A Minimally Invasive Model of Aortic Stenosis in Swine
Summary
This protocol describes a minimally invasive surgical procedure for ascending aortic banding in swine.
Abstract
Large animal models of heart failure play an essential role in the development of new therapeutic interventions due to their size and physiological similarities to humans. Efforts have been dedicated to creating a model of pressure-overload induced heart failure, and ascending aortic banding while still supra-coronary and not a perfect mimic of aortic stenosis in humans, closely resembling the human condition.
The purpose of this study is to demonstrate a minimally invasive approach to induce left ventricular pressure overload by placing an aortic band, precisely calibrated with percutaneously introduced high-fidelity pressure sensors. This method represents a refinement of the surgical procedure (3Rs), resulting in homogenous trans-stenotic gradients and reduced intragroup variability. Additionally, it enables swift and uneventful animal recovery, leading to minimal mortality rates. Throughout the study, animals were followed for up to 2 months after surgery, employing transthoracic echocardiography and pressure-volume loop analysis. However, longer follow-up periods can be achieved if desired. This large animal model proves valuable for testing new drugs, particularly those targeting hypertrophy and the structural and functional alterations associated with left ventricular pressure overload.
Introduction
Heart failure (HF) is a life-threatening disease that affects millions of people worldwide, causing major social and economic impacts1. One of its significant etiologies is aortic valve disease or aortic stenosis (AS). Aortic stenosis is more prevalent in advanced age and ranks as the second most common valvular lesion in the United States. AS-related mortality has also increased in Europe, particularly in countries without access to recent interventional procedures2. Given the complexity of HF and the scarcity of therapeutic innovations, there is a pressing need for reliable animal models that can replicate the human condition and facilitate the testing of new interventions3. While rodent models outnumber large animal models, the latter offers several advantages due to their size and physiological similarities, allowing the testing of drug doses and medical devices intended for human use.
The aim of this method is to establish a reproducible model of ascending aortic banding (AAB) applicable to most large animal species used in biomedical research. In this study, the procedure is demonstrated in swine using a minimally invasive approach, adhering to the 3Rs principles (replacement, reduction, and refinement4). This approach ensures the creation of an accurate pressure gradient, resulting in high reproducibility (potentially reducing the number of required animals). Additionally, the small surgical incision (2-3 cm) minimizes surgical insult, improving animal well-being compared to more aggressive approaches like sternotomy and larger thoracotomies5 (refinement). Furthermore, providing a video demonstration of the method, along with detailed descriptions in the literature, could potentially reduce the need for animals used solely for training purposes (replacement), further decreasing animal usage. This model can be adapted for different swine strains/breeds with distinct growth rates and induces sustained pressure overload, leading to significant hypertrophy after 1 or 2 months of follow-up.
Current methods employ fixed stenosis6, disregarding animal size variability, or calculate gradient using fluid-filled pressure readings7, which are less reliable than high-fidelity pressure sensors and are susceptible to signal damping8. Another approach uses a single pressure measurement distal to the stenosis5. However, calibrating the stenosis through simultaneous proximal and distal pressure signals using percutaneously delivered high-fidelity pressure sensors represents a substantial optimization of the protocol, resulting in improved group homogeneity. By visually demonstrating this method, other researchers should be able to replicate it without significant obstacles, increasing the availability of this model while promoting the application of the 3Rs principles.
Protocol
The animal experiments were performed at the Experimental Surgery laboratory in the University of Porto, Cardiovascular Research and Development Centre (UnIC, Porto, Portugal). The institutional animal ethics committee approved the study in accordance with the National Authority for Animal Health (Direcção-Geral de Alimentação e Veterinária, DGAV, Ref: 2021-07-30 011706 0421/000/000/2021). The experimenters were either licensed (FELASA-equivalent Laboratory Animal Sciences authorization) or were cardiothoracic surgeons or anesthesiologists. Animals used in this work were males from a Landrace x Pietrain background and were acquired from a breeder licensed by DGAV (PTAH03). Starting weight of the animals was 20-25 kg, which allowed for a maximum of 2-month follow-up (70-80 kg, Figure 1). Longer follow periods are compromised due to the significant animal growth, which our infrastructures were unable to handle.
1. Anesthesia and vital sign monitoring
- Fast the selected animal overnight with water ad libitum.
- Weigh the conscious animal (allow it to walk on an animal scale), or use an estimate based on arrival weight and expected growth rate.
- Prepare a cocktail of ketamine (15 mg/kg), midazolam (0.5 mg/kg), and azaperone (4 mg/kg) in a 20 mL Luer lock syringe connected to an extension line (100 cm) followed by a 21 G needle (see Table of Materials). Ensure to have enough anesthetic to account for the dead volume of the injection tubing.
- Isolate the pig in a calm and safe environment (usually an empty animal maintenance room, if possible) and anesthetize the animal through an intramuscular injection in the neck or hind leg muscle (following institutional protocol). Ensure the animal is in a quiet and dark place and ensure it cannot injure itself while losing recumbency, which should take 10-15 min.
NOTE: The use of an extension line avoids the need to restrain the animal. - Once the animal is in recumbency, place the anesthetized animal on a stretcher and transport the animal to the operating room (measure the animal's weight at this point if conscious weighing was not an option).
- Position the animal in a right or left lateral decubitus depending on the ear to be cannulated. Clean the ear several times with chlorhexidine and alcohol in a circular motion. Then, canulate the marginal ear vein using a 20 G intravenous catheter and secure it using non-woven adhesive (see Table of Materials). Connect the IV catheter to a 3-port stopcock manifold pre-flushed with saline.
NOTE: Alternatively, compared to a standard IV infusion line (drip line), an infusion pump enables precise adjustment of the flow rate (2 mL/kg/h). - Place a transdermal fentanyl patch (50 µg/h) (see Table of Materials) in the contralateral ear.
- Place the animal on a radiotransparent surgical table, in dorsal recumbency, on top of a warming mattress, and secure it in place ( straps securing the paws to the surgical table).
- Connect a propofol perfusion line to the 3-port stopcock manifold. Anesthesia maintenance will be provided by propofol administered through a 50 mL syringe mounted on a syringe perfusion pump (see Table of Materials) at a rate of 10-20 mg/kg/h.
- Administer a bolus of propofol (4 mg/kg) and fentanyl (10 µg/kg) (see Table of Materials) to induce apnea and allow intubation.
NOTE: The animal will be in apnea from this moment, and mechanical ventilation must be established. Before proceeding, ensure that the oxygen source is available, and the ventilator (see Table of Materials) is calibrated and ready for ventilation. - After ensuring loss of reactivity, and with one user holding the mouth of the pig open while simultaneously pulling the tongue outwards, use a laryngoscope with a number 4 Miller blade (see Table of Materials) to identify and gently mobilize the epiglottis, gaining a view of the vocal cords. Either introduce the endotracheal tube directly or advance a bougie and the endotracheal tube over it first. Some atraumatic intestinal forceps can aid in mobilizing the soft palate and gaining access to the epiglottis.
- Inflate the endotracheal tube cuff and connect it to the anesthesia machine/ventilator. Adjust the ventilatory parameters to 8-10 mL/kg of tidal volume, respiratory rate of 15-25 respirations per minute, and 5 cm H2O of PEEP (positive end-expiratory pressure). Adjust ventilatory parameters to maintain end-tidal CO2 between 35 and 45 mmHg.
- Position the SpO2 sensor on the tongue or ear (where the best signal is obtained), place the esophageal temperature probe, and attach the ECG electrodes (see Table of Materials).
- Apply sterile ophthalmic lubricating ointment to prevent corneal injury.
2. Arterial cannulation
- After ensuring proper anesthetic depth by the absence of palpebral reflex and steady heart rate and blood pressure, thoroughly clean and disinfect the groin area with chlorhexidine and alcohol in a circular motion. Cover the animal with fenestrated sterile drapes (see Table of Materials), with the hole positioned in the femoral artery area (previously confirmed by palpation or ultrasound). Administer cefazolin (25 mg/kg) as antibiotics prophylaxis.
- If the procedure involves animal recovery (aortic banding), use the aseptic technique from this point forward.
NOTE: A strict aseptic technique is not required if it is a terminal procedure (PV Loop analysis). However, working in a sterile fashion is advantageous to avoid infection that could affect hemodynamic measurements. - Identify the puncture site and infiltrate the area with subcutaneous 1% lidocaine.
- Identify the common femoral artery using the vascular probe (see Table of Materials) and confirm the position of the ultrasound marker and correct depth.
NOTE: Femoral artery puncture can be performed using a short axis, long axis, or a combination of both techniques, utilizing a bi-plane modality in certain systems. However, our team more frequently employs the short-axis approach. - Prepare the introducer sheath (see Table of Materials) by flushing the introducer and dilator with heparinized saline before assembling it. Ensure the 3-way stopcock in the introducer side port is in the off position towards the animal to avoid blood loss when removing the dilator.
- Advance an arterial needle (preferably an echogenic one, see Table of Materials) into the femoral artery while monitoring its trajectory using ultrasound. Once the arterial lumen is reached, which can be confirmed by pulsating arterial blood exiting the needle hub, advance a J-tip guidewire into the artery. The correct introduction of the guidewire can be confirmed with ultrasound.
- Remove the needle, keep pressure on the puncture site to avoid additional bleeding, and advance the introducer + dilator (size 6 Fr, 10 cm length) assembly into the artery. Remove the dilator and confirm the position of the introducer by aspirating from its side port and sequentially flushing with sterile saline.
- Connect an arterial pressure line to the side-port of the femoral artery introducer for blood pressure monitoring. Ensure that the pressure transducer height is at the level of the right atria and that atmospheric pressure is zero.
- Cover the introducer with a sterile drape until left ventricular catheterization.
3. Ascending aortic banding (preparation)
- Adjust the animal's position to a slight right lateral decubitus and elevate the left front paw.
- Locate the position of the ascending aorta using the cardiac ultrasound transducer (see Table of Materials) and mark the incision site before thoroughly disinfecting the chest of the animal with chlorhexidine and alcohol in a circular motion.
- Cover the animal with sterile drapes.
- Administer a fentanyl bolus (10 µg/kg) to ensure sufficient analgesia. To confirm the depth of anesthesia and analgesia, observe for the lack of palpebral reflexes and no changes in heart rate or blood pressure upon making the first incision.
- Make a 2-3 cm skin incision at the level of the 3/4th intercostal space and dissect the underlying fascia and muscle layers until the intercostal space is reached.
- Enter the thorax using blunt scissors while the animal is in forced expiration without PEEP to avoid damaging the lung.
- Increase the incision to allow placement of the retractor blades to a maximum of 3 cm.
- Retract the ribs and visualize the underlying structures. If the incision is made in the correct place, the pulmonary artery should be readily visible. The aorta will be posterior to it.
- Using minimally invasive cardiac surgery forceps and scissors, open the pericardium and retract the left atria and any lung tissue covering the view of the aorta using wet sterile gauze.
NOTE: Avoid manipulating the left atria too much, as it will lead to atrial fibrillation. If that occurs and does not resolve spontaneously, apply electrical cardioversion. - Carefully separate the aorta from the pulmonary artery until the transverse pericardial sinus is reached. This will be the channel through which the banding material will be passed.
NOTE: For banding of the ascending aorta, several materials can be used, depending on the animal size and follow-up period. For animals with limited growth and/or a short follow-up period, a nylon cable zip tie can be used (cheaper option), while for faster-growing animals and/or a longer follow-up period, a vascular prostheses graft fixed with titanium clips can be used (more expensive option), avoiding band internalization (discussed in detail in the sections below) (see Table of Materials). - Option 1 (nylon cable zip tie):
- Cut a ~10 cm segment of sterile plastic tubing with a lumen small enough to snuggly fit the tip of the nylon band.
NOTE: The sterile tubing and nylon band are previously sterilized by ethylene oxide sterilization or submersion in formaldehyde for at least 24 h. - Use 90° curved forceps to pass the plastic tubing (which serves as an atraumatic guide to the nylon band) around the aorta, through the previously created path, from the space between the aorta and pulmonary artery (proximal) towards the space between the aorta and right atria (distal). Palpation with the finger can help guide the forceps through the correct path.
- Take care not to apply too much strain on the pulmonary artery or right atria, as this can lead to hemodynamic instability. Pay close attention to vital signs during this step to avoid prolonged periods of systemic hypotension.
- Once the plastic guide is seen on the distal side, grasp it with tissue forceps and carefully pull it around the aorta, bringing the nylon band with it. Connect the two ends of the nylon band without constricting the aorta.
- Cut a ~10 cm segment of sterile plastic tubing with a lumen small enough to snuggly fit the tip of the nylon band.
- Option 2 (ePTFE graft)
- Cut a ~10 cm sterile ePTFE graft of a 5 mm 40 cm long graft.
- Use a 90° curved forceps to handle the graft and pass it around the aorta. See steps 3.11.2 and 3.11.3.
- Place a radiopaque marker (see Table of Materials) in the banding area to facilitate aorta catheterization.
- Cover the intercostal space with wet gauze and sterile drapes.
4. Left ventricle (LV)/Aorta catheterization
- Administer Heparin (200 U.kg-1).
NOTE: Endovascular procedures are associated with a risk of clot formation and distal embolization, while heparin administration would lead to excessive bleeding during the surgical access to the aorta. Therefore, LV/aorta catheterization is performed after the aorta is accessed and the banding put in place. - Connect a dual hemostasis valve adapter or a star-shaped hemostasis valve to a 6 Fr MP1 guide catheter (see Table of Materials) and flush with heparinized saline. Preload the guide catheter with a 260 cm 0.035 in J-tip guidewire. Introduce this assembly through the femoral arterial sheath.
NOTE: There may be a risk of bleeding when advancing two high-fidelity pressure sensors (HFPS) through a standard cross-cut hemostasis valve. An alternative approach could be using two separate guide catheters, but this would require a second arterial access site. To address both the bleeding issue and the need for additional access sites, one can opt for either a dual port hemostasis valve or a star-shaped hemostasis valve. These alternatives solve the bleeding problem and eliminate the requirement for extra access points. Once the guide catheter is advanced through the arterial sheath, it's important to note that the side port of the sheath will not allow for blood pressure measurement. To measure blood pressure, it is necessary to connect the arterial line to the side port of the hemostasis valve adapter of the guide catheter instead. - Advance the guidewire and guide the catheter into the ascending aorta under fluoroscopic guidance. When the aortic valve is identified, carefully cross it with the guidewire, and introduce the guide catheter into the LV. If needed, use contrast to facilitate anatomical positioning. Check pressure traces to confirm LV positioning.
- Remove the guidewire while leaving the guide catheter in the LV. Flush the catheter after aspirating and ensure no air bubbles are present in the catheter.
- Advance an already calibrated HFPS, through one of the ports of the dual hemostasis valve, into the LV. A mark with a sterile pen can be placed on the catheter body to know when it comes out of the guide catheter tip. Alternatively, confirmation of a clear ventricular pressure signal is a sign of exiting the guide catheter (signal interference is observed while the HFPS is inside the guide catheter).
- Advance a second HFPS through the other port of the dual hemostasis valve and into the LV.
- Pull the guide catheter back into the ascending aorta distally to the radiopaque marker placed on the banding site while leaving one of the HFPS in the LV. Confirm catheter position using pressure traces.
NOTE: The HFPS should be connected to the recording system and placed in sterile saline for at least 30 min before use to allow the pressure sensor to equilibrate. Before introducing the HFPS into the guide catheter, ensure to zero the pressure by placing the sensor at the surface of sterile saline. - Cover the vascular access site with a sterile drape and move to the thorax to constrict the aorta.
5. Ascending aortic banding (constriction)
- Slightly pull on the nylon band (option 1) or the ePTFE graft (option 2) to ensure that the HFPS is properly positioned – LV pressure should go up, while aortic pressure distal to the banding (radiopaque marker) should not increase.
- If catheter positioning is incorrect, adjust the position of the HFPS to ensure that proximal and distal pressure to the constriction site is clearly recorded.
- Option 1: close the nylon band until it is snug around the aorta.
- Close the nylon band one click at a time while closely monitoring pressures. After each click, allow the pressures to stabilize.
- Close the nylon band gradually until the desired pressure gradient is reached. The target is a gradient of approximately 100 mmHg, while ensuring that the left ventricular end-diastolic pressures do not exceed 25 mmHg.
NOTE: If the gradient achieved is just under 100 mmHg (i.e., between 90-95 mmHg), refrain from tightening the nylon band further. It is crucial not to overtighten in this situation. However, if the nylon band is accidentally tightened excessively during the procedure or after stabilization, one can use bone cutters (see Table of Materials) to cut the nylon band and then repeat the previous procedures (step 3.11 and step 5.3) to readjust and achieve the appropriate pressure gradient. - Place a piece of sterile plastic tubing on the nylon band end to avoid any accidental damage to the surrounding structures.
- Option 2: approximate the ePTFE ends and constrict the band using 45° forceps, while monitoring pressures, to estimate the relative location of the constriction.
- Place a titanium hemoclip on the forceps position (clamping the two ends of the ePTFE graft will leave a mark on it, serving to guide the hemoclip position).
- Check the pressure gradient. If the gradient is optimal, confirm the previous hemoclip position by placing a second hemoclip immediately above the previous one (this will not increase the constriction but will avoid any distal slippage of the clip).
- If the gradient is not sufficient, place an additional clip below the previous clip (constricting the aorta further). Do this until the gradient is optimal. If the gradient is too large, use the clip applier to remove the clip and place another one more distally.
- Trim the ends of the ePTFE graft to avoid having too much graft material in the chest and suture the graft to the proximal side of the aorta using a 5.0 suture (see Table of Materials) to avoid distal movement of the graft.
- Wait 15 min after placing the banding to stabilize pressures and determine if the gradient remains optimal or if decompensation and acute failure will install. If hypotension occurs without spontaneous resolution, it is very likely that the LV is decompensating, and banding alleviation is required.
- Close the pericardium using a 3-0 PDS II suture (see Table of Materials).
- Place a chest drain and connect it to a surgical aspirator. Increase PEEP to 10 cmH2O to start recruiting any lung that suffered atelectasis.
- Close the chest wall in layers using a 3-0 PDS II suture.
- Close the last muscle stitch while removing the chest drain and with ventilation stopped at end-expiration with high pressure (manually adjusted to 20-30 cmH2O).
- Resume normal ventilation and close the skin using a 3-0 PDS II suture with an intradermal pattern after flushing the surgical wound with iodine-povidone.
- Remove the Mikro-caths (see Table of Materials) and check surface pressure to account for pressure drift during the procedure.
- Remove the guide catheter.
- Remove the introducer sheath and apply manual compression to close the arteriotomy. Apply compression to the access site for at least 10 min. Assess the site for hemostasis by slowly removing pressure and confirming the absence of bleeding or hematoma formation.
- Place a stitch on the puncture site using an absorbable suture if needed.
NOTE: If needed, at this time, a quick transthoracic echocardiogram can help determine if cardiac function is good and allow estimation of aortic pressure gradient. While not necessary (as the pressure gradient was measured with high-fidelity pressure sensors), an echo-derived gradient can be used to compare the model with clinical data. Note that due to the surgery, image quality will be compromised. - Stop anesthesia and extubate the animal once spontaneous ventilation is detected. Disconnect the animal from the ventilator and ensure that proper airflow is felt through the endotracheal tube and that peripheral oxygenation is not compromised.
- Extubate and place a Guedel if necessary.
- Remove the peripheral vein catheter.
- Monitor the animal for at least 15 min while monitoring ECG/heart rate and peripheral oxygenation.
- If stable, take the animal to a clean recovery pen with increased ambient temperature. Use a portable vital sign device (see Table of Materials) to continuously monitor heart rate and saturation until the animal regains consciousness.
- Follow the animals for the desired period and perform transthoracic echocardiography or pressure-volume loop analysis to determine cardiac function.
Representative Results
During the initial development of the model, the mortality rate was approximately 30%, with animals dying from acute heart failure after banding and surgical complications. However, after the model was established, surgical complications became less common, and the mortality rate dropped to around 15%. The two deaths that occurred were due to aortic rupture during dissection.
The use of high-fidelity pressure sensors enables obtaining high-quality pressure signals (Figure 2), allowing real-time and accurate calibration of the stenosis. This ensures that all operated animals experience a similar degree of left ventricular pressure overload, reducing variability within the group. Moreover, the catheter itself has a 2.3 F shaft, which has minimal impact on flow obstruction compared to larger fluid-filled catheters. After an initial investment, the catheters can be reused multiple times, and if sterilization is needed, ethylene oxide can be used (usually available through collaboration with surgical departments at the hospital).
The trans-stenotic gradient can be calculated in real-time by the software, which measures the pressure difference between the left ventricle (proximal pressure) and the distal aorta (distal pressure). A few minutes of stabilization between each constriction step ensures that the left ventricle has time to adapt. After determining the desired constriction degree, a 15 min stabilization period should be applied to ensure that the banding degree remains stable and the animal is compensated (Figure 2A).
This approach is superior to other methodologies that do not measure trans-stenotic gradient in real-time and lack both the homogeneity of having a similar gradient between all animals (92.3 ± 2.3 mmHg, mean and standard error of the mean, respectively, for 7 operated animals) and tight monitoring of left ventricular pressures. Additionally, this approach avoids the difficulties associated with performing transthoracic echocardiography in swine, particularly in certain breeds like the Vietnamese potbellied pig, which has a more significantly protruding sternum.
Transthoracic echocardiography can confirm aortic banding both immediately after the surgery and during follow-up time points (Figure 3). The banding surgery results in significant stenosis of the aorta with turbulent flow, which can be qualitatively evaluated or quantified using continuous wave Doppler. Figure 2 displays representative images of 2-month follow-up echocardiography, showing significant aortic stenosis (upper row) and left ventricular concentric hypertrophy (middle and bottom rows). Two months after banding, the animals develop significant cardiac hypertrophy. The macroscopic evaluation revealed larger hearts and a thicker left ventricular wall (Figure 4). The two-month follow-up period was determined based on the growth rate of the used animals, as a longer follow-up period would result in animals too large to be handled by our infrastructures.
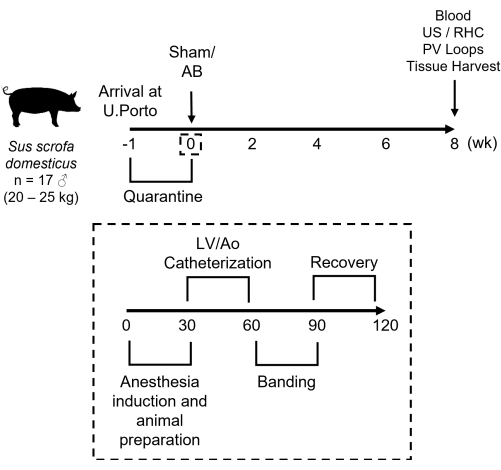
Figure 1: Schematics of the aortic banding protocol. After receiving 20-25 kg male pigs, the animals are submitted to a 1 week quarantine period. On the day of the procedure, the animals are anesthetized, the LV and aorta are catheterized, and high-fidelity pressure sensors are placed, followed by aortic banding and animal recovery. The whole procedure, once mastered, lasts around 2 h. Two months after surgery, the animals are submitted to a terminal evaluation, including collection of samples and measurement of physiological variables. AB-aortic banding, Ao-aorta, LV-left ventricle, PV-pressure-volume, RHC-right heart catheterization, US-ultrasound. Please click here to view a larger version of this figure.
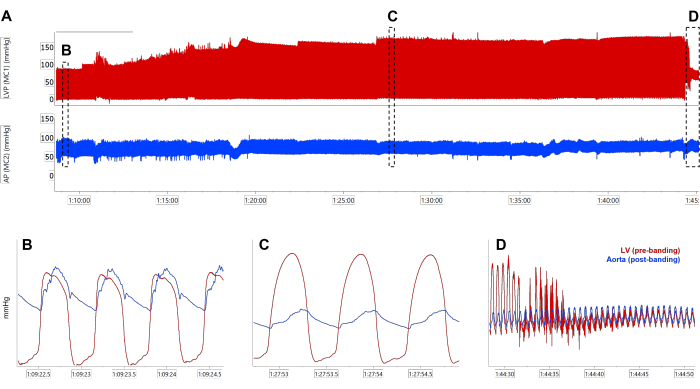
Figure 2: Pressure measurements during aortic banding. (A) Representative traces of LV and aortic (distal to the banding) pressures during aortic banding. Zoom in on LV and aortic pressure before (B) and after (C) constriction, showing the gradient creation (difference between peak systolic LV and aortic pressure). (D) Pull-off of the ventricular pressure sensor, transitioning from the aorta proximal to the banding to the aorta distal to the banding. AP-arterial pressure, LVP-left ventricle pressure, MC-high-fidelity pressure sensor. Please click here to view a larger version of this figure.
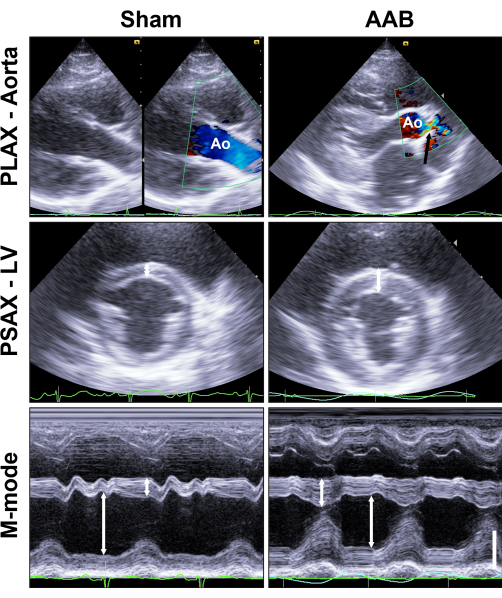
Figure 3: Transthoracic echocardiography. Follow-up at 2 months after surgery reveals significant stenosis of the aorta (black arrow, upper row). LV hypertrophy is apparent, both in 2D (white arrows, middle row), as well as in M-mode, which also demonstrates concentric hypertrophy (white arrows, bottom row). The vertical bar corresponds to 3 cm, and 2D PSAX images were acquired at a depth of 15 cm. Please click here to view a larger version of this figure.
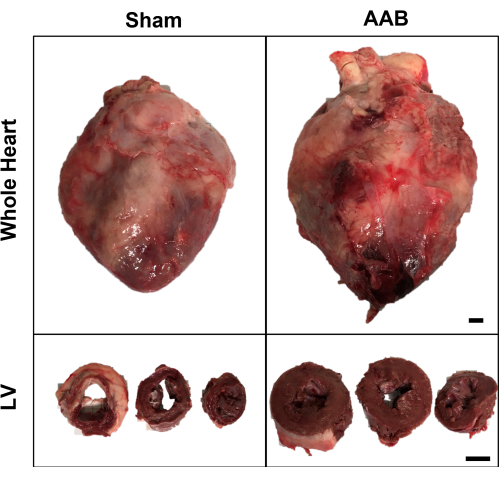
Figure 4: Post-mortem macroscopic analysis of the heart. Aortic banding leads to cardiomegaly, with clear hypertrophy of the LV wall. Heart slices are base, mid-cavity, and apex from left to right. Pericardial adhesions can be seen throughout the epicardium. Scale bars represent 1 cm (upper row) and 4 cm (lower row). Please click here to view a larger version of this figure.
Discussion
In recent years, several studies have utilized surgical aortic banding as a model for left ventricular pressure overload and heart failure (descending9 to the ascending aorta10), allowing researchers to obtain various phenotypes tailored to their specific needs. Although using such models requires costly equipment and specialized knowledge, the information they provide is invaluable. Swine, due to its size and similarity to the human heart, serves as an ideal model11, gaining ethical acceptance as organ donors for xenotransplantation.
The main critical step in this method is the dissection of the aorta and the placement of the banding material (nylon cable or ePTFE graft) around it. During this step, several complications can occur, including laceration or rupture of the surrounding structures or the aorta itself. Controlling such complications can be achieved by placing a pursue-string suture or a mattress suture with pledgets on the hole if bleeding can be controlled to properly visualize the wound. It is strongly recommended to have the procedure performed by a cardiothoracic surgeon, which significantly reduces complication and mortality rates.
Another critical step is the constriction of the aorta, which should be done in sequential steps with stabilization periods in between. Paying close attention to systemic peripheral pressures is crucial, as sustained significant hypotension (mean arterial pressure below 60 mmHg) may result from the LV’s inability to cope with the current stenosis. If not resolved, especially when ventricular pressures start to drop as well, acute heart failure will lead to the loss of the animal. The removal of the nylon cable or titanium clip is necessary when hypotension does not spontaneously resolve.
However, the main limitation of this model, and many aortic banding models, is the band’s location relative to the coronary ostia. Supra-coronary banding placement does not entirely mimic aortic stenosis and may lead to increased blood pressure in the coronary circulation, which might be protective12. Limited evidence suggests no differences between sub-coronary and supra-coronary aortic banding in pigs13, indicating that the increased complications associated with sub-coronary banding surgery may not be worthwhile.
Depending on the animal strain used and the follow-up time, band internalization may become an issue. Although mainly described in rodents14, it has also been observed in the pulmonary artery of pigs15. Using ePTFE graft segments significantly increases the contact area and eliminates the occurrence of band internalization. However, ePTFE grafts are more expensive, and when using slow-growing breeds, such as the Vietnamese pot-bellied pig, band internalization is not an issue when using nylon zip ties. Researchers should choose their approach based on the animal breed used.
For fast-growing breeds, long-term follow-up might be challenging due to animal size (availability of infrastructure and equipment large enough to handle >100 kg animals) and prohibitive maintenance costs.
Another limitation of this model, as well as all models requiring pericardial space access, is the presence of significant pericardial adhesions after surgery. Our experience shows no difference between closing or not closing the pericardial incision after band placement. While it does not affect function, dissecting the heart and identifying different structures becomes more time-consuming, and the epicardium is likely to be damaged if the pericardium is fully separated.
This minimally invasive method represents a significant refinement of the typical surgical procedure, leading to an uneventful and speedier recovery. The use of two high-fidelity catheters for simultaneous pressure measurement and real-time gradient measurement significantly improves the accuracy of the procedure and the reproducibility of the model, leading to a reduction in the number of animals required. The model can be applied to the study of new therapeutic interventions or devices aimed at left ventricular hypertrophy, as well as the determination of new pathophysiological mechanisms associated with left ventricular pressure overload.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported and funded under the QREN project 2013/30196, the "la Caixa" Banking Foundation, the Fundação para a Ciência e Tecnologia (FCT) project, LCF/PR/HP17/52190002. JS and EB were supported by the European Union's Horizon 2020 research and innovation program under the Marie Sklodowska-Curie grant agreement no. 813716. PdCM was supported by the Stichting Life Sciences Health (LSH)-TKI project MEDIATOR (LSHM 21016).
Materials
| 3-0 PDS II suture | Ethicon | Z683G | Aorta banding |
| 5-0 prolene | Ethicon | 7472H | Aorta banding |
| ACUSON NX2 Ultrasound System | Siemens | (240)11284381 | Vascular Access and Echocardiography |
| Arterial Extension 200 cm | PMH | 303.0666 | Anesthesia Maintenance |
| Atlan A300 Ventilator | Draeger | 8621300 | Ventilation |
| Bone cutters | Fehling | AMP 367.00 | Aorta banding |
| Cefazolin 1000 mg | Labesfal | 100063 | Antibiotic |
| Chlorhexidine 4% Wash Solution | AGA | 19110008 | Cleaning |
| Doyen Intestinal Forceps | Aesculap | EA121R | Intubation |
| Echogenic Introducer Needle | Teleflex | AN-04318 | Vascular Access |
| Endotracheal tube | Intersurgical | 8040070 | Intubation |
| ePTFE vascular graft (5 mm x 40 cm) | GORE-TEX | S0504 | Aorta banding |
| Extension line 100 cm | PMH | 303.0394 | Anesthesia Induction |
| F.O. Laryngoscope | Luxamed | E1.317.012 | Intubation |
| F.O. Miller Blade 4 204 x 17 mm | Luxamed | 3 | Intubation |
| Fenestrated Sterile Drape | Bastos Viegas | 4882-256 | Aseptic Technique |
| Fentanyl 0.5 mg/10 mL | B.Braun | 5758883 | Anesthesia / Analgesia |
| Guidewire 260 cm J-tip | B.Braun | J3 FC-FS 260-035 | Left Ventricle catheterization |
| Infusomat Space Infusion Pump | B.Braun | 24101800 | Fluids / Drug administration |
| Intercostal retractor | Fehling Surgical | MRP-1 | Thoracotomy |
| Introcan Certo IV Catheter 20G | B.Braun | 4251326 | Fluids / Drug administration |
| Isotonic Saline Solution 0.9% | B.Braun | 5/44929/1/0918 | Fluids / Drug administration |
| Ketamidor 100 mg/mL | Richter pharma | 1121908AB | Anesthesia Induction |
| L10-5v Linear Transducer | Siemens | 11284481 | Vascular Access |
| Midazolam 15 mg/3 mL | Labesfal | PLB762-POR/2 | Anesthesia Induction |
| Mikro-cath | Millar | 63405(1) | Pressure recording |
| MP1 guide catheter 6 Fr | Cordis | 67027000 | Left Ventricle catheterization |
| Needle Holder | Fehling Surgical | ZYY-5 | Aorta banding |
| Non-woven adhesive | Bastos Viegas | 442-002 | Fluids / Drug administration |
| P4-2 Phased Array Transducer | Siemens | 11284467 | Echocardiography |
| Perfusor Compact Syringe Perfusion Pump | B.Braun | 8717030 | Fluids / Drug administration |
| Pressure Signal Conditioner | ADinstruments | PCU-2000 | Pressure recording |
| Propofol Lipuro 2% | B.Braun | 357410 | Anesthesia Maintenance |
| Radifocus Introducer II Standard Kit B – Introducer Sheath | Terumo | RS+B60K10MQ | Vascular Access |
| Radiopaque marker | Scanlan | 1001-83 | Aorta banding |
| Scissors | Fehling Surgical | Thoracotomy | |
| Skinprep (Chlorhexidine 2% / 70% Isopropyl alcohol) | Vygon | SKPC015ES | Disinfection |
| Stopcock manifold (3 ports) | PMH | 310.0489 | Fluids / Drug administration |
| Straight forceps | Fehling Surgical | ZYY-1 | Thoracotomy |
| Stresnil 40 mg/mL | ecuphar | 572184.2 | Anesthesia Induction |
| Syringe Luer Lock 20 cc | Omnifix B.Braun | 4617207V | Anesthesia Induction |
| Syringe Luer Lock 50 cc | Omnifix B.Braun | 4617509F | Anesthesia Maintenance |
| Transdermal fentanyl Patch 50 mcg/h | Mylan | 5022153 | Analgesia |
| Ultravist | Bayer | KT0B019 | Angiography |
| Universal Hemostasis Valve Adapter | Merit Medical | UHVA08 | Left Ventricle catheterization |
| Velcro Limb Immobilizer | PMH | SU-211 | Animal stabilization |
| Venofix A, 21 G | B.Braun | 4056337 | Anesthesia Induction |
| Vista 120S Patient Monitor | Draeger | MS32997 | Monitoring |
| Weck titanium clip | Teleflex | 523760 | Aorta banding |
| Weck titanium clip applier | Teleflex | 523166 | Aorta banding |
| Zhiem Vision | Iberdata | N/A | Fluoroscopy |
References
- Savarese, G., et al. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovascular Research. 118 (17), 3272-3287 (2023).
- Hartley, A., et al. Trends in mortality from aortic stenosis in Europe: 2000-2017. Frontiers in Cardiovascular Medicine. 8, 748137 (2021).
- Silva, K. A. S., Emter, C. A. Large Animal models of heart failure: a translational bridge to clinical success. Journal of the American College of Cardiology: Basic to Translational Science. 5 (8), 840-856 (2020).
- Brink, C. B., Lewis, D. I. The 12 Rs framework as a comprehensive, unifying construct for principles guiding animal research ethics. Animals (Basel). 13 (7), 1128 (2023).
- Choy, J. S., Zhang, Z. D., Pitsillides, K., Sosa, M., Kassab, G. S. Longitudinal hemodynamic measurements in swine heart failure using a fully implantable telemetry system. PLoS One. 9 (8), 103331 (2014).
- Ishikawa, K., et al. Increased stiffness is the major early abnormality in a pig model of severe aortic stenosis and predisposes to congestive heart failure in the absence of systolic dysfunction. Journal of the American Heart Association. 4 (5), 001925 (2015).
- Emter, C. A., Baines, C. P. Low-intensity aerobic interval training attenuates pathological left ventricular remodeling and mitochondrial dysfunction in aortic-banded miniature swine. American Journal of Physiology-Heart and Circulatory Physiology. 299 (5), H1348-H1356 (2010).
- Brito, J., Raposo, L., Teles, R. C. Invasive assessment of aortic stenosis in contemporary practice. Frontiers in Cardiovascular Medicine. 9, 1007139 (2022).
- Tan, W., et al. A Porcine model of heart failure with preserved ejection fraction induced by chronic pressure overload characterized by cardiac fibrosis and remodeling. Frontiers in Cardiovascular Medicine. 8, 677727 (2021).
- Bikou, O., Miyashita, S., Ishikawa, K. Pig model of increased cardiac afterload induced by ascending aortic banding. Methods in Molecular Biology. 1816, 337-342 (2018).
- Lelovas, P. P., Kostomitsopoulos, N. G., Xanthos, T. T. A comparative anatomic and physiologic overview of the porcine heart. Journal of the American Association for Laboratory Animal Science. 53 (5), 432-438 (2014).
- Tian, L., et al. Supra-coronary aortic banding improves right ventricular function in experimental pulmonary arterial hypertension in rats by increasing systolic right coronary artery perfusion. Acta Physiologica (Oxf). 229 (4), 13483 (2020).
- Sorensen, M., Hasenkam, J. M., Jensen, H., Sloth, E. Subcoronary versus supracoronary aortic stenosis. An experimental evaluation. Journal of Cardiothoracic Surgery. 6, 100 (2011).
- Lygate, C. A., et al. Serial high resolution 3D-MRI after aortic banding in mice: band internalization is a source of variability in the hypertrophic response. Basic Research in Cardiology. 101 (1), 8-16 (2006).
- Jalal, Z., et al. Unexpected Internalization of a Pulmonary Artery Band in a Porcine Model of Tetralogy of Fallot. World Journal for Pediatric and Congenital Heart Surgery. 8 (1), 48-54 (2017).

