A GMP-Compliant Procedure for the Generation of Gene-Modified T cells
Summary
This protocol outlines the process of transducing primary human T cells with a gene of interest, ensuring compatibility with Good Manufacturing Practice (GMP) standards.
Abstract
The field of Adoptive Cell Therapy (ACT) has been revolutionized by the development of genetically modified cells, specifically Chimeric Antigen Receptor (CAR)-T cells. These modified cells have shown remarkable clinical responses in patients with hematologic malignancies. However, the high cost of producing these therapies and conducting extensive quality control assessments has limited their accessibility to a broader range of patients. To address this issue, many academic institutions are exploring the feasibility of in-house manufacturing of genetically modified cells, while adhering to guidelines set by national and international regulatory agencies.
Manufacturing genetically modified T cell products on a large scale presents several challenges, particularly in terms of the institution’s production capabilities and the need to meet infusion quantity requirements. One major challenge involves producing large-scale viral vectors under Good Manufacturing Practice (GMP) guidelines, which is often outsourced to external companies. Additionally, simplifying the T cell transduction process can help minimize variability between production batches, reduce costs, and facilitate personnel training. In this study, we outline a streamlined process for lentiviral transduction of primary human T cells with a fluorescent marker as the gene of interest. The entire process adheres to GMP-compliant standards and is implemented within our academic institution.
Introduction
The emergence of Adoptive Cell Therapies (ACT) and Chimeric Antigen Receptor (CAR)-T cells have brought about a revolutionary shift in modern clinical practice, establishing a new paradigm of personalized medicine. Particularly, CAR-T cells targeting CD19 have demonstrated exceptional clinical responses and represent the most advanced T cell therapy for B cell malignancies1,2,3,4,5. However, the current commercialized gene-modified T cell therapies heavily rely on viral vectors for gene transfer. The implementation of viral vectors in ACT, especially under Good Manufacturing Practice (GMP) conditions within an academic setting, presents significant challenges due to scalability limitations, specialized equipment requirements, the need for highly skilled personnel, and the use of GMP-compliant reagents for plasmid-mediated viral particle production6,7,8.
Consequently, the manufacturing process for gene-modified T cell therapies is intricate and typically commences with the isolation of Peripheral Blood Mononuclear Cells (PBMCs) from a leukapheresis product. T cells are often enriched from the PBMC pool and activated using anti-CD3/CD28 micro-complexes7,9. Subsequently, T cells are genetically modified with either viral or non-viral vectors, which can be produced in situ or outsourced. To attain the required cell numbers for infusion, gene-modified cells are expanded before final formulation and/or cryopreservation. Finally, the cell product must satisfy various release criteria based on multiple quality control assays9.
This protocol presents a comprehensive methodology utilizing simple techniques for the ex vivo manipulation of healthy PBMCs to manufacture a gene-modified T cell product under GMP-compliant conditions. The protocol incorporates the use of a lentiviral vector, which can be outsourced to a viral production company providing all necessary quantitative/functional, purity, and safety tests (e.g., viral titer, detection of host cell DNA replication-competent lentivirus). For this study, the vector employed is a VSV-g (Vesicular Stomatitis Virus G Protein)-based second-generation lentiviral vector with Green Fluorescent Protein (GFP), as the gene of interest, in constitutive expression.
Protocol
This study was approved by the Ethics Committee of the University of Patras and the Institutional Review Board of the University General Hospital of Patras. Prior to obtaining biological specimens, informed consent was obtained from healthy individuals. The eligibility assessment of participants was conducted according to institutional procedures and in compliance with JACIE standards for leukapheresis. If this protocol is to be implemented in a clinical ACT setting, all procedures must adhere to Good Manufacturing Practice (GMP) guidelines and be performed in GMP-compliant clean rooms. Special safety precautions are necessary when working with viral vectors, and these procedures should be conducted within a dedicated biosafety cabinet. In this study, the described procedures were conducted within a validated H35 HEPA Hypoxystation (see Table of Materials) to ensure a controlled and monitored environment. Decontamination of all hazardous liquids should be carried out using 10% bleach solution.
1. PBMC isolation and T cell activation (Day 0)
- Collect cells from the donor apheresis bag (leukopaks, see Table of Materials) into 50 mL tubes, depending on the initial volume.
- Perform two washes of the cell bag by infusing 15 mL of complete hematopoietic media (+5% HSA, commercial serum-free hematopoietic media, see Table of Materials).
- Conduct polysaccharide-based density gradient centrifugation at a 1:2 (reagent:sample) dilution. Centrifuge at 800 x g for 30 min at room temperature with breaks off (A/D, 9/0).
- Using a micropipette, collect PBMCs into a clean 50 mL tube.
- Wash the cells twice with 1x PBS, filling up to 50 mL for each wash (400 x g, 10 min, room temperature). Resuspend the cells in 5 mL of complete media.
- Count the cells using trypan blue using either a standard hemocytometer or an automated cell counter (see Table of Materials).
- Dilute the cells with complete media to achieve a concentration of 2 x 106 cells/mL.
- Activate 20 x 106 PBMCs by adding 10 µL of T cell stimulation reagent (see Table of Materials) per 2 x 106 starting cells.
NOTE: This protocol is designed for the activation of 20 x 106 PBMCs but can be adjusted for larger cell numbers as needed. - Add rhIL-2 (20 U/mL) (see Table of Materials), mix gently, and transfer the mixture into an appropriate vessel (e.g., 6-well plate).
- Incubate the cells at 37 °C, 5% CO2 for 72 h.
2. Lentiviral T cell transduction (Day 3)
- Transfer the cell culture into a suitable vessel, such as a 50 mL conical tube. Mix well and centrifuge at 300 x g for 10 min at room temperature (RT) to remove the activation reagent. Carefully discard the supernatant completely and resuspend the pellet in 1 mL of complete medium.
NOTE: At this stage, it is possible to thaw the virus on ice. - Count the cells using trypan blue.
- Resuspend the cells in complete medium to achieve a concentration that allows for plating 0.5 x 106 cells in a total volume of 400 µL per well in a 24-well plate. Add rhIL-7 (155 U/mL) and rhIL-15 (290 U/mL) (see Table of Materials).
NOTE: Ensure to account for the volume of viruses to be added. Refer to step 2.4. - In a separate tube, add Vectofusin-1 (see Table of Materials) to Opti-MEM medium at a concentration of 10 µg/mL, with a volume equal to the viral volume to be used.
NOTE: Consider the total cell culture volume when calculating the concentration of Vectofusin-1. See step 2.3. Alternatively, Vectofusin-1 GMP can be used as a GMP-grade reagent. - Combine the concentrated virus at a Multiplicity of Infection (MOI) of 40 with the Vectofusin-1/Opti-MEM mixture (1:1 ratio) and mix thoroughly. Add this mixture to the cell culture and adjust the total volume to 400 µL per well using complete medium, if necessary.
- Place the lid on the plate, seal it with paraffin film, and centrifuge at 1000 x g for 2 h at 32 °C. Subsequently, incubate the plate at 37 °C, 5% CO2 overnight.
3. Virus removal and T cell expansion (Day 4)
- Collect the cells and pool them into a suitable vessel, such as a 50 mL tube.
- Centrifuge the cells at 400 x g at room temperature (RT) for 5 min. Carefully remove the supernatant completely and resuspend the pellet in 1 mL of complete media.
- Count the cells using trypan blue. Resuspend the cells in complete media at a concentration of 1 x 106 cells/mL. Add rhIL-7 (155 U/mL) and rhIL-15 (290 U/mL) (see Table of Materials).
- Transfer the cells to an appropriate vessel, such as a T25 flask, at a concentration of 0.3-0.5 x 106 cells/cm2 and incubate them at 37 °C, 5% CO2, for 2 days.
NOTE: If a larger cell population is required, cells can be subcultured (1:2) in the presence of rhIL7 and rhIL-15 every 2 days, following the same incubation conditions mentioned above, until the desired cell numbers are achieved.
4. Cryopreservation (Day 14)
NOTE: Ice will be required to transfer cryovials to the storage location during this step. The timing of cryopreservation depends on the desired cell numbers. For this protocol, cryopreservation is performed on Day 14 after achieving a 25-fold expansion.
- Once the desired cell number is reached, harvest the cells and transfer them to 50 mL tubes (adjust the number of tubes based on the total volume).
- Centrifuge the tubes at 400 x g at room temperature (RT) for 5 min. Carefully remove the supernatant completely and resuspend the pellet in 10 mL of media. If necessary, pool the cells from multiple tubes.
- Count the cells using trypan blue. Keep an aliquot of 1 x 106 cells for each quality control (QC) testing (refer to Section 5) to assess Vector Copy Number and Transduction Efficiency.
- Centrifuge the tubes at 400 x g for 10 min at RT. At this point, label the cryovials accordingly.
- Completely remove the supernatant and resuspend the pellet in a cryopreservation solution consisting of 50% HSA and 40% HBSS at a concentration of 1 x 107 cells per mL per cryovial. Transfer the cell suspension into the appropriate number of cryovials and add 10% dimethyl sulfoxide (DMSO) to each cryovial.
- Place the cryovials on ice and promptly transfer them to a controlled-rate freezer for cryopreservation. After cryopreservation, transfer the cryovials to a monitored liquid nitrogen tank for long-term storage.
5. Quality control (Day 14)
NOTE: For release testing, the product underwent several tests, including sterility tests for microbial growth, and endotoxin level assessment. These tests were conducted in certified laboratories at the University General Hospital of Patras and the University of Patras. Mycoplasma presence was determined in-house using a biochemical detection assay and measured on a luminometer following the manufacturer's instructions (see Table of Materials). Cell phenotype (Figure 1) as well as cell number and viability were assessed in-house by flow cytometry (see section 5.1) and an automated cell counter (see Table of Materials), respectively. Release criteria were established based on published data and recommendations10,11for GMP-produced cell products (Table 1).
- Perform transduction efficiency-flow cytometry staining (Day 14)
NOTE: An aliquot of 1 x 106 cells will be stained for viability (7-AAD) and CD3 marker2 to evaluate the transduction efficiency (see Table of Materials). If the gene of interest, such as in the case of CAR T cells, needs to be detected, the specific antibody should be in a different color from that of the CD3 marker and 7-AAD to avoid compensation. Appropriate control samples should be taken into account.- Place the sample in a FACS tube and add 500 µL of FACS buffer.
- Centrifuge the tube at 400 x g at RT for 5 min.
- Completely discard the supernatant with a pipette and resuspend the pellet in a 1:5 diluted solution of CD3:FACS buffer (see Table of Materials).
- Vortex the sample and incubate for 30 min on ice in the dark.
- Add 500 µL of FACS buffer. Centrifuge at 400 x g for 5 min (RT).
- Completely discard the supernatant and resuspend the pellet in a 1:20 diluted solution of 7-AAD:FACS buffer (see Table of Materials).
- Vortex the sample and incubate for 10 min on ice in the dark.
- Add 200 µL of FACS buffer and analyze the sample using a flow cytometer (see Table of Materials).
Representative Results
Assessment of transduction efficiency
The transduced cells were evaluated for the expression of the gene of interest (GFP) using flow cytometry. Representative results are depicted in Figure 1. On Day 14, more than 95% of the cells were CD3+, indicating successful T cell activation and expansion. The transduction efficiency within the CD3+ population was measured at 58.7% (TD, Figure 1C) compared to the non-transduced cells (UNTD, Figure 1B) which exhibited a transduction efficiency of 0.51%. Viability analysis showed that more than 95% of the cells were alive after excluding 7-AAD+ cells (Figure 1A). FlowJo Software (see Table of Materials) was used for data analysis.
Assessment of expansion profile
The transduced cells were expanded in the presence of rhIL-7 (155 U/mL) and rhIL-15 (290 U/mL) from Day 4 until the day of cryopreservation (Day 14). Subculturing was performed every 2 days. The cells underwent a 25-fold expansion, as shown in Figure 2, reaching clinically relevant cell numbers based on the initial cell count.
Assessment of quality control
The product underwent various quality control tests, including sterility testing, mycoplasma detection, and assessment of endotoxin levels. Additionally, functional characterization was performed according to the criteria outlined in Table 1. The product met all the set criteria, indicating its suitability for further use.
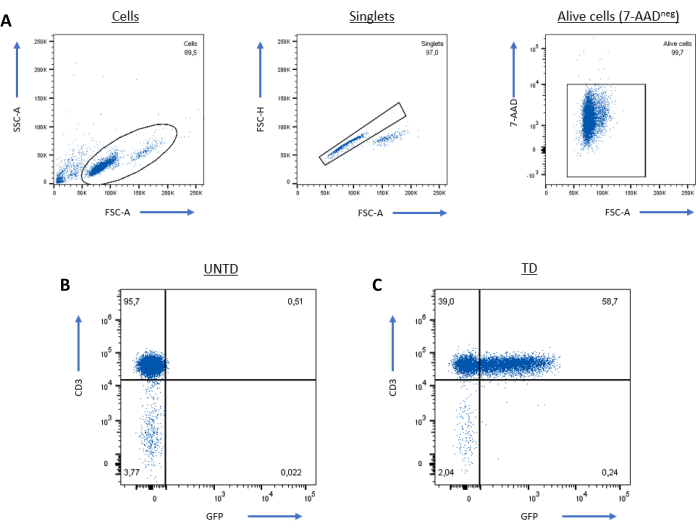
Figure 1: Representative results showing the expression of the GFP transgene on human primary CD3+ T cells. Analysis was performed on singlet alive (7-AADneg) cells (A). The positivity threshold of the transduced (TD) cells (C) was determined based on the non-transduced (UNTD) population (B). Please click here to view a larger version of this figure.
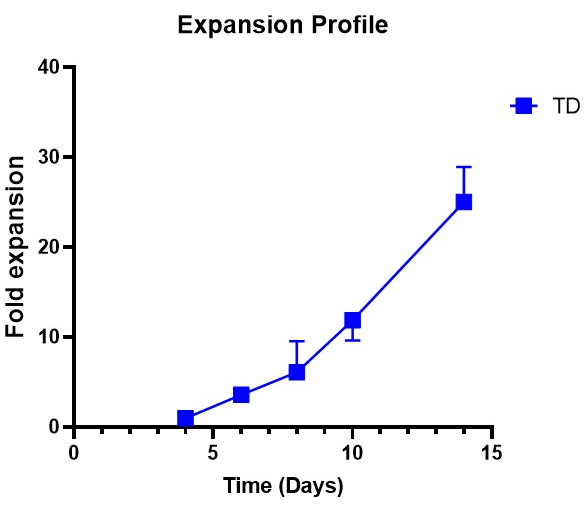
Figure 2: Expansion profile of transduced T cells. Transduced (TD) T cells were expanded in the presence of rhIL-7 and rhIL-15 for 10 days with subculturing every 2 days (n = 3). Please click here to view a larger version of this figure.
| Assessment | Method | Criteria |
| Sterility | Development of microorganisms on TSA (Tryptic Soy Agar) or SDA (Sabouraud Dextrose Agar) plates (outsourced or collaborating certified Microbiology Lab) | Negative after 7 days |
| Mycoplasma | Mycoalert Plus Detection kit | Negative |
| Endotoxin | Gel Clot Limulus Amobecyte Lysate (LAL) test (outsourced or collaborating certified laboratory within University’s Department of Public Health) | <0.5 EU/mL |
| Cell phenotype | Flow cytometry (in house) | CD3+ ≥80% GFP+ ≥30% (Transduction efficiency) |
| Cell number | Automated counter (in house) | >1 x 108 cells |
| Viability | Trypan blue stain (in house) | ≥70% alive cells |
| Transgene copies/cell | Real-time PCR (in house) | ≤50 copies/µg DNA |
Table 1: Release criteria for the cell product.
Discussion
Lentiviral vectors as gene delivery vehicles are important tools in the field of cell and gene therapy because of their ability to transduce both dividing and non-dividing cells12. However, large-scale production of lentiviral vectors using GMP-compatible methods is still challenging due to various parameters, such as transfection methods and purification steps, which can result in variability in lentivirus batches. Academic centers often obtain viral stocks from viral production biotechnology companies, which provide documentation on the functionality and safety of the virus to meet release criteria13,14.
A successful transduction protocol relies not only on the quality of the viral stock but also on the fitness of the target cells. Efficient stimulation of PBMCs is crucial as a failure in the activation step may lead to low transduction efficiency. Additionally, the choice of promoter and envelope protein can impact the efficiency of the transduction protocol15,16. Different transduction enhancers possess distinct properties that facilitate the successful incorporation of viral particles into target cells17,18,19,20. Vectofusin-1, a non-toxic histidine-rich amphipathic peptide, serves as a transduction enhancer that streamlines the transduction process by eliminating the need for pre-coating on cell culture surfaces18. Furthermore, methods like spinoculation have been shown to increase the susceptibility of target cells to viruses21. Cytokines play a major role as they provide costimulatory signals for full T cell activation and have pleiotropic effects on the proliferation, survival, and function of T cells. IL-7 and IL-15 supplementation creates a favorable environment for CAR T cell expansion due to higher proliferation capacity and lower apoptotic rate22,23,24. In this protocol, a high MOI (MOI = 40) was observed for achieving transduction of T cells (>50%), consistent with other transduction protocols25,26.
The quality of the cell product manufactured under GMP conditions largely depends on the manufacturing facilities, equipment, and availability and quality of GMP-grade ancillary materials. The current product was produced in a GMP facility equipped with a building monitoring system (BMS) that controls and logs environmental conditions throughout all the clean rooms (Grades D, C, B, and A). For release testing, this product underwent sterility tests for microbial growth, mycoplasma detection, and endotoxin level assessment along with functional characterization tests, either performed in certified laboratories or in-house, meeting all the set criteria (Table 1).
Currently, most T cell therapies are manufactured in automated closed systems such as the CliniMACS Prodigy (Miltenyi Biotec, Germany) or Cocoon (Lonza, Switzerland), among many others under development, which offer ease of handling and consistency between lots11,27,28. However, this limits the production of gene therapies to specialized centers and one product per run. The current protocol has been developed as an open process to minimize costs associated with the procurement of specialized equipment while meeting the set release criteria. In conclusion, a simplified, cost-effective, and GMP-compliant protocol is outlined for generating a gene-modified T cell product with an adequate transduction efficiency (>50%), applicable to CAR T cell production in an academic setting.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This research has been co-financed by the European Union and Greek national funds through the Operational Program Competitiveness, Entrepreneurship, and Innovation, under the call RESEARCH – CREATE – INNOVATE (project code: T2EDK – 00474). We would also like to express our gratitude to the Choose Life Foundation for providing continuous support to the GMP Lab "Dimitris Lois" of the Institute of Cell Therapy.
Materials
| 24-well TC-treated plate | Corning | 353047 | |
| 5 mL FACS tube | Corning | 352054 | |
| 50 mL Falcon tubes | Greiner biomedica | 227261 | |
| 6-well TC-treated plate | Corning | 353046 | |
| 7-AAD | BD Biosciences | 559925 | |
| BD FACS CANTO II | BD Biosciences | V96300084 | |
| BSA | Applichem | A1391 | |
| Cell Counter | Corning Cell Cytosmart | J21E0081 | |
| Cryovials | Greiner biomedica | 122263 | |
| Dimethyl Sulfoxide (DMSO) | Sigma-Aldrich | D2438 | |
| Dulbecco’s Phosphate Buffered Saline (D-PBS) | Life Technologies | 14190 | |
| EDTA | Invitrogen | 15575-038 | |
| FACS Buffer (1x D-PBS, 0.5% BSA, 2 mM EDTA) | – | – | |
| FlowJo Software v10.6.2 | TreeStar Inc | – | |
| Gibco CTS Opti-MEM I Medium | ThermoFisher Scientific | A4124801 | |
| GMP rhIL-15 | Miltenyi Biotec | 170-076-114 | |
| GMP rhIL-2 | Miltenyi Biotec | 170-076-146 | |
| GMP rhIL-7 | Miltenyi Biotec | 170-076-111 | |
| Hanks’s Balanced Salt Solution (HBSS) | Life Technologies | 14175 | |
| HEPA Whitley H35 Hypoxystation | Don Whitley Scientific | HA0315172H | |
| Human Serum Albumin (HSA) | Baxter | B05AA01 | |
| Junior LB 9509 Portable Luminometer | Berthold Technologies | 6506 | |
| Lenti-X Provirus Quantitation Kit | Takara Bio | 631239 | |
| Lymphoprep | Stemcell Technologies | 7811 | |
| MACS GMP T cell TransAct | Miltenyi Biotec | 170-076-156 | |
| Mouse Anti-Human CD3 (Clone: UCHT-1) | BD Biosciences | 557706 | |
| MycoAlert Plus Detection kit | Lonza Bioscience | LT07-703 | |
| Parafilm ultra-expandable closing film tape 5 cm x 75 m | D.Dutscher | 90261 | |
| PeriStem PS-650-DB (apheresis bag) | Biomed Device | SC00816 | |
| Planer Kryo10 Series III | Planer | ||
| T75 flasks | Greiner biomedica | 658175 | |
| Trypan blue, 0.4% solution | Invitrogen | T10282 | |
| Vectofusin-1 GMP | Miltenyi Biotec | 170-076-165 | |
| X-VIVOTM 15 Serum-free Hematopoietic Cell Medium | Lonza Bioscience | A1048501 |
References
- Maude, S. L., Teachey, D. T., Porter, D. L., Grupp, S. A. CD19-targeted chimeric antigen receptor T-cell therapy for acute lymphoblastic leukemia. Blood. 125 (26), 4017-4023 (2015).
- Brentjens, R., et al. CD19-targeted T cells rapidly induce molecular remissions in adults with chemotherapy-refractory acute lymphoblastic leukemia. Science Translational Medicine. 5 (177), 177ra38 (2013).
- Maude, S. L., et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. The New England Journal of Medicine. 371 (16), 1507-1517 (2014).
- Grupp, S. A., et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. The New England Journal of Medicine. 368 (16), 1509-1518 (2013).
- Schuster, S. J., et al. Sustained remissions following chimeric antigen receptor modified T cells directed against CD19 (CTL019) in patients with relapsed or refractory CD19+ lymphomas. Blood. 126 (23), 183 (2015).
- Levine, B. L., Miskin, J., Wonnacott, K., Keir, C. Global manufacturing of CAR T cell therapy. Molecular Therapy – Methods & Clinical Development. 4, 92-101 (2017).
- Wang, X., Rivière, I. Clinical manufacturing of CAR T cells: foundation of a promising therapy. Molecular Therapy – Oncolytics. 3, 16015 (2016).
- Iancu, E. M., Kandalaft, L. E. Challenges and advantages of cell therapy manufacturing under Good Manufacturing Practices within the hospital setting. Current Opinion in Biotechnology. 65, 233-241 (2020).
- Kaiser, A. D., et al. Towards a commercial process for the manufacture of genetically modified T cells for therapy. Cancer Gene Therapy. 22 (2), 72-78 (2015).
- . Guidelines on quality, non-clinical and clinical aspects of medicinal products containing genetically modified cells. European Medicines Agency. , (2020).
- Castella, M., et al. Point-of-care CAR T-Cell production (ARI-0001) using a closed semi-automatic bioreactor: experience from an academic phase I clinical trial. Frontiers in Immunology. 11, 482 (2020).
- Naldini, L. Lentiviruses as gene transfer agents for delivery to non-dividing cells. Current Opinion in Biotechnology. 9 (5), 457-463 (1998).
- Ausubel, L. J., et al. Production of CGMP-grade lentiviral vectors. Bioprocess International. 10 (2), 32-43 (2012).
- Van der Loo, J. C. M., Wright, J. F. Progress and challenges in viral vector manufacturing. Human Molecular Genetics. 25 (R1), R42-R52 (2016).
- Rad, S. M. . A. H., Poudel, A., Tan, G. M. Y., McLellan, A. D. Promoter choice: Who should drive the CAR in T cells. PLoS One. 15 (7), e0232915 (2020).
- Sandrin, V., et al. Lentiviral vectors pseudotyped with a modified RD114 envelope glycoprotein show increased stability in sera and augmented transduction of primary lymphocytes and CD34+ cells derived from human and nonhuman primates. Blood. 100 (3), 823-832 (2002).
- Costello, E., et al. Gene transfer into stimulated and unstimulated T lymphocytes by HIV-1-derived lentiviral vectors. Gene Therapy. 7 (7), 596-604 (2000).
- Radek, C., et al. Vectofusin-1 improves transduction of primary human cells with diverse retroviral and lentiviral pseudotypes, enabling robust, automated closed-system manufacturing. Hum Gene Therapy. 30 (12), 1477-1493 (2019).
- Hanenberg, H., et al. Colocalization of retrovirus and target cells on specific fibronectin fragments increases genetic transduction of mammalian cells. Nature Medicine. 2 (8), 876-882 (1996).
- Lamers, C. H. J., et al. Retronectin-assisted retroviral transduction of primary human T lymphocytes under good manufacturing practice conditions: tissue culture bag critically determines cell yield. Cytotherapy. 10 (4), 406-416 (2008).
- O’Doherty, U., Swiggard, W. J., Malim, M. H. Human immunodeficiency virus type 1 spinoculation enhances infection through virus binding. Journal of Virology. 74 (21), 10074-10080 (2000).
- Cieri, N., et al. IL-7 and IL-15 instruct the generation of human memory stem T cells from naive precursors. Blood. 121 (4), 573-584 (2013).
- Zhou, J., et al. Chimeric antigen receptor T (CAR-T) cells expanded with IL-7/IL-15 mediate superior antitumor effects. Protein Cell. 10 (10), 764-769 (2019).
- Ghassemi, S., et al. Rapid manufacturing of non-activated potent CAR T cells. Nature Biomedical Engineering. 6 (2), 118-128 (2022).
- Wang, R. -. N., et al. Optimized protocols for γδ T cell expansion and lentiviral transduction. Molecular Medicine Reports. 19 (3), 1471-1480 (2019).
- Yuan, W., et al. Comparative analysis and optimization of protocols for producing recombinant lentivirus carrying the anti-Her2 chimeric antigen receptor gene. The Journal of Gene Medicine. 20 (7-8), e3027 (2018).
- Zhu, F., et al. Closed-system manufacturing of CD19 and dual-targeted CD20/19 chimeric antigen receptor T cells using the CliniMACS Prodigy device at an academic medical center. Cytotherapy. 20 (3), 394-406 (2018).
- Mock, U., et al. Automated manufacturing of chimeric antigen receptor T cells for adoptive immunotherapy using CliniMACS Prodigy. Cytotherapy. 18 (8), 1002-1011 (2016).

