Establishing Human Lung Organoids and Proximal Differentiation to Generate Mature Airway Organoids
Summary
The protocol presents a method to derive human lung organoids from primary lung tissues, expand the lung organoids and induce proximal differentiation to generate 3D and 2D airway organoids that faithfully phenocopy the human airway epithelium.
Abstract
The lack of a robust in vitro model of the human respiratory epithelium hinders the understanding of the biology and pathology of the respiratory system. We describe a defined protocol to derive human lung organoids from adult stem cells in the lung tissue and induce proximal differentiation to generate mature airway organoids. The lung organoids are then consecutively expanded for over 1 year with high stability, while the differentiated airway organoids are used to morphologically and functionally simulate human airway epithelium to a near-physiological level. Thus, we establish a robust organoid model of the human airway epithelium. The long-term expansion of lung organoids and differentiated airway organoids generates a stable and renewable source, enabling scientists to reconstruct and expand the human airway epithelial cells in culture dishes. The human lung organoid system provides a unique and physiologically active in vitro model for various applications, including studying virus-host interaction, drug testing, and disease modeling.
Introduction
Organoids have become a robust and universal tool for in vitro modeling of organ development and studying biology and disease. When cultured in a growth factor-defined culture medium, adult stem cells (ASC) from a variety of organs can be expanded in 3-dimension (3D) and self-assembled into organ-like cellular clusters composed of multiple cell types, termed organoids. Clevers' laboratory reported the derivation of the first ASC-derived organoid, human intestinal organoid, in 20091,2. Afterward, ASC-derived organoids have been established for a variety of human organs and tissues, including prostate3,4, liver5,6, stomach7,8,9, pancreas10, mammary gland11, and lung 12,13. These ASC-derived organoids retained the critical cellular, structural, and functional properties of the native organ and maintained genetic and phenotypic stability in long-term expansion culture14,15.
Organoids can also be derived from pluripotent stem cell (PSC), including embryonic stem (ES) cells and induced pluripotent stem (iPS) cell16. While PSC-derived organoids exploit the mechanisms of organ development for their establishment, ASCs can be coerced to form organoids by rebuilding conditions that mimic the stem cell niche during physiological tissue self-renewal or tissue repair. PSC-derived organoids are favorable models to explore development and organogenesis, albeit unable to reach the comparable maturation level of ASC-derived organoids. The fetal-like maturation status of PSC-derived organoids, and complexity for establishing these organoids substantially prevent their broad applications for studying biology and pathology in mature tissues.
The human respiratory tract, from nose to terminal bronchiole, is lined with the airway epithelium, also called the pseudostratified ciliated epithelium, which consists of four major cell types, i.e., ciliated cell, goblet cell, basal cell, and club cell. We established the ASC-derived human lung organoid from human lung tissues in collaboration with Clevers' lab12,13. These lung organoids are consecutively expanded in the expansion medium for over a year; the precise duration varies among different organoid lines obtained from different donors. However, compared to the native airway epithelium, these long-term expandable lung organoids are not mature enough since ciliated cells, the major cell population in the human airway, are under-represented in these lung organoids. Thus, we developed a proximal differentiation protocol and generated 3D and 2D airway organoids that morphologically and functionally phenocopy the airway epithelium to a near-physiological level.
Here we provide a video protocol to derive human lung organoids from the primary lung tissues, expand the lung organoids and induce proximal differentiation to generate 3D and 2D airway organoids.
Protocol
All experimentation using human tissues described herein was approved by the Institutional Review Board of the University of Hong Kong/Hospital Authority Hong Kong West Cluster (UW13-364 and UW21-695). Informed consent was obtained from patients before tissue collection.
1. Derivation of human lung organoid
- Preparation of experimental materials
- Prepare basal medium by supplementing advanced DMEM/F12 medium with 2 mM glutamine, 10 mM HEPES, 100 U/mL of penicillin, and 100 µg/mL of streptomycin.
- Prepare human lung organoid expansion medium by supplementing basal medium with 10% R-spondin 1 conditioned medium, 10% Noggin conditioned medium, 1x B27 supplement, 1.25 mM N-acetylcysteine, 10 mM nicotinamide, 5 µM of Y-27632, 500 nM of A-83-01, 1 µM of SB202190, 5 ng/mL of fibroblst growth factor (FGF)-7, 20 ng/mL of FGF-10, and 100 µg/mL of primocin (see Table of Materials).
NOTE: The R-spondin 1 and Noggin conditioned medium can be replaced with commercial recombinant R-spondin 1 (500 ng/mL) and Noggin (100 ng/mL). - Prewarm a 24-well suspension culture plate in a cell culture incubator. Use a standard cell culture incubator with 5% CO2 and humidified atmosphere at 37 °C. Thaw basement matrix in a 4 °C fridge. Keep the basement matrix and culture medium on ice during experimentation.
- Cell isolation from human lung tissues for 3D organoid culture
- Procure freshly resected human lung tissues sized around 0.5 cm from patients who undergo surgical resection due to various diseases. Transport the lung tissues in 30 mL of basal medium at room temperature and process as quickly as possible inside a biosafety hood.
- Mince lung tissues into small pieces (0.5-1 mm) with a sterile scalpel in a 10 cm cell culture dish. Wash tissue pieces with 10 mL of cold basal medium in a 15 mL centrifuge tube, followed by centrifugation at 400 x g for 5 min at 4 °C.
- Discard the supernatant and resuspend the pellet in 8 mL of basal medium supplemented with collagenase at a final concentration of 2 mg/mL. Digest the tissue pieces by shaking the tube at 120 rpm for 30-40 min at 37 °C.
- Pipet up and down for 20x to shear the digested tissue pieces using a 10 mL serological pipette. Stack a 100 µm strainer on a 50 mL centrifuge tube and filter the suspension.
- Recover tissue pieces on the strainer with the basal medium and transfer them to a 15 mL centrifuge tube, followed by a second round of shearing and filtering. Additional shearing-filtering can be performed 1x-2x to isolate more cells, especially when a small piece of tissue (e.g., <0.5 cm) is procured.
- Add FBS to the flow-through with a final concentration of 2% to terminate digestion, followed by centrifugation at 400 x g for 5 min at 4 °C.
- Resuspend the cell pellet in 10 mL of basal medium, followed by centrifugation at 400 x g for 5 min at 4 °C. Discard the supernatant.
- (Optional) If many erythrocytes are seen in the pellet (estimated by the color of the pellet), resuspend the cell pellet in 2 mL of red blood cell lysis buffer and incubate at room temperature for 5 min. Then, add 10 mL of basal medium to the tube, followed by centrifugation at 400 x g for 5 min at 4 °C. Discard the supernatant.
- Resuspend the pellet in cold basement matrix and keep on ice. Add 80-160 µL of basement matrix for cells recovered from a lung tissue of size around 0.5 cm; the amount is sufficient to seed 2-4 droplets.
- Dispense 40 µL of suspension to each well of a pre-warmed 24-well suspension culture plate. Incubate the culture plate at 37 °C for 10-15 min. Let the basement matrix solidify to form a droplet.
- Add 500 µL of human lung organoids expansion medium supplemented with 5 nM of Heregulin beta-1 to each well and incubate the plate in a cell culture incubator.
- Refresh the medium every 3 days. Remove the old medium while keeping the droplet intact and add fresh medium with caution. Passage the organoids after incubation for 10-14 days. Use Heregulin beta-1 only in the initial culture before the first passage.
- Observe the organoids under a microscope to ensure the organoids are not embedded with a very high cell density. If a droplet disintegrates due to an overly high cell density, recover and re-embed the organoids and cells with a higher volume of basement matrix to make more droplets with a lower and desirable cell density.
2. Expansion of human lung organoids
- Prepare a Pasteur pipette by burning the tip of a Pasteur pipette onto a flame, such as a Bunsen burner, to narrow the opening from 1.5 mm to around 1.0 mm in diameter. Cool down the pipettes, followed by autoclaving to sterilize. Wet the pipettes with the basal medium to avoid cell attachment and loss during mechanical shearing.
- Lung organoids passage with mechanical shearing
- Use a 1 mL tip and pipet up and down to break the droplets obtained above. Then, transfer the organoids along with the medium to a 15 mL centrifuge tube and adjust the volume to 10 mL with cold basal medium. Discard the supernatant after centrifugation at 300 x g for 5 min at 4 °C
- Wash the organoids with 10 mL of cold basal medium once again.Resuspend the organoids in 2 mL of cold basal medium. Pipet up and down to shear the organoids into small pieces with a Pasteur pipette.
- Supplement with basal medium to a total volume of 10 mL, followed by centrifugation at 300 x g for 5 min at 4 °C. Resuspend the organoid fragments with cold basement matrix sufficient to enable a 1:3 to 1:5 expansion. Keep on ice.
- Place 40 µL of organoid suspension in each well of a pre-warmed 24-well plate. Incubate the culture plate at 37 °C for 10-15 min. Let the basement matrix solidify.
- Add 500 µL of lung organoid expansion medium to each well and incubate in a cell culture incubator. Refresh the medium every 3 days. Passage the organoids every 2 weeks with a ratio of 1:3 to 1:5.
- Lung organoids passage with trypsinization
NOTE: Trypsinization is preferred when it is difficult to shear the lung organoid into small pieces using a Pasteur pipet, or the size of the organoids is highly variable, or the subsequent experimentations require organoids of more uniform size.- Harvest the lung organoids as shown in step 2.2.1. Resuspend the organoid in 1 mL of dissociation enzyme and incubate in a 37 °C water bath for 3-5 min.
- Add 1 mL of basal medium to the tube. Shear the organoids mechanically by pipetting the organoids up and down into small pieces using a Pasteur pipet. Check the size of organoid pieces under a microscope at 4x magnification. Then, add 40 µL of FBS to terminate digestion.
NOTE: Determine the size of organoid fragments under the microscope according to the experimental arrangement. If an experiment needs more organoids or organoids of more uniform size, shear organoids into smaller pieces or even single cells. Then, it takes a longer time, probably 3 weeks, before the organoids are ready for experimentation. - Top up with basal medium to a final volume of 10 mL, followed by centrifugation at 300 x g for 5 min at 4 °C. Resuspend the organoid pieces in cold basement matrix with a volume sufficient to passage with a ratio of 1:5-1:10. Keep on ice.
- Place 40 µL of organoid suspension in each well of a pre-warmed 24-well plate. Incubate the culture plate at 37 °C for 10-15 min. Let the basement matrix solidify.
- Supplement with 500 µL of lung organoid expansion medium per well and incubate in a cell culture incubator. Refresh the expansion medium every 3 days. Passage the organoids after 2-3 weeks.
NOTE: Around 100 organoids are mounted within a 40 µL droplet of basement matrix. The lung organoids normally grow better with a relatively high cell density. Re-embed the organoids with a lower cell density if droplets disintegrate or the growing organoids attach together due to the overly high cell density.
3. Proximal differentiation to generate mature airway organoids
- Prepare proximal differentiation medium (PD medium) by supplementing the air liquid interface basal medium with 1x air liquid interface supplement, 1x air liquid interface maintenance supplement, 4 µg/mL of heparin, 1 µM of hydrocortisone, 10 µM of Y-27632, and 10 µM of DAPT (see Table of Materials).
- 3D airway organoids
- Incubate lung organoids in the expansion medium for 7-10 days after passaging via mechanical shearing. Replace the expansion medium with the PD medium. Incubate the organoids in the PD medium for 14 days in a cell culture incubator.
- Discard the PD medium in each well. Add cell lysate buffer to harvest the differentiated airway organoid for RNA extraction and detection of cellular gene expression by RT-qPCR assay.
- Alternatively, incubate the organoids at 37 °C for 60 min after addition of 10 mM EDTA to disassociate the organoids into single cells, followed by flow cytometry analysis to examine cell populations. The organoids are ready for various experimental manipulations.
- 2D airway organoid
- Prepare sufficient 3D lung organoids for 2D differentiation culture. A total of 1.3 x 105 and 4.5 x 105 cells are required for a 24-well permeable support insert and a 12-well permeable support insert, respectively.
- After 3D lung organoids grow in the expansion medium for 2 weeks, digest the organoids into single cells and seed into the 24-well and 12-well inserts to generate 2D airway organoids.
- Pre-incubate the inserts with the basal medium overnight in a cell culture incubator. Add 250 µL and 500 µL of basal medium in the top and bottom chamber of a 24-well plate, respectively. For a 12-well plate, add 500 µL and 1,000 µL of basal medium in the top and bottom chamber, respectively.
- Harvest 3D lung organoids as described in step 2.2.1. Resuspend the organoids with 1 mL of dissociation enzyme and incubate in a 37 °C water bath for 3-5 min.
- Add 1 mL of basal medium to the tube. Pipet up and down to shear the organoids into single cells with a Pasteur pipet and check the cells under a microscope. Then, add 40 µL of FBS to terminate digestion.
- Filter the cells through a 40 µm strainer into a 50 mL centrifuge tube. Transfer the filtered cell suspension to a 15 mL tube. Top up with basal medium to a total volume of 10 mL, followed by centrifugation at 300 x g for 5 min at 4 °C.
- Resuspend the pellet, collected from 24 droplets (40 µL), in 1-2.5 mL of lung organoid expansion medium depending on the cell density in the droplets. Count the number of cells with a cell counter under a microscope. Adjust the cell concentration to 1.3 x 106/mL (for 24-well inserts) or 9 x 105/mL (for 12-well inserts).
- Remove the basal medium from the top and bottom chambers. Add 500 µL and 1,000 µL of expansion medium in the bottom chamber of the 24-well insert and 12-well insert, respectively. Seed 100 µL and 500 µL of cell suspension prepared in step 3.3.7 onto the apical chamber of the 24-well insert and 12-well insert, respectively.
- Incubate in a cell culture incubator for 2 days. Replace the expansion medium with the PD medium in both apical and bottom chamber of the plates. Incubate the organoids in cell culture incubator for 14 days and refresh the PD medium every 3 days.
NOTE: Mobile cilia are discernible in the 3D and 2D organoids under a microscope from day 7 after incubation in the PD medium. After 14 days of differentiation culture in the PD medium, the airway organoids are mature for various experimental manipulations. - Measure trans-epithelial electrical resistance (TEER) every other day using an electrical resistance measurement systemaccording to a standard protocol described in17.
Representative Results
This protocol enables the derivation of human lung organoids with a high success rate. Fresh human lung tissue is minced into small pieces, and then decomposed with collagenase. The resultant single cells are embedded in the basement matrix and incubated in the lung organoid expansion medium supplemented with a cocktail of niche factors for the outgrowth of epithelial stem cells (step 1.1.2). Figure 1 shows the microphotograph of freshly isolated lung cells embedded in reduced growth factor basement membrane matrix Type 2 (BME; Figure 1A, left). Cystic organoids appear and enlarge over time (Figure 1A, right). Meanwhile, the unrelated cells undergo cell death gradually. Fibroblasts are present in the culture during the first or second passages. Afterward, the culture contains epithelial organoids exclusively, which are lung organoids derived from epithelial stem cells present in the primary lung tissues. These lung organoids are passaged every 2-3 weeks by mechanical shearing at a ratio of 1:3 to 1:5 or by trypsinization at a ratio of 1:5 to 1:10 (steps 2.2-2.3). The representative microphotographs of lung organoids after the fourth passage are shown in Figure 1B. After mechanical shearing, the organoid fragments embedded in BME form cystic domains within a couple of hours (Figure 1B, left). A microphotograph of the same field on day 5 (Figure 1B, right) shows organoids growing over time. These expanding human lung organoids harbor all the four major airway epithelial cell types, including ACCTUB+ or FOXJ1+ ciliated cell, P63+ basal cell, CC10+ club cell, and MUC5AC+ goblet cell18 (Figure 1C), in a premature state. Notably, these human lung organoids can be consecutively and stably passaged for over 1 year. When maintained within the basement matrix, lung organoids are most likely to show an apical-in polarity, less than 2%-3% of lung organoids show an apical-out polarity13. As a result, cell apexes are not readily accessible unless the 3D organoids are sheared open.
However, compared to the native human airway epithelium, these long-term expandable lung organoids are not sufficiently mature since the dominant cell population in the native human airway epithelium, ciliated cell, is under-represented in the lung organoid. We then defined a proximal differentiation (PD) medium to improve the maturation status of human lung organoids. The organoids incubated in the expansion medium and the PD medium developed distinct morphology over time (Figure 2A). Motile cilia were more abundant in the organoids in the PD medium than those in the expansion medium. After 2 weeks of differentiation culture in the PD medium, motile cilia are discernible in every single organoid (Figure 2B and Supplementary Video 1). Interestingly, the beating cilia drive the cell debris and excreted mucin inside the organoid lumen to swirl unidirectionally, which adequately recapitulates the mucociliary escalator to remove the inhaled particles (Supplementary Video 1), an important self-clearing mechanism of the human airways. We demonstrate that the ciliated cells increased dramatically to around 50% in the differentiated organoids as compared to the original lung organoids. To assess the percentages of four types of epithelial cells, the 2D airway organoids were analyzed by flow cytometry. Briefly, the organoids were dissociated with 10 mM EDTA for 60 min at 37 °C, fixed with 4% PFA, and permeabilized with 0.1% surfactant. Subsequently, the cells were incubated with primary antibodies (see Table of Materials) for 1 h at 4 °C followed by staining with secondary antibodies. A FACS system was used to analyze the samples. The flow cytometry analysis demonstrated differentiated organoids accommodate four airway epithelial cell types (Figure 2C). Therefore, we developed a proximal differentiation protocol to generate airway organoids that can faithfully simulate the human airway epithelium to a near-physiological level.
To enable the organoid apical surface to be readily accessible and better model the human airway epithelium exposure to respiratory pathogens, we generated 2D monolayers of airway organoids. After 2 weeks of differentiation culture, 2D airway organoids developed an intact epithelial barrier (Figure 3A,B). We also performed a dextran blockage assay to assess the integrity of epithelial barrier formed in 2D airway organoids. On day 10 after culture in transwell inserts, fluorescein isothiocyanate-dextran (MW 10,000) was added in the medium of the top chamber and incubated at 37 °C for 4 h. The media in the top and bottom chambers were harvested for a fluorescence assay. Dextran blockage index refers to the fluorescence intensity of the medium in the top chamber versus that in the bottom chamber (Figure 3B). These 2D airway organoids also contain abundant ciliated cells (Figure 3C). Ciliated cells were labeled by anti-β-Tubulin IV antibody (ACCTUB) and goat anti-mouse 488 secondary antibody. Confocal images were acquired using a confocal microscope. Multi-channel images were acquired using the lasers 405 nm for DAPI, 488 nm for ACCTUB, and 633/640 nm for Phalloidin. Imaging parameters were adjusted according to the user manual of the confocal microscope. Briefly, the pinhole size was set to 1 AU, the master gain was set to 650 V to 750 V with digital gain of 1.0, and the laser power was adjusted for each channel within the range of 0.2% to 5%. Image processing was performed by using the analysis software provided.
The human respiratory tract is lined with two distinct types of epitheliums, i.e., airway epithelium and alveolar epithelium. The former lines the airways from the nasal cavity to the terminal bronchiole and consists of four major types of epithelial cell, i.e., ciliated cell, goblet cell, club cell, and basal cell. In addition, the airway epithelium lining the proximal and distal airways shows a variable cellular composition along the proximal-distal axis. The proximal airway epithelium is pseudostratified, consisting of abundant ciliated cells and mucus-secreting goblet cells; whereas the distal airway epithelium is a single layer of cuboidal ciliated and club cells with less basal and goblet cells19. Human lung tissues used for deriving lung organoids have been procured from patients who underwent surgical resections due to various diseases. We use normal lung tissues adjacent to the diseased tissues for organoid culture. These lung tissues typically contain bronchioles of variable size surrounded by alveolar sacs. During the initial culture, airway epithelial stem cells or airway progenitor cells in the lung tissues survive and proliferate due to the niche factors in the expansion medium. The expansion medium enables initial derivation and long-term expansion of lung organoids by directing the organoids toward an immature state, while the airway differentiation protocol generates airway organoids phenocopying the native airway epithelium morphologically and functionally. The model system, including derivation, expansion, and differentiation of lung organoids, is outlined in Figure 4. The cellular composition in the proximal and distal airway epithelium is also illustrated in Figure 4.
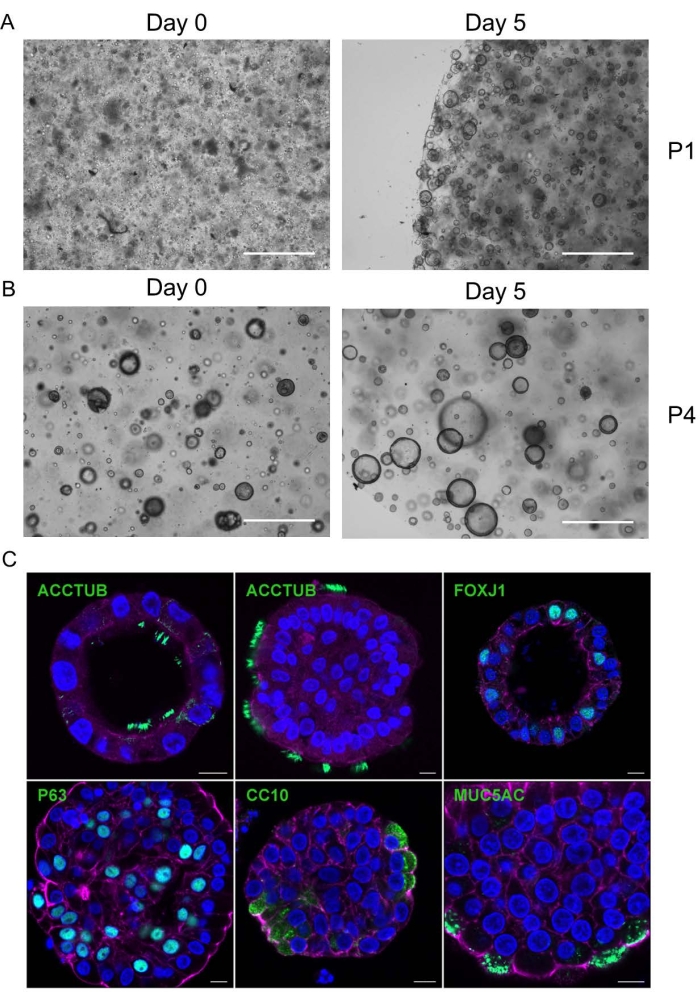
Figure 1: Derivation, expansion, and characterization of human lung organoids. (A) A representative microphotograph shows single cells embedded in the basement matrix after isolation from lung tissues on day 0 (left). On day 5, cystic organoids are growing (right). Scale bar is 0.5 mm. (B) A representative microphotograph of lung organoids on the day of the fourth passage and day 5 after passage. Scale bar is 0.5 mm. P1 and P4 represent the first and fourth passages. The images were taken at 10x magnification. (C) Confocal images of four airway epithelial cell types in human lung organoids. Four lineages of airway epithelial cells are present in the lung organoids, including ACCUB+ and FOXJ1+ ciliated cells, P63+ basal cells, CC10+ club cells, and MUC5AC+ goblet cells. Nuclei and cellular actin filaments are counterstained with DAPI (blue) and Phalloidin-647 (purple), respectively. Scale bar is 10 µm. This figure has been adopted from13. Please click here to view a larger version of this figure.
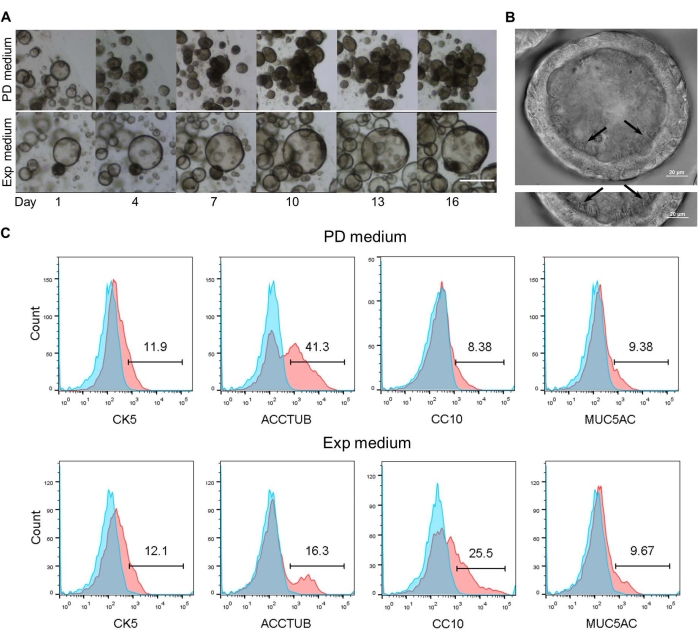
Figure 2: Proximal differentiation of human lung organoids. (A) Human lung organoids were cultured in the PD medium or the expansion (Exp) medium in parallel for 16 days. Bright-field microphotographs of organoids at the indicated days are shown. Scale bar is 0.4 mm. (B) Cilia in the differentiated airway organoids are shown (black arrow). Scale bar is 20 µm. (C) The percentages of individual cell types in organoids incubated in PD medium (top) and expansion medium (bottom) as detected by FACS analysis. The representative histograms of one organoid line are shown. The experiment was performed in three different organoid lines. This figure has been adopted from13. Please click here to view a larger version of this figure.
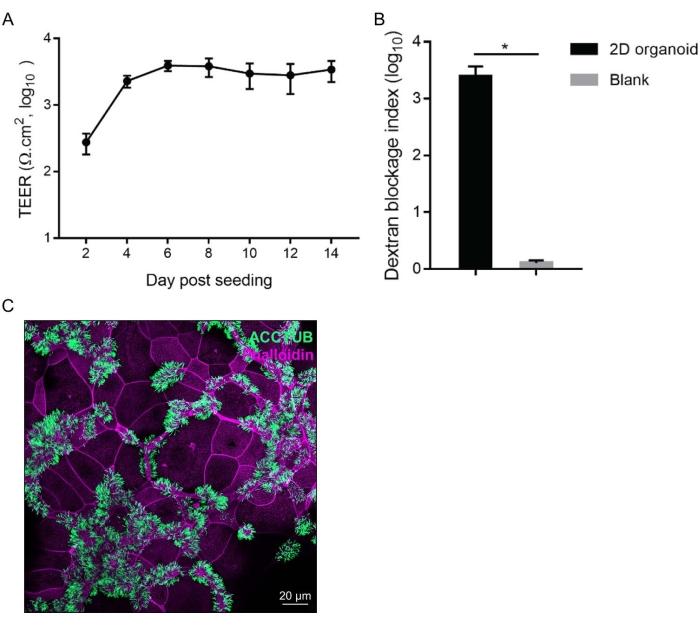
Figure 3: Generation of 2D differentiated airway organoids. (A) Trans-epithelial electronic resistance (TEER) was measured at the indicated day after incubation in the PD medium. Data show mean ± standard deviation (SD) of 2D monolayers in 10 inserts. (B) On day 10 after culture in permeable support plates, fluorescein isothiocyanate-dextran was added and the media in the top and bottom chambers were harvested for a fluorescence assay after 4 h. Dextran blockage index refers to the fluorescence intensity of the medium in the top chamber versus that in the bottom chamber. The diameter of permeable support inserts used in our experiment is 0.4 µm. Without seeding any cells, the dextran can freely penetrate the normal 2D inserts. Thus, the dextran blockage index of a normal 2D (the bar labeled with Blank) should be 1. Data represent the mean ± SD of 10 inserts seeded with 2D airway organoids (2D organoid) and those in two blank inserts (blank). (C) Confocal images of abundant ACCTUB+ ciliated cells (green) in 2D airway organoids. Cellular actin filaments are counterstained with Phalloidin-647 (purple). Scale bar is 20 µm. This figure has been adopted from13. Please click here to view a larger version of this figure.
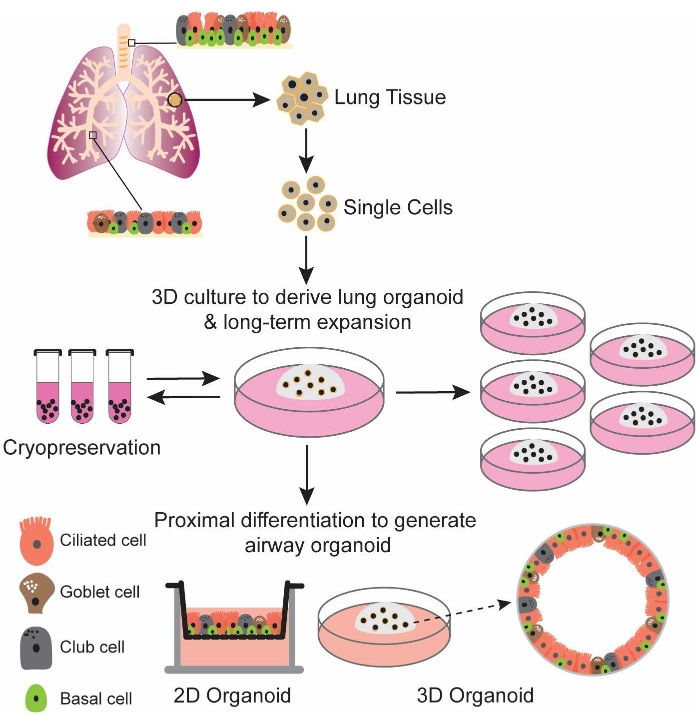
Figure 4: Schematic illustration of derivation, expansion, and differentiation of human lung organoids. Single cells isolated from human lung tissues are directly embedded in the basement matrix and incubated in the lung organoid expansion medium. The derived human lung organoids can be long-term expanded with high stability and readily recovered from cryopreserved stocks. Upon differentiation, the generated airway organoids can faithfully simulate human airway epithelium. 2D and 3D airway organoids have been developed for various experimental manipulations. Please click here to view a larger version of this figure.
Supplementary Video 1. The synchronously beating cilia drive the cell debris to swirl unidirectionally in the differentiated airway organoids13. This video has been adopted from13. Please click here to download this Video.
Discussion
The human airways are lined with the airway epithelium, also known as the pseudostratified ciliated epithelium. The major cell types of the upper airway epithelium are ciliated cells that enable the coordinated movement of their apical cilia to expel mucus and inhaled particles from the airways, goblet cells that produce and secrete mucus, and basal cells that line the basement membrane and are implicated in regeneration. In the small airway such as bronchioles, the cuboidal airway epithelium contains secretory club cells and fewer ciliated cells than in the upper airway regions. We describe a robust protocol to derive human lung organoids from the epithelial stem cells in human lung tissues. These human lung organoids are maintained in the expansion medium and consecutively passaged for over 1 year with high stability. Key growth factors in the expansion medium include R-spondin, a Wnt agonist20; and Noggin, which is an inhibitor of BMP signaling21, as well as FGF7 and FGF10. Prior studies have revealed a crucial role of Wnt, FGF, and BMP signaling in the homeostasis of respiratory epithelium22,23,24. The expansion medium enables initial derivation and long-term expansion of lung organoids by directing the organoids toward an immature state. We further develop a proximal differentiation method to generate 3D and 2D airway organoids, that accommodate four major airway cell types and simulate the human airway epithelium to a near-physiological level. During the whole procedure, including initial derivation, long-term expansion, and proximal differentiation, neither tedious cell purification nor feeder and stromal cells are required. Thus, we establish an organoid model of the human airway epithelium. The two phases of culture, expansion culture and differentiation culture, are mutually exclusive. The lung organoids provide a stable source for long-term expansion, while differentiated airway organoids faithfully phenocopy the human airway epithelium. These organoids are amenable to various experimental manipulations, including imaging, RNA sequencing, flow cytometry analysis, genetic editing, etc.13,14,25,26,27.
To ensure high efficiency to derive lung organoids, the expansion medium must be reconstituted accurately and meticulously, which is essential for the high establishment rate enabled by the protocol. A major limitation of this organoid model is the pure epithelial composition, lack of stromal cells, and immune cells present in the human respiratory mucosa, which may cause the airway organoids to deviate from the native airway epithelium to some extent. Thus, we strive to generate the next generation of respiratory organoids by incorporating immune cells and other biologically relevant components into our current organoid model.
The airway organoids we established faithfully simulate the multi-cellular composition and functionality of the native human respiratory epithelium to a near-physiological level, which is impossible in any homogenous cell lines. Our organoid models enable scientists to reconstruct and stably expand the native human airway epithelium in culture plates. These airway organoids are a universal tool to study the biology and pathology of the human airways. Primary airway epithelial cells used in research labs are not expandable due to the limited replicative capacity and hardly serve as a reproducible and readily-accessible research tool.
Organoids, including human respiratory organoids, have revealed their uniqueness and strength for studying human pathogens, including SARS-CoV-228,29,30,31,32,33,34. As a universal and physiological-active tool, human lung organoids can be widely utilized to explore the biology and pathology of the human respiratory tract.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank the Center of PanorOmic Sciences and Electron Microscope Unit, Li Ka Shing Faculty of Medicine, University of Hong Kong, for assistance in confocal imaging and flow cytometry. This work was partly supported by funding from Health and Medical Research Fund (HMRF, 17161272 and 19180392) of the Food and Health Bureau; General Research Fund (GRF, 17105420) of the Research Grants Council; and Health@InnoHK, Innovation and Technology Commission, the Government of the Hong Kong Special Administrative Region.
Materials
| Reagents for lung organoid culture | |||
| Advanced DMEM/F12 | Invitrogen | 12634010 | – |
| A8301 | Tocris | 2939 | 500nM |
| B27 supplement | Invitrogen | 17504-044 | 1x |
| Cultrex Reduced Growth Factor Basement Membrane Matrix, Type 2 (BME 2) | Trevigen | 3533-010-0 | 70-80% |
| FGF-10 | Peprotech | 100-26 | 20 ng/mL |
| FGF-7 | Peprotech | 100-19 | 5 ng/mL |
| GlutaMAX (glutamine) | Invitrogen | 35050061 | 1x |
| HEPES 1M | Invitrogen | 15630-056 | 10 mM |
| Heregulin β-1 | Peprotech | 100-03 | 5 nM |
| N-Acetylcysteine | Sigma-Aldrich | A9165 | 1.25 mM |
| Nicotinamide | Sigma-Aldrich | N0636 | 10 mM |
| Noggin (conditional medium) | home made | – | 10x |
| Penicillin-Streptomycin (10,000 U/mL) | Invitrogen | 15140-122 | 1x |
| Primocin | Invivogen | ant-pm-1 | 100 µg/mL |
| Rspondin1 (conditional medium) | home made | – | 10x |
| SB202190 | Sigma-Aldrich | S7067 | 1 µM |
| Y-27632 | Tocris | 1254 | 5 µM |
| Proximal differentiation medium | |||
| DAPT | Tocris | 2634 | 10 µM |
| Heparin Solution | StemCell Technology | 7980 | 4 µg/mL |
| Hydrocortisone Stock Solution | StemCell Technology | 7925 | 1 µM |
| PneumaCult-ALI 10X Supplement | air liquid interface supplement | ||
| PneumaCult-ALI Basal Medium | StemCell Technology | 05001 | air liquid interface basal medium |
| PneumaCult-ALI Maintenance Supplement | air liquid interface maintenance supplement | ||
| Y-27632 | Tocris | 1254 | 10 µM |
| Equipment | |||
| Biological safety cabinet | Baker | 1-800-992-2537 | |
| Carl Zeiss LSM 780 or 800 | Zeiss | confocal microscope | |
| CO2 Incubator | Thermo Fisher Scientific | 42093483 | |
| Stereo-microscope | Olympus Corporation | CKX31SF | |
| Centrifuge | Eppendorf | 5418BG040397 | |
| Serological pipettor | Eppendorf | ||
| Micropipette | Eppendorf | ||
| ZEN black or ZEN blue software | Zeiss | analysis software | |
| Consumables | |||
| 12mm Trans-well | StemCell Technology | #38023 | |
| 12-well cell culture plate | Cellstar | 665970 | |
| 15- and 50 ml conical tubes | Thermo Fisher Scientific | L6BF5Z8118 | |
| 24-well cell culture plate | Cellstar | 662160 | |
| 6.5mm Trans-well | StemCell Technology | #38024 | |
| Medical Syringe Filter Unit, 0.22 µm | Sigma-Aldrich | SLGPR33RB | |
| Microfuge tubes | Eppendorf | ||
| Micropipette tips | Thermo Fisher Scientific | TFLR140-200-Q21190531 | |
| Pasteur pipette glass | Thermo Fisher Scientific | 22-378893 | |
| Serological pipettes(5ml, 10ml, 25ml) | Thermo Fisher Scientific | BA08003, 08004, 08005 | |
| Antibodies | |||
| Goat Anti-Mouse Alexa Fluor 594 | Invitrogen | A11005 | |
| Goat Anti-Mouse, Alexa Fluor 488 | Invitrogen | A11001 | |
| Goat Anti-Rabbit Alexa Fluor 488 | Invitrogen | A11034 | |
| Goat Anti-Rabbit Alexa Fluor 594 | Invitrogen | A11037 | |
| Goat Anti-Rat Alexa Fluor 594 | Invitrogen | A11007 | |
| Mouse Anti-Cytokeratin 5 | Abcam | ab128190 | |
| Mouse Anti-FOX J1 | Invitrogen | 14-9965-82 | |
| Mouse Anti-Mucin 5AC | Abcam | ab3649 | |
| Mouse Anti-β-tubulin 4 | Sigma | T7941 | |
| Rabbit Anti-p63 | Abcam | ab124762 | |
| Rat Anti-Uteroglobin/CC-10 | R&D Systems | MAB4218-SP | |
| Other reagent | |||
| TrypLE Select Enzyme (10X) | Thermo Fisher Scientific | A1217701 | dissociation enzyme |
References
- Sato, T., et al. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology. 141 (5), 1762-1772 (2011).
- Sato, T., et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. 459 (7244), 262-265 (2009).
- Karthaus, W. R., et al. Identification of multipotent luminal progenitor cells in human prostate organoid cultures. Cell. 159 (1), 163-175 (2014).
- Chua, C. W., et al. Single luminal epithelial progenitors can generate prostate organoids in culture. Nature Cell Biology. 16 (10), 951-954 (2014).
- Hu, H., et al. Long-term expansion of functional mouse and human hepatocytes as 3D organoids. Cell. 175 (6), 1591-1606 (2018).
- Huch, M., et al. In vitro expansion of single Lgr5+ liver stem cells induced by Wnt-driven regeneration. Nature. 494 (7436), 247-250 (2013).
- Schlaermann, P., et al. A novel human gastric primary cell culture system for modelling Helicobacter pylori infection in vitro. Gut. 65 (2), 202-213 (2016).
- Bartfeld, S., et al. In vitro expansion of human gastric epithelial stem cells and their responses to bacterial infection. Gastroenterology. 148 (1), 126-136 (2015).
- Wroblewski, L. E., et al. Helicobacter pylori targets cancer-associated apical-junctional constituents in gastroids and gastric epithelial cells. Gut. 64 (5), 720-730 (2015).
- Huch, M., et al. Unlimited in vitro expansion of adult bi-potent pancreas progenitors through the Lgr5/R-spondin axis. The EMBO Journal. 32 (20), 2708-2721 (2013).
- Sachs, N., et al. A living biobank of breast cancer organoids captures disease heterogeneity. Cell. 172 (1-2), 373-386 (2018).
- Sachs, N., et al. Long-term expanding human airway organoids for disease modeling. The EMBO Journal. 38 (4), 100300 (2019).
- Zhou, J., et al. Differentiated human airway organoids to assess infectivity of emerging influenza virus. Proceedings of the National Academy of Sciences of the United States of America. 115 (26), 6822-6827 (2018).
- Clevers, H. Modeling development and disease with organoids. Cell. 165 (7), 1586-1597 (2016).
- Fatehullah, A., Tan, S. H., Barker, N. Organoids as an in vitro model of human development and disease. Nature Cell Biology. 18 (3), 246-254 (2016).
- Lancaster, M. A., Huch, M. Disease modelling in human organoids. Disease Model Mechanisms. 12 (7), (2019).
- . Millicell ERS-2 User Guide Available from: https://www.merckmillipore.com/HK/en/life-science-research/cell-culture-systems/cell-analysis/millicell-ers-2-voltohmmeter/FiSb.qB.LDgAAAFBdMhb3.r5 (2021)
- Dye, B. R., et al. In vitro generation of human pluripotent stem cell derived lung organoids. eLife. 4, 05098 (2015).
- Dye, B. R., Miller, A. J., Spence, J. R. How to grow a lung: Applying principles of developmental biology to generate lung lineages from human pluripotent stem cells. Current Pathobiology Reports. 4, 47-57 (2016).
- Glinka, A., et al. LGR4 and LGR5 are R-spondin receptors mediating Wnt/beta-catenin and Wnt/PCP signalling. EMBO Reports. 12 (10), 1055-1061 (2011).
- Groppe, J., et al. Structural basis of BMP signalling inhibition by the cystine knot protein Noggin. Nature. 420 (6916), 636-642 (2002).
- Tadokoro, T., Gao, X., Hong, C. C., Hotten, D., Hogan, B. L. BMP signaling and cellular dynamics during regeneration of airway epithelium from basal progenitors. Development. 143 (5), 764-773 (2016).
- Mou, H., et al. Dual SMAD signaling inhibition enables long-term expansion of diverse epithelial basal cells. Cell Stem Cell. 19 (2), 217-231 (2016).
- Balasooriya, G. I., Goschorska, M., Piddini, E., Rawlins, E. L. FGFR2 is required for airway basal cell self-renewal and terminal differentiation. Development. 144 (9), 1600-1606 (2017).
- Bar-Ephraim, Y. E., Kretzschmar, K., Clevers, H. Organoids in immunological research. Nature Reviews. Immunology. 20 (5), 279-293 (2019).
- Drost, J., Clevers, H. Translational applications of adult stem cell-derived organoids. Development. 144 (6), 968-975 (2017).
- Dutta, D., Heo, I., Clevers, H. Disease modeling in stem cell-derived 3D organoid systems. Trends in Molecular Medicine. 23 (5), 393-410 (2017).
- Zhou, J., et al. Infection of bat and human intestinal organoids by SARS-CoV-2. Nature Medicine. 26 (7), 1077-1083 (2020).
- Salahudeen, A. A., et al. Progenitor identification and SARS-CoV-2 infection in human distal lung organoids. Nature. 588 (7839), 670-675 (2020).
- Han, Y., et al. Identification of SARS-CoV-2 inhibitors using lung and colonic organoids. Nature. 589 (7841), 270-275 (2020).
- Mykytyn, A. Z., et al. SARS-CoV-2 entry into human airway organoids is serine protease-mediated and facilitated by the multibasic cleavage site. eLife. 10, 64508 (2021).
- Jacob, F., et al. Human pluripotent stem cell-derived neural cells and brain organoids reveal SARS-CoV-2 neurotropism predominates in choroid plexus epithelium. Cell Stem Cell. 27 (6), 937-950 (2020).
- Lamers, M. M., et al. SARS-CoV-2 productively infects human gut enterocytes. Science. 369 (6499), 50-54 (2020).
- Mallapaty, S. The mini lungs and other organoids helping to beat COVID. Nature. 593 (7860), 492-494 (2021).

