Permanent Ligation of the Left Anterior Descending Coronary Artery in Mice: A Model of Post-myocardial Infarction Remodelling and Heart Failure
Summary
Heart failure is the leading cause of hospitalization and a major cause of mortality. A model of permanent ligation of the left anterior descending coronary artery in mice is applied to investigate ventricular remodelling and cardiac dysfunction post-myocardial infarction. The technique of invasive hemodynamic measurements in mice is presented.
Abstract
Heart failure is a syndrome in which the heart fails to pump blood at a rate commensurate with cellular oxygen requirements at rest or during stress. It is characterized by fluid retention, shortness of breath, and fatigue, in particular on exertion. Heart failure is a growing public health problem, the leading cause of hospitalization, and a major cause of mortality. Ischemic heart disease is the main cause of heart failure.
Ventricular remodelling refers to changes in structure, size, and shape of the left ventricle. This architectural remodelling of the left ventricle is induced by injury (e.g., myocardial infarction), by pressure overload (e.g., systemic arterial hypertension or aortic stenosis), or by volume overload. Since ventricular remodelling affects wall stress, it has a profound impact on cardiac function and on the development of heart failure. A model of permanent ligation of the left anterior descending coronary artery in mice is used to investigate ventricular remodelling and cardiac function post-myocardial infarction. This model is fundamentally different in terms of objectives and pathophysiological relevance compared to the model of transient ligation of the left anterior descending coronary artery. In this latter model of ischemia/reperfusion injury, the initial extent of the infarct may be modulated by factors that affect myocardial salvage following reperfusion. In contrast, the infarct area at 24 hr after permanent ligation of the left anterior descending coronary artery is fixed. Cardiac function in this model will be affected by 1) the process of infarct expansion, infarct healing, and scar formation; and 2) the concomitant development of left ventricular dilatation, cardiac hypertrophy, and ventricular remodelling.
Besides the model of permanent ligation of the left anterior descending coronary artery, the technique of invasive hemodynamic measurements in mice is presented in detail.
Introduction
Heart failure is a syndrome in which the heart fails to pump blood at a rate commensurate with the cellular oxygen requirements at rest or during stress. It is characterized by fluid retention, shortness of breath, and fatigue, in particular on exertion. Heart failure is a growing public health problem, the leading cause of hospitalization, and a major cause of mortality. Ischemic heart disease is the main cause of heart failure1.
Ventricular remodelling refers to changes in structure, size, and shape of the left ventricle. In other words, ventricular remodelling concerns an alteration of the left ventricular architecture. This architectural remodelling of the left ventricle is induced by injury (e.g., myocardial infarction), by pressure overload (e.g., systemic arterial hypertension or aortic stenosis), or by volume overload (e.g., mitral insufficiency). Since ventricular remodelling affects wall stress, it has a profound impact on cardiac function and on the development of heart failure.
Loss of myocardial tissue following acute myocardial infarction results in a decreased systolic ejection and an increased left ventricular end-diastolic volume and pressure. The Frank-Starling mechanism, implying that an increased end-diastolic volume results in an increased pressure developed during systole, may help to restore cardiac output. However, the concomitant increased wall stress may induce regional hypertrophy in the non-infarcted segment, whereas in the infarcted area expansion and thinning may occur. Experimental animal studies show that the infarcted ventricle hypertrophies and that the degree of hypertrophy is dependent on the infarct size2.
The loss of myocardial tissue following acute myocardial infarction results in a sudden increase in loading conditions. Post-infarct remodelling occurs in the setting of volume overload, since the stretched and dilated infarcted tissue increases the left ventricular volume. An increased ventricular volume not only implies increased preload (passive ventricular wall stress at the end of diastole) but also increased afterload (total myocardial wall stress during systolic ejection). Afterload is increased since the systolic radius is increased. Therefore, ventricular remodelling post-myocardial infarction is characterized by mixed features of volume overload and pressure overload.
The myocardium consists of 3 integrated components: cardiomyocytes, extracellular matrix, and the capillary microcirculation. All 3 components are involved in the remodelling process. Matrix metalloproteinases produced by inflammatory cells induce degradation of intermyocyte collagen struts and cardiomyocyte slippage. This leads to infarct expansion characterized by the disproportionate thinning and dilatation of the infarct segment3. In later stages of remodelling, interstitial fibrosis is induced, which negatively affects the diastolic properties of the heart.
The vascular and cardiomyocyte compartment in the myocardium should remain balanced in the process of ventricular remodelling to avoid tissue hypoxia4,5. Whether hypertrophy progresses to heart failure or not may be critically dependent on this balance between the vascular and cardiomyocyte compartment in the myocardium.
A model of permanent ligation of the left anterior descending coronary artery in mice is used to investigate ventricular remodelling and cardiac function post-myocardial infarction. This model is fundamentally different in terms of objectives and pathophysiological relevance compared to the model of transient ligation of the left anterior descending coronary artery. In this latter model of ischemia/reperfusion injury, the initial extent of the infarct may be modulated by factors that affect myocardial salvage following reperfusion6. In contrast, the infarct area at 24 hours after permanent ligation of the left anterior descending coronary artery is fixed. Cardiac function in this model will be affected by 1) the process of infarct expansion, infarct healing, and scar formation; and 2) the concomitant development of left ventricular dilatation, cardiac hypertrophy, and ventricular remodelling.
Protocol
NOTE: All experimental procedures described in this section were approved by the Institutional Animal Care and Research Advisory Committee of the Katholieke Universiteit Leuven (Project: 154/2013-B De Geest).
1. Permanent Ligation of the Left Anterior Descending Coronary Artery
- Anesthetize the mouse by intraperitoneal administration of 40 mg/kg to 70 mg/kg of sodium pentobarbital. Ensure the mouse reaches its proper plane of anesthesia when it no longer reacts a firm toe pinch. Always confirm proper anesthetization this way prior to any surgical procedure or intervention. Use lubricating ophthalmic ointment to prevent dryness of the cornea while under anesthesia. Provide preoperative analgesia 2-4 hr before the start of the procedure (buprenorphine 0.05 mg/kg SQ).
- Apply consistent aseptic techniques during survival surgery. Implement procedures that inhibit to a maximally possible extent microbial contamination so that significant infection or suppuration does not occur. These procedures include use of sterile instruments and sterile materials, disinfection of the surgical area, and removal of fur/hair over the surgical site and disinfection of this site.
- Intubate the mouse with a self-prepared blunted 20-gauge needle.
- Put the mouse in supine position with hyperextension of the head.
- Focus the light on the neck region. Lift the tongue with a blunted pincet. The entrance of the larynx can be clearly seen.
- Pass the blunted needle through the larynx into the trachea under direct vision. Assess correct intubation by connecting the mouse to the ventilator (stroke volume in µl: 3 x body weight (g) + 155; frequency: 120 strokes per min).
- Alternatively, enhance visualization of endotracheal intubation by first carefully exposing the trachea.
- Make a 5 mm mid-neck incision and retract muscle tissue just above the trachea.
- Perform the intubation using a surgical stereomicroscope for direct visualization of the trachea. Lift the tongue and enter the self-prepared blunted 20 gauge needle into the trachea. Confirm correct intubation by connecting the mouse to the ventilator (stroke volume in µl: 3 x body weight (g) + 155; frequency: 120 strokes per min).
- Put the mouse in supine position with hyperextension of the head.
- Keep the mouse in supine position and fix the mouse with tape. Perform surgery on a heating pad to prevent hypothermia.
- Shave and disinfect the skin with Betadine. Take care that the left hind limb crosses the right hind limb in order to obtain a better view on the left ventricle during surgery.
- Make a small transverse cutaneous incision up to the sternum and separate the underlying skin and muscles.
- Pull aside the m. pectoralis minor and m. pectoralis major with a 5-0 silk suture.
- Make an incision in the third intercostal space by inserting a blunted pincet.
- Move the pincet under the intercostal muscles from lateral to medial till the sternum is reached. Puncture the thoracic wall by pushing the pincet from inside to the skin. Complete the thoracotomy by carefully cutting the intercostal muscle just above the pincet with a small scissor. Use this technique to prevent puncturing of the lungs.
- Put a sponge immersed in 0.9% NaCl into the cavity to protect the lungs. Introduce a wound spreader (chest retractor) into the intercostal space to obtain exposure of the left side of the heart. As of now, the left atrium, the left ventricle, and the left anterior descending coronary artery are visible under the stereomicroscope.
- Perform a ligation of the left anterior descending coronary artery with a single 6-0 prolene ligature about 1 mm under the tip of the left atrium. This is distal from the first diagonal branch.
NOTE: Alternatively, 7-0 (0.05 mm diameter) or 8-0 threads (0.04 mm diameter) may be used. The needle is a C-1 13 mm 3/8 circle Taper Point needle. Successful ligation of the left anterior descending coronary artery induces immediate discoloration, resulting in a pale appearing myocardium in the affected territory. - Remove the wound spreader (chest retractor).
- Place three 6-0 Ti-Cron sutures around the intercostal space. Before tightening the sutures, remove the sponge from the chest cavity and re-expand the lungs by blocking the outflow of the ventilator. By doing so, the lungs reconnect with the parietal pleura.
- Subsequently, pull the sutures tight and repeat re-expansion by pressing down on the chest. Confirm the successful closure of the thorax using a small amount of saline (no air bubbles must be seen when applying pressure on the chest).
- Look through the intercostal muscle to confirm normal expansion of the lungs. Reposition both pectoralis muscles, serving as an extra barrier for prevention of a pneumothorax.
- Close the skin with 5-0 silk-sutures.
- Disconnect the mouse from the ventilator and allow recovery on the heating pad. Do not leave an animal unattended until it has regained sufficient consciousness to maintain sternal recumbency. Do not return an animal that has undergone surgery to the company of other animals until fully recovered.
- Consistently provide postoperative analgesia (buprenorphine 0.05 mg/kg SQ BID for at least 48 hr after surgery).
2. In vivo Invasive Hemodynamic Measurements in Mice
- Before the procedure, submerge the 1.0 French Millar pressure catheter in sterile water at 37 °C for at least 30 min to minimize signal drift. Electronically calibrate the pressure sensor at 0 mm Hg and 100 mm Hg and record data at 2,000 Hz.
- Perform anesthesia by intraperitoneal administration of 1.4 g/kg urethane. Check that the mouse reaches its proper plane of anesthesia when it no longer reacts a firm toe pinch.
- Place the anesthetized mouse in supine position. Secure its limbs with tape. Maintain body temperature with a heating pad and monitor with a rectal probe. Shave the neck region and make a midline incision in the neck region to expose the thyroid gland.
- Fix the neck with bent needles.
- Pull aside the salivary gland and expose the right common carotid artery. The vagus nerve, which resembles a white thread, lies along the artery. Carefully separate the carotid artery from the vagus nerve using a curved forceps.
- Pass a curved forceps under the right common carotid artery to separate it from other tissues. Remove connective tissue around the artery.
- Pass two 6-0 silk wires under the right common carotid artery. Make a tight knot on the top wire, which is placed towards the head, close, and fix with a kocher (distal occlusive ligation). Pass the proximal wire twice from left to right and fix with 2 kochers (proximal non-occlusive wire).
- Keep the carotid artery moist by dropping sterile 0.9% NaCl. Dry off excess liquid with cotton buds.
- Make an incision in the right common carotid artery with a 26-gauge needle between the distal ligation and the proximal non-occlusive wire.
- Introduce the pressure sensor into the artery. Verify that there is no blood loss. Gently push the 1.0 French Millar pressure catheter forward and adjust the proximal non-occlusive wire in such a way that the catheter can pass carefully through the wire under the clavicle.
- Minimize blood loss during adjustment of the proximal non-occlusive wire. Do not compress the pressure sensor too much while advancing since it is very fragile. Since the proximal wire must not occlude the artery, the vessel should remain filled with blood.
- Start recording the pressure signal. An arterial pressure signal fluctuates in a healthy mouse between a diastolic pressure of 60 – 70 mm Hg and a systolic pressure of 100 – 120 mm Hg.
- Direct the catheter via the innominate artery and via the aorta into the left ventricle. The ventricular pressure fluctuates between 0 mm Hg and 100 – 120 mm Hg. Allow the catheter to stabilise inside the left ventricle. Record the signal for 30 min to 60 min depending on the experimental requirements.
- After completion of the experiment, soak the catheter in Alconox 1% for 30 min. Wash the catheter with Milli-Q water. Store the catheter in a foam block.
- Retrieve data from the recording software for further analysis.
- For data analysis, consider a time interval where the pressure signal is stable. Choose at least 10 consecutive cardiac cycles of the recorded data of interest.
- Use LabChart software version 8.0 or similar to analyse the heart rate, the maximal systolic left ventricular pressure, the minimal diastolic left ventricular pressure, the peak rate of isovolumetric left ventricular contraction (dP/dtmax), the peak rate of isovolumetric left ventricular relaxation (dP/dtmin), the end-diastolic left ventricular pressure, and the time constant of the isovolumetric left ventricular pressure fall (tau)7.
NOTE: The end-diastolic pressure corresponds to the pressure at the time-point immediately before the pressure surge induced by isovolumetric contraction. The calculation of tau is based on fitting the left ventricular pressure into a monoexponential decay curve, expressed as P(t)=P0e-t/tau + b, In this formula, P(t) is the left ventricular pressure at time t after the maximal negative value of dP/dt has been reached. The parameter b corresponds to the theoretical asymptote, which in a simplified approach can be assumed to be zero. Enhanced isovolumetric relaxation results in a smaller value of tau.
Representative Results
The extent of myocardial infarction can be assessed by Evans blue/2,3,5-triphenyltetrazolium chloride (TTC) double staining. TTC is a redox indicator, which is converted to deep-red 1,3,5-triphenylformazan in living tissues due to the activity of various dehydrogenases in the presence of NADH8. Figure 1 shows a representative section of the heart at 24 hr after ligation of the left anterior descending coronary artery. Blue-stained areas indicate non-ischemic/normal regions. The myocardial area at risk is defined as the myocardial tissue within the perfusion bed distally from the ligation of the left anterior descending coronary artery. Deep-red-stained areas indicate ischemic but viable regions (non-infarcted area at risk) regions whereas negatively stained areas (pale red) indicate infarcted regions. In this model, the non-infarcted area at risk is negligible and not visible in this image, which reflects the permanent character of the ligation of the left anterior descending coronary artery. The infarcted area is typically between 50 – 60% of the total left ventricular wall area9,10. Two components in the evolution of the infarct zone should be discerned: 1) the three-dimensional geometry of the infarct may alter as a consequence of infarct expansion leading to a thinner but more extended myocardial infarction, and 2) the total volume of the infarct may decrease, which reflects the healing process with wound contraction and scar formation.
Infarct expansion can be quantified by evaluating the time-course of infarct length and infarct thickness10. A representative Sirius Red stained cross-section of a heart at day 28 after permanent ligation of the left anterior descending coronary artery is shown in Figure 2. The image shows an infarct that has been substantially stretched. In a cross-section, this expansion corresponds to an increase of the absolute infarct length and a decrease of infarct thickness.
Besides considerations on the three-dimensional geometry and the volume of the infarct, another caveat with regard to interpretation of infarct parameters should be considered. Since the viable myocardial tissue will also undergo hypertrophy, it is clear that the ratio of infarct area versus total left ventricular wall area will decrease as a function of time. Assessment of longitudinal changes of the infarct zone requires therefore a clear insight in the difference between absolute parameters and relative parameters.
An arterial and ventricular pressure register is shown in Figure 3. After stabilization of the catheter, heart rate, maximal systolic left ventricular pressure, minimal diastolic left ventricular pressure, the peak rate of isovolumetric left ventricular contraction (dP/dtmax), the end-diastolic left ventricular pressure, and the peak rate of isovolumetric left ventricular relaxation (dP/dtmin) are determined. The time constant of isovolumetric left ventricular pressure fall (tau) is quantified using the method of Weiss et al.7.

Figure 1. Assessment of area at risk and infarct size 1 day after myocardial infarction. The images from left to right go from the cardiac apex to the base of the heart. 24 hr after permanent ligation of the left anterior descending coronary artery, 2 ml of Evans blue dye was injected as a bolus into the aorta to quantify the volume of the perfused myocardium. Hearts were subsequently arrested in diastole by injection of CdCl (100 µl; 0.1 N), were flushed with physiological saline to wash out excess blue dye, and were embedded in 5% low gelling temperature agarose. Afterwards, 500 µm thick cross-sections were made using a HM 650 V Vibration Microtome and slices were then incubated in a 1.5% 2,3,5-triphenyltetrazolium chloride containing isotonic phosphate buffer (pH 7.4) for 30 min at 37 °C. Images were made using a SteREO Lumar V.12 microscope. The negatively stained area of infarcted tissue, the red-stained non-infarcted area at risk, and the blue-stained non-ischemic healthy ventricular wall area were quantified using Image J software. Scale bar represents 0.5 mm. Please click here to view a larger version of this figure.
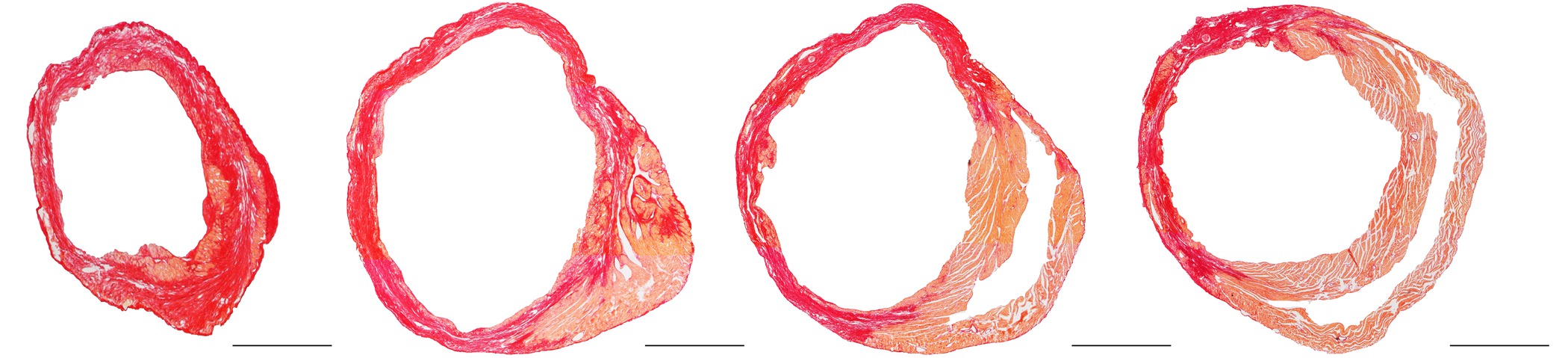
Figure 2. Representative Sirius Red stained cross-section of a heart at day 28 after ligation of the left anterior descending coronary artery. The images from left to right go from the cardiac apex to the base of the heart. The standard protocol of Junqueira et al.11 was applied for Sirius Red staining. This histochemical method stains fibrotic infarct tissue dark red and healthy tissue orange. Scale bar represents 1 mm. Please click here to view a larger version of this figure.
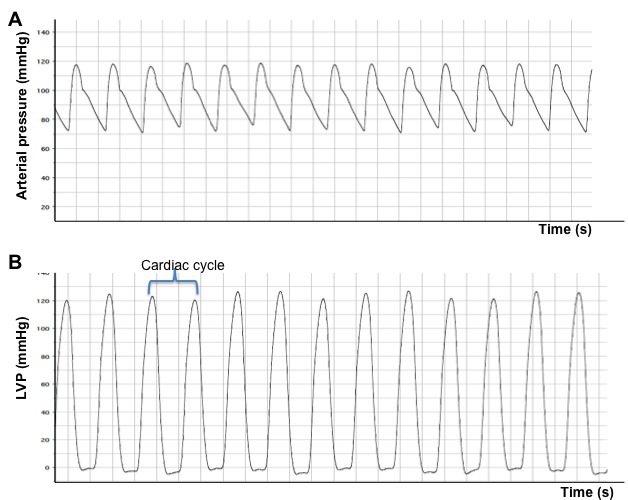
Figure 3. Arterial (A) and left ventricular (B) pressure registers obtained after catheterization of normal mice. The obvious difference between an arterial signal and a ventricular signal is that the latter signal drops to approximately 0 mm Hg during diastole. LVP: left ventricular pressure.
Discussion
Chronic changes in myocardial structure and function, the development of left ventricular dysfunction, and progression to heart failure can be investigated in several murine models12. Cardiac remodelling and dysfunction can be induced by myocardial injury or by pressure overload secondary to transverse aortic constriction, or may be investigated in genetic models of dilated cardiomyopathy12. Obviously, the most pronounced benefit of murine models is the availability of a great number of transgenic and knock-out strains including cell-type specific and inducible transgenic models. Evaluation of cardiac remodelling in these models has been greatly facilitated by the development of technologies like ultrahigh resolution 2D- and 3D-echocardiography, micro-magnetic resonance imaging, hemodynamic evaluation using micromanometry, and micromanometer conductance technology for pressure volume loop analyses. In this report, we have focused on the model of myocardial injury induced by permanent ligation of the left anterior descending coronary artery and evaluation of left ventricular function by left ventricular micromanometry.
The first murine model of myocardial infarction was described in 1978 by Zolotareva et al13. This model of permanent occlusion should be clearly discerned from the murine model of transient occlusion of the left anterior descending coronary artery that was first described by Michael et al14. This latter model is used to investigate ischemia-reperfusion injury and myocardial salvage whereas permanent ligation is applied to study the pathophysiology of post-myocardial infarction cardiac remodelling.
The coronary artery tree of mice is different compared to humans15-17. The septal coronary artery in the mouse is variable in origin15-17. It arises classically as a branch of the right coronary artery18,19, but may also arise from a separate ostium in the right sinus of Valsalva17, or exceptionally from the left coronary artery16. The anatomy of the coronary artery tree may slightly differ between different strains20. However, ligation of the left anterior descending coronary artery as it emerges immediately under the left auricle results in reproducible large infarctions involving the anterolateral, posterior, and apical regions of the heart as demonstrated by histology and echocardiography15,17. Importantly, the septum is spared from infarction, which reflects the presence of the distinct septal coronary artery. In contrast to mice, blood supply of the septum in humans is mainly provided by branches of the left anterior descending coronary artery21. This difference in anatomy and the resulting sparing of the septum in mice will affect remodelling of the heart.
As stated in the results section, ligation of the left anterior descending coronary artery in mice induces a large myocardial infarction that comprises 50% to 60% of the left ventricle9,10. The occurrence of ventricular rupture in the first two weeks after myocardial infarction in mice is frequent22,23. This complication of myocardial infarction is strain-dependent and occurs significantly more often in male mice than in female mice22. Furthermore, ventricular rupture happens more frequently in hypercholesterolemic mice than under conditions of normocholesterolemia9. The occurrence of ventricular rupture in mice should be put in the right perspective. First of all, this complication is a manifestation of infarct expansion. This disproportionate thinning and dilatation of the infarct segment not only induces myocardial rupture but is also a trigger for cardiac remodelling and for congestive heart failure24-27. Thus, the frequency of myocardial rupture is an indicator for the degree of infarct expansion and thus for the extent of cardiac remodelling and development of cardiac dysfunction in surviving mice.
The second important consideration with regard to ventricular rupture is the necessity to take survival bias into account when functional or structural data are compared between two groups in an intervention study10. It is reasonable to assume that mice with the most pronounced infarct expansion will succumb to ventricular rupture. In case of a significant difference of survival rate between two groups, one should be aware that died mice are excluded from the functional or structural analysis. Consequently, bias is introduced in any comparison of mice surviving at a specific time-point.
As discussed above, many different technologies are available to evaluate cardiac remodelling after myocardial infarction. However, the availability of advanced and complex technologies may be limited for several investigators. Hemodynamic evaluation using a micromanometer pressure sensor introduced via the common carotid artery is relatively straightforward and significantly less expensive than other technologies. However, it should be stressed that there are inherent limitations to the interpretation of left ventricular micromanometry. In contrast, analysis of ventricular pressure-volume relationships (e.g., using conductance micromanometry) constitutes the most rigorous and comprehensive approach to evaluate intact cardiac function28. The unique advantage of pressure volume methodology to measure cardiac function is that it enables more specific measurements of the left ventricular performance independently of loading conditions and of the heart rate29.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by Onderzoekstoelagen grant OT/13/090 of the KU Leuven and by grant G0A3114N of the FWO-Vlaanderen.
Materials
| Reagents | |||
| Buprenorphine (Buprenex®) | Bedford Laboratories | ||
| Sodium Pentobarbital (Nembutal®) | Ceva | ||
| Betadine® | VWR internationals | 200065-400 | |
| 5 – 0 silk suture | Ethicon, Johnson & Johnson Medical | K890H | |
| 6 – 0 prolene suture | Ethicon, Johnson & Johnson Medical | F1832 | |
| 6 – 0 Ti- Cron suture | Ethicon, Johnson & Johnson Medical | F1823 | |
| Urethane | Sigma | 94300 | |
| Alconox | Alconox Inc. | ||
| Equipment | |||
| Ventilator, MiniVent Model 845 | Hugo Sachs | 73-0043 | |
| Chest retractor or Thorax retractor | Kent Scientific corporation | INS600240 | ALM Self-retaining, serrated, 7cm long, 4 x 4 "L" shaped prongs, 3mm x 3mm |
| 1.0 French Millar pressure catheter | Millar Instruments | SPR – 1000/NR | |
| Powerlab | ADInstruments Pty Ltd. | ||
| LabChart® software | ADInstruments Pty Ltd. | ||
| Rectal probe | ADInstruments Pty Ltd. |
References
- He, J., et al. Risk factors for congestive heart failure in US men and women: NHANES I epidemiologic follow-up study. Arch Intern Med. 161, 996-1002 (2001).
- Anversa, P., Sonnenblick, E. H. Ischemic cardiomyopathy: pathophysiologic mechanisms. Prog Cardiovasc Dis. 33, 49-70 (1990).
- Erlebacher, J. A., Weiss, J. L., Weisfeldt, M. L., Bulkley, B. H. Early dilation of the infarcted segment in acute transmural myocardial infarction: role of infarct expansion in acute left ventricular enlargement. J Am Coll Cardiol. 4, 201-208 (1984).
- Shimizu, I., et al. Excessive cardiac insulin signaling exacerbates systolic dysfunction induced by pressure overload in rodents. J Clin Invest. 120, 1506-1514 (2010).
- Tirziu, D., et al. Myocardial hypertrophy in the absence of external stimuli is induced by angiogenesis in mice. J Clin Invest. 117, 3188-3197 (2007).
- Theilmeier, G., et al. High-density lipoproteins and their constituent, sphingosine-1-phosphate, directly protect the heart against ischemia/reperfusion injury in vivo via the S1P3 lysophospholipid receptor. Circulation. 114, 1403-1409 (2006).
- Weiss, J. L., Frederiksen, J. W., Weisfeldt, M. L. Hemodynamic determinants of the time-course of fall in canine left ventricular pressure. J Clin Invest. 58, 751-760 (1976).
- Bohl, S., et al. Refined approach for quantification of in vivo ischemia-reperfusion injury in the mouse heart. Am J Physiol Heart Circ Physiol. 297, 2054-2058 (2009).
- Van Craeyveld, E., Jacobs, F., Gordts, S. C., De Geest, B. Low-density lipoprotein receptor gene transfer in hypercholesterolemic mice improves cardiac function after myocardial infarction. Gene Ther. 19, 860-871 (2012).
- Gordts, S. C., et al. Beneficial effects of selective HDL-raising gene transfer on survival, cardiac remodelling and cardiac function after myocardial infarction in mice. Gene Ther. 20, 1053-1061 (2013).
- Junqueira, L. C., Bignolas, G., Brentani, R. R. Picrosirius staining plus polarization microscopy, a specific method for collagen detection in tissue sections. Histochem J. 11, 447-455 (1979).
- Patten, R. D., Hall-Porter, M. R. Small animal models of heart failure: development of novel therapies, past and present. Circ Heart Fail. 2, 138-144 (2009).
- Zolotareva, A. G., Kogan, M. E. Production of experimental occlusive myocardial infarction in mice. Cor Vasa. 20, 308-314 (1978).
- Michael, L. H., et al. Myocardial ischemia and reperfusion: a murine model. Am J Physiol. 269, 2147-2154 (1995).
- Salto-Tellez, M., et al. Myocardial infarction in the C57BL/6J mouse: a quantifiable and highly reproducible experimental model. Cardiovasc Pathol. 13, 91-97 (2004).
- Fernandez, B., et al. The coronary arteries of the C57BL/6 mouse strains: implications for comparison with mutant models. J Anat. 212, 12-18 (2008).
- Kumar, D., et al. Distinct mouse coronary anatomy and myocardial infarction consequent to ligation. Coron Artery Dis. 16, 41-44 (2005).
- Clauss, S. B., Walker, D. L., Kirby, M. L., Schimel, D., Lo, C. W. Patterning of coronary arteries in wildtype and connexin43 knockout mice. Dev Dyn. 235, 2786-2794 (2006).
- Icardo, J. M., Colvee, E. Origin and course of the coronary arteries in normal mice and in iv/iv mice. J Anat. 199, 473-482 (2001).
- Yoldas, A., Ozmen, E., Ozdemir, V. Macroscopic description of the coronary arteries in Swiss albino mice (Mus musculus). J S Afr Vet Assoc. 81, 247-252 (2010).
- James, T. N., Burch, G. E. Blood supply of the human interventricular septum. Circulation. 17, 391-396 (1958).
- Gao, X. M., Xu, Q., Kiriazis, H., Dart, A. M., Du, X. J. Mouse model of post-infarct ventricular rupture: time course, strain- and gender-dependency, tensile strength, and histopathology. Cardiovasc Res. 65, 469-477 (2005).
- Muthuramu, I., Jacobs, F., Singh, N., Gordts, S. C., De Geest, B. Selective homocysteine lowering gene transfer improves infarct healing, attenuates remodelling, and enhances diastolic function after myocardial infarction in mice. PLoS One. 8, 63710 (2013).
- Eaton, L. W., Weiss, J. L., Bulkley, B. H., Garrison, J. B., Weisfeldt, M. L. Regional cardiac dilatation after acute myocardial infarction: recognition by two-dimensional echocardiography. N Engl J Med. 300, 57-62 (1979).
- Erlebacher, J. A., et al. Late effects of acute infarct dilation on heart size: a two dimensional echocardiographic study. Am J Cardiol. 49, 1120-1126 (1982).
- Schuster, E. H., Bulkley, B. H. Expansion of transmural myocardial infarction: a pathophysiologic factor in cardiac rupture. Circulation. 60, 1532-1538 (1979).
- Jugdutt, B. I., Michorowski, B. L. Role of infarct expansion in rupture of the ventricular septum after acute myocardial infarction: a two-dimensional echocardiographic study. Clin Cardiol. 10, 641-652 (1987).
- Pacher, P., Nagayama, T., Mukhopadhyay, P., Batkai, S., Kass, D. A. Measurement of cardiac function using pressure-volume conductance catheter technique in mice and rats. Nat Protoc. 3, 1422-1434 (2008).
- Vanden Bergh, A., Flameng, W., Herijgers, P. Parameters of ventricular contractility in mice: influence of load and sensitivity to changes in inotropic state. Pflugers Arch. 455, 987-994 (2008).

