Chronic Unpredictable Mild Stress in Rats based on the Mongolian medicine
Summary
This protocol outlines a chronic unpredictable mild stress (CUMS) model for depression based on Mongolian medical theory, along with methods for validating behavioral tests.
Abstract
Depression is a prevalent affective disorder and constitutes a leading cause of global disability. The limitations of current pharmacological interventions contribute to the substantial health burden attributed to this condition. There is a pressing need for a deeper understanding of the underlying mechanisms of depression, making pre-clinical models with translational potential highly valuable. Mongolian medicine, a subset of traditional medicine, posits that disease occurrence is closely tied to the equilibrium of wind, bile, and Phlegm. In this study, we introduce a protocol for the chronic unpredictable mild stress (CUMS) method in rats. Within this framework, rats are subjected to a series of fluctuating, mild stressors to induce a depression-like phenotype, mimicking the pathogenesis of human depression. Behavioral assays employed in this protocol include the sucrose preference test (SPT), indicative of anhedonia-a core symptom of depression; the open field test (OFT), which measures anxiety levels; and the Morris water maze test (MWM), which evaluates spatial memory and learning abilities. The CUMS method demonstrates the capability to induce anhedonia and to cause long-term behavioral deficits. Furthermore, this protocol is more aligned with Mongolian medical theory than other animal models designed to elicit depression-like behavior. The development of this animal model and subsequent research provide a robust foundation for future innovative studies in the realm of Mongolian medicine.
Introduction
Major depressive disorder (MDD) is a prevalent mental illness, ranking as the third leading cause of disability globally and affecting over 300 million people1,2,3. Notably, it is estimated that at least half of the affected individuals do not receive adequate treatment4. Given this gap, animal models serve as a crucial tool for investigating the etiology of depression. To date, more than 20 different animal models for depression exist5. Among these, the chronic unpredictable mild stress (CUMS) model, refined by Paul Winer in 1987, is the most frequently utilized6. The CUMS model operates on the premise that exposing rodents to a diverse range of socio-environmental stressors leads to symptoms akin to anxiety, tension, and depression. The methodology involves exposing animals to various mild stressors over several weeks, culminating in a range of behavioral alterations, including anhedonia and depressive-like behaviors7,8. These changes are accompanied by shifts in endocrine and neurotransmitter profiles, such as a reduction in 5-HT9,10. These outcomes closely mirror those observed in humans diagnosed with MDD, thereby validating the model’s utility. The CUMS model is particularly valued for its effectiveness in evaluating antidepressants, manifesting high levels of surface, structural, and predictive validity11,12. Unlike other models, CUMS is sensitive to the effects of chronic administration of monoaminergic antidepressants. For instance, selective serotonin reuptake inhibitors (SSRIs) such as citalopram, paroxetine, and fluoxetine have been shown to prevent and reverse anhedonia under conditions of chronic stress12,13. Additionally, new rapid-acting antidepressants, such as ketamine, have also demonstrated efficacy in this model14,15. In contrast, other tests like the forced swimming test (FST) and tail suspension test (TST) are less reliable for modeling long-term behavioral changes, often reflecting adaptations to acute stress rather than enduring symptoms of depression16. These characteristics underscore the robust validity of the CUMS model in depression research. One of the most salient features of the CUMS model, recognized for its high reliability in classical studies, is anhedonia-the incapacity to experience pleasure or interest in everyday activities17,18. This phenomenon is commonly evaluated using sucrose preference tests, and many antidepressants have been shown to reverse reduced sucrose consumption. Several other metrics are also commonly employed in the CUMS literature, including the open field test (OFT), which assesses voluntary motor behavior, exploratory tendencies, and tension, thereby gauging the severity of depression19. Other tests such as the elevated plus maze (EPM) evaluate anxiety-like behaviors, the Morris water maze test (MWM) examines cognitive functioning20, and the FST assesses susceptibility to negative emotions and behavioral despair20. Furthermore, the majority of stressors that impact humans are social in nature. Individuals with suboptimal social relationships, characterized by limited social activities, networks, and support, are at a heightened risk for various diseases21,22. This is also relevant in the case of rodents, who are social animals living in groups. For example, rats that are housed in isolation exhibit characteristics of what is termed isolation syndrome, which induces social stress and expedites the onset of depression23.
Mongolian medicine, a significant branch of Chinese medicine, posits that the onset of disease is a complex interplay between intrinsic and external factors. These external factors, referred to as the four auxiliary conditions, encompass climate change, diet, lifestyle, and sudden events such as infections, startling incidents, and psychological disorders. The disease process is conceptualized as an ongoing interaction between three elements-referred to as the three types of homors-and the Seven Bodily Constituents in concert with the four auxiliary conditions24. Mongolian medicine holds that the human body functions as an integrated entity, maintained by a relative balance among the three homors. A disruption in this balance is considered to be a precursor to illness24. Given the pivotal role of animal experimentation in bridging traditional and modern medicine, it is crucial to develop animal models that are relevant for research in the field of Mongolian medicine. Accordingly, we employed a 28-day isolation methodology coupled with CUMS to simulate these physiological and psychological stressors. We selected nine specific unpredictable stressors and sought to underpin this modeling method through the Three homors theory of Mongolian medicine. Establishing a robust animal model is fundamental for advancing basic research in Mongolian medicine and will contribute significantly to its foundational studies.
Protocol
The experimental protocols received approval from the Ethics of Animal Experiment Care Committee of Inner Mongolia Medical University (YKD202301172) and adhered to the National Institutes of Health guidelines for animal care and ethics. The license number for our animal center is NO.110324230102364187. Twenty-four male Sprague-Dawley (SD) rats, each 8 weeks old (200 g ± 20 g), were acquired and housed in a controlled environment with a temperature of 22°C ± 2°C and humidity of 55% ± 15%. Feed the rats with a rodent maintenance-feed diet and pure water with corn cobs for bedding. The rats were subjected to a 12 h/12 h light/dark cycle for 1 week prior to experimentation.
1. Establishment of CUMS rat model
- Grouping
- Divide the 24 rats randomly into 2 groups: the control (CON) group, which will not be exposed to isolation or stress, and the model (MOD) group. Each group contains 12 rats.
- House the rats in standard cages measuring 55 cm x 40 cm x 20 cm, with 6 rats per cage. Retain the cage assignment throughout the acclimatization period unless otherwise indicated.
- Fill each rearing cage with fresh bedding and replace it twice a week.
- Conduct a 1-week acclimatization period. Allow the rats unrestricted access to food and water, except during CUMS stressor application. Maintain a constant environment with a temperature of 22°C ± 2°C, humidity of 55% ± 15%, and a 12 h/12 h light/dark cycle from 08:00 to 20:00, unless otherwise stated.
- Prior to initiating the experiment, handle the rats daily to acclimate them to the researcher and to minimize additional stress during the experimental phase.
- Isolation with chronic unpredictable mild stress
- Concurrently place the MOD and CON groups in separate rooms. House the MOD group rats individually, while keeping the CON group rats together. Maintain all other conditions constant.
- Implement a 28-day stressor regimen25. To prevent habituation and to ensure stressor unpredictability, administer one random stressor per day, avoiding the use of the same stressor on consecutive days.
- Randomly apply one of the following nine stressors26,27 on different days: 24 h water deprivation, 24 h food deprivation, wet padding, cage incline, inversion of the light/dark cycle, cold exposure at 4 °C, heat exposure at 45 °C, 1 min tail clamping, or 15 min shaking at 160 rpm. The specific design is outlined in Table 1.
- During the application of the stressor, restrict access to food and water to the MOD group until the stress was over, except during the inversion of the light/dark cycle. The CON group was not required to restrict water and diet.
- Stress methods
- Initiate the experiment by applying depression stimuli in conjunction with 28-day isolation to all rats, excluding the control group. House these rats in individual cages. Refer to Table 2 for conditions related to depression stimuli.
- To perform the tail clamp method, secure the tail of a rat from the MOD group by clamping the tail with a standard paper clip at a distance of 1-2 cm from the root of the rat's tail. Measure the clamping time for a duration of 1 min (n = 12).
- In the water deprivation method, withhold water from a MOD group rat by removing its water bottle and record for 24 h.
NOTE: The time of the start of the water deprivation was recorded so that the end time could be accurately calculated; the behavior of the rats during this period was observed, including activity, appetite and mental state. - In the food deprivation method, withhold food from a MOD group rat and record for 24 h.
NOTE: Record the time when food deprivation begins so that the end time can be accurately calculated; ensure that the rats are adequately hydrated during this period; and observe the behavior of the rats during this period, including activity and mental status, etc. - For cold stimulation at 4 °C, place a rat from the MOD group in a bucket of cold water and record for 5 min. Ensure that the water temperature remains consistent throughout the test. At the conclusion of the experiment, dry the rat with a blower and return it to its original cage.
NOTE: Maintain the cold water temperature at 4 °C using a thermometer and ice cubes to regulate the water temperature when it rises. The water should be changed regularly to ensure that the water quality is clean and the temperature is constant. When swimming, all limbs and trunks of the rats should be immersed in cold water except the head. The depth of the water must be greater than the length of the rat's body to prevent the rat from jumping out of the water due to contact with the bottom of the bucket. - To administer 45 °C heat stress, place a MOD group rat in an incubator and record for 5 min, ensuring that the temperature remains stable throughout the test.
- In the inversion of the light-dark cycle, envelop the cage in a black cloth for 1 h to simulate daytime darkness. Subsequently, illuminate the cage for 12 h during the night to mimic daylight. Record the rat's behavior, food and water intake, and sleep patterns for 24 h.
- For the wet padding experiment, introduce 200 mL of water into a cage containing 100 g of padding. House a MOD group rat in the wet cage and record the behavior of the rats under wet bedding stress, including activity, appetite, water intake, etc. Observe whether there are any abnormal behaviors or uncomfortable reactions, such as the skin and hair condition of the rats, which may be caused by the wet padding, and record them in time for subsequent analysis for 24 h. Following the test, dry the rat with a blower and return it to a cage with fresh wood shavings.
- In the cage incline method, situate a MOD group rat in a cage tilted at a 45° angle against a wall and record for 24 h. Use the cage frame structure to adjust the angle and secure the cage in place.
NOTE: Calculate the time from start to finish and observe the behavior of the rats during the period of tilting the cage, including activity, appetite, and mental state, and ensure that the angle of the tilted cage is set correctly and remains stable to ensure the accuracy and reproducibility of the experiment. - For high-speed shaking, place a MOD group rat in a mechanical shaker set to 160 rpm and record the rat for 15 min. Behavioral testing methods are subsequently utilized to evaluate the successful establishment of the model.
- After applying stressors, relocate the MOD group cages from the CUMS room back to the housing room. During the 4-week stress exposure period, maintain the CON group in their home cages situated in the housing room.
- Precautions during experiments
- Transfer cages of the MOD group back to the general housing room after applying the stressor in the CUMS room.
- Animal monitoring during CUMS modeling
- During tail clamping, the animal is likely to struggle due to the induced stimulation. Throughout this period, continuously monitor the clamp. If it becomes dislodged, pause the timer, reapply the clamp, and then resume the timer for 1 min.
- Do not impose water deprivation and wet bedding stressors simultaneously.
NOTE: Avoiding the simultaneous imposition of wet padding and water deprivation helps maintain experimental integrity, reduces confounding variables, and promotes animal welfare. - The animal's body temperature and ambient room temperature may raise the water temperature during cold water swimming. Therefore, adjust by adding ice water or ice cubes to maintain a constant water temperature.
- Observe the rats at 30 min intervals during the application of stressors, except during diurnal reversal. Pay special attention to signs of unusual distress, such as shivering, lethargy, or lack of movement. If such symptoms are observed-particularly potential hypothermia during 4 °C cold water swimming and wet bedding-immediately remove the rat from the stressor.
NOTE: Remove the experimental animals from the study when they have health problems, such as infection, severe trauma, aggressive behaviours, abnormal mobility, etc.,The conditions for removing the animals from the study typically revolve around protecting their health and safety while ensuring the integrity of experimental outcomes. - Conduct daily inspections for wounds or other physical or behavioral abnormalities in each rat. If any abnormalities are observed, consult the laboratory veterinarian to determine whether the rat should be excluded from the experiment.
- Weigh each rat every 3 days. If an animal loses more than 20% of its pre-feeding basal body weight, it must be excluded from the experiment.
2. Behavioral tests
- To begin, administer the depression stimuli in conjunction with isolation for 28 days to all rats, excluding the control group. House the rats in individual cages. Refer to Table 2 for specifics on the depression stimuli conditions.
- For the open field test, partition a black box into 25 square sections of equal area. Install a video tracking analysis system in the box. Place the rat in the central square and monitor its horizontal and vertical activities for a duration of 5 min.
NOTE: The dimensions of the box are 500 mm x 500 mm x 300 mm. Activity data will be gathered using the video tracking system to assess anxiety-related behaviors in the rodents when exposed to a novel environment. - Subsequently, tally the number of squares traversed by the rat, using all paws, to quantify horizontal activity. Count instances of standing and grooming as indicators of vertical activity. After each test, sanitize the box using 75% alcohol to eliminate any residual rat odors for subsequent tests.
- Next, anhedonia is assessed through the sucrose preference test. Place two bottles on the cage lid: bottle A contains pure water, while bottle B contains 1% sucrose solution. Allow ad libitum access to both solutions for the rat. Weigh the bottles before and after consumption to calculate the 60 min sucrose preference rates on days 0, 7, 14, 21, and 28. The formula is as follows:
Sucrose consumption =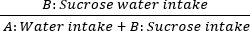 × 100%
× 100% - To measure spatial memory and learning abilities, employ the Morris water maze test. Divide the pool into four quadrants, numbering them from one to four. Place a submerged resting platform 1 cm below the water surface in the third quadrant.
- Introduce milk to the pool to increase water opacity and maintain a water temperature of approximately 23 °C throughout the experimental procedure.
- Place each rat in various quadrants of the maze, allowing them 120 s to locate the hidden platform. The rats must rely on spatial memory and learning skills to remember the platform's location. Once they learn the platform's location, they can swim directly to it. Record the latency time using the Morris water maze video tracking system.
- Position the rat at a fixed location in the pool. If the subject fails to locate the hidden platform within 120 s, record the latency as 120 s.
- Finally, remove the hidden platform, place the rat back into the water, and record the number of zone-crossings over a 120 s period.
3. Statistical analysis
- To assess significant differences in biochemical parameters, employ one-way analysis of variance (ANOVA) followed by Duncan's post hoc test. Present the data as mean ± standard error (SE) and regard a p-value of less than 0.05 as statistically significant.
Representative Results
Results from the behavioral tests in the CUMS-induced rat depression model
To corroborate the efficacy of the CUMS procedure for inducing depression-like behaviors, a manipulation check was performed. Male Sprague-Dawley (SD) rats were randomly allocated to either the MOD or CON group for a period of 4 weeks, as outlined in step 1.2.3. Subsequently, the rats were sacrificed, and their hippocampi were completely dissected for assessment of 5-HT, a neurotransmitter strongly associated with depression pathophysiology10, using enzyme-linked immunosorbent assay (ELISA)28.
Before CUMS induction, no significant differences were observed among the tested groups in the OFT score, sucrose preference, or MWM analysis.
After establishing the CUMS model, a one-way analysis of variance (ANOVA) revealed a significant impact of CUMS on rat anxiety behaviors. Specifically, in the OFT, significant differences were observed between the groups. The MOD group exhibited lower vertical and horizontal scores compared to the CON group (**P < 0.01; Figure 1A,B,E), suggesting that the CUMS procedure was effective in inducing anxiety.
Subsequently, the sucrose preference test (SPT) was administered over 5 days, as described in step 2.4, to evaluate whether the rats showed signs of anhedonia post-CUMS exposure. Results indicated no significant differences in sucrose preference percentage between the groups on day 0. However, one-way ANOVA revealed a significant difference in sucrose intake between the groups on day 28. Specifically, the sucrose preference percentage in the MOD group was lower than that in the CON group (**P < 0.01; Figure 1C), confirming the effectiveness of the CUMS protocol in inducing anhedonia.
A one-way ANOVA revealed that CUMS significantly impacted the spatial memory and learning abilities of rats, as assessed by the MWM test. The latency for the MOD group to locate the platform was notably longer than that for the CON group (**P < 0.01). In terms of crossing times, the MOD group crossed fewer times than the CON group (**P < 0.01; Figure 1D,F). These findings support the efficacy of the CUMS protocol in inducing memory impairment in rats.
Results of hippocampal 5-HT levels in the CUMS-induced rat depression model
One-way ANOVA on hippocampal 5-HT levels indicated a significant difference among the groups (**P < 0.01). The MOD group exhibited reduced hippocampal 5-HT levels when compared to the CON group (Figure 2). These results suggest that the CUMS protocol effectively decreased hippocampal 5-HT levels, a phenomenon commonly observed in human depression29.
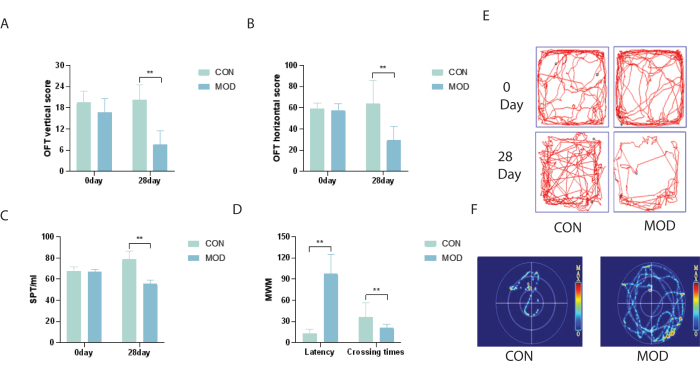
Figure 1: Effects of the CUMS-induced rat depression model. (A) Open field test (OFT) vertical scores. (B) Open field test horizontal scores. (C) Sucrose consumption levels (%) on days 0 and 28. CUMS-induced anhedonia and reduced sucrose preference in rats. (D) Latency time and zone crossing numbers in the MWM test. Results are presented as the mean ± SE (n = 12 rats per group). **P < 0.01 indicates significant changes in the model (MOD) group compared to the control (CON) group calculated using one-way analysis of variance (ANOVA). (E) OFT tracks for rats in each group. (F) MWM tracks for each group of rats. Please click here to view a larger version of this figure.
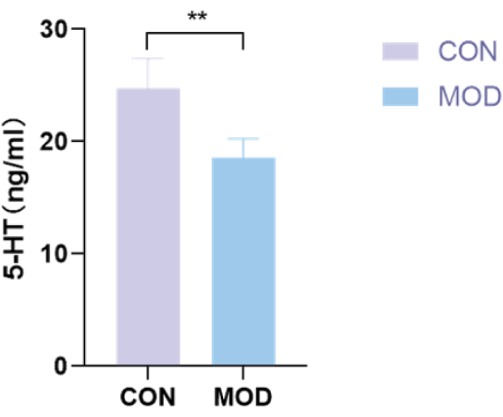
Figure 2: Effect on brain 5-HT levels in CUMS-induced depression model in rats. Rats subjected to 4 weeks of CUMS demonstrated a significant reduction in hippocampal 5-HT levels compared to the control (CON) group. Results are presented as the mean ± SE (n = 12 rats per group). **P < 0.01 indicates significant changes in the model (MOD) group compared to the CON group calculated using one-way analysis of variance (ANOVA). Please click here to view a larger version of this figure.
| Stressors | Days |
| Tail clamp 1 min | 1, 7, 20 |
| Water deprivation 24 h | 3, 18, 28 |
| Food deprivation 24 h | 2, 16, 25 |
| Swimming in 4 °C cold water 5 min | 4, 11, 19, 23 |
| 45 °C heat stress 5 min | 5, 14, 17, 27 |
| Inversion of light dark cycle | 6, 12, 22 |
| Wet padding | 9, 15, 26 |
| Cage incline 24 h | 10, 21 |
| High-speed shaking 15 min | 8, 13, 24 |
Table 1: Chronic unpredictable mild stress (CUMS) schedule. Stressors and execution dates for the induction of CUMS in rat subjects.
| Stressors | Four Auxiliary Conditions | Three Homors change |
| Tail clamp 1 min | Sudden factors | Wind↑ Bile↑ |
| Water deprivation 24 h | Diet | Wind↑ |
| Food deprivation 24 h | Diet | Wind↑ |
| Swimming in 4 °C cold water 5min | Living | Phlegm↑ |
| 45 °C heat stress 5min | Living, Climate change | Bile↑ |
| Inversion of light dark cycle | Living | Wind↑ |
| Wet padding | Living | Phlegm↑ |
| Cage incline 24 h | Living | Wind↑ |
| High-speed shaking 15min | Sudden factors | Wind↑ |
Table 2. Nine stressors with the four auxiliary conditions and the theory of three homors in Mongolian medicine. The intrinsic relationship between the nine stressors, the theory of three homors, and four auxiliary conditions in Mongolian medicine.
Discussion
Depression is a mental disorder characterized by symptoms such as low mood, lack of pleasure, and reduced energy30. In the field of depression research, the establishment of a reliable animal model is crucial for advancing therapeutic interventions. Among various animal models, the CUMS model is particularly noteworthy for its high reliability, validity, and its congruence with the characteristics of human depression31. It is well-suited to mimic the cumulative effects of low-level stressors over an extended period across various settings. In this study, we established a CUMS-induced depression model in rats, employing a range of stressors outlined in step 1.2.3 and Table 2. Several aspects of the stress protocol are critical for the model’s success. Firstly, rats in all MOD groups must be housed individually, unlike those in the CON group. Secondly, the CUMS stress regimen should be carefully planned to maintain unpredictability. Specifically, rats should be subjected to a randomized selection of seven out of the nine possible stressors on a weekly basis. The variability and unpredictability of these stressors are essential for the model’s efficacy32,33. Lastly, repeated use of the same stressor should be minimized to prevent the rats from adapting, which could compromise the integrity of the experimental outcomes. Moreover, the stressor settings were designed to be adjustable throughout the course of the study.
The CUMS model offers several advantages as a depression model. It closely aligns with the pathogenesis of human depression and accurately represents most clinical symptoms and underlying mechanisms34. One of its key strengths is that it uses anhedonia, or the lack of pleasure, as a quantifiable indicator6,7. The model also accounts for the various factors that contribute to the onset of depression35,36. Importantly, the CUMS model is designed for long-term application, spanning several months. This duration is consistent with the needs of a depression model and allows for the assessment of drugs in a chronic dosing regimen, as well as the identification of compounds with rapid mechanisms of action36. Due to its comprehensive nature, the CUMS model has become widely accepted in the scientific literature.
Despite these advantages, the CUMS model is not without limitations5,37. One of the primary drawbacks is its resource-intensive nature, requiring significant investments in environment, manpower, and time, which consequently results in low reproducibility38. There are also inconsistencies in the literature regarding the selection and intensity of stressors, as well as the sequence in which they are applied38. Such inconsistencies make it challenging to replicate the model across different laboratories. Furthermore, the literature lacks a detailed time schedule for applying stressors, adding another layer of ambiguity39,40. Another issue arises from the variation in combination methods employed across different studies; these often lack explicit rationale, creating difficulties in model replication. Subject animals are highly sensitive to minor alterations in the stressor design, which also contributes to low reproducibility40. Moreover, using a limited number of stressors can lead to habituation among the subject animals, making the stressor design predictable. Finally, animals from different laboratories show varied responses to stressors, contributing to the poor reproducibility across studies. In the existing literature, Wistar Kyoto (WKY) rats have been identified as particularly susceptible to a depression-like phenotype following exposure to CUMS25,41. This suggests that inter-strain variability can significantly affect susceptibility to stress-induced behavioral changes. Similarly, the SPT, a key metric often used for assessing the efficacy of depression models, presents its own set of challenges. Specifically, individual rats show variability in their consumption of a 1% sucrose solution20,42. Under stress conditions, both stress-prone and stress-resistant animals exhibit alterations in their consumption of sweet solutions. Consequently, it becomes challenging to ascertain the proportion of animals within a given batch that are genuinely stress-resistant or belong to the opposite category. This variability may result in some animals displaying SPT outcomes that do not accurately reflect their disease state.
To enhance the reliability of the CUMS model, we propose several methodological refinements. First, it is crucial to segregate animals in the CON group from those in the MOD group to mitigate the impact of olfactory, visual, and auditory stimuli, which can induce stress in the animals43,44. Second, to minimize variability, it is advisable to designate one or two experienced laboratory personnel to conduct all experiments. This approach serves to reduce errors attributable to individual factors. In addition, prior research has identified stress-resilient test animals13; hence, it would be prudent to screen for such groups either prior to formal experimentation or post hoc to better interpret the results. Regarding the SPT test, which is a reward-based behavioral assessment that gauges intrinsic anhedonia, it is essential to determine the optimal sucrose concentration before initiating the CUMS protocol. Preliminarily establishing preference based on sucrose concentration can markedly enhance the test’s discriminative sensitivity20. However, it should be noted that rats subjected to 24 h food and water deprivation may indiscriminately consume any available liquid due to extreme thirst35,45. To obtain a more accurate measure of sucrose preference, we recommend assessing consumption during the nocturnal phase of the circadian cycle, when rodents are most active. Stressors applied during the diurnal (inactive) phase could result in chronic sleep deprivation, thereby introducing an unintended stressor46,47. Finally, it is imperative to meticulously control for the influence of manageable factors described above to ensure that the data collected are as reliable as possible.
The current Western clinical treatment regimens for depression often come with pronounced toxic side effects and a high likelihood of relapse upon cessation of medication. Investigating treatment approaches rooted in traditional medicine and delving into the pathogenesis of depression through the lens of Mongolian medicine theory could offer innovative insights into the disease’s etiology. In this context, the development of animal models of depression that align with traditional medicine principles stands as a pivotal step in Mongolian medicine research. The selection of the nine stressors in the solitary-culture CUMS modeling method is particularly noteworthy; it is informed by Mongolian medicine’s theory of the three homors and the inherent relationship between the four auxiliary conditions and the three homors. This approach exhibits a high degree of compatibility with the unique tenets of Mongolian medicine. Phenotypic analyses of depression, in conjunction with external factors, reveal significant correlations with the Mongolian medical concepts of Wind and Phlegm (see Table 2). Specifically, wind-related dysfunctions manifest clinically as symptoms such as loss of energy, insomnia, forgetfulness, fatigue, and pain. Dysfunctions related to Phlegm, on the other hand, result in clinical manifestations like unresponsiveness, blurred consciousness, pessimism, and negativity. These symptoms align closely with the core symptoms of MDD48,49. Therefore, the aforementioned CUMS model serves as a pathological model that effectively simulates the complex interactions among external conditions and the balance of the three types of homors, as posited by Mongolian medicine theory, thereby contributing to the onset of disease.
The strengths of this model are multifaceted. Firstly, it adheres to contemporary medical etiological theories, satisfying the criteria set for animal models within the realm of modern medicine. Secondly, it incorporates principles from traditional Mongolian medicine to replicate specific symptoms of human diseases in animals, thus embodying characteristics unique to Mongolian medical practices. Given its alignment with both modern and traditional medical theories, this model serves as an effective platform for foundational research in Mongolian medicine. Therefore, the integration of diseases and symptoms within this animal model represents a confluence of research in Mongolian and modern medicine. While the model has its limitations, its ongoing refinement will significantly contribute to the future of basic research in Mongolian medicine.
Animal models have become indispensable in the exploration of depression due to the ethical and practical challenges associated with direct human brain study. Despite their inherent limitations in fully replicating the complexities of depression, the evolution and validation of these models remain a continual process. In this study, we have combined principles from both Mongolian and modern medicine to develop an animal model of depression. This collaborative approach offers valuable insights for constructing a more comprehensive animal model that encompasses both traditional Mongolian and modern medical theories.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We are grateful for the instrumentation and laboratory provided byextend our gratitude to the Mongolian medical faculty of the Inner Mongolian Medical University, China.This study was supported by, for providing the necessary instrumentation and laboratory facilities. This study received financial support from the National Natural Sciences Foundation of China (Grant No. 81760762) and), the Inner Mongolia Medical University Project of China (Grant No. YKD2022MS074), and the Scientific Research Project of Higher Education in Inner Mongolia, China (Grant No. NJZY22661) and the Open Fund Project of Key Laboratory of Chinese and Mongolian Medicine in Inner Mongolia Autonomous Region,China (Grant No. MYX2023-K07).
Materials
| 1.5 mL centrifuge tube | service Biotechnology Co., Ltd | EP-150-M | |
| 1000 µL Pipette | service Biotechnology Co., Ltd | IC021198160223 | |
| 10 µL pipette tip | service Biotechnology Co., Ltd | IC012395160823 | |
| 10 µL pipette tip | service Biotechnology Co., Ltd | TP-10 | |
| 1250 µL pipette tip | service Biotechnology Co., Ltd | TP-1250 | |
| 2 mL centrifuge tube | service Biotechnology Co., Ltd | EP-200-M | |
| 200 µL pipette tip | service Biotechnology Co., Ltd | TP-200 | |
| 200 µL pipette tip | service Biotechnology Co., Ltd | IC021029160323 | |
| 300 µL Multi-Channel Pipette | service Biotechnology Co., Ltd | IC091006161022 | |
| 50 µL Pipette | service Biotechnology Co., Ltd | DS35110 | |
| Automatic plate washing machine | rayto Life Sciences Co., Ltd | RT-3100 | |
| Benchtop High-Speed Freezing Centrifuge | dalong construction Co., Ltd | D3024R | |
| electronic balance | Mettler Toledo International Trade (Shanghai) Co., Ltd | ME203E/02 | |
| Electrothermal blast drying oven | Labotery Experimental Instrument Equipment Co., Ltd | GEL-70 | |
| Enzyme Label Detector | BioTeK Co., Ltd | Epoch | |
| High Speed Tissue Grinder | service Biotechnology Co., Ltd | KZ- -F -F |
|
| Horizontal Freezer | Mellow Group Co., Ltd | BCD-318AT | |
| Laboratory Ultrapure Water Machine | Jinan Aiken Environmental Protection Technology Co., Ltd | AK-RO-C2 | |
| Morris water maze video trail analysing system | Tai Meng Tech Co., Ltd | WMT-200 | |
| Rat 5-HT ELISA Kit | Lian Ke bio Co., Ltd,China | 96T/48T | |
| SPF grade Sprague Dawley (SD) rats | SPF (Beijing) Biotechnology Co | SCXK(JING)2019-0010 | |
| Sprague Dawley rats | Beijing Biotechnology Co., Ltd, China | SCXK (JING) 2019-0010 | |
| Vertical Refrigerated Display Cabinet | Xingx Group Co., Ltd | LSC-316C | |
| video tracking system | Tai Meng Tech Co., Ltd | ZH-ZFT | |
| vortex mixer | Servicebio technology Co., Ltd | MV-100 |
References
- Alqurashi, G. K., et al. The impact of chronic unpredictable mild stress-induced depression on spatial, recognition and reference memory tasks in mice: Behavioral and histological study. Behav Sci. 12 (6), 166 (2022).
- Yu, S., Wang, L., Jing, X., Wang, Y., An, C. Features of gut microbiota and short-chain fatty acids in patients with first-episode depression and their relationship with the clinical symptoms. Front Psychol. 14, 1088268 (2023).
- Duda, P., Hajka, D., Wójcicka, O., Rakus, D., Gizak, A. Gsk3β: A master player in depressive disorder pathogenesis and treatment responsiveness. Cells. 9 (3), 727 (2020).
- Correia, A. S., Vale, N. Tryptophan metabolism in depression: A narrative review with a focus on serotonin and kynurenine pathways. Int J Mol Sci. 23 (15), 8493 (2022).
- Hao, Y., Ge, H., Sun, M., Gao, Y. Selecting an appropriate animal model of depression. Int J Mol Sci. 20 (19), 4827 (2019).
- Willner, P., Towell, A., Sampson, D., Sophokleous, S., Muscat, R. Reduction of sucrose preference by chronic unpredictable mild stress, and its restoration by a tricyclic antidepressant. Psychopharmacology (Berl). 93 (3), 358-364 (1987).
- Nakase, S., Kitayama, I., Soya, H., Hamanaka, K., Nomura, J. Increased expression of magnocellular arginine vasopressin mrna in paraventricular nucleus of stress-induced depression-model rats. Life Sci. 63 (1), 23-31 (1998).
- Wu, X., et al. Involvement of kynurenine pathway between inflammation and glutamate in the underlying etiopathology of cums-induced depression mouse model. BMC Neurosci. 23 (1), 62 (2022).
- Zhang, C., et al. Minocycline ameliorates depressive behaviors and neuro-immune dysfunction induced by chronic unpredictable mild stress in the rat. Behav Brain Res. 356, 348-357 (2019).
- Ma, J., Wang, R., Chen, Y., Wang, Z., Dong, Y. 5-HT attenuates chronic stress-induced cognitive impairment in mice through intestinal flora disruption. J Neuroinflammation. 20 (1), 23 (2023).
- Nollet, M. Models of depression: Unpredictable chronic mild stress in mice. Curr Protoc. 1 (8), e208 (2021).
- Becker, M., Pinhasov, A., Ornoy, A. Animal models of depression: What can they teach us about the human disease. Diagnostics (Basel). 11 (1), 123 (2021).
- Petkovic, A., Chaudhury, D. Encore: Behavioural animal models of stress, depression and mood disorders. Front Behav Neurosci. 16, 931964 (2022).
- Okine, T., Shepard, R., Lemanski, E., Coutellier, L. Sex differences in the sustained effects of ketamine on resilience to chronic stress. Front Behav Neurosci. 14, 581360 (2020).
- Fitzgerald, P. J., et al. Sex- and stress-dependent effects of a single injection of ketamine on open field and forced swim behavior. Stress. 24 (6), 857-865 (2021).
- Doron, R., Burstein, O. The unpredictable chronic mild stress protocol for inducing anhedonia in mice. J Vis Exp. (140), e58184 (2018).
- De Vry, J., Schreiber, R. The chronic mild stress depression model: Future developments from a drug discovery perspective. Psychopharmacology (Berl). 134 (4), 349-350 (1997).
- Tong, J., et al. Antidepressant effect of helicid in chronic unpredictable mild stress model in rats. Int Immunopharmacol. 67, 13-21 (2019).
- Liu, H., et al. Tnf signaling pathway-mediated microglial activation in the pfc underlies acute paradoxical sleep deprivation-induced anxiety-like behaviors in mice. Brain Behav Immun. 100, 254-266 (2022).
- He, L. W., et al. Optimization of food deprivation and sucrose preference test in sd rat model undergoing chronic unpredictable mild stress. Animal Model Exp Med. 3 (1), 69-78 (2020).
- Ma, W., Wu, B., Gao, X., Zhong, R. Association between frailty and cognitive function in older chinese people: A moderated mediation of social relationships and depressive symptoms. J Affect Disord. 316, 223-232 (2022).
- Geng, C., et al. Systematic impacts of chronic unpredictable mild stress on metabolomics in rats. Sci Rep. 10 (1), 700 (2020).
- Holmes, T. H., Rahe, R. H. The social readjustment rating scale. J Psychosom Res. 11 (2), 213-218 (1967).
- Zhang, M., et al. Shuxie-1 decoction alleviated cums -induced liver injury via il-6/jak2/stat3 signaling. Front Pharmacol. 13, 848355 (2022).
- Antoniuk, S., Bijata, M., Ponimaskin, E., Wlodarczyk, J. Chronic unpredictable mild stress for modeling depression in rodents: Meta-analysis of model reliability. Neurosci Biobehav Rev. 99, 101-116 (2019).
- Hu, C., et al. Re-evaluation of the interrelationships among the behavioral tests in rats exposed to chronic unpredictable mild stress. PLoS One. 12 (9), e0185129 (2017).
- Li, Y., et al. Increased hippocampal fissure width is a sensitive indicator of rat hippocampal atrophy. Brain Res Bull. 137, 91-97 (2018).
- Hu, R. L. B. G., et al. Effects of the Mongolian medicine Zadi-5 on monoamine neurotransmitters in the brain of rats with chronic stress depression model. Info Traditional Chinese Med. 34 (06), 5-8 (2017).
- Zhou, Y., Cong, Y., Liu, H. Folic acid ameliorates depression-like behaviour in a rat model of chronic unpredictable mild stress. BMC Neurosci. 21 (1), 1 (2020).
- Zhuang, Y., Zeng, R., Liu, X., Yang, L., Chan, Z. Neoagaro-oligosaccharides ameliorate chronic restraint stress-induced depression by increasing 5-ht and bdnf in the brain and remodeling the gut microbiota of mice. Mar Drugs. 20 (11), 725 (2022).
- Socała, K., et al. The role of microbiota-gut-brain axis in neuropsychiatric and neurological disorders. Pharmacol Res. 172, 105840 (2021).
- Song, J., Kim, Y. K. Animal models for the study of depressive disorder. CNS Neurosci Ther. 27 (6), 633-642 (2021).
- Abelaira, H. M., Réus, G. Z., Quevedo, J. Animal models as tools to study the pathophysiology of depression. Braz J Psychiatry. 35 Suppl 2, S112-S120 (2013).
- Strekalova, T., et al. Chronic mild stress paradigm as a rat model of depression: Facts, artifacts, and future perspectives. Psychopharmacology (Berl). 239 (3), 663-693 (2022).
- Markov, D. D. Sucrose preference test as a measure of anhedonic behavior in a chronic unpredictable mild stress model of depression: Outstanding issues. Brain Sci. 12 (10), 1287 (2022).
- Czéh, B., Fuchs, E., Wiborg, O., Simon, M. Animal models of major depression and their clinical implications. Progress in Neuro-Psychopharmacology and Biological Psychiatry. 64, 293-310 (2016).
- Gururajan, A., Reif, A., Cryan, J. F., Slattery, D. A. The future of rodent models in depression research. Nat Rev Neurosci. 20 (11), 686-701 (2019).
- Markov, D. D., Novosadova, E. V. Chronic unpredictable mild stress model of depression: Possible sources of poor reproducibility and latent variables. Biology (Basel). 11 (11), 1621 (2022).
- Willner, P. The chronic mild stress (cms) model of depression: History, evaluation and usage. Neurobiol Stress. 6, 78-93 (2017).
- Lages, Y. V. M., Rossi, A. D., Krahe, T. E., Landeira-Fernandez, J. Effect of chronic unpredictable mild stress on the expression profile of serotonin receptors in rats and mice: A meta-analysis. Neurosci Biobehav Rev. 124, 78-88 (2021).
- Willner, P., et al. Validation of chronic mild stress in the wistar-kyoto rat as an animal model of treatment-resistant depression. Behavioural Pharmacology. 30 (2 and 3), 239-250 (2019).
- Slattery, D. A., Cryan, J. F. Modelling depression in animals: At the interface of reward and stress pathways. Psychopharmacology (Berl). 234 (9-10), 1451-1465 (2017).
- Sterley, T. L., et al. Social transmission and buffering of synaptic changes after stress. Nat Neurosci. 21 (3), 393-403 (2018).
- Brechbühl, J., et al. Mouse alarm pheromone shares structural similarity with predator scents. Proc Natl Acad Sci U S A. 110 (12), 4762-4767 (2013).
- Beck, R. C., Self, J. L., Carter, D. J. Sucrose preference thresholds for satiated and water-deprived rats. Psychol Rep. 16, 901-905 (1965).
- Hawkins, P., Golledge, H. D. R. The 9 to 5 rodent – time for change? Scientific and animal welfare implications of circadian and light effects on laboratory mice and rats. J Neurosci Methods. 300, 20-25 (2018).
- Daut, R. A., Ravenel, J. R., Watkins, L. R., Maier, S. F., Fonken, L. K. The behavioral and neurochemical effects of an inescapable stressor are time of day dependent. Stress. 23 (4), 405-416 (2020).
- Hu, R. L. B. G., et al. Experimental research on nutmeg wuwei pills against of depression model rats behavior and hippocampus monoamine neurotransmitters. Chinese J Exp Traditional Medical Formulae. 21 (11), 146-149 (2015).
- Hu, R. L. B. G., et al. Effects of Rou kou Wuwei Pill on the learning and memory abilities and the expression of BDNF and TrkB in hippocampus of depression rats. Chinese J Traditional Chines Med Pro. 32 (8), 3797-3800 (2017).

