Large-Scale Cell Production Based on GMP-Grade Dissolvable Porous Microcarriers
Summary
Here, we present a protocol to achieve the large-scale manufacturing of adherent cells through a fully closed system based on GMP-grade dissolvable microcarriers. The cultivation of human mesenchymal stem cells, HEK293T cells, and Vero cells was validated and met both quantity demands and quality criteria for the cell and gene therapy industry.
Abstract
Researchers in the cell and gene therapy (CGT) industry have long faced a formidable challenge in the efficient and large-scale expansion of cells. To address the primary shortcomings of the two-dimensional (2D) planar culturing system, we innovatively developed an automated closed industrial scale cell production (ACISCP) platform based on a GMP-grade, dissolvable, and porous microcarrier for the 3D culture of adherent cells, including human mesenchymal stem/stromal cells (hMSCs), HEK293T cells, and Vero cells. To achieve large-scale expansion, a two-stage expansion was conducted with 5 L and 15 L stirred-tank bioreactors to yield 1.1 x 1010 hMSCs with an overall 128-fold expansion within 9 days. The cells were harvested by completely dissolving the microcarriers, concentrated, washed and formulated with a continuous-flow centrifuge-based cell processing system, and then aliquoted with a cell filling system. Compared with 2D planar culture, there are no significant differences in the quality of hMSCs harvested from 3D culture. We have also applied these dissolvable porous microcarriers to other popular cell types in the CGT sector; specifically, HEK293T cells and Vero cells have been cultivated to peak cell densities of 1.68 x 107 cells/mL and 1.08 x 107 cells/mL, respectively. This study provides a protocol for using a bioprocess engineering platform harnessing the characteristics of GMP-grade dissolvable microcarriers and advanced closed equipment to achieve the industrial-scale manufacturing of adherent cells.
Introduction
The CGT industry has witnessed an exponential expansion over the past two decades. The evolution of next-generation medicines is anticipated to treat and cure numerous refractory diseases1. Since the first Food and Drug Administration (FDA) approval of a CGT product, Kymriah, in 2017, CGT-related research and development in the world has continued to grow at a fast rate, with the FDA seeing active investigational new drug applications for CGT increased to 500 in 20182. It had been predicted that the number of approvals of CGT products will likely be 54-74 in the United States by 20302.
While the rapid growth in CGT research and innovation is exciting, there is still a large technological gap between lab research and industrial-scale manufacturing that could deliver these promising medicines to reach as many patients as needed at affordable costs. The current processes adopted for these clinical trials were established in labs for small-scale experiments, and significant efforts are needed to improve and innovate on CGT manufacturing3. There are many types of CGT products, most of them based on live cells, which can be allogenic, autologous, engineered, or natural. These living drugs are much more complex than small molecular entities or biologics, hence making large-scale manufacturing a significant challenge4,5,6. In this work, we demonstrate a large-scale cell production protocol for three anchorage-dependent cells that are widely applied in CGTs. These include human mesenchymal stem/stromal cells (hMSCs), which have been used for cell-based therapy, and HEK293T cells and Vero cells, both of which are used to produce viruses for the genetic engineering of the final therapeutic cell product. Anchorage-dependent cells are commonly cultured on planar systems, which require manual processing. However, manual culture methods require a significant amount of labor and are prone to contamination, which can compromise the quality of the end product. Furthermore, there is no in-line process control, leading to substantial variability in quality between batches7. Taking stem cell therapy as an example, with a promising pipeline of over 200 stem-cell therapy candidates, it is estimated that 300 trillion hMSCs would be needed per year to meet the demands of clinical applications8. Hence, the large-scale manufacturing of therapeutic cells has become a prerequisite to perform these therapeutic interventions with such a high cell demand9.
To preclude the setbacks of planar systems, efforts have been made in developing large-scale manufacturing processes in stirred-tank bioreactors with conventional non-dissolvable microcarriers10,11,12,13, but these suffer from complicated preparation procedures and low cell-harvesting efficiency14. Recently, we have innovated a dissolvable microcarrier for stem cell expansion, aiming to circumvent the challenges of cell harvesting from conventional non-dissolvable commercial microcarriers15. This novel, commercially available GMP-grade 3D dissolvable porous microcarrier, 3D TableTrix, has shown great potential for large-scale cell production. Indeed, 3D culture based on these porous microcarriers could potentially recreate favorable biomimetic microenvironments to promote cell adhesion, proliferation, migration, and activation16. The porous structures and interconnected pore networks of microcarriers could create a larger cell adhesion area and promote the exchange of oxygen, nutrients, and metabolites, thus creating an optimal substrate for in vitro cell expansion17. The high porosity of these GMP-grade 3D dissolvable porous microcarriers enables large-scale expansion of hMSCs, and the ability for the cells to be fully dissolved allows for the efficient harvesting of these expanded cells18. It is also a GMP-grade product and has been registered as a pharmaceutical excipient with the Chinese Center for Drug Evaluation (filing numbers: F20210000003 and F20200000496)19 and the FDA of the United States (FDA, USA; Drug Master File number: 35481)20.
Here, we illustrate an automated closed industrial scale cell production (ACISCP) system18 using these dispersible and dissolvable porous microcarriers for hMSC, HEK293T cell, and Vero cell expansion. We achieved a successful two-tiered expansion of hMSCs (128 cumulative fold expansion in 9 days) from a 5 L bioreactor to a 15 L bioreactor and finally obtained up to 1.1 x 1010 hMSCs from a single batch of production. The cells were harvested by completely dissolving the microcarriers, concentrated, washed and formulated with a continuous flow centrifuge-based cell processing system, and then aliquoted with a cell filling system. Furthermore, we assessed the quality of hMSC products to confirm compliance. We also demonstrated the application of these dissolvable microcarriers for the scaled-up production of two other types of anchorage cells, HEK293T cells and Vero cells, that are extensively applied in the CGT industry. The peak cell density of HEK293T cells reached 1.68 x 107 cells/mL, whereas the peak density of Vero cells reached 1.08 x 107 cells/mL. The ACISCP system could be adapted to culture a variety of adherent cells, and it could potentially become a powerful platform contributing to expediting the industrialization of CGT.
Protocol
The human umbilical cord was obtained from Beijing Tsinghua Changgeng Hospital. All procedures and protocols regarding the acquisition, isolation, and culture of human umbilical cord mesenchymal stem cells (UCMSCs) were conducted with informed consent and with the approval of the Ethics Committee of Beijing Tsinghua Changgeng Hospital (filing number 22035-4-02), and the procedures and protocols complied with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
1. Monolayer culture of hMSCs, HEK293T cells, and Vero cells
- Isolate primary human UCMSCs from Wharton's jelly according to the reported method21. Use serum-free (SF) hMSC culture medium for the isolation and outgrowth of primary UCMSCs, harvest the cells on passage 2, and cryopreserve as a master cell bank (MCB).
- Inoculate 6 x 105 human UCMSCs (preferably passage 2 or passage 3 cells) in one T75 flask (i.e., the seeding density on 2D planar vessels is 8,000 cells/cm2) for monolayer culture with 15 mL of complete SF hMSC culture medium. Ensure uniform seeding through a figure-eight movement of the flask. Culture at 37 °C in a 5% CO2 cell culture incubator to 80%-90% confluency.
- To harvest, decant the cell culture medium, and rinse the cells with 3-5 mL of phosphate buffered saline (PBS) twice. Then, add 2 mL of 0.25% Trypsin-EDTA, and incubate at 37 °C for 1-2 min until most cells have become round and have detached from the flask, as observed under a bright-field inverted microscope with a 4x objective. Add 3 mL of SF hMSC culture medium to terminate digestion.
- Transfer the cell suspension to a 15 mL conical centrifuge tube, and centrifuge at 179 x g for 5 min. Decant the supernatant, rinse the cell pellet with PBS twice, and resuspend in around 3-5 mL of SF hMSC culture medium for seeding to microcarriers in bioreactors.
NOTE: Check the cell viability, and make sure the cells are >90% viable. Only prepare the cells after all the preparation work for the bioreactors has been completed (steps 2.1-3.6). - For the monolayer culture of HEK293T cells and Vero cells, inoculate at 2 x 106-3 x 106 cells into each T75 flask, and culture with DMEM containing 10% fetal bovine serum (FBS) and 10% newborn calf serum (NBS), respectively.
- Follow steps 1.3-1.4 for harvesting the cells. Resuspend in 3-5 mL of DMEM containing 10% FBS.
2. Preparation of the stirred-tank bioreactor before cell seeding
- On day −1, rinse the glass vessel thoroughly with running deionized (DI) water once.
- Disassemble all the stainless tubes and impellers from the headplate, immerse them in DI water, and sonicate with an ultrasonic cleaner at 40 kHz and 60 °C for 1 h to clean thoroughly.
NOTE: The preparations and procedures will be the same for first-tier expansion in the 5 L bioreactor and the second-tier expansion in the 15 L bioreactor. The parameters for the 15 L bioreactor are given in brackets throughout. If no parameters are given in brackets, these parameters are the same for the 5 L and 15 L bioreactors. - Carefully check all the O-rings are intact and in place, and then install the disassembled parts back onto the headplate according to the manufacturer's instructions.
NOTE: Replace any worn or torn O-rings before assembly if necessary. Worn O-rings will compromise the airtightness and, hence, the sterility of the vessel. - Add 7 L (18 L for the 15 L bioreactor) of 0.5 M NaOH solution into the 5 L vessel, and place the headplate with the tubes assembled on the rim of the vessel to immerse the tubes in the NaOH solution. Let it stand for 12 h or overnight to remove endotoxins from the tubes and glass vessel.
NOTE: Do not exceed 7 L (18 L) as this is the maximum volume of the 5 L (15 L) glass vessel. Wear proper personal protective equipment when handling the NaOH solution, as it is corrosive. - Remove the headplate and decant the NaOH solution into a proper waste bottle. Discard according to lab safety regulations. Rinse the vessel, the headplate, and its parts thoroughly with endotoxin-free DI water or water for injection to remove residual alkaline.
- Add 2 L (5 L) of PBS to the vessel, reassemble all the parts, and bolt the headplate to the metal frame of the vessel according to the manufacturer's instructions.
- Fit a Closed System Consumable Pack to the appropriate stainless steel (SS) parts/tubes/ports on the headplate according to the manufacturer's instructions. Clamp all the Robert clamps on the tubes, and reinforcing by clamping with hemostats is recommended. Leave one of the tubes, with an 0.22 μm air vent filter at the end that is connected to the condenser, unclamped.
NOTE: Slip the silicone tubes onto the tips onto the hemostats to prevent piercing the silicone tubes. Clamp only on the silicone tubes, and avoid clamping the C-flex tubes included in this kit. Leaving one tube on the condenser unclamped will allow for pressure relief during autoclaving. - Prepare 250 mL of 0.1 M NaOH solution in an autoclavable 500 mL GL 45 glass bottle, and fit with a GL 45 Multiport Connector Screw Cap with two ports. Connect a 180 mm long, 0.13 in x 0.25 in (internal diameter x outer diameter) silicone tube onto one of the ports on the lower side of the cap; the length should be long enough to just touch the bottom of the bottle.
- Connect another piece of silicone tube, with a length 500 mm longer than the previous one, to the same port on the upper side of the cap, and connect the other end to a drip feed port on the 5 L vessel headplate. Connect a 0.22 μm air vent filter to the other port on the screw cap.
- Perform a two-point calibration on the pH probe as per the manufacturer's instructions. Then, insert the pH, dissolved oxygen (DO), and live cell count probes into the vessel in the appropriate PG13,5 ports, and screw tight onto the headplate.
- Perform an airtightness check of the fully assembled vessel according to the manufacturer's instructions. Maintain a pressure of 0.4 bar ± 0.01 bar for at least 30 min. Slowly release the pressure after the system passes the test. If the airtightness test fails, carefully search for leak points, and reinforce the vessel assembly.
- Autoclave the fully assembled vessel with the consumable kit at 15 psi and 121 °C for 60 min. Following autoclaving, check all the air vent filters to make sure they are intact and dry. Remove all the hemostats.
NOTE: A wet air vent filter will not filter the gas efficiently and might compromise the sterility of the vessel during cell culture. Change the air vent filters if they are wet, and autoclave the entire vessel again. - Transfer the autoclaved vessel into a cleanroom, and wait for it to completely cool to room temperature. Insert the temperature probe into the thermowell tube, and connect the cables for the motor, pH probe, and DO probe from the controller to the respective parts on the vessel. Wrap the heat mat around the vessel tightly.
- Unclamp the Robert clamps on the flexible tubes on the exhaust gas condenser, ring sparger, drip feed port for NaOH, and air overlay port. Turn on the controller, log in, and enter the parameters listed in Table 1 into the system according to the manufacturer's instructions. These tubes should not be clamped at any time during the culture process.
- Click on Start, and name the experiment to start the bioreactor. Allow the bioreactor to run on these settings for at least 12 h, or overnight, to saturate the PBS with oxygen in order to perform a one-point calibration for the DO probe later.
- Calibrate the liquid handling volume speed of pump 1 and pump 2 on the bioreactor controller. Install a 0.13 in x 0.25 in silicone tube on each pump separately, with one end dipping into a bottle of water and the other end of the tube inserted into a measuring cylinder.
- Set the pump rotation speed to 300 rpm, and time for 1 min. Note the amount of water dispensed into the measuring cylinder for both pumps. These numbers will be needed for the subsequent steps.
- Prepare 500 mL of SF hMSC culture medium as per the manufacturer's instructions, and replace the cap of the cell culture medium bottle with a disposable C-Flex EZ Top container closure and compatible cap. Carry out the cap replacement inside a biosafety cabinet (BSC).
- Weld the outlet tube on the bottle of medium to the inlet C-flex tube on the bottle of 10 g of pre-sterilized W01 microcarriers for MSC. Install a section of the tube onto pump 1 of the bioreactor controller, set the pump rotation to 300 rpm, and pump in all the culture medium from the bottle into the bottle of microcarriers. Store these microcarriers at 4 °C until use on day 0.
NOTE: The microcarriers should be prepared on day −1. For 15 L bioreactor culture, prepare four bottles (i.e., 40 g, with 4 x 500 mL of SF medium) in total.
3. Cell seeding, culture, and harvest in a 5 L stirred-tank bioreactor
- On day 0, perform a one-point calibration, specifically 100%, for the DO probe on the bioreactor controller as per the manufacturer's instructions. The stirred-tank bioreactor is now ready for cell culture.
- Prepare a 2 L (5 L) glass bottle for waste collection, fit it with a GL 45 Multiport Connector Screw Cap with two ports, with 30 mm, 0.25 in x 0.44 in C-Flex tubes connected to both ports, and autoclave to sterilize. Weld the C-flex tube from one of the ports from the waste bottle to the harvest tube on the headplate of the bioreactor vessel with a tube welder.
- Stop the temperature, pH, and DO control on the bioreactor controller temporarily, install the silicone tube attached to the harvest tube onto peristaltic pump 2 in the correct liquid flow direction, and fully dispense all the PBS from the vessel into the waste bottle. Dislodge the tube from pump, and seal and disconnect the waste bottle.
NOTE: Always unclamp any Robert clamps on the flexible tubes involved in liquid flow in each step. Clamp back after the liquid transfer is completed before sealing and disconnecting. Clamp nearer toward the headplate side. Unclamping and clamping should be performed for all the subsequent steps unless otherwise stated. - Prepare 7 L (30 L) of complete SF hMSC medium, and replace each original bottle cap with a disposable C-Flex EZ Top container closure and compatible cap. Weld a Single-Use Filtration Module to one of the ports on the 10 L (50 L) Single-Use Storage Bag. Perform the operation of replacing the caps inside a BSC.
- Filter and transfer the culture medium to the storage bag, one bottle at a time, by welding the bottles of cell culture medium to the other end of the capsule filter and using the pump on the bioreactor controller. Designate as the feed bag.
- Weld one outlet port from the feed bag to the C-flex tube on the feed tube of the headplate, and install the tube to pump 1 on the bioreactor controller. Weld the other outlet port from the feed bag to the bleed tube, and install the tube to pump 2 on the bioreactor controller in the correct direction of liquid flow.
NOTE: Install the section on the fluid flow line, which is made of silicone tubing, on the pump for better resistance to wear and tear as compared to C-Flex tubes. Ensure the tubes are installed in the correct direction. - Set the pump 1 speed to 300 rpm, and stop the pump after dispensing 1 L (5 L) of medium from the feed bag into the vessel, based on the volume dispensing speed noted in step 2.17. Start the temperature, pH, and DO control again, with the same settings as described in step 2.14.
- Seal and disconnect the tube connecting the feed bag to the feed tube on the headplate. Weld the C-Flex tube from the bottle of the dispersed microcarriers prepared in step 2.18 to the feed tube, and pump all the content from the bottle into the vessel. Seal and disconnect the empty bottle of microcarrier suspension. Disinfect the entire surface of the bottle with 75% ethanol, and place in a BSC to use as a cell suspension transfer bottle.
NOTE: Leave a longer piece of C-Flex tube on the headplate side when sealing and disconnecting the disposable accessories, as multiple welding and disconnecting steps will be performed on the same tube in the following steps. - Ensure that the temperature shown on the controller reaches a steady state of 37 °C, which usually takes about 30 min, before proceeding to prepare the seed cells as in steps 1.3-1.4. Resuspend 2.5 x 108 hMSCs in 500 mL of SF medium, and transfer to the empty bottle previously containing the microcarrier suspension.
NOTE: The seed cells for the 15 L bioreactor expansion should be prepared according to step 3.21, with the aim of inoculating 1.0 x 109 cells in 500 mL. - Weld the C-Flex tube from the bottle now containing the cell suspension to the feed port on the headplate, and pump all of the contents from the bottle into the vessel to initiate cell inoculation to the microcarriers. Seal and disconnect the empty bottle. Re-weld the feed port from the feed bag to the feed tube on the headplate.
- Adjust the agitation settings on the controller to allow intermittent agitation for enhanced inoculation efficiency of hMSCs by setting the following parameters: V1 = 35 (30) rpm/5 min; V2 = 0 rpm/25 min; number of cycles = 12 (24); final agitation speed = 35 (30) rpm.
NOTE: The agitation speed may be adjusted to 40 (35) rpm or 45 (40) rpm if non-uniform distribution or sedimentation of the microcarriers are observed during the culture process. - Set up an automated medium supplement and exchange regime on the bioreactor controller in the Medium Exchange interface. Set the parameters for the hMSC cultivation as stated in Table 2.
NOTE: Unclamp any Robert clamps on the flexible tubes involved in liquid flow at this stage, especially for the feed and bleed tube. Keep these unclamped through the entire culture process. - Take the samples through the sampling tube by connecting the disposable sampling bags via Luer connectors at the desired time points, usually once every day, and aliquoting 10 mL each time into the sampling bag. Aliquot 2-3 mL of sample into the first sampling bag, discard, and then aliquot the actual 10 mL of sample into a new sampling bag each time.
NOTE: The initial 2-3 mL sample could be liquid left in the tube from the previous sampling time point and may not accurately represent the condition in the vessel. Always perform these steps with extra aseptic measures by sanitizing the Luer connectors before and after making or breaking connections with 75% ethanol. - Transfer the samples taken to a 15 mL conical centrifuge tube. Perform the following tests on the samples.
- Take 200 μL of the cell-laden microcarrier suspension, and stain with Calcein-AM/PI Cell Double Staining according to the manufacturer's instructions. Observe under a fluorescence microscope.
- Sediment the microcarriers by gravitation, which usually takes 5-10 min. Aspirate the supernatant, and test the glucose concentration with a glucose meter as per the manufacturer's instructions. Plot a glucose level graph.
- Incubate the sedimented cell-laden microcarriers with a freshly prepared 3-5 mL of 1 mg/mL digest solution at 37 °C for 20-30 min to fully dissolve the microcarriers. Count the cells after staining with AO/PI with an automated fluorescence cell analyzer. Plot a cell growth curve. Correlate with the real-time live cell growth curve generated on the controller.
- On day 4 or day 5 of culture, prepare 50 mL (200 mL) of digest solution at 30 mg/mL. The working mass ratio between the digest solution and dissolvable microcarriers ranges from 3:20 to 5:20. Sterile-filter with a 0.22 μm capsule filter, and further dilute in 500 mL (2 L) of Hank's balanced salt solution (HBSS) with calcium and magnesium. Transfer to a 3 L disposable storage bag.
NOTE: Only prepare the digest solution just before cell harvesting. Try to aim for at least 1.0 x 109-1.2 x 109 cells for the first-tier expansion and 1.0 x 1010 cells for the second-tier expansion. If the cells grow faster, harvest at day 4; if not, harvest at day 5. It is not advised to go beyond day 5. - Set agitation speed to 0 on the controller for the microcarriers to settle, which usually happens within 20 min. Turn off the automated medium exchange protocol, and stop the temperature, pH, and DO control on the bioreactor controller. Manually start pump 2 at 300 rpm on the controller, and dispense supernatant into the feed regime to leave about 1 L (2 L) of culture suspension in the vessel (using the volume dispensing speed noted in step 2.17 or gauging from the graduation marks on the body of the vessel). Seal and totally disconnect the feed bag.
- Weld the bag of freshly prepared digest solution from step 3.15 to the feed tube, and pump all the contents into the vessel. Start the agitation speed at 45 (40) rpm. Ensure that the temperature control is still maintaining the temperature at 37 °C. Microcarriers will dissolve within 40-60 min. Take 1 mL of the sample with a sterile Luer lock syringe at 30 min and subsequently every 5-10 min to observe under a bright-field microscope to check the dissolution of the microcarriers.
- After the microcarriers have dissolved, weld one 3 L storage bag (two 3 L storage bags for the 15 L bioreactor) to the harvest tube, and pump out the cell suspension completely using pump 2 at 300 rpm. Prepare 1 L (3.5 L) of saline containing 1% human serum albumin (HSA) as a wash buffer and 500 mL of complete SF medium (500 mL of cell cryopreservation buffer, a formulation of 10% dimethyl sulfate (DMSO) and 90% SF medium is taken as an example) in separate disposable storage bags using the methods and accessories mentioned in previous steps for other liquid transfers.
- Turn on the cell processing system, install a Disposable Cell Processing Kit to the instrument according to the manufacturer's instructions, weld the bags of cell suspension, wash buffer, and SF medium (cryopreservation buffer) to the Disposable Cell Processing Kit, and carefully follow all the instructions displayed on the user interface to initialize and check the integrity and proper installation of the kit.
- Once the installation is complete, enter the parameters listed in Table 3 for cell washing with wash buffer and resuspension in SF medium (cryopreservation buffer) to the software to start the wash and resuspension process. The whole procedure will be performed automatically with several manual check points.
- Take a sample of the cells from the centrifuge chamber, as indicated by the cell processing system, and calculate the cell density with an automated cell analyzer according to the manufacturer's instructions. Adjust the resuspension volume for a density of 2 x 106 cells/mL or a maximum total volume of 250 mL.
- Seal and disconnect the cell transfer bag from the Disposable Cell Processing Kit after the system has aspirated the cells out from the chamber. Readjust the cell density to 2 x 106 cells/mL with complete SF medium in a larger cell transfer bag/medium storage bag if necessary. These will be the seed cells for the second-tier expansion in the 15 L bioreactor.
4. Cell formulation, fill, and finish
- Start preparing the 15 L bioreactor 1 day before cell harvest from the 5 L bioreactor. Follow the steps in protocol section 2 and 3 completely except with the parameters for 15 L indicated in brackets.
- Resuspend the cells in 250 mL of cryopreservation buffer, and seal and disconnect the cell transfer bag from the Disposable Cell Processing Kit after the system has aspirated the cells out from the chamber.
- Transfer 2,000 mL of cryopreservation buffer together with the 250 mL of resuspended cells from step 4.2 to a 3 L cell transfer bag. Weld this transfer bag to a Disposable Fill&Finish Consumable Kit.
NOTE: The additional volume of cryopreservation buffer used here should be determined by the specification of formulation and total cell number calculation as per the counting results gained in step 3.21 - Assemble the Disposable Fill&Finish Consumable Kit to the cell filling system, and turn on the pre-cooling system according to the manufacturer's instructions. Follow the instructions on the system, and set to fill 20 mL/bag with a total of 20 bags per batch. Seal and disconnect these bags from the kit, and follow standard cryopreservation procedures. Use cryostorage bags.
- Weld a new set of 20 cryopreservation bags to the Disposable Fill&Finish Consumable Kit, and repeat the fill and finish procedure until all the cells are aliquoted. Follow standard procedures to cryopreserve these cells.
5. Characterization of the cell quality
- Sample the harvested hMSCs from the dissolvable microcarriers, and use a cell processing system for characterization by flow cytometry for the determination of the cell identity following the methods described in literature18.
- Thaw the cryopreserved cells, and replate on conventional 2D flasks or microplates for further characterization of the cell features, including the cell morphology and tri-lineage differentiation capability, following methods described in literature18.
6. HEK293T expansion on G02 microcarriers in the stirred-tank bioreactor
- Prepare a 5 L bioreactor as mentioned in step 2. For HEK293T cells, culture the cells with sterile G02 microcarriers instead of W01. The culture process is similar to in protocol section 3, although some parameters are different, as will be detailed in the following steps. Install a special perfusion tube in the bioreactor in replacement of the bleed tube, as perfusion is needed for the culture of HEK293T cells.
- Different from the protocol detailed in protocol section 3, prepare 20 g of G02 microcarriers (i.e., two bottles) in DMEM with 10% FBS for the 5 L bioreactor, with the day 0 culture volume set at 3.5 L (i.e., a final concentration of about 5.71 g/L). Dispense 2 L of medium into the vessel before the two bottles of G02 microcarriers, and inoculate 500 mL of 1.75 x 109 HEK293T cells (i.e., at a final density of 5 x 105 cells/mL).
- Perform an agitation regime and medium exchange regime (based on the amount of medium required to be exchanged per day, expressed as culture volume per day, CVD), which is different from protocol section 3 and is detailed in Table 4.
NOTE: Activate pump 1 and pump 2 manually so that both can pump continuously for perfusion to occur. Adjust the pump speed every day to meet the perfusion rate based on the calibration of the pumps' volume dispensing speed. Usually, this will be in the single digit range. For HEK293T cells, use fresh medium as feed, and discard spent medium totally into a waste bottle, unlike with hMSCs, for which the medium is circulated. - For the second tier expansion to the 15 L bioreactor, use bead-to-bead transfer of the HEK293T cells instead of full dissolution of the microcarriers as used with hMSCs. Specifically, stop the agitation to sediment the microcarriers after desired cell density is reached, and then aspirate as much supernatant as possible out of the perfusion tube, feed PBS with 3 times the volume of remaining suspension in to wash the microcarriers, and repeat three times. Aspirate and discard as much PBS as possible in the final wash.
- Feed in 3.5 L of 1x recombinant trypsin to detach the cells from the microcarriers while the G02 microcarriers remain intact. Set the temperature to 37 °C and the agitation speed to 60 rpm. Terminate the detachment within 45 min, when more than 80% of the cells have detached, with an equal amount of complete medium.
- Transfer directly from the harvest port to a prepared 15 L vessel for second-tier expansion.
7. Vero cell expansion on V01 microcarriers in the stirred-tank bioreactor
- Prepare V01 microcarriers by transferring the lyophilized microcarriers into the bioreactor vessel together with PBS to a concentration of 20 mg/mL. Assemble and sterilize the vessel as mentioned in protocol section 2, but autoclave for 30 min instead.
- After sterilization, V01 microcarriers naturally settle down. Exchange the supernatant with basic DMEM two times using the bleed and feed tubes, and add the complete DMEM containing 10% NBS to 50%-75% of the volume of the vessel before proceeding to protocol section 3 for cell inoculation and culture.
- Different from the protocol detailed in protocol section 3, use 24 g of V01 microcarriers for a 4 L culture of Vero cells (i.e., a final concentration of 6 g/L), and inoculate 500 mL of 4 x 109 Vero cells (i.e., at a final density of 1 x 106 cells/mL).
- Follow steps 6.3-6.6 for the culture process parameters and passaging to second-tier expansion in the 15 L stirred-tank bioreactor.
Representative Results
The ACISCP platform is a fully closed system that employs a series of stirred-tank bioreactors for scale-up expansion, a cell processing system for automated cell harvesting and formulation, and a cell filling system (Figure 1). Adherent cells attach to the porous microcarriers, which can be dispersed into the bioreactor, thus achieving the suspended cultivation of adherent cells.
Following the protocol as described, 2.5 x 108 hMSCs with 10 g of W01 microcarriers were firstly inoculated in a 5 L stirred-tank bioreactor for an initial expansion. Around 2.9 x 109 cells were concentrated and washed by the cell processing system on day 4, out of which 1.0 x 109 harvested cells were passaged to the 15 L stirred-tank bioreactor with 40 g of W01 microcarriers for the second phase of cultivation. Eventually, 1.1 x 1010 MSCs were harvested, formulated, and aliquoted into over 70 formulation bags on day 9 (Figure 2A). The cell number, viability, and glucose consumption were monitored daily (Figure 2B,C). A cumulative 128-fold expansion was achieved; meanwhile, the cell viability was kept higher than 90%. The intake of glucose exhibited a negative correlation with cell expansion, showing metabolism associated with healthy cell growth.
The dissolvable porous microcarriers can be pre-sterilized and come in a single-use closed system packaging, suitable for GMP (Figure 3A). The cells could attach to the surface of the microcarriers and the inner wall of the macropores (Figure 3B). By using a fluorescent dye, such as Calcein-AM/PI Cell Double Staining Kit, the cells inside the porous microspheres could be visualized (Figure 3C). Merging bright-field images with fluorescent images helped to analyze the uniformity of the cell distribution. The live cell staining also showed a strong association with cell counting measured by either manual sampling or the online live cell counting probe.
In addition to the tremendous demand for high cell quantities, critical quality attribute (CQA) assessments and release tests of MSCs are indispensable, as without these, cells could not be sterilized or extensively purified post-culturing. To assure the cell quality, 3D-expanded MSCs can be sampled at each stage of the ACISCP platform. MSCs freshly harvested from the ACISCP platform retained classical phenotypic markers22, with an expression of >95% and <2% for the positive markers (CD73, CD90, CD105) and negative markers (HLA-DR, CD34, CD45, CD19, CD14), respectively (Figure 4A). The cryopreserved cells were thawed and inoculated to a 2D planar flask and showed good adhesion ability, normal expansion behavior (Figure 4B), and a typical spindle shape with spiral growth (Figure 4C). Furthermore, the cells also maintained their capability to differentiate into osteogenic, adipogenic, and chondrogenic lineages (Figure 4D).
Moreover, HEK293T cells and Vero cells were tested in the ACISCP platform as well. For HEK293T cell culture, the inoculum cell concentration was 5 x 105 cells/mL in the 5 L bioreactor. After the bead-to-bead transfer process, the peak cell concentration reached 1.68 x 107 cells/mL in the 15 L bioreactor (Figure 5A), and the HEK293T cells maintained high viability (Figure 5B). For the Vero cell culture, the inoculum cell concentration was 2 x 105 cells/mL in the 5 L bioreactor. After the bead-to-bead transfer process, the peak cell concentration achieved was 1.08 x 107 cells/mL in the 15 L bioreactor (Figure 6A), and the Vero cells maintained high viability as well (Figure 6B).
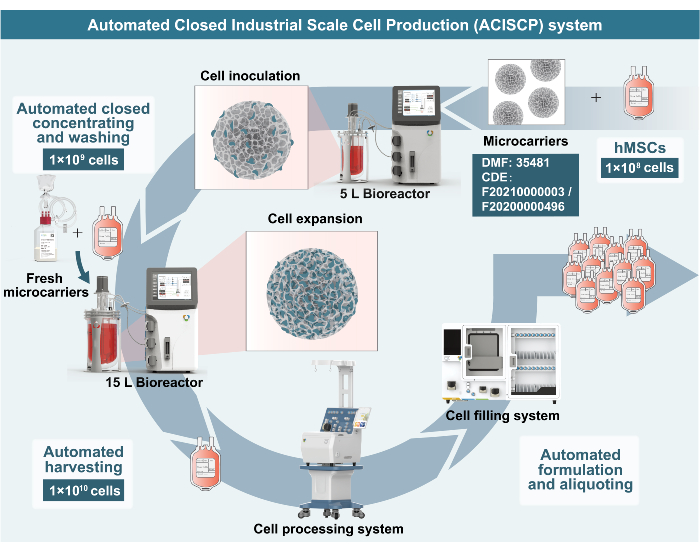
Figure 1: Schematic roadmap of hMSC production via the ACISCP platform. This figure has been modified from published literature18. Please click here to view a larger version of this figure.
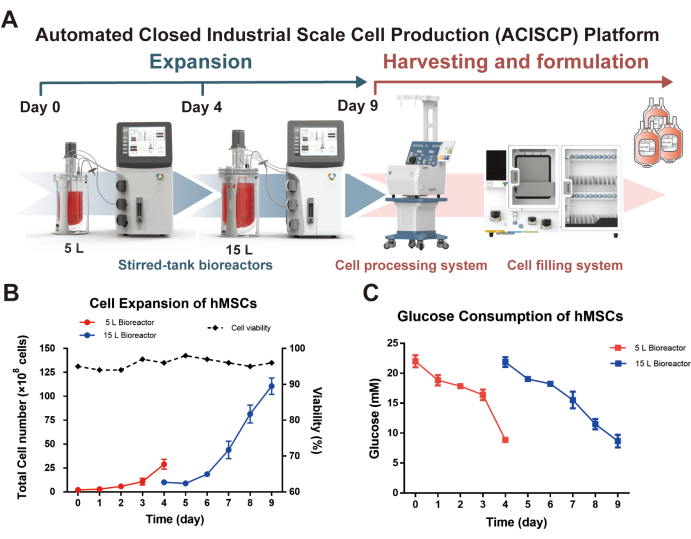
Figure 2: Cell expansion of hMSCs via the ACISCP platform. (A) The ACISCP platform consists of a series of stirred-tank bioreactors, a cell processing system, and a cell filling system. (B) Growth curve of hMSCs in the two-tiered expansion process, (C) and the glucose consumption rate during the process. Please click here to view a larger version of this figure.
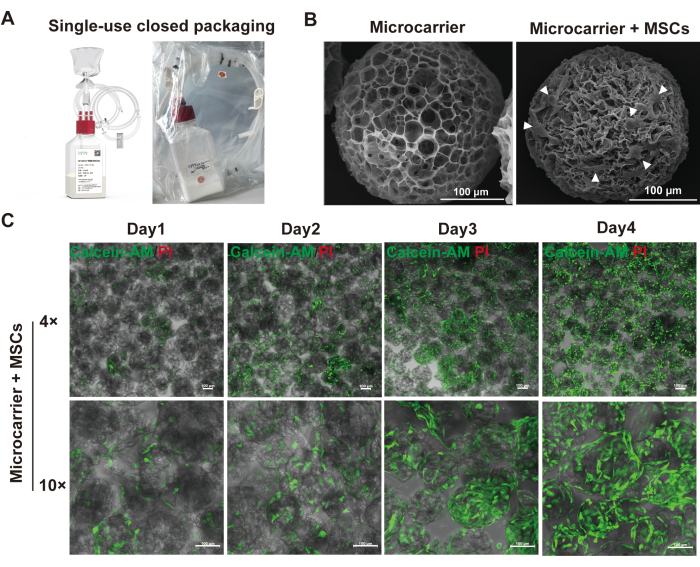
Figure 3: Characterization of hMSC growth on the W01 microcarrier. This figure has been modified from published literature18. (A) W01 microcarriers are prepared with single-use closed-system packaging. (B) Scanning electron microscope images of empty microcarriers and microcarriers cultured with adherent hMSCs. The white triangular heads indicate cells on the surface of microcarriers; scale bar = 100 µm. (C) Representative merged bright-field and fluorescence (live cell staining) images of hMSCs cultured on microcarriers from day 1 to day 4; scale bar = 100 µm. Please click here to view a larger version of this figure.
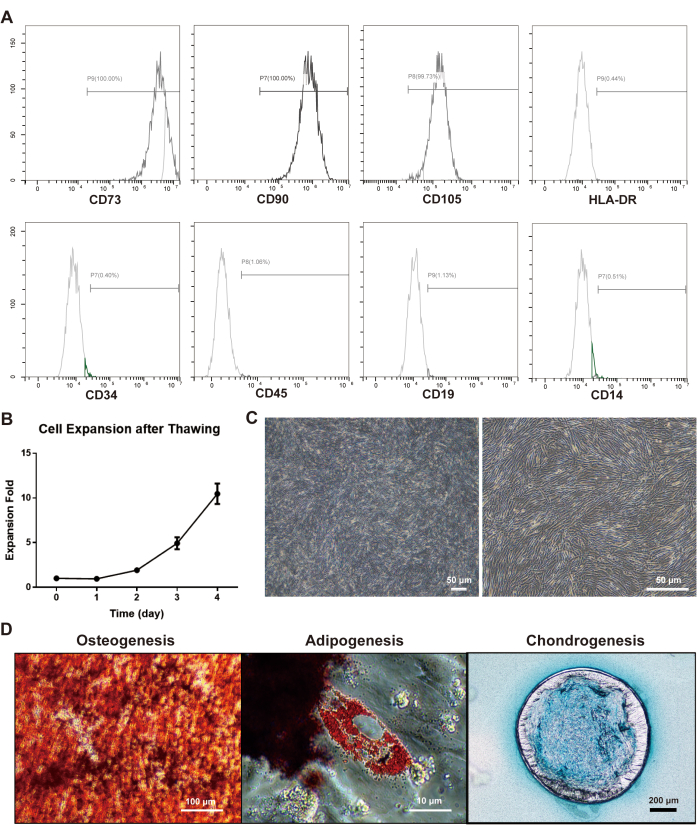
Figure 4: Cell identity assessment of hMSCs harvested from the ACISCP platform. (A) The surface markers of hMSCs were characterized by flow cytometry. All the positive markers (>95%) and negative markers (<2%) met the criteria. (B) Cryopreserved 3D-expanded hMSCs exhibited normal expansion behavior after thawing and re-plating to 2D flasks. (C) Morphology of the 3D-expanded hMSCs; scale bar = 50 µm. (D) Tri-lineage differentiation of the 3D-expanded hMSCs. Osteogenic, adipogenic, and chondrogenic capabilities were assessed by Alizarin Red, Oil Red, and Alcian Blue staining, respectively. Please click here to view a larger version of this figure.
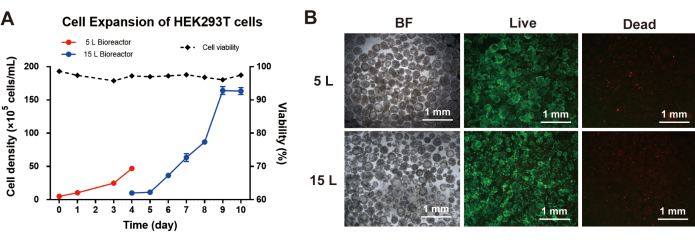
Figure 5: Cell expansion of HEK293T cells via the ACISCP platform. (A) HEK293T cell density in the 5 L and 15 L bioreactors. (B) Bright-field (BF) image, Calcein-AM-stained (Live), and PI-stained (Dead) HEK293T cells cultured on G02 microcarriers; scale bar = 1 mm. Please click here to view a larger version of this figure.
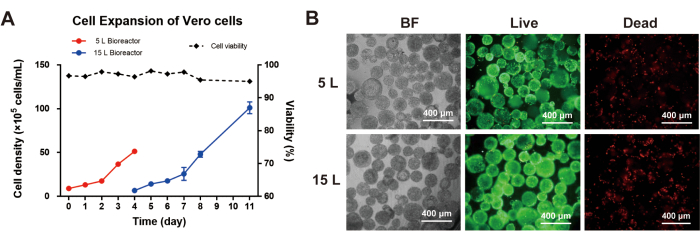
Figure 6: Cell expansion of Vero cells via the ACISCP platform. (A) Vero cell density in the 5 L and 15 L bioreactors. (B) Bright-field (BF) image, Calcein-AM-stained (Live), and PI-stained (Dead) Vero cells cultured on V01 microcarriers; scale bar = 400 µm. Please click here to view a larger version of this figure.
Table 1: Parameter settings of the stirred-tank bioreactor. Please click here to download this Table.
Table 2: Parameter settings of automated medium exchange for hMSC cultivation. Please click here to download this Table.
Table 3: Parameter settings of the cell processing system for hMSC passage and formulation. Please click here to download this Table.
Table 4: Medium exchange regime and agitation regime for HEK293T and Vero cells. Please click here to download this Table.
Discussion
Both immunotherapy and stem cell therapy utilize live cells as drugs; however, their final products should not be purified or sterilized in the same way as small molecules or viruses. Therefore, the principle of Quality by Design (QbD) should always be kept in mind and practically applied to the Chemical Manufacturing and Control (CMC) process during cell production23. A fully closed cell culture system, as well as a processing system and a filling system, are preferentially considered to meet the requirements. In this study, we have presented a protocol for utilizing an ACISCP platform based on GMP-grade, dissolvable, and porous 3D microcarriers to perform a two-tiered expansion of hMSCs. Some researchers have explored the feasibility of using bioreactors together with microcarriers to expand hMSCs in vitro, but the majority of reported studies have shown that an increase in scale would lead to a decrease in hMSC yield, from 6.1 x 105 cells/mL or even 12.5 x 105 cells/mL in 100 mL of working volume to 2.7 x 105 cells/mL in 2 L of working volume24,25,26. Even though stirred-tank bioreactors exhibit the highest scalability, key issues such as the structural characteristics of the vessel, the impeller type, the tip speed, the volumetric mass transfer coefficient, kLa, of oxygen, and the power number, should be thoroughly considered24. Distinct from previous stirred-tank bioreactors, which were usually designed for the cultivation of microbes or domesticated anchorage-independent cells like CHO cells, the bioreactor used in this study was specifically designed for microcarrier-based culture and is, thus, able to satisfy the different process needs of nutrient exchange strategies. In addition to differences in the process of cell cultivation, the ACISCP platform employs a continuous flow centrifuge-based cell processing system together with a cell filling system, in contrast to conventional harvesting procedures for producing large amounts of cells performed by repeated manual work, thus guaranteeing a high handling efficacy. Advanced automated devices have a high yield in one-batch production.
Most stem cells are anchorage-dependent; thus, they demand microcarriers for cell manufacturing. Conventionally, previous commercial microcarriers were formulated to leverage different surface characteristics, thus achieving different cell expansion fold numbers. In previous research, the use of Cytodex type 1 microcarriers for porcine bone marrow MSCs produced a cell density of about 4 x 105 cells/mL, while the use of gelatin-coated Cytodex type 3 produced comparable cell numbers (3.8 x 105 cells/mL) to human placental MSCs27. Nevertheless, Cytodex type 1 and type 3 are non-dissolvable, and, thus, the trypsinization process during cell harvesting impacts not only the cell recovery yield but also the viability and quality. Hence, there are no successful cases of using conventional microcarriers for CGT products, where cells as the final products cannot undergo extensive downstream purification processes to eliminate the contamination of non-dissolvable microcarriers28. By contrast, W01 microcarriers are fully dissolvable, meaning that the cell products can be easily harvested by degradation of the microcarrier. This microcarrier product also comes in fully closed system packaging, meaning an operation process compliant with GMP could be achieved easily. We have shown that passaging with a two-tiered expansion process can be easily achieved with W01 microcarriers, and as high as 1.1 x 1010 total cells can be harvested in a single batch; indeed, this could only be achieved in 50 L bioreactors in previous reports29,30. In this work, the peak cell density inside the stirred-tank bioreactor reached approximately 11.6 x 105 cells/mL before harvest. Thus, it could be expected that one could easily achieve 1 x 1011 cells per batch with a 100-200 L bioreactor in the ACISCP platform, which would mean that thousand batches per year could easily meet the demand for 300 trillion hMSCs each year.
Since cells have varied characteristics, different types of 3D microcarriers are produced and are available for testing the compatibility between a specific cell and a microcarrier. For non-cell end products in CGT, microcarrier-based scale-ups for HEK293T cells and Vero cells in bioreactors have been reported to reach 1.5 x 106 cells/mL and 3.3 x 106 cells/mL, respectively31. We used dissolvable porous microcarriers, G02 and V01, in the ACISCP platform to expand HEK293T cells and Vero cells, respectively, and the peak cell densities of both cells reached above 1 x 107 cells/mL. Furthermore, the degradability of 3D microcarriers can be beneficial for the production of biological products of intracellular viruses, as the cells could be easily separated from the microcarriers. Critically, the ACISCP platform represents an established new biomanufacturing process that meets both the quantity and quality requirements for cell and gene therapy products. We anticipate that our large-scale cell production platform will act as an essential tool to enable the development of cell and gene therapies.
Despite the advantages of the platform discussed above, several constraints remain to be circumvented in the future. Firstly, the bioreactors employed in the ACISCP platform in the current form are still equipped with glass tanks, which require complicated washing procedures and cleaning verification to meet the criteria of GMP. A single-use stirred-tank bioreactor system could be potentially employed in the future to produce an intact set of single-use consumable kits. Secondly, different from domesticated cell lines, hMSCs are primary cell strains isolated from individual donors and varied tissues, which means hMSCs exhibit heterogeneity. Therefore, the protocol described in this paper serves as a reference, while systematic process developments are required to achieve the technical migration of the ACISCP platform. Thirdly, even though our co-workers have preliminarily tested the feasibility of producing the vaccinia virus with Vero cells32, whether good performance for viral production in the 3D expansion of HEK293T cells could be achieved remains to be further validated. In conclusion, the large-scale cell production method of the ACISCP platform, based on dissolvable porous microcarriers and a series of automated closed systems, offers an opportunity to enhance production efficiency and refine quality control at an industrial scale for the manufacturing of CGT products.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was financially supported by the National Science Foundation for Distinguished Young Scholars (82125018).
Materials
| 0.25% trypsin EDTA | BasalMedia | S310JV | Used for 2D cell harvest. |
| 3D FloTrix Digest | CytoNiche Biotech | R001-500 | This is a reagent that specifically dissolves 3D TableTrix microcarriers. |
| 3D FloTrix MSC Serum Free Medium | CytoNiche Biotech | RMZ112 | This is a serum-free,animal-free medium for mesenchymal stem cell expansion and maintenance in 2D planar culture as well as 3D culture on 3D TableTrix microcarriers. |
| 3D FloTrix Single-Use Filtration Module | CytoNiche Biotech | R020-00-10 | This module contains 0.22 μm capsule filters used for filtration of culture medium and digest solution. |
| 3D FloTrix Single-Use Storage Bag (10 L) | CytoNiche Biotech | R020-00-03 | Used as feed bag for 5 L bioreactor. |
| 3D FloTrix Single-Use Storage Bag (3 L) | CytoNiche Biotech | R020-00-01 | Used as cell seeding or transfer bags. |
| 3D FloTrix Single-Use Storage Bag (50 L) | CytoNiche Biotech | R020-00-04 | Used as feed bag for 15 L bioreactor. |
| 3D FloTrix vivaPACK Disposable Fill&Finish Consumable Kit | CytoNiche Biotech | PACK-01-01 | This is a standard kit adapted to 3D vivaPACK fill and finish system. |
| 3D FloTrix vivaPACK fill and finish system for cells | CytoNiche Biotech | vivaPACK | This system is a closed liquid handling device, with automated mixing and gas exhausting functions. Cells resuspended in cryopreservation buffer can be rapidly and evenly aliquoted into 20 bags per batch. |
| 3D FloTrix vivaPREP PLUS cell processing system | CytoNiche Biotech | vivaPREP PLUS | This system is a continuous flow centrifuge-based device.Cells can be concentrated, washed, and resuspended under completely closed procedures. |
| 3D FloTrix vivaPREP PLUS Disposable Cell Processing Kit | CytoNiche Biotech | PREP-PLUS-00 | This is a standard kit adapted to 3D vivaPREP PLUS cell processing. |
| 3D FloTrix vivaSPIN bioreactor 15 L | CytoNiche Biotech | FTVS15 | This bioreactor product employs a controller, a 15 L glass stirred-tank vessel, and assessories. A special perfusion tube is available. |
| 3D FloTrix vivaSPIN bioreactor 5 L | CytoNiche Biotech | FTVS05 | This bioreactor product employs a controller, a 5 L glass stirred-tank vessel, and assessories.A special perfusion tube is available. |
| 3D FloTrix vivaSPIN Closed System Consumable Pack (10/15 L) | CytoNiche Biotech | R020-10-10 | This is a standard tubing kit adapted to 3D vivaSPIN bioreactor 15 L, containing sampling bags. |
| 3D FloTrix vivaSPIN Closed System Consumable Pack (2/5 L) | CytoNiche Biotech | R020-05-10 | This is a standard tubing kit adapted to 3D vivaSPIN bioreactor 5 L, containing sampling bags. |
| 3D TableTrix microcarriers G02 | CytoNiche Biotech | G02-10-10g | These porous and degradable microcarriers are suitable for HEK293T cell culture. They come pre-sterilized in 10g/bottle with C-Flex tubings for welding to tubes on bioreactors. |
| 3D TableTrix microcarriers V01 | CytoNiche Biotech | V01-100-10g | These porous and degradable microcarriers are suitable for adherent cell culture, they come as non-sterilized microcarriers that need to be autoclaved in PBS before use. They are especially suitable for vaccine production. |
| 3D TableTrix microcarriers W01 | CytoNiche Biotech | W01-10-10g (single-use packaging); W01-200 (tablets) |
These porous and degradable microcarriers are suitable for adherent cell culture, especially for cells that need to be harvested as end products. They come pre-sterilized in 10g/bottle with C-Flex tubings for welding to tubes on bioreactors.The product has obtained 2 qualifications for pharmaceutical excipients from CDE, with the registration numbers of [F20200000496; F20210000003]. It has also received DMF qualification for pharmaceutical excipients from FDA, with the registration number of [DMF:35481] |
| APC anti-human CD45 Antibody | BioLegend | 368512 | Used in flow cytometry for MSC identity assessment |
| Calcein-AM/PI Double Staining Kit | Dojindo | C542 | Calcein-AM/PI Double Staining Kit is utilized for simultaneous fluorescence staining of viable and dead cells. This kit contains Calcein-AM and Propidium Iodide (PI) solutions, which stain viable and dead cells, respectively. |
| Cap for EZ Top Container Closures for NALGENE-containers (500mL) | Saint-Gobain | CAP-38 | Brands and catalogue numbers are only for example, similar products are available from various suppliers and as long as they have the same functionality, items could be substituted with other brands. |
| C-Flex Tubing, Formulation 374 (0.25 in x 0.44 in) | Saint-Gobain | 374-250-3 | Used for tube welding and disconnection. |
| CryoMACS Freezing Bag 50 | Miltenyi Biotec | 200-074-400 | Used for expanding the 3D FloTrix vivaPACK Disposable Fill&Finish Consumable Kit. |
| Dimethyl Sulfate (DMSO) | Sigma | D2650-100mL | Used for preparation of cryopreservation solution. |
| Dulbecco's Modified Eagle Medium (DMEM) | BasalMedia | L120KJ | Used for cultivation of HEK293T and Vero cells. |
| DURAN Original GL 45 Laboratory bottle (2 L) | DWK life sciences | 218016357 | Used for waste collection from the 5 L bioreactor. |
| DURAN Original GL 45 Laboratory bottle (5 L) | DWK life sciences | 218017353 | Used for waste collection from the 15 L bioreactor. |
| DURAN Original GL 45 Laboratory bottle (500 mL) | DWK life sciences | 218014459 | Used for supplementary bottle of 0.1 M NaOH. |
| EZ Top Container Closures for NALGENE-containers (500mL) | Saint-Gobain | EZ500 ML-38-2 | Brands and catalogue numbers are only for example, similar products are available from various suppliers and as long as they have the same functionality, items could be substituted with other brands. |
| Fetal bovine serum (FBS) superior quality | Wisent | 086-150 | Used for cultivation of HEK293T cells. |
| FITC anti-human CD14 Antibody | BioLegend | 301804 | Used in flow cytometry for MSC identity assessment. |
| FITC anti-human CD34 Antibody | BioLegend | 343504 | Used in flow cytometry for MSC identity assessment. |
| FITC anti-human CD90 (Thy1) Antibody | BioLegend | 328108 | Used in flow cytometry for MSC identity assessment. |
| Flow cytometry | Beckman Coulter | CytoFLEX | Used for cell identity assessment. |
| Fluorescence Cell Analyzer | Alit life science | Countstar Rigel S2 | Used for cell counting. Cell viability can be calculated by staining with AO/PI dyes. |
| GL 45 Multiport Connector Screw Cap with 2 ports | DWK life sciences | 292632806 | Brands and catalogue numbers are only for example, similar products are available from various suppliers and as long as they have the same functionality, items could be substituted with other brands. |
| Glucose Meter | Sinocare | 6243578 | Used for detecting glucose concentration in cell culture medium and supernatant. |
| Hank's Balanced Salt Solution (HBSS), with calcium and magnesium | Gibco | 14025092 | Used for preparation of digest solution. |
| Human Albumin 20% Behring (HSA) | CSL Behring | N/A | Used for preparation of wash buffer. |
| Inverted fluorescent microscope | OLYMBUS | CKX53SF | Used for brifgt field and fluorescent observation and imaging. |
| Nalgene Measuring Cylinder (500 mL) | Thermo Scientific | 3662-0500PK | Used for calibrating the liquid handling volume speed of peristaltic pumps. |
| Newborn calf serum (NBS) superfine | MINHAI BIO | SC101.02 | Used for cultivation of Vero cells. |
| OriCell human mesenchymal stem cell adipogenic differentiation and characterization kit | Cyagen | HUXUC-90031 | Used for tri-lineage differentiation of hUCMSCs. |
| OriCell human mesenchymal stem cell chondrogenic differentiation and characterization kit | Cyagen | HUXUC-90041 | Used for tri-lineage differentiation of hUCMSCs. |
| OriCell human mesenchymal stem cell osteogenic differentiation and characterization kit | Cyagen | HUXUC-90021 | Used for tri-lineage differentiation of hUCMSCs. |
| PE anti-human CD105 Antibody | BioLegend | 800504 | Used in flow cytometry for MSC identity assessment. |
| PE anti-human CD19 Antibody | BioLegend | 302208 | Used in flow cytometry for MSC identity assessment. |
| PE anti-human CD73 (Ecto-5'-nucleotidase) Antibody | BioLegend | 344004 | Used in flow cytometry for MSC identity assessment. |
| PE anti-human HLA-DR Antibody | BioLegend | 307605 | Used in flow cytometry for MSC identity assessment. |
| Phosphate Buffered Saline (PBS) | Wisent | 311-010-CL | Used in autoclaving of glass vessel and V01 microcarriers, and replacement of culture medium. |
| Sani-Tech Platinum Cured Sanitary Silicone Tubing (0.13 in x 0.25 in) | Saint-Gobain | ULTRA-C-125-2F | Used for solution transfering driven by peristaltic pumps. |
| Sterile Saline | Hopebiol | HBPP008-500 | Used for preparation of wash buffer. |
| Trypzyme Recombinant Trypsin | BasalMedia | S342JV | This reagent is used for bead-to-bead transfer of HEK293T and Vero cells. |
| Tube Sealer | Yingqi Biotech | Tube Sealer I | This sealer is compatible with both C-Flex tubing and PVC tubing. |
| Tube Welder for PVC tubing | Chu Biotech | Tube Welder Micro I | Used for welding of PVC tubing. |
| Tube Welder for TPE tubing | Yingqi Biotech | Tube Welder I-V2 | Used for welding of TPE tubing. |
| ViaStain AO / PI Viability Stains | Nexcelom | CS2-0106-25mL | Dual-Fluorescence Viability, using acridine orange (AO) and propidium iodide (PI), is the recommended method for accurate viability analysis of primary cells, such as PBMCs, and stem cells in samples containing debris. |
References
- Golchin, A., Farahany, T. Z. Biological products: Cellular therapy and FDA approved products. Stem Cell Reviews and Reports. 15 (2), 166-175 (2019).
- Young, C. M., Quinn, C., Trusheim, M. R. Durable cell and gene therapy potential patient and financial impact: US projections of product approvals, patients treated, and product revenues. Drug Discovery Today. 27 (1), 17-30 (2022).
- Elverum, K., Whitman, M. Delivering cellular and gene therapies to patients: solutions for realizing the potential of the next generation of medicine. Gene Therapy. 27 (12), 537-544 (2020).
- Blache, U., Popp, G., Dünkel, A., Koehl, U., Fricke, S. Potential solutions for manufacture of CAR T cells in cancer immunotherapy. Nature Communications. 13 (1), 5225 (2022).
- Lee, B., et al. Cell culture process scale-up challenges for commercial-scale manufacturing of allogeneic pluripotent stem cell products. Bioengineering. 9 (3), 92 (2022).
- Emerson, J., Glassey, J. Bioprocess monitoring and control: Challenges in cell and gene therapy. Current Opinion in Chemical Engineering. 34, 100722 (2021).
- Robb, K. P., Fitzgerald, J. C., Barry, F., Viswanathan, S. Mesenchymal stromal cell therapy: progress in manufacturing and assessments of potency. Cytotherapy. 21 (3), 289-306 (2019).
- Olsen, T. R., Ng, K. S., Lock, L. T., Ahsan, T., Rowley, J. A. Peak MSC-Are we there yet. Frontiers in Medicine. 5, 178 (2018).
- . Roots Analysis. Stem Cell Therapy Contract Manufacturing (CMO) Market, 2019 – 2030 Available from: https://www.rootsanalysis.com/reports/view_document/stem-cell-therapy-contract-manufacturing-market-2019-2030/271.html (2019)
- Elseberg, C. L., et al. Microcarrier-based expansion process for hMSCs with high vitality and undifferentiated characteristics. The International Journal of Artificial Organs. 35 (2), 93-107 (2012).
- de Soure, A. M., Fernandes-Platzgummer, A., Silva, d. a., L, C., Cabral, J. M. Scalable microcarrier-based manufacturing of mesenchymal stem/stromal cells. Journal of Biotechnology. 236, 88-109 (2016).
- Rafiq, Q. A., Coopman, K., Nienow, A. W., Hewitt, C. J. Systematic microcarrier screening and agitated culture conditions improves human mesenchymal stem cell yield in bioreactors. Biotechnology Journal. 11 (4), 473-486 (2016).
- Tavassoli, H., et al. Large-scale production of stem cells utilizing microcarriers: A biomaterials engineering perspective from academic research to commercialized products. Biomaterials. 181, 333-346 (2018).
- Mizukami, A., et al. Technologies for large-scale umbilical cord-derived MSC expansion: Experimental performance and cost of goods analysis. Biochemical Engineering Journal. 135, 36-48 (2018).
- Yan, X., et al. Dispersible and dissolvable porous microcarrier tablets enable efficient large-scale human mesenchymal stem cell expansion. Tissue Engineering. Part C, Methods. 26 (5), 263-275 (2020).
- Carletti, E., Motta, A., Migliaresi, C., Haycock, J. W. Scaffolds for tissue engineering and 3D cell culture. 3D Cell Culture: Methods and Protocols. , 17-39 (2011).
- Loh, Q. L., Choong, C. Three-dimensional scaffolds for tissue engineering applications: Role of porosity and pore size. Tissue Engineering. Part B Reviews. 19 (6), 485-502 (2013).
- Zhang, Y., et al. GMP-grade microcarrier and automated closed industrial scale cell production platform for culture of MSCs. Journal of Tissue Engineering and Regenerative Medicine. 16 (10), 934-944 (2022).
- NMPA. Pharmaceutical Excipient Registration Database: Microcarrier tablets for cells. Center for Drug Evaluation Available from: https://www.cde.org.cn/main/xxgk/listpage/ba7aed094c29ae314670a3563a716e (2023)
- List of Drug Master Files (DMFs). US Food and Drug Administration Available from: https://www.fda.gov/drug-mater-files-dmfs/list-drug-master-files-dmfs (2023)
- Beeravolu, N., et al. Isolation and characterization of mesenchymal stromal cells from human umbilical cord and fetal placenta. Journal of Visualized Experiments. (122), e55224 (2017).
- Xie, Y., et al. The quality evaluation system establishment of mesenchymal stromal cells for cell-based therapy products. Stem Cell Research & Therapy. 11 (1), 176 (2020).
- Maillot, C., Sion, C., De Isla, N., Toye, D., Olmos, E. Quality by design to define critical process parameters for mesenchymal stem cell expansion. Biotechnology Advances. 50, 107765 (2021).
- Silva Couto, P., et al. Expansion of human mesenchymal stem/stromal cells (hMSCs) in bioreactors using microcarriers: Lessons learnt and what the future holds. Biotechnology Advances. 45, 107636 (2020).
- Chen, S., et al. Facile bead-to-bead cell-transfer method for serial subculture and large-scale expansion of human mesenchymal stem cells in bioreactors. Stem Cells Translational Medicine. 10 (9), 1329-1342 (2021).
- Tsai, A. C., Pacak, C. A. Bioprocessing of human mesenchymal stem cells: From planar culture to microcarrier-based bioreactors. Bioengineering. 8 (7), 96 (2021).
- Hewitt, C. J., et al. Expansion of human mesenchymal stem cells on microcarriers. Biotechnology Letters. 33 (11), 2325-2335 (2011).
- Mawji, I., Roberts, E. L., Dang, T., Abraham, B., Kallos, M. S. Challenges and opportunities in downstream separation processes for mesenchymal stromal cells cultured in microcarrier-based stirred suspension bioreactors. Biotechnology and Bioengineering. 119 (11), 3062-3078 (2022).
- Schirmaier, C., et al. Scale-up of adipose tissue-derived mesenchymal stem cell production in stirred single-use bioreactors under low-serum conditions. Engineering in Life Sciences. 14 (3), 292-303 (2014).
- Lawson, T., et al. Process development for expansion of human mesenchymal stromal cells in a 50L single-use stirred tank bioreactor. Biochemical Engineering Journal. 120, 49-62 (2017).
- Yang, J., et al. Large-scale microcarrier culture of HEK293T cells and Vero cells in single-use bioreactors. AMB Express. 9 (1), 70 (2019).
- Fang, Z., et al. Development of scalable vaccinia virus-based vector production process using dissolvable porous microcarriers. 25th Annual Meeting of the American Society of Gene & Cell Therapy. Molecular Therapy. 30, 195-196 (2022).

