329, TRPA15, NMDA receptors8). Activation of Gq-type receptors entails phosphatidylinositol 4,5-bisphosphate (PIP2) lipolysis to generate inositol trisphosphate (IP3) and diacylglycerol for [Ca2+]i mobilization that can then mediate K+ channel activation for changes in Vm15. K+ channel activity, and thereby Vm alone, can also be studied directly under physiological conditions2 or via select pharmacological targeting30. Moreover, the arteriolar endothelial tube enables enhanced fluorescent visualization of select plasma membrane remodeling (e.g., Filipin-III), intracellular organelles (e.g., ER tracker), and cell-to-cell coupling (e.g., propidium iodide)18,19. Thus, another feature of the study model entails effective pairing of cellular morphology and histology with parameters of function, such as Ca2+ and electrical signaling, in aging and the development of chronic diseases.
The components of the protocol that deserve special attention are arteriolar dissection, enzymatic digestion, trituration, and securing of the arteriolar endothelial tube for measurements. During dissection from the brain, damage to the parenchymal arteriole should be avoided at all costs to ensure successful digestion and trituration for isolating an intact and functional endothelial layer. Further, excessive stretching while securing the endothelial tube in the perfusion chamber will also destroy the preparation, effectively negating all prior protocol steps.
Viability tests should be applied to ensure that arteriolar endothelial tubes are functional with respect to key physiological components such as Ca2+ signaling (influx and/or intracellular release) following stimulation of G-protein coupled receptors, the extent of Vm hyperpolarization, and robust intercellular coupling through gap junctions18,23. Examples of such controls include reversible purinergic stimulation using MTA (1 µM; Δ[Ca2+]i ~ 300 nM; ≥10 mV hyperpolarization; Figure 4B,C), reversible SKCa/IKCa activation using NS309 (1 µM; ≥20 mV hyperpolarization), and/or correspondence between current injection (e.g., ~-1 nA) into one cell and Vm responses (~-10 mV) of another at a distance of ≥250 µm18,31. With practice, care, and experimental rigor, the entire protocol is reproducible if the learner has patience and perseverance.
This method has key limitations to address, such as the insufficient abundance of samples (e.g., nucleic acids, proteins, and lipids) and relative fragility for biochemical experiments and live-cell recordings, respectively. However, this molecular biology caveat can be avoided by pooling from several animals and/or devising quantitation methods of higher sensitivity and throughput. If the physiological temperature is to be maintained at 37 °C, short experimental timeframes (≤1 h) will be required with focused pharmacological interventions only. In addition, it would be extremely challenging to isolate arteriolar endothelial tubes from near-term or newborn (<1-month-old) animals, potentially precluding studies of cerebral vascular development.
Other considerations encompass the precise composition of cerebral vascular endothelial tubes, potentially requiring select approaches of immunofluorescence and electrophysiology. For example, the basement membrane (composed primarily of collagen IV, laminin, and fibronectin) runs along the cerebral vascular tree. It is known for its contribution to the integrity of the BBB formed by brain capillary endothelial cells32. It should be noted that this study model may not be appropriate for BBB permeability experiments as basement membrane components are substrates of the enzyme cocktail used for partial digestion of arteriolar endothelial tubes, in particular, the collagenase H blend. Further, application of a selective enzyme cocktail, removal of the internal elastic lamina, and clear identification of endothelial cells per structure (parallel arrangement vs. circumferential for smooth muscle cells), morphology ("teardrop" shape of endothelial cells vs. spindle-shaped smooth muscle cells), and electrophysiology (e.g., hyperpolarization to GPCR stimulation), eliminates physiological contributions of smooth muscle cells and pericytes. Finally, the experimenter must take note of endothelial cell heterogeneity (gene expression profile, structure, function) per organ type (e.g., brain, heart, lung33) in addition to the brain region (e.g., cortex9,34, hippocampus10) and respective vascular segmentation (e.g., arteries, arterioles, and capillaries9,34,35). Thus, a comprehensive study of cerebral blood flow regulation will also require complementary ex vivo (e.g., intravascular pressure myography, capillary-parenchymal arteriole preparation) and in vivo (e.g., intracerebral Laser Doppler) approaches of intact vascular segments and networks.
In summary, this paper presents an advanced technique demonstrating how to prepare an intact endothelial tube freshly isolated from mouse brain parenchymal arterioles. We anticipate that this approach will complement and/or build from in vivo and intact vascular study models. The overall goal is to generate new information for understanding mechanisms underpinning cerebral blood flow at the fundamental level for physiology and select transitions towards pathology for therapy.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This research has been supported by grants from the National Institutes of Health (R00AG047198 & R56AG062169 to EJB; R00HL140106 to PWP) and the Alzheimer's Association (AZRGD-21-805835 to PWP). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the Alzheimer's Association.
Materials
| Amplifiers | Molecular Devices, Sunnyvale, CA, USA | Axoclamp 2B & Axoclamp 900A | |
| Audible baseline monitors | Ampol US LLC, Sarasota, FL, USA | BM-A-TM | |
| Bath Chiller (Isotemp 500LCU) | ThermoFisher Scientific | 13874647 | |
| Borosilicate glass capillaries (Pinning) | Warner Instruments | G150T-6 | |
| Borosilicate glass capillaries (Sharp Electrodes) | Warner Instruments | GC100F-10 | |
| Borosilicate glass capillaries (Trituration) | World Precision Instruments (WPI), Sarasota, FL, USA | 1B100-4 | |
| BSA: Bovine Serum Albumin | Sigma | A7906 | |
| CaCl2: Calcium Chloride | Sigma | 223506 | |
| Collagenase (Type H Blend) | Sigma | C8051 | |
| Cover Glass (2.4 × 5.0 cm) | ThermoFisher Scientific | 12-548-5M | |
| Data Acquision Digitizer | Molecular Devices, Sunnyvale, CA, USA | Digidata 1550A | |
| Dissection Dish (Glass Petri with Charcoal Sylgard bottom) | Living Systems Instrumentation, St. Albans City, VT, USA | DD-90-S-BLK | |
| Dithioerythritol | Sigma | D8255 | |
| DMSO: Dimethyl Sulfoxide | Sigma | D8418 | |
| Elastase (porcine pancreas) | Sigma | E7885 | |
| Endoplasmic Reticulum Tracker (ER-Tracker Red, BODIPY TR Glibenclamide) | ThermoFisher Scientific | E34250 | |
| Fiber optic light sources | Schott, Mainz, Germany & KL200, Zeiss | Fostec 8375 | |
| Flow Control Valve | Warner Instruments | FR-50 | |
| Fluorescence system interface, ARC lamp & power supply, hyperswitch and PMT | Molecular Devices, Sunnyvale, CA, USA | IonOptix Systems | |
| Forceps (Fine-tipped, sharpened) | FST | Dumont #5 & Dumont #55 | |
| Function Generator | EZ Digital, Seoul, South Korea | FG-8002 | |
| Fura-2 AM dye | Invitrogen, Carlsbad, CA, USA | F14185 | |
| Glucose | Sigma-Aldrich (St. Louis, MO, USA) | G7021 | |
| HCl: Hydrochloric Acid | ThermoFisher Scientific (Pittsburgh, PA, USA) | A466250 | |
| Headstages | Molecular Devices | HS-2A & HS-9A | |
| HEPES: (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid) | Sigma | H4034 | |
| Inline Solution Heater | Warner Instruments | SH-27B | |
| KCl: Potassium Chloride | Sigma | P9541 | |
| MgCl2: Magnesium Chloride | Sigma | M2670 | |
| Microforge | Narishige, East Meadow, NY, USA | MF-900 | |
| Micromanipulator | Siskiyou | MX10 | |
| Micropipette puller (digital) | Sutter Instruments, Novato, CA, USA | P-97 or P-1000 | |
| Microscope (Nikon-inverted) | Nikon Instruments Inc, Melville, NY, USA | Ts2 | |
| Microscope (Nikon-inverted) | Nikon Instruments Inc | Eclipse TS100 | |
| Microscope objectives | Nikon Instruments Inc | 20X (S-Fluor) and 40X (Plan Fluor) | |
| Microscope platform (anodized aluminum; diameter, 7.8 cm) | Warner Instruments | PM6 or PH6 | |
| Microscope Stage (Aluminum) | Siskiyou, Grants Pass, OR, USA | 8090P | |
| Microsyringe Pump Controller | World Precision Instruments (WPI), Sarasota, FL, USA | SYS-MICRO4 | |
| MTA: 2-Methylthioadenosine diphosphate trisodium salt | Tocris | 1624 | |
| NaCl: Sodium Chloride | Sigma | S7653 | |
| NaOH: Sodium Hydroxide | Sigma | S8045 | |
| Nuclear Stain (NucBlue Live ReadyProbes Reagent; Hoechst 33342) | ThermoFisher Scientific | R37605 | |
| Oscilloscope | Tektronix, Beaverton, Oregon, USA | TDS 2024B | |
| Papain | Sigma | P4762 | |
| Phase contrast objectives | Nikon Instruments Inc | (Ph1 DL; 10X & 20X) | |
| Plasma Membrane Stain (CellMask Deep Red) | ThermoFisher Scientific | C10046 | |
| Plexiglas superfusion chamber | Warner Instruments, Camden, CT, USA | RC-27 | |
| Scissors (3 mm & 7 mm blades) | Fine Science Tools (or FST), Foster City, CA, USA | Moria MC52 & 15000-00 | |
| Scissors (Vannas style; 9.5 mm & 3 mm blades) | World Precision Instruments | 555640S, 14364 | |
| Stereomicroscopes | Zeiss, NY, USA | Stemi 2000 & 2000-C | |
| Syringe filter (0.22 µm) | ThermoFisher Scientific | 722-2520 | |
| Temperature Controller (Dual Channel) | Warner Instruments | TC-344B or C | |
| Valve Control System | Warner Instruments | VC-6 | |
| Vibration Isolation Table | Technical Manufacturing, Peabody, MA, USA | Micro-g |
References
- Fernandez-Klett, F., Offenhauser, N., Dirnagl, U., Priller, J., Lindauer, U. Pericytes in capillaries are contractile in vivo, but arterioles mediate functional hyperemia in the mouse brain. Proceedings of the National Academy of Sciences of the United States of America. 107 (51), 22290-22295 (2010).
- Longden, T. A., et al. Capillary K+-sensing initiates retrograde hyperpolarization to increase local cerebral blood flow. Nature Neuroscience. 20 (5), 717-726 (2017).
- Kelleher, R. J., Soiza, R. L. Evidence of endothelial dysfunction in the development of Alzheimer’s disease: Is Alzheimer’s a vascular disorder. American Journal of Cardiovascular Disease. 3 (4), 197-226 (2013).
- Hakim, M. A., Behringer, E. J. Development of Alzheimer’s disease progressively alters sex-dependent KCa and sex-independent KIR channel function in cerebrovascular endothelium. Journal of Alzheimers Disease. 76 (4), 1423-1442 (2020).
- Pires, P. W., Earley, S. Neuroprotective effects of TRPA1 channels in the cerebral endothelium following ischemic stroke. elife. 7, 35316 (2018).
- Mughal, A., Harraz, O. F., Gonzales, A. L., Hill-Eubanks, D., Nelson, M. T. PIP2 improves cerebral blood flow in a mouse model of Alzheimer’s disease. Function. 2 (2), (2021).
- Zhao, L., et al. Pharmacologically reversible zonation-dependent endothelial cell transcriptomic changes with neurodegenerative disease associations in the aged brain. Nature Communications. 11 (1), 4413 (2020).
- Peters, E. C., et al. Amyloid-beta disrupts unitary calcium entry through endothelial NMDA receptors in mouse cerebral arteries. Journal of Cerebral Blood Flow and Metabolism. , (2021).
- De Silva, T. M., Modrick, M. L., Dabertrand, F., Faraci, F. M. Changes in cerebral arteries and parenchymal arterioles with aging: Role of rho kinase 2 and impact of genetic background. Hypertension. 71 (5), 921-927 (2018).
- Fontaine, J. T., Rosehart, A. C., Joutel, A., Dabertrand, F. HB-EGF depolarizes hippocampal arterioles to restore myogenic tone in a genetic model of small vessel disease. Mechanisms of Ageing and Development. 192, 111389 (2020).
- Pires, P. W., Dabertrand, F., Earley, S. Isolation and cannulation of cerebral parenchymal arterioles. Journal of Visualized Experiments: JoVE. (111), e53835 (2016).
- Rosehart, A. C., Johnson, A. C., Dabertrand, F. Ex vivo pressurized hippocampal capillary-parenchymal arteriole preparation for functional study. Journal of Visualized Experiments: JoVE. (154), e60676 (2019).
- Hakim, M. A., Behringer, E. J. Simultaneous measurements of intracellular calcium and membrane potential in freshly isolated and intact mouse cerebral endothelium. Journal of Visualized Experiments: JoVE. (143), e58832 (2019).
- Hakim, M. A., Chum, P. P., Buchholz, J. N., Behringer, E. J. Aging alters cerebrovascular endothelial GPCR and K+ channel function: Divergent role of biological sex. Journals of Gerontology, Series A: Biological Sciences and Medical Sciences. 75 (11), 2064-2073 (2020).
- Behringer, E. J., Hakim, M. A. Functional interaction among KCa and TRP channels for cardiovascular physiology: Modern perspectives on aging and chronic disease. International Journal of Molecular Sciences. 20 (6), 1380 (2019).
- Marrelli, S. P., Eckmann, M. S., Hunte, M. S. Role of endothelial intermediate conductance KCa channels in cerebral EDHF-mediated dilations. American Journal of Physiology-Heart and Circulatory Physiology. 285 (4), 1590-1599 (2003).
- Hannah, R. M., Dunn, K. M., Bonev, A. D., Nelson, M. T. Endothelial SKCa and IKCa channels regulate brain parenchymal arteriolar diameter and cortical cerebral blood flow. Journal of Cerebral Blood Flow and Metabolism. 31 (5), 1175-1186 (2011).
- Hakim, M. A., Buchholz, J. N., Behringer, E. J. Electrical dynamics of isolated cerebral and skeletal muscle endothelial tubes: Differential roles of G-protein-coupled receptors and K+ channels. Pharmacological Research and Perspectives. 6 (2), 00391 (2018).
- Hakim, M. A., Behringer, E. J. Methyl-beta-cyclodextrin restores KIR channel function in brain endothelium of female Alzheimer’s disease Mice. Journal of Alzheimers Disease Reports. 5 (1), 693-703 (2021).
- Behringer, E. J., Shaw, R. L., Westcott, E. B., Socha, M. J., Segal, S. S. Aging impairs electrical conduction along endothelium of resistance arteries through enhanced Ca2+-activated K+ channel activation. Arteriosclerosis Thrombosis and Vascular Biology. 33 (8), 1892-1901 (2013).
- Attems, J., Jellinger, K. A. The overlap between vascular disease and Alzheimer’s disease–lessons from pathology. BMC Medicine. 12, 206 (2014).
- Fisher, C. M. The arterial lesions underlying lacunes. Acta Neuropathologica. 12 (1), 1-15 (1968).
- Behringer, E. J. Calcium and electrical signaling in arterial endothelial tubes: New insights into cellular physiology and cardiovascular function. Microcirculation. 24 (3), (2017).
- Dunn, K. M., Nelson, M. T. Neurovascular signaling in the brain and the pathological consequences of hypertension. American Journal of Physiology-Heart and Circulatory Physiology. 306 (1), 1-14 (2014).
- Cipolla, M. J., et al. Increased pressure-induced tone in rat parenchymal arterioles vs. middle cerebral arteries: role of ion channels and calcium sensitivity. Journal of Applied Physiology. 117 (1), 53-59 (2014).
- Cipolla, M. J., Smith, J., Kohlmeyer, M. M., Godfrey, J. A. SKCa and IKCa Channels, myogenic tone, and vasodilator responses in middle cerebral arteries and parenchymal arterioles: effect of ischemia and reperfusion. Stroke. 40 (4), 1451-1457 (2009).
- Chen, Y. L., et al. Calcium signal profiles in vascular endothelium from Cdh5-GCaMP8 and Cx40-GCaMP2 mice. Journal of Vascular Research. 58 (3), 159-171 (2021).
- Bando, Y., Sakamoto, M., Kim, S., Ayzenshtat, I., Yuste, R. Comparative evaluation of genetically encoded voltage indicators. Cell Reports. 26 (3), 802-813 (2019).
- Pires, P. W., Sullivan, M. N., Pritchard, H. A., Robinson, J. J., Earley, S. Unitary TRPV3 channel Ca2+ influx events elicit endothelium-dependent dilation of cerebral parenchymal arterioles. American Journal of Physiology-Heart and Circulatory Physiology. 309 (12), 2031-2041 (2015).
- Behringer, E. J., Segal, S. S. Tuning electrical conduction along endothelial tubes of resistance arteries through Ca2+-activated K+ channels. Circulation Research. 110 (10), 1311-1321 (2012).
- Behringer, E. J., Socha, M. J., Polo-Parada, L., Segal, S. S. Electrical conduction along endothelial cell tubes from mouse feed arteries: confounding actions of glycyrrhetinic acid derivatives. British Journal of Pharmacology. 166 (2), 774-787 (2012).
- Thomsen, M. S., Routhe, L. J., Moos, T. The vascular basement membrane in the healthy and pathological brain. Journal of Cerebral of Blood Flow and Metabolism. 37 (10), 3300-3317 (2017).
- Jambusaria, A., et al. Endothelial heterogeneity across distinct vascular beds during homeostasis and inflammation. elife. 9, 51413 (2020).
- Diaz-Otero, J. M., Garver, H., Fink, G. D., Jackson, W. F., Dorrance, A. M. Aging is associated with changes to the biomechanical properties of the posterior cerebral artery and parenchymal arterioles. American Journal of Physiology-Heart and Circulatory Physiology. 310 (3), 365-375 (2016).
- Chen, M. B., et al. Brain endothelial cells are exquisite sensors of age-related circulatory cues. Cell Reports. 30 (13), 4418-4432 (2020).

