Synovial Fluid Analysis to Identify Osteoarthritis
Summary
Synovial fluid analysis under transmitted, polarized, and compensated light microscopy is used to evaluate the inflammatory or non-inflammatory nature of a sample through simple steps. It is particularly useful in osteoarthritis to detect calcium crystals and identify a more severe subset of osteoarthritis.
Abstract
Synovial fluid (SF) analysis is important in diagnosing osteoarthritis (OA). Macroscopic and microscopic features, including total and differential white blood cell (WBC) count, help define the non-inflammatory nature of SF, which is a hallmark of OA. In patients with OA, WBC in SF samples usually does not exceed 2000 cells per microliter, and the percentage of inflammatory cells, such as neutrophils, is very low or absent. Calcium crystals are frequent in SF collected from OA patients. Although their role in the pathogenesis of OA remains unclear, they have been associated with a mild inflammatory process and a more severe disease progression. Recently, calcium crystals have been described in both the early and late stages of OA, indicating that they may play a vital role in diagnosing different clinical subsets of OA and pharmacological treatment. The overall goal of SF analysis in OA is two-fold: to ascertain the non-inflammatory degree of SF and to highlight the presence of calcium crystals.
Introduction
Osteoarthritis (OA) is a complex and multifactorial chronic joint disease, with an estimated pooled global prevalence of 16% in subjects aged 15 and over, and 23% in subjects aged 40 and over1. The incidence of OA is expected to increase due to an aging population and an increase in risk factors, such as obesity and metabolic syndrome2.
Among the major issues associated with OA are the difficulty in diagnosing the disease in its early stages and currently available treatments limited to pain management and symptomatic slow-acting drugs (SYSADOAs) such as glycosaminoglycans. The diagnosis of OA is based on clinical symptoms and imaging findings. However, the lack of correlation between the two assessments can cause years of delay in OA diagnosis and treatment initiation2. OA is characterized by degenerative processes and low-grade inflammation, which can be easily investigated via synovial fluid (SF) analysis. SF is a viscous plasmatic dialysate rich in hyaluronic acid, which lubricates the joint space, provides nutrients and oxygen to cartilage, and removes metabolic waste. SF also acts as a shock absorber, thus protecting the joints during stress and strain3.
SF analysis is a simple and reliable method that must always be performed during the initial evaluation of patients with musculoskeletal symptoms and joint effusions3. Recommendations from the American College of Rheumatology, the British Society for Rheumatology, and others include SF analysis among the diagnostic testing for rheumatic diseases that must be undertaken mainly in evaluating acute monoarthritis4,5. Total and differential leucocyte counts obtained from SF analysis provide a snapshot of the pathological process occurring in the joint, thus classifying the degree of inflammation. The identification of pathogenic crystals, such as monosodium urate (MSU) and calcium pyrophosphate (CPP) crystals, under polarized light, is vital in the diagnosis and treatment of crystal arthritis (e.g., gout and pseudogout). Furthermore, the presence of microorganisms suggests a diagnosis of septic arthritis.
Calcium crystals are frequent in samples collected from patients with OA6. Basic calcium phosphate (BCP) crystals and CPP have been reported in approximately 22% and 23% of SF samples, respectively, from patients with OA6. Although their role remains unclear, these crystals have been associated with more severe forms of OA7 and are considered an epiphenomenon of the pathological process itself. Calcium crystals have been detected in 100% of tissue samples from OA patients undergoing knee replacement8. Furthermore, it has been hypothesized that calcium crystals may be involved in the pathogenesis of OA owing to their inflammatory effects, demonstrated by several studies9 and mediated, at least in part, by the NLRP3 inflammasome10.
More recently, calcium crystals have been described in both early and late stages6 of OA, indicating that they may play a vital role in diagnosing different clinical subsets of OA and pharmacological treatment.
The overall goal of SF analysis is twofold: to determine the inflammatory degree of SF and to diagnose crystal or septic arthritis by identifying specific crystals or microorganisms. It is a particularly useful, simple, and reliable tool in diagnosing OA, owing to the typical non-inflammatory pattern with a very low percentage or absence of neutrophils.
Advantages over alternative techniques
SF analysis consists of simple procedures that include total and differential leucocyte counts and crystal search. Manual cell counting performed by expert laboratory technicians3,11 remains the gold standard for the cytological analysis of synovial and other body fluids. However, due to the time-consuming limitation and the inter- and intra-observer variability of this method, automated cell counters have gradually replaced manual counting in large routine clinical laboratories where blood and urine analyzers have been adapted to enable SF analysis11,12. Nevertheless, manual counting presents some advantages in specific settings: (1) in the ambulatory, to obtain a total and differential WBC value on time; (2) to identify cell types such as cytophagocytic mononuclear (Reiter) cells or non-hematopoietic cells such as synoviocytes, that automated counters cannot recognize; (3) when the sample is too small to be handled by the instrument; (4) to create local laboratory registries that are readily accessible for research purposes. Another advantage of manual counting is seen insupravital staining, a method that allows the differentiation of cells very quickly and immediately after glass slide preparation. By contrast, traditional stainings, such as the Wright and the May-Grünwald-Giemsa procedures, require air-dried SF smears and time to stain the cells13. Although not suitable for time-sensitive routine analyses, these staining methods reveal more detailed cell populations in the sample, including erythrocytes, basophils, eosinophils, polymorphonuclear leukocytes, lymphocytes, and platelets.
Finally, routine SF analysis for crystals is performed using a simple technique based on polarized light microscopy, which provides fast results14. Alternative methods, such as scanning and electron microscopy, yield more accurate and sensitive results, but their use in everyday clinical practice is not feasible due to high costs and time-consuming sample preparation and analysis.
Protocol
The present protocol complies with the guidelines of the Ethics committee of Padova University Hospital. SF was collected with patient consent from the knee joints of patients receiving therapeutic arthrocentesis for joint effusion at their initial presentation to the clinic or in response to an arthritic flare. Contraindications to the procedure were: coagulopathy, anticoagulant medications, skin lesions, dermatitis, or cellulitis overlying the joint. All SF samples were deidentified.
1. Synovial fluid preparation
- Transfer the SF samples collected during arthrocentesis of swollen joints15 into a tube containing anticoagulant (preferably EDTA; see Table of Materials) and to a second tube containing no additive (Figure 1).
- In suspected cases of infection, aseptically transfer a portion of the SF sample (~ 1 mL) into culture bottles for microbiological testing.
NOTE: Infection can be suspected as a result of both clinical features (severe tumor, dolor, calor, functio laesa, and at times fever) and macroscopic appearance of the SF (turbidity, color). In this case, the physician prescribes a microbiological analysis, which is performed by a microbiology laboratory. - Carry out SF analysis (steps 2-5) as soon as the sample is collected.
2. Macroscopic examination
NOTE: Macroscopic evaluation of SF samples consists of subjective, qualitative, and semiquantitative assessments. There are no reference standard scales, and no control samples are used.
- Annotate the SF volume expressed in milliliters. Define the color of the sample, which can range from pale to dark yellow. Record the presence of blood.
- Define the degree of clarity by observing the sample in the backlight and determining the ease of reading a printed page through the fluid tube (Figure 2A).
- Assess the viscosity by dripping the sample from a disposable pipette. A long string or drop formation indicates good or poor viscosity, respectively (Figure 2B).
- Assess the presence of particulate materials, including tissue fragments or fibrin, by observing the sample through the tube and in the backlight.
3. Microscopic examination
NOTE: The SF microscopic analysis consists of cytological examination, including total and differential white blood cell (WBC) counts and the search for crystals.
- Determine the total cell count following the steps below.
- Determine the total number of leukocytes in the SF collected in the EDTA tube using a hemocytometer (see Table of Materials).
- Draw 0.5 µL of the SF from the EDTA tube using a Malassez-Potain pipette (mark 0.5; see Table of Materials) (Supplementary Figure 1). With the same pipette, draw 9.5 µL of 0.1% methylene blue solution (mark 11). The final dilution is 20-fold.
- Mix the pipette by gentle inversion, discard the first drops, and pour the synovial fluid + methylene blue solution into the counting chamber.
- Count the cells in two consecutive squares out of nine, and multiply the obtained number by 100 (simplified formula; Supplementary Figure 2). Express WBCs as the number of leukocytes per cubic millimeter.
NOTE: Simplified formula: Leukocytes (N/mm3) = the number of counted cells in two consecutive squares x 100. - Classify the SF according to the total number of WBCs following previously published reports16,17 (Table 1).
- Perform differential cell count.
- Determine the percentage of polymorphonuclear (PMN) cells, monocytes, and lymphocytes using supravital or traditional staining13,18.
- For supravital staining, place one small drop of SF in the center of a ready-to-use slide pre-coated with cresyl violet and methylene blue (Supplementary Figure 3; see Table of Materials).
- Cover with a coverslip, and place a drop of microscope immersion oil.
- Prepare an air-dried SF smear for traditional staining (less suitable for routine analysis). Put a small drop of SF at the end of a clean slide. Using a second slide or a coverslip, spread it along the slide with a forward movement. Let the smear air-dry.
- Stain the sample according to a standard May-Grünwald-Giemsa procedure18.
- Examine the slide under the oil immersion objective (1,000x).
- Count 100 consecutive cells and distinguish between polymorphonuclear cells, monocytes, and lymphocytes (Figure 3, Supplementary Figure 4).
- Express the total number in each category as a percentage.
NOTE: To obtain accurate results, cytological examinations must be performed as soon as the SF is collected or within 24-48 h of arthrocentesis when properly stored (4 °C).
4. Crystal detection
- Determine the presence of pathological crystals using a polarized light microscope equipped with an additional polarizing filter and a red compensator19 (see Table of Materials).
- Perform slide preparation for crystal detection.
- Carefully clean a glass slide with optical paper or soft paper imbibed with ethanol. Apply a small drop of SF on the glass slide.
- Cover with a coverslip and place the slide under the microscope.
- Identify the crystals.
- Focus the slide under the microscope using a low-power magnification objective. Carefully examine the slide under a bright field with a 40x objective. Look inside and outside the cells.
- Identify the crystals according to their size and shape (e.g., rhomboidal, needle-shaped, rod-shaped).
- Insert the polarizing filter between the light source and the specimen. Rotate the polarizer until its optical axis is perpendicular to the analyzer (placed above the objectives) to create a dark background.
- Once a birefringent crystal has been identified, insert the compensator between the polarizer and the specimen and move it parallel or perpendicular to the optical axis of the crystal.
- Observe the color revealed by the crystal (Table 2).
NOTE: MSU crystals appear needle-shaped, brightly birefringent in the dark field, and display a yellow/orange color when their axis is parallel to the compensator. Conversely, they are blue when positioned perpendicular (negative sign) (Table 2; Figure 4). CPP crystals are rod- or rhomboid-like crystals, weakly birefringent under polarized light, and display a blue or yellow color when they are aligned parallel or perpendicular to the axis of the compensator (positive sign), respectively (Table 2; Figure 5).
5. Alizarin red staining
- Prepare a 2% solution of alizarin red S (pH 4.1-4.3), filter the solution through a 0.22 µm filter, and store it away from light.
- For preparing the slide, mix a drop of SF with the same volume of alizarin red solution (1:1) on a clean slide. Cover with a coverslip and examine at 400x magnification.
- Identify the crystals under the microscope.
NOTE: The test is positive when bright, dark red precipitates are visible under transmitted light, with a round shape and concentric layers. Under polarized light, they appear strongly birefringent20 (Figure 6).
Representative Results
Large joints affected by OA are often swollen and produce significant amounts of SF, which are drained via arthrocentesis15. The macroscopic characteristics of SF evaluated immediately after arthrocentesis essentially include the quantity, color, clarity, and viscosity17. Despite their low specificity, they provide preliminary data on the degree of inflammation. The color depends on SF cellularity and the degree of fragmentation of extracellular matrix macromolecules. The SF collected from patients with OA is pale yellow, a shade generally associated with non-inflammatory samples, whereas dark yellow is associated with inflammatory effusions (Figure 2). The presence of small amounts of blood in the sample, for instance, due to capillary ruptures, results in slightly orange color. Bloody effusions must be carefully considered as they might indicate a potential hemarthrosis. Clarity is a hallmark of SF collected from patients with OA. In fact, non-inflammatory SF is poor in cells, debris, hyaluronic acid, and fibrin fragments, unlike the turbid appearance of inflammatory SF (Figure 2).
Viscosity represents a good inflammatory index as it is associated with the integrity of matrix macromolecules, mainly hyaluronic acid (Figure 2). These molecules depolymerize during inflammation, resulting in a less viscous SF. Depending on the disease stage and severity, the viscosity may be preserved or slightly reduced in OA.
The number of leukocytes never exceeds 500-1,000 cells/mm3 in OA, and they are mostly monocytes (Supplementary Figure 5A) and other mononuclear cells like synoviocytes (Supplementary Figure 5B). One of the most relevant and frequent findings in OA SF concerns CPP and BCP crystals. CPP crystals are easily detected under compensated polarized light microscopy (Figure 5), whereas the identification of BCP crystals is rendered more difficult due to their submicroscopic size (Figure 6). In fact, the length of a single BCP crystal is less than 1 µm, and although these crystals form aggregates, the clumps appear as non-birefringent amorphous-looking globules. Although nonspecific, positive alizarin red staining generally reveals the presence of BCP crystals.
Contrary to pseudogout, wherein CPP crystals are numerous and associated with high leukocyte counts and inflammatory clinical features, in OA, the number of CPP crystals in SF is very low and not linked to any altered humoral response. Nevertheless, SF samples with CPP or BCP crystals display higher levels of inflammatory cytokines when compared with OA without crystals21. Regardless of WBC count, SFs with crystals are also associated with increased percentages of PMN cells5. From a clinical point of view, the relationship between calcium crystals and inflammation may help define a particular subset of patients at an increased risk of developing more severe disease.
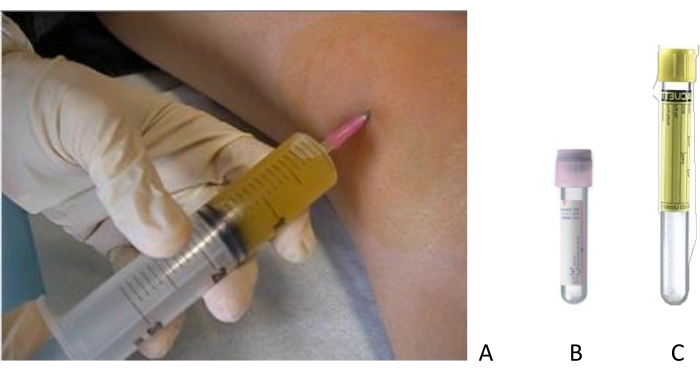
Figure 1: Joint aspiration of a swollen knee. (A) Arthrocentesis is performed after accurate disinfection of the skin with sterile materials. The SF is collected in (B) EDTA tubes for total cell count and (C) no-additive tubes for differential cell count and crystal search. Please click here to view a larger version of this figure.

Figure 2: Characteristics of inflammatory (left) and non-inflammatory (right) SF samples. (A) Color and clarity of inflammatory (left) and non-inflammatory (right) SF samples. (B) The viscosity of a non-inflammatory SF evaluated through the "string" test. The laboratorist evaluates the length of the "string" formed by a falling drop of SF from a syringe or a pipette. The sample in the picture produces a long string. Please click here to view a larger version of this figure.
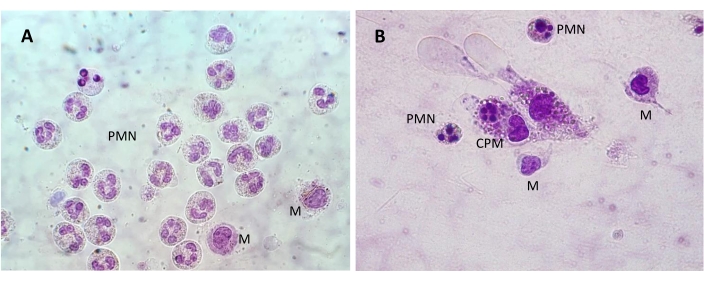
Figure 3: SF cells observed in a pre-stained slide. (A) A group of polymorphonuclear (PMN) cells with multilobed nuclei and two monocytes with their bulky nuclei in the lower right side of the image. (B) PMN cells and monocytes (M) with a cytophagocytic mononuclear (CPM) cell in the center of the image. Bright field; 1,000x magnification. Please click here to view a larger version of this figure.
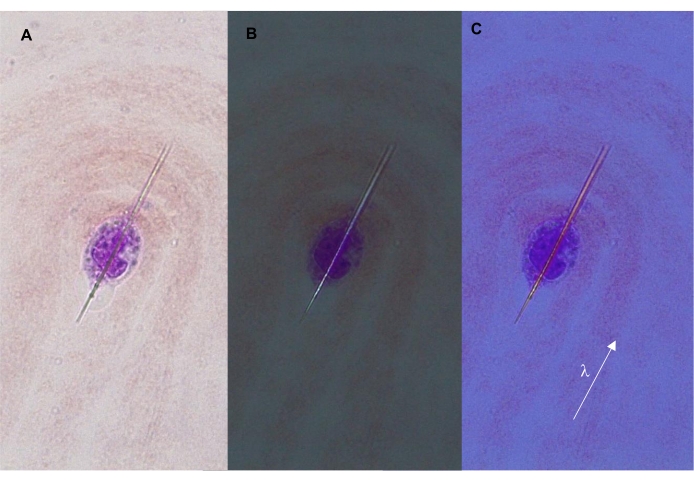
Figure 4: Intracellular MSU crystal. The crystal forms a typical needle shape shown under (A) transmitted, (B) polarized, and (C) compensated polarized light (400x). The arrow indicates the orientation of the λ filter (compensator), which is parallel to the long axis of the crystal. In this configuration, the crystal appears yellow/orange. Please click here to view a larger version of this figure.
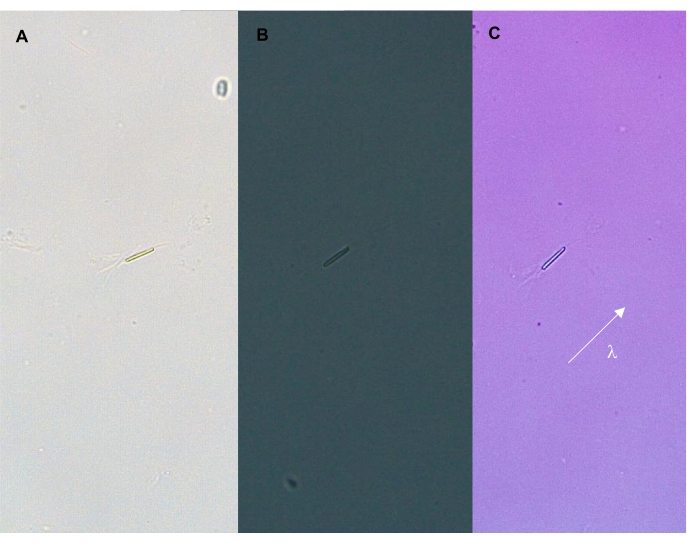
Figure 5: Intracellular CPP crystal. The crystal is shown under (A) transmitted, (B) polarized, and (C) compensated light (400x). The arrow indicates the orientation of the λ filter (compensator), which is parallel to the long axis of the crystal. In this configuration, the crystal appears blue. Please click here to view a larger version of this figure.
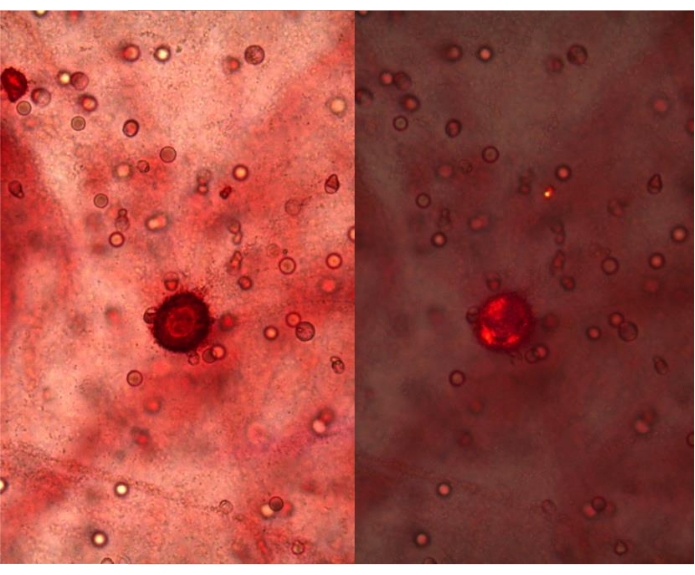
Figure 6: Positivity to the alizarin red test of an SF from OA, 400x. Red precipitates are visible under transmitted light (left panel) and polarized light (right panel), 400x magnification. Please click here to view a larger version of this figure.
Table 1: Degree of inflammation of SF according to the total number of leucocytes. Please click here to download this Table.
Table 2: Colors exhibited by MSU and CPP crystals under compensated polarized light and signs of birefringence. Please click here to download this Table.
Supplementary Figure 1: Blood diluting pipettes according to Malassez-Potain for leucocytes. Please click here to download this File.
Supplementary Figure 2: The layout of the chamber grid for WBC count. Please click here to download this File.
Supplementary Figure 3: Pre-stained, ready-to-use slides for a quick and easy differential WBC morphology evaluation. Please click here to download this File.
Supplementary Figure 4: May-Grünwald-Giemsa staining of an SF smear. Eosinophilic granules are clearly visible (arrow), 1,000x magnification. Please click here to download this File.
Supplementary Figure 5: Monocyte and synoviocytes collected from an SF sample. (A) Monocyte from an SF sample collected from a patient with osteoarthritis (1,000x). (B) Synoviocytes from an SF sample collected from a patient with osteoarthritis (1,000x). Please click here to download this File.
Discussion
In OA, SF analysis helps define disease characteristics through simple steps: total and differential leukocyte counts and searching for crystals, including CPP and BCP. Furthermore, the detection of MSU crystals may highlight important comorbidities.
Despite low costs and simple execution, the sensibility of the tests and reliability of the results may be affected due to inexperienced analysts-mainly as it pertains to crystal identification. Training and experience of the analyst are crucial in distinguishing the shape and birefringence of CPP and MSU crystals and matching their shape to their degree of birefringence. Some studies have reported discrepancies in crystal identification between different laboratories; thus, training laboratory technicians is essential to obtain reliable and consistent results14. Crystal identification is also affected by misinterpretation of birefringent particles that may be confused for pathogenic crystals. These may derive from external sources (i.e., dust or fibers) or may be present inside the joint cavity (i.e., corticosteroids or lipids). A thorough cleaning of the slide can resolve the former, but the latter can only be overcome through training and experience14.
Another limitation of SF analysis regards the use of alizarin red staining for BCP identification. As mentioned earlier, this is not a specific test, and thus caution must be exercised when interpreting results. The analyst must be able to recognize red precipitates with a peculiar morphology (Figure 11). More sensitive techniques, such as scanning electron microscopy (SEM), can be used to identify BCP crystals, but they are not used in routine examinations of SF in everyday clinical practice. Importantly, it has been shown that results obtained with alizarin red staining show a good, albeit not optimal, concordance with those obtained by SEM22, thus enhancing the value of the former.
Another salient issue to consider regards what to do when arthrocentesis yields very small amounts of SF and how to store the sample. When only a few drops of SF are collected from small joints, such as inter- and metacarpophalangeal joints and the wrist, keeping the material in the syringe and spreading it directly onto a clean glass slide is recommended. Using a micropipette and some tricks, it is possible to prepare more than one slide and perform a complete analysis. In detail, the analyst draws the SF with the counting pipette from the slide prepared for crystal searching and prepares the hemacytometer chamber, whereas some microliters can be aspirated for the supravital staining.
As far as the storage is concerned, SF samples degrade over time, and their analysis requires fresh preparations to obtain reliable results. Alternatively, SF can be preserved at 4 °C and used within 24 h, and no later than 48 h, for cytological examination. To avoid cell clumping and SF coagulation, attention must be paid to collecting the specimen in at least one EDTA tube. Frozen SF samples stored at -20 °C for a longer period of time can be used only for crystal search.
SF analysis is an irreplaceable pathognomonic test in rheumatology, owing to its utility, simplicity of execution, and affordability. The main challenge for future applications of SF analysis is a more accurate diagnosis of rheumatic diseases. This will be possible by integrating this method with more advanced and innovative technologies23, making it possible to characterize SF composition and identify specific molecular biomarkers and proteomics at the site of inflammation.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The Authors wish to acknowledge Professor Leonardo Punzi for his precious mentorship in the field of synovial fluid analysis and Padova University Hospital for its support.
Materials
| Alizarin red S | Merck | A5533 | For BCP crystal search |
| Burker chamber | Merck | BR718905 | For total white blood cell count |
| Cover glasses | Merck | C7931 | For microscopic examination |
| EDTA tubes | BD | 368861 | For SF collection |
| Glass slides | Merck | S8902 | For crystal search |
| Lambda filter (compensator) | Any | Refer to microscope company | For crystal identification |
| Malassez-Potain pipette | Artiglass | 54830000 | For dilution of synovial fluid |
| Methylene blue solution | Merck | 3978 | For total white blood cell count |
| Polarized microscope | Leica, Nikon, others | Depending on the model and company | For complete synovial fluid analysis |
| Polarizing lens | Any | Refer to microscope company | For crystal identification |
| Testsimplet | Waldeck | 14386 | Supravital staining for cell differentiation |
References
- Cui, A., et al. Global, regional prevalence, incidence and risk factors of knee osteoarthritis in population-based studies. EClinicalMedicine. 29, 100587 (2020).
- Hunter, D. J., Bierma-Zeinstra, S. Osteoarthritis. Lancet. 393 (10182), 1745-1759 (2019).
- Mandell, B. F. . Synovial Fluid Analysis and the Evaluation of Patients with Arthritis. , (2022).
- Schmerling, R. H. Guidelines for the initial evaluation of the adult patient with acute musculoskeletal symptoms. Arthritis & Rheumatism. 39 (1), 1-8 (1996).
- Coakley, G., et al. RCGP and BSAC guidelines for management of the hot swollen joint in adults. Rheumatology. 45 (8), 1039-1041 (2006).
- Frallonardo, P., et al. Basic calcium phosphate and pyrophosphate crystals in early and late osteoarthritis: relationship with clinical indices and inflammation. Clinical Rheumatology. 37 (10), 2847-2853 (2018).
- Nalbant, S., et al. Synovial fluid features and their relations to osteoarthritis severity: new findings from sequential studies. Osteoarthritis Cartilage. 11 (1), 50-54 (2003).
- Fuerst, M., et al. Calcification of articular cartilage in human osteoarthritis. Arthritis & Rheumatism. 60 (9), 2694-2703 (2009).
- Campillo-Gimenez, L., et al. Inflammatory potential of four different phases of calcium pyrophosphate relies on NF-κB activation and MAPK pathways. Frontiers in Immunology. 9, 2248 (2018).
- Pazár, B. Basic calcium phosphate crystals induce monocyte/macrophage IL-1β secretion through the NLRP3 inflammasome in vitro. The Journal of Immunology. 186 (4), 2495-2502 (2011).
- Martín, M. J. A., et al. Automated cell count in body fluids: a review. Advances in Laboratory Medicine. 2, 149-161 (2021).
- Seghezzi, , et al. Optimization of cellular analysis of synovial fluids by optical microscopy and automated count using the Sysmex XN body fluid mode. Clinica Chimica Acta. 462, 41-48 (2016).
- Reginato, A. J., Maldonado, I., Reginato, A. M., Falasca, G. F., O’Connor, C. R. Supravital staining of synovial fluid with Testsimplets. Diagnostic Cytopathology. 8 (2), 147-152 (1992).
- Lumbreras, B., et al. Analysis for crystals in synovial fluid: training of the analysts results in high consistency. Annals of the Rheumatic Diseases. 64 (4), 612-615 (2005).
- Tieng, A., Franchin, G. Knee arthrocentesis in adults. Journal of Visualized Experiments. , e63135 (2022).
- Punzi, L., Oliviero, F. Arthrocentesis and synovial fluid analysis in clinical practice: value of sonography in difficult cases. Annals of the New York Academy of Sciences. 1154 (1), 152-158 (2009).
- Schumacher, H. R., Reginato, A. J. . Atlas of Synovial Fluid Analysis and Crystal Identification. , (1991).
- Mopin, A., Driss, V., Brinster, C. A. Detailed protocol for characterizing the murine C1498 cell line and its associated leukemia mouse model. Journal of Visualized Experiments. (116), e54270 (2016).
- Oliviero, F., Punzi, L. Basics of Polarized Light Microscopy. Synovial Fluid Analysis and the Evaluation of Patients with Arthritis. , 79-90 (2022).
- Paul, H., Reginato, A. J., Schumacher, H. R. Alizarin red S staining as a screening test to detect calcium compounds in synovial fluid. Arthritis & Rheumatism. 26 (2), 191-200 (1983).
- Favero, M., et al. Synovial fluid fetuin-A levels in patients affected by osteoarthritis with or without evidence of calcium crystals. Rheumatology. 58 (4), 729-730 (2019).
- Frallonardo, P., et al. Detection of calcium crystals in knee osteoarthritis synovial fluid: a comparison between polarized light and scanning electron microscopy. Journal of Clinical Rheumatology. 22 (7), 369-371 (2016).
- Peffers, M. J., Smagul, A., Anderson, J. R. Proteomic analysis of synovial fluid: current and potential uses to improve clinical outcomes. Expert Review of Proteomics. 16 (4), 287-302 (2019).

