Summary
Protocol details are provided for in vitro labeling human embryonic stem cells with second harmonic generating nanoparticles. Methodologies for hESC investigation by multi-photon microscopy and their differentiation into cardiac clusters are also presented.
Abstract
In this visualized experiment, protocol details are provided for in vitro labeling of human embryonic stem cells (hESC) with second harmonic generation nanoparticles (HNPs). The latter are a new family of probes recently introduced for labeling biological samples for multi-photon imaging. HNPs are capable of doubling the frequency of excitation light by the nonlinear optical process of second harmonic generation with no restriction on the excitation wavelength.
Multi-photon based methodologies for hESC differentiation into cardiac clusters (maintained as long term air-liquid cultures) are presented in detail. In particular, evidence on how to maximize the intense second harmonic (SH) emission of isolated HNPs during 3D monitoring of beating cardiac tissue in 3D is shown. The analysis of the resulting images to retrieve 3D displacement patterns is also detailed.
Introduction
Nonlinear microscopy systems, thanks to their inherent three dimensional sectioning capabilities, have increasingly triggered the demand for photo-stable fluorophores with two-photon absorption bands in the near-infrared. Only in the last couple of years, to complement the development of fluorescence-based labels (dyes, quantum dots, up-converting nanoparticles), a different imaging methodology has been exploiting the use of a novel family of inherently nonlinear nanoparticles as labels, i.e. harmonic nanoparticles (HNPs) which have been specifically developed for multi-photon microscopy. These labels, based on inorganic noncentrosymmetric crystals, exert optical contrast generating the SH of the excitation frequency: for example by converting a fraction of near infrared pulsed excitation light (λ = 800 nm) into visible blue light (λ/2 = 400 nm). Several authors in the recent past have tested different materials, including iron iodate Fe(IO3)3 1, potassium niobate (KNbO3)2, lithium niobate (LiNbO3)3, barium titanate (BaTiO3)4,5, potassium titanyl phosphate (KTiOPO4, KTP)6-8, and zinc oxide (ZnO)5,9,10. Compared to fluorescent probes, HNPs possess a series of attractive properties, such as complete absence of bleaching and blinking, narrow emission bands, excitation-wavelength tunability (from ultraviolet to infrared), orientation retrieval capability, and coherent optical response. These unique properties have been recently explained in two comprehensive review papers11,12. The possibility of working in the infrared spectral region, which increases imaging depth by minimizing scattering and absorption, also drastically limits sample photo degradation13,14. Moreover, the infinitely photo-stable signal guaranteed by HNPs makes them ideal probes for long-term cell tracking, which is particularly appealing for regenerative medicine applications15.
In this visualized experiment, protocol details are provided for in vitro labeling of human embryonic stem cells (hESC) with unfunctionalized HNPs. The synthesis and preparation of colloidal suspensions is detailed in a previous publication and in references therein16 and is beyond the scope of this work. Methodologies for hESC investigation by multi-photon microscopy and their differentiation into cardiac clusters (maintained as long term air-liquid cultures) are presented. Human ESC can be let to differentiate within so called embryoid bodies (EBs) in two different ways, either by EB formation of colony fragments in suspension or, alternatively, forced aggregation of single cells into EBs using the Aggrewell plate, as illustrated in Figure 1A. Culturing beating clusters of cardiac cells on polytetrafluoroethylene (PTFE) porous filters facilitates their long-term maintenance for further studies (for example electrophysiological measurements of action potentials).
The excitation source of the scanning microscope should be able to deliver ultrashort pulses (with a pulse duration smaller than 300 fsec at the sample) in order to reach the peak power needed to perform second harmonic imaging of HNPs. For instance, the most common fsec-source used for imaging are tunable Ti:Sapphire lasers. Alternatively, other ultrafast lasers can be employed, for instance erbium ion17, chrome forsterite18 or Ti:sapphire pumped infrared optical parametric oscillators. The microscope can be equipped with an objective with preferably a rather high numerical aperture. Very importantly, prior to measurements, and each time the objective is replaced, it is mandatory to minimize the dispersion present in the set-up (lenses) by optimizing the settings of the laser pulse pre-compressor at the working wavelength of choice. This procedure, detailed in the protocol, ensures that the laser pulse is as close as possible to the transform limited duration (i.e. shortest as possible) at the focal plane and maximizes the sample nonlinear response.
The goal of the image analysis described at the end of the protocol is to identify and track in 3D HNPs movements associated with the rhythmic contractions of beating cardiac clusters. Tracking nanoparticles in the image plane is simply realized by identifying their positions in successive movie frames. To extract information on axial movement, a prior calibration of the nonlinear intensity response as a function of axial displacement is mandatory. Note that for long-term measurements, an active interferometric control of sample axial position is required to maintain the validity of the calibration curve in the presence of thermal and/or mechanical drifts.
The HNPs used here to trace beating cells within aggregates are based on potassium niobate oxide (KNbO3), but other available nonlinear nanomaterials are reviewed in detail in the work of Staedler et al16.
The nonlinear optical efficiencies of most of the nanomaterials investigated so far are very comparable. The choice for KNbO3 was essentially motivated by the good stability of the colloidal solution and its good biocompatibility, tested on several human cell lines even at fairly high concentration and long exposition times16.
Given the novelty of the nanomaterial employed for this work, the main characteristics of HNPs as compared to fluorescent/luminescent bio-markers are shown in a short original computer video animation realized by the authors.
Protocol
1. Culture and Expansion of Human ESC
- Prepare the cell propagation medium (called PM) containing Knockout DMEM supplemented with 20% Knockout Serum, 1% MEM Non-Essential Amino Acids, 1% L-glutamine 200 mM, 1% penicillin-streptomycin, and 3.5 µl β-mercaptoethanol.
- Thaw human ESC (hESC) in 8 ml PM medium and centrifuge them 5 min at 115 x g to discard DMSO-supplemented freezing medium.
- Add 1 ml of PM medium containing 10 mM Rock inhibitor to prevent apoptosis and improve cell survival upon thawing (24 hr incubation only).
- Add basic fibroblast growth factor (bFGF) to the PM at the desired concentration (4-10 ng/ml) for maintenance of pluripotency.
- Plate the hESC on irradiated human foreskin feeder cells (γHFF) (Figure 1A), previously plated at a concentration of 2 x 104 cells/cm2.
- Change medium daily and, when necessary, remove portions of colonies starting to differentiate by aspiration using an elongated sharply cut Pasteur pipette.
- Once a week, passage colonies enzymatically:
- Remove medium and wash colonies with 2 ml PBS.
- Remove PBS and incubate them with 0.5 ml per well of warm collagenase IV at 1 mg/ml for 10 min at 37 °C, 5% CO2.
- Collect colony fragments in 2 ml new PM and centrifuge at 115 x g for 3 min.
- Alternatively, colonies can be passaged mechanically by scraping:
- Remove medium and wash colonies with 2 ml PBS.
- Manually scrape colonies using the StemPro EZPassage roller scraper tool (Figure 1B).
- Collect colony fragments and centrifuge then for 3 min at 115 x g to remove the supernatant.
- Plate fragments on γHFF or process them for differentiation into embryoid bodies (EBs).
2. Human ESC Differentiation Protocol
- Prepare the differentiation medium (DM) using Knockout DMEM supplemented with 2% Hyclone serum, 1.0% MEM Non-Essential Amino Acids, 1.0% L-glutamine 200 mM, 1% penicillin-streptomycin, 3.5 µl β-mercaptoethanol.
- Remove medium and wash colonies with 2 ml PBS.
- EB formation of colony fragments in suspension:
- Remove PBS and incubate them with 0.5 ml per well of warm collagenase IV at 1 mg/ml for 10 min at 37 °C, 5% CO2.
- Collect colony fragments in 2 ml new DM and centrifuge at 115 x g for 3 min.
- Culture them in suspension in ultra-low attachment 6-well plates (2 ml per well) for 4 days (Figure 1C).
- Collect and plate the newly formed EBs on 0.1% gelatin-coated 24-well plates (about 3 EBs/well) (Figure 1D).
- Alternatively, forced aggregation of single cells into EBs using the Aggrewell plate:
- Remove PBS and dissociate colonies into single cells using accutase (0.5 ml/well).
- Centrifuge and resuspend cells in DM at 1.2 x 106/ml.
- Add 2 ml per Aggrewell chamber for 1 day (Figure 1B').
- Collect newly formed EBs (Figure 1C') and plate them on 0.1% gelatin-coated 24-well plates (about 3 EBs/well).
- Change DM every 2-3 days until the appearance of beating clusters of cardiomyocytes (15-30 days).
3. Air-liquid 3D Cultures of Beating Clusters and Labeling with HNPs
- Identify and manually dissect clusters of beating cardiomyocytes (Figure 1D), normally appearing after 2-4 weeks of culture, using an elongated sharply cut Pasteur pipette.
- Deposit 4 PTFE filters per insert that will be placed on a 6-well plate (Figure 1F).
- Add 1 ml of DM medium on top of each well.
- Add one dissected beating cluster on each PTFE filter (Figure 1E), maximum 4/well, i.e. 24 aggregates per plate (Figure 1F').
- Change DM medium every 2-3 days.
- To label the clusters, remove the PTFE filter containing the beating cluster and put it on 3.5 cm glass bottom dish (170 µm thick).
- Add 1 ml of HNP at a concentration of 50 µg/ml for 30 min.
4. Non-linear Optical Imaging
- Sample preparation. After HNP labeling (see Step 3.6), transfer either whole differentiating early hEBs or cardiac beating clusters (dissected upon contraction appearance) on PTFE filters over a 170 µm thick glass-bottom dish compatible with the working distance of high numerical aperture objectives. Alternatively, apply a direct coating of beating clusters to the glass-bottom dish pre-coated with 0.1% gelatin.
- Transfer samples to an all-in-one microscope equipped with incubating chamber ensuring stable temperature, humidity and CO2 control over extended periods of time.
- After initial inspection, image the sample via wide-field imaging under white light illumination to identify structures of interest within differentiated EBs or active beating clusters that will be selected for further investigations.
- If cell autofluorescence from NADH and SH from HNPs have to be simultaneously acquired, set a shorter laser excitation wavelength (i.e. 720 nm) to match the molecular absorption. Use one narrow band-pass spectral filter centered at half the excitation wavelength (i.e., λ = 360 ± 10 nm) to detect SH, and another one to detect autofluorescence.
- Firstly perform a fast, low resolution, three-dimensional scan to rebuild the overall morphology of the cardiac cluster and select an image plane within the cluster volume with several visible HNPs.
- If necessary, change the excitation wavelength freely to best match the sample's optical properties or to avoid photobleaching, photo damage and to image deeper into tissues with an IR wavelength (i.e., 790 nm) and replace the second harmonic filter accordingly (i.e., 395 nm). Optimize the settings of the laser pulse pre-compressor at the working wavelength of choice by maximizing the second harmonic signal of HNPs deposited on the sample.
- To monitor in real time the cardiac cluster contractions, set the microscope optical scanner in the fastest mode such as resonant mode, allowing high speed acquisition of two-dimensional images.
- Choose the settings as best compromise between image contrast and acquisition speed, such as:
- Scan averaging: 4
- Pixel dwell time: 0.1 µsec
- Image rate: 3.75 frames per sec (fps)
- Laser power is adjusted according to focal spot size and scanning speed. 50 mW (measured at the entrance of the objective) is a typical value for 0.1 µsec pixel dwell time and Plan APO 20X N.A. 0.75 objective preserving cell viability.
5. Image Analysis
- Axial calibration procedure. Select a single HNP deposited on a bare substrate. Adjust the focus (i.e. the axial position of the nanoparticle) for maximizing the SH intensity. Vary in a controlled way the axial position of the microscope stage to obtain the calibration curve relating SH intensity as a function of the HNP offset from the focal plane. The output of this calibration procedure for the Plan APO 20X N.A. 0.75 objective is reported in Figure 4C.
- Select HNPs present in all video frames and determine their periodic movements. This procedure can be easily automated, for example by a custom code seeking for signal maximum intensity spots in a defined region of interest and connecting them among different frames (an example code written in Matlab R2009 b using the function 'region props' of the Image Processing Toolbox is presented).
- Assess the quantification of the out-of-plane displacements, associated with non-continuously traceable HNPs moving along the optical axis, by converting their intensity modulations throughout different frames into axial displacement using the calibration curve established at Step 5.1. Note that this procedure gives access to axial movements but cannot be used to determine the absolute direction (upwards or downwards).
Representative Results
Prior to assessing the beating activity by confocal imaging, a careful characterization of the nonlinear optical response of the PTFE filters was performed, either alone or in the presence of HNPs at high concentration (1 mg/ml). It was ensured that: i) the bare substrate two-photon excited fluorescence is very weak and cannot prevent measuring the relevant biological samples, and ii) the SH emission from isolated HNPs can be easily acquired by imaging through the substrate in epi-detection mode (Figure 2). The aim was to have reference controls for the quantification of 3D displacement.
In Figure 3, HNP-labeled cardiac structures and EBs are displayed. The red (Figure 3A), yellow (Figures 3B and 3C) and, green (Figure 3D) colors correspond to NADH autofluorescence, while the intense blue SH spots stem from isolated HNP. It is interesting to point out the very good optical contrast achievable by this technique and the relatively sparse labeling as compared to other nanoparticles-based methods, like quantum dots or up-conversion nanoparticles. In the present study, having very intense isolated labels was advantageous for tracking several independent individual particle movements and reconstructing collective movement of the stem cell-derived structures.
Figure 3B illustrates a slice view of a HNP-labeled cardiac beating cluster. Such 3D structures can then be recorded at high speed to monitor the contraction pattern in the cell-HNP aggregate as reported in Movie 1. In this case, to maintain high acquisition speed rate for resolving the NP motion, the overall acquisition sensitivity was not sufficient for recording cell autofluorescence along with SH emission from HNPs.
The application of image analysis described in Section 5 of the protocol applied to the ensemble of movie frames enables the extract of information about individual HNP motion (direction, in-plane and out-of-plane displacement, frequency). The result of this analysis (Figure 4) indicates that, within the same cardiac cluster, the frequency is constant for in- and out-of-plane motion (see Fourier transform in Figure 4B) and displacements are of the order of a few micrometers (the lengths of the arrows indicating the exemplary displacements of five NPs in Figure 4A correspond to the maximal elongation of the oscillations).
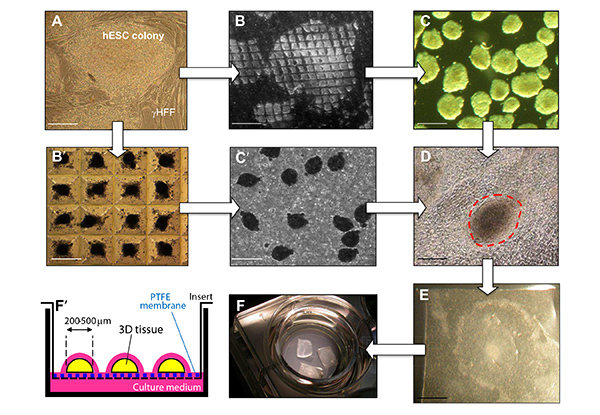
Figure 1. Differentiation of hESC into cardiac beating clusters and labeling with HNPs nanoparticles for second harmonic imaging microscopy. Undifferentiated hESC colonies (A) were either 1) cut down into smaller pieces using the StemPro EZPassage tool (B) and let in suspension to form irregular EBs (C), or 2) dissociated into single cells and reclustered using the Aggrewell system (B') to form regular EBs (C'). Both types of EBs (C or C') were cultured for 2 days in suspension and then adhered to gelatin-coated dishes. Beating clusters of cardiomyocytes (D) were then manually dissected using a scalpel and cultured on PTFE filters (E) deposited on inserts to allow air-liquid 3D cultures (F and F').(scale bars for panels A, B, B',C and C': 100 µm; for panels D and E: 250 µm). Please click here to view a larger version of this figure.
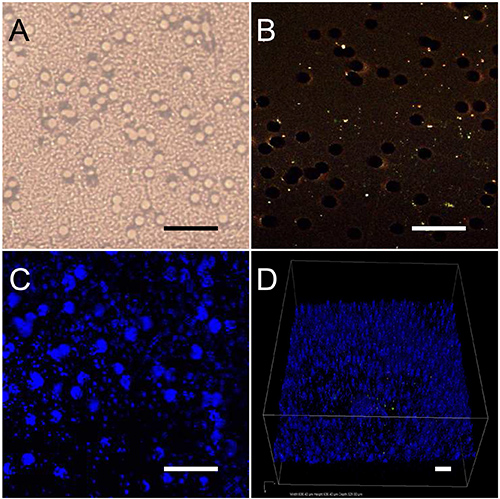
Figure 2. PTFE substrate optical characterization. (A) Bright field image of the PTFE filter alone. (B) Two-photon image of the PTFE filter, which displays a weak fluorescence compared to the rather high laser intensity applied for this control measurement (13 mW). (C and D) After adding to the bare substrate a water drop containing HNPs at 1 mg/ml concentration, their SH emission can be easily acquired through the filter at comparatively low laser intensity (2 mW). In D, a 3D image of the nanoparticles spread on the filter is shown. (scale bars: 50 µm).
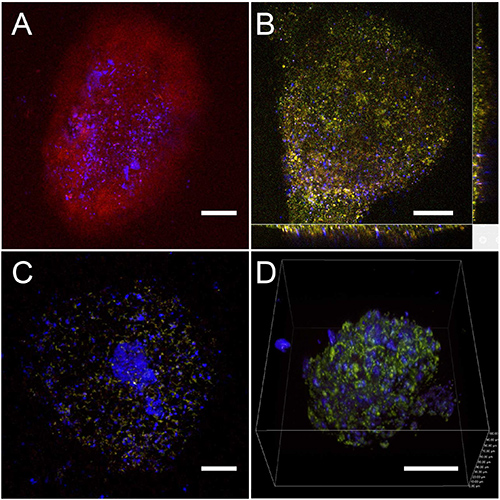
Figure 3. Multiphoton imaging of HNP labeled cardiac structures and EBs. (A) The two-photon fluorescence signal of a beating cluster is displayed in red and the SH signal from the HNPs is shown in blue. The colors correspond to the intensity measured by the four nondescanned photomultipliers equipped with different spectral filters. (Blue 395 ± 11 nm, green 485 ± 20 nm, yellow 531 ± 40 nm, and red 607 ± 70 nm). This cluster was imaged directly through the PTFE porous filter with a 10X objective with an excitation wavelength of 720 nm and a mean power of 8.8 mW. (B) A slice view of a HNP labeled cardiac structure extracted from a z-stack. (C) An EB imaged through the PTFE (scale bars for A, B and C: 100 μm). (D) A 3D image of an EB labeled with HNPs, reconstructed from z-scans using an apochromatic 40X N.A 1.25 water immersion objective (scale bar: 50 µm). The HNPs are well spread around the whole EB and the cardiac cluster, allowing the movement analysis.
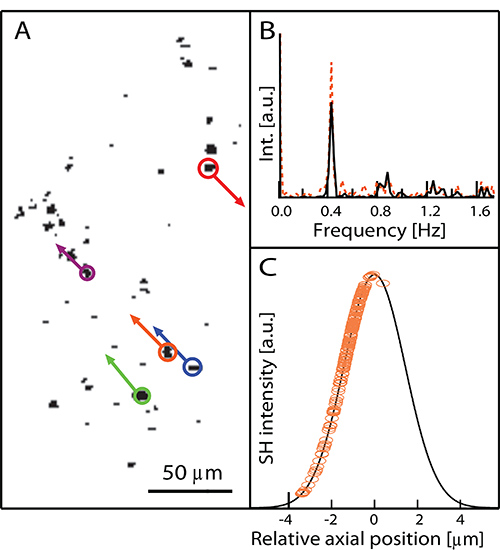
Figure 4. (A) Individual frame of the movie of the beating cluster and vectorial analysis of in-plane HNPs displacements (SH signal shown in black). The lengths of the arrows correspond to the maximal elongation of the oscillations. (B) Fourier transform of NPs oscillations showing that within the same cardiac cluster, the frequency is constant for in- (black line) and out-of-plane motion (orange dotted line) and corresponds to 0.4 Hz. (C) Calibration curve used to convert HNP intensity into axial displacement (See Section 5 of the protocol). Circles: experimental datapoints.
Discussion
The application of nanotechnology to stem cell research is a relatively new but rapidly expanding field. As pointed out by various review articles on the subject, the use of nanoparticles can be applied to accomplish different research tasks, ranging from cell tracking (both in vitro and in vivo), to intracellular delivery of proteins and genes, not least the creation of biomimetic cellular environments for preferential stimulation/inhibition of specific differentiation pathways19,20. The approach described in this protocol is limited to optical tracking and, although the possibility of using infrared excitation leads to increased penetration depth with respect to fluorescence based techniques; optical imaging cannot be extended to whole body and therefore should be complemented by magnetic detection21. Previous published work showed several approaches to assess structure and movement of cardiac cell clusters, including labeling by fluorescent dyes22, quantum dots23,24, super-paramagnetic iron oxide nanoparticles25, and up-conversion fluorescent particles26-28. The advantages of HNPs with respect to these nanoprobes are associated with absence of bleaching over extended periods of time, increased imaging penetration, wavelength tunability for increased contrast with respect to autofluorescence. The peculiar sparse cell labeling (i.e. in Figure 3D we count approximately 160 NPs in a 100 µm side cube) that is obtained by this approach turns out to be very well suited to trace cell contractions in 3D human ESC-derived cardiac clusters at high spatial resolution.
The choice of the nanomaterial is not limited to PEG-coated KNbO3, but can encompass a rich family of nanoparticles displaying noncentrosymmetric crystal structures, which can possibly incorporate other functionalities (magnetic, radioactive) allowing multi-modal detection. Moreover, such PEG-coated HNPs can further be functionalized to be specifically directed against epitopes or binding proteins in order to selectively target specific cell subpopulations. On the other hand, the imaging approach proposed here requires a scanning microscope equipped with an ultrafast laser source. A natural follow up that can be envisaged from this work is monitoring the integration of labeled cells into 3D biomaterials and scaffolding structures in vitro and further into native tissues, to follow the incorporation and the functionality of transplanted cells over extended periods.
An advantage of using PTFE filters bearing 3D aggregates, as in our case, is the possibility to perform multiple measurements over days and weeks, as filters can be restored back on their insert supports without risks of contaminations. More importantly, PTFE filters also enable the repetitive assessment of action potentials using multi-electrode arrays.
Cell damage thresholds for multi-photon excitation in the near-infrared has been previously determined to less than 1 TW/cm2 by König et al29. In our study, using a 0.75 NA 20X objective, the applied intensity remains under the TW/cm2 (0.26 TW/cm2) and the short pixel dwell time (0.1 μsec) as compared to König's work (80 μsec) ensures the possibility to preserve the sample alive and differentiating.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors would like to acknowledge the partial funding from the European FP7 Research Project NAMDIATREAM (NMP4-LA-2010-246479, http://www.namdiatream.eu) and the INTERREG IV France-Switzerland NAOMI project.
Materials
| Microscope Incubator | OKO LAB | UNO package (top stage) | 37°C, 5% CO2, moisturized |
| Multiphoton microscope | Nikon | AR1-MP | |
| Fast scanning, four non photomultiplier descanned detectors | |||
| Filters SHG and autofluorescence | Semrock | FF01-360/12-25 | |
| FF01-395/11-25 | |||
| FF02-485/20-25 | |||
| Microscope objectives | Nikon | CFI Plan Fluor 10x | NA 0.30, WD 16 mm |
| CFI Plan Apo 20x | NA 0.75, WD 1.0 mm, VC | ||
| CFI Apo 40x | NA 1.25, WD 0.18mm λS, Nano-Crystal Coat | ||
| Rhock inhibitor | Sigma | Y-27632 | |
| Knockout DMEM | Invitrogen | 10829 | |
| Knockout Serum | Invitrogen | 10828 | |
| MEM Non-Essential Amino Acids | Invitrogen | 1140 | |
| L-glutamine 200mM | Invitrogen | 25030 | |
| Penicillin-streptomycin | Invitrogen | 15140 | |
| β-mercaptoethanol | Sigma | M7522 | |
| Collagenase IV | Gibco | 17104-019 | |
| Roller scraper tool | StemPro EZPassage, Invitrogen | 23181-010 | |
| StemPro Accutase | Gibco | S11105-01 | |
| Aggrewell system | StemCell Technologies | 27845 | |
| Hyclone serum | Thermo Scientific | SH30070.03 | |
| Gelatin | Sigma | G9391 | |
| 6-well plates | Falcon | 353046 | |
| 24-well plates | Nunclon | 142475 | |
| Polytetrafluoroethylene (PTFE) filters | Millipore | NA76/25 | |
| Inserts | Millipore | PICM03050 |
References
- Bonacina, L., et al. Polar Fe(IO3)3 nanocrystals as local probes for nonlinear microscopy. Applied Physics B-Lasers and Optics. 87, 399-403 (2007).
- Nakayama, Y., et al. Tunable nanowire nonlinear optical probe. Nature. 447, 1098-1101 (2007).
- Aufray, M., et al. New Synthesis of Nanosized Niobium Oxides and Lithium Niobate Particles and Their Characterization by XPS Analysis. J Nanosci Nanotechno. 9, 4780-4785 (2009).
- Hsieh, C. L., Grange, R., Pu, Y., Psaltis, D. Three-dimensional harmonic holographic microcopy using nanoparticles as probes for cell imaging. Opt Express. 17, 2880-2891 (2009).
- Pantazis, P., Maloney, J., Wu, D., Fraser, S. E. Second harmonic generating (SHG) nanoprobes for in vivo imaging. Proc Natl Acad Sci U S A. 107, 14535-14540 (2010).
- Baumner, R., et al. Evanescent-field-induced second harmonic generation by noncentrosymmetric nanoparticles. Opt Express. 18, 23218-23225 (2010).
- Le Xuan, L., et al. Photostable second-harmonic generation from a single KTiOPO4 nanocrystal for nonlinear microscopy. Small. 4, 1332-1336 (2008).
- Sandeau, N., et al. Defocused imaging of second harmonic generation from a single nanocrystal. Opt Express. 15, 16051-16060 (2007).
- Johnson, J. C., et al. Near-field imaging of nonlinear optical mixing in single zinc oxide nanowires. Nano Lett. 2, 279-283 (2002).
- Kachynski, A. V., Kuzmin, A. N., Nyk, M., Roy, I., Prasad, P. N. Zinc oxide nanocrystals for nonresonant nonlinear optical microscopy in biology and medicine. J Phys Chem C. 112, 10721-10724 (2008).
- Bonacina, L. Nonlinear Nanomedecine: Harmonic Nanoparticles toward Targeted Diagnosis and Therapy. Mol Pharmaceut. 10, 783-792 (2013).
- Dempsey, W. P., Fraser, S. E., Pantazis, P. SHG nanoprobes: advancing harmonic imaging in biology. Bioessays. 34, 351-360 (2012).
- Campagnola, P. J., Loew, L. M. Second-harmonic imaging microscopy for visualizing biomolecular arrays in cells, tissues and organisms. Nat Biotechnol. 21, 1356-1360 (2003).
- Denk, W., Strickler, J. H., Webb, W. W. 2-Photon Laser Scanning Fluorescence Microscopy. Science. 248, 73-76 (1990).
- Magouroux, T., et al. High-Speed Tracking of Murine Cardiac Stem Cells by Harmonic Nanodoublers. Small. 8, 2752-2756 (2012).
- Staedler, D., et al. Harmonic Nanocrystals for Biolabeling: A Survey of Optical Properties and Biocompatibility. Acs Nano. 6, 2542-2549 (2012).
- Extermann, J., et al. Nanodoublers as deep imaging markers for multi-photon microscopy. Optics Express. 17, 15342-15349 (2009).
- Chen, I. H., et al. Wavelength dependent damage in biological multi-photon confocal microscopy: A micro-spectroscopic comparison between femtosecond Ti : sapphire and Cr : forsterite laser sources. Opt. Quantum Electron. 34, 1251-1266 (2002).
- Ferreira, L., Karp, J. M., Nobre, L., Langer, R. New opportunities: The use of Nanotechnologies to manipulate and track stem cells. Cell Stem Cell. 3, 136-146 (2008).
- Kaur, S., Singhal, B. When nano meets stem: The impact of nanotechnology in stem cell biology. J Biosci Bioeng. 113, 1-4 (2012).
- Hong, H., Yang, Y. N., Zhang, Y., Cai, W. B. Non-Invasive Imaging of Human Embryonic Stem Cells. Curr Pharm Biotechno. 11, 685-692 (2010).
- Tumbar, T., et al. Defining the epithelial stem cell niche in skin. Science. 303, 359-363 (2004).
- Shah, B. S., Clark, P. A., Moioli, E. K., Stroscio, M. A., Mao, J. J. Labeling of mesenchymal stem cells by bioconjugated quantum dots. Nano Lett. 7, 3071-3079 (2007).
- Zimmer, J. P., et al. Size series of small indium arsenide-zinc selenide core-shell nanocrystals and their application to in vivo imaging. J Am Chem Soc. 128, 2526-2527 (2006).
- Vallee, J. P., et al. Embryonic stem cell-based cardiopatches improve cardiac function in infarcted rats. Stem Cells Transl Med. 1, 248-260 (2012).
- Idris, N. M., et al. Tracking transplanted cells in live animal using upconversion fluorescent nanoparticles. Biomaterials. 30, 5104-5113 (2009).
- Haase, M., Schafer, H. Upconverting nanoparticles. Angew Chem Int Ed Engl. 50, 5808-5829 (2011).
- Cheng, L., et al. Multifunctional Upconversion Nanoparticles for Dual-Modal Imaging-Guided Stem Cell Therapy under Remote Magnetic Control. Adv Funct Mater. 23, 272-280 (2013).
- Konig, K., So, P. T. C., Mantulin, W. W., Gratton, E. Cellular response to near-infrared femtosecond laser pulses in two-photon microscopes. Optics Letters. 22, 135-136 (1997).

