The Pilocarpine Model of Temporal Lobe Epilepsy and EEG Monitoring Using Radiotelemetry System in Mice
Summary
This manuscript describes a method of inducing status epilepticus by systemic pilocarpine injection and of monitoring spontaneous recurrent seizures in live animals using a wireless telemetry video and electroencephalogram system. This protocol can be utilized for studying the pathophysiologic mechanisms of chronic epilepsy, epileptogenesis, and acute seizures.
Abstract
Temporal lobe epilepsy (TLE) is a common neurological disorder in adulthood. For translational studies of chronic epilepsy, pilocarpine-induced status epilepticus (SE) is frequently selected to recapitulate spontaneous recurrent seizures (SRS). Here we present a protocol of SE induction by intraperitoneal (i.p.) injection of pilocarpine and monitoring of chronic recurring seizures in live animals using a wireless telemetry video and electroencephalogram (EEG) system. We demonstrated notable behavioral changes that need attention after pilocarpine injection and their correlation with hippocampal neuronal loss at 7 days and 6 weeks post-pilocarpine. We also describe the experimental procedures of electrode implantation for video and EEG recording, and analysis of the frequency and duration of chronic recurrent seizures. Finally, we discuss the possible reasons why the expected results are not achieved in each case. This provides a basic overview of modeling chronic epilepsy in mice and guidelines for troubleshooting. We believe this protocol can serve as a baseline for suitable models of chronic epilepsy and epileptogenesis.
Introduction
TLE is one of the most common acquired epilepsies1. People with epilepsy experience recurrent seizures as a result of abnormal neuronal activities in the brain2,3. Given that TLE is often intractable, it is crucial to understand the basic mechanisms underlying the development of epilepsy.
Animal models that can recapitulate the key characteristics of human TLE can offer better appreciation of TLE pathophysiology, allowing us to readily monitor and manipulate critical factors in epileptogenesis. Among them, chemoconvulsants-induced SE has been widely used4,5. Unlike other epilepsy models, such as electrical stimulation which shows no hippocampal sclerosis and robust SRS6,7,8, the systemic injection of chemoconvulsants can mimic clinical pathogenesis of human TLE, i.e., initial brain injury, a latent period, and a chronic epileptic stage manifesting SRS5,9,10. Therefore, this technique can be utilized in various studies explaining the mechanisms of acute brain damage, epileptogenesis, or seizure suppression. Moreover, histopathological alterations induced by chemoconvulsants are similar to those seen in human TLE, providing an additional rationale for use of TLE rodent models10,11,12. Notably, structural damages involving the hippocampus have been consistently reproduced in both kainic acid- and pilocarpine-induced SE models. However, compared to kainic acid injection, the pilocarpine model can produce more robust SRS in mice, which can offer sizable advantages for studying chronic epilepsy when considering the wide availability of transgenic mouse lines5,13,14,15. Moreover, seizure progression after pilocarpine injection is generally faster than in the kainic acid model, providing additional evidence for the effective use of a pilocarpine model of epilepsy.
Here, we demonstrate a method of inducing SE by the i.p. injection of pilocarpine and by performing video and EEG monitoring in chronic epilepsy.
Protocol
All experimental procedures were approved by the Ethics Committee of the Catholic University of Korea and were carried out in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publications No. 80-23).
1. SE Induction
- Purchase 8-week-old male C57BL/6NHsd mice and weigh each mouse. Then, use a marker pen to mark the tails of all mice for their easy identification during SE induction.
- Calculate the amount of scopolamine methyl bromide (scopolamine; 2 mg/kg), terbutaline hemisulfate (terbutaline; 2 mg/kg), and pilocarpine hydrochloride (pilocarpine; 280 mg/kg) based on the mouse weight and add saline (0.9% NaCl, 10 mL/kg) to make solutions.
Note: For example, if the weight of the mouse is 25 g, the following amounts are applied: Scopolamine and terbutaline: 2 mg/kg * (25 g/1,000 g) = 0.05 mg, Saline: 10 mL/kg * (25 g/1,000 g) = 0.25 mL, Pilocarpine: 280 mg/kg * (25 g/1,000 g) = 7 mg, Saline: 10 mL/kg * (25 g/1,000 g) = 0.25 mL. - Load the scopolamine and terbutaline mixture in a 1 mL syringe with a 30 G needle. Inject the solution intraperitoneally into each mouse, and then return the mice to their cages. At 30 min after scopolamine and terbutaline administration, inject pilocarpine solution into each mouse (i.p.; 1 mL syringe; 30 G needle). Immediately after pilocarpine injection, place the mice in an incubator (28-30°C) for observation.
- Carefully monitor the behavior of the mice until SE is induced. If limbic motor seizures that correspond to stage 3 or higher according to Racine's scale are detected, record the time and monitor the mice to determine whether the seizures occur more frequently. Once continuous motor seizures last more than 2 min, place the mouse in a new cage at room temperature and keep monitoring for 3 h to determine whether their convulsive seizures continue and SE is induced. Euthanize the mice that failed to enter SE at about 2 h after pilocarpine injection.
Note: Racine's scale; stage 1, mouth and facial movement; stage 2, head nodding; stage 3, forelimb clonus; stage 4, rearing with forelimb clonus; stage 5, rearing and falling with forelimb clonus16. If the mouse is not transferred to the cage at room temperature immediately after SE induction, the mouse can die due to hyperthermia. - Terminate behavioral acute seizures at 3 h after SE onset by injecting diazepam solution (i.p.; 10 mg/kg; 1 mL syringe; 30 G needle). Make the diazepam solution by diluting 5 mg/mL of diazepam in 10% polyoxyl 35 hydrogenated castor oil by adding saline (i.p.; 10 mL/kg; 1 mL syringe; 30 G needle).
Note: 10% polyoxyl 35 hydrogenated castor oil solution: 1 mL polyoxyl 35 hydrogenated castor oil solution + 9 mL saline. Store the solution at room temperature. For example, if the mouse body weight is 25 g, inject 250 µL of diazepam solution: 50 µL 5 mg/mL diazepam + 200 µL 10% polyoxyl 35 hydrogenated castor oil solution = 250 µL 1 mg/mL diazepam. - For sham mice, perform an i.p. injection of the scopolamine methyl bromide and terbutaline hemisulfate mixture (i.p.; both 2 mg/kg, 10 mL/kg; 1 mL syringe; 30 G needle). 30 min later, inject the saline intraperitoneally (i.p.; 10 mL/kg; 1 mL syringe; 30 G needle).
Note: If the mouse body weight is 25 g, inject 250 µL of saline instead of pilocarpine. - After diazepam injection (i.p.; 1 mL syringe; 30 G needle), administer 1 mL of 5% dextrose per individual mouse (i.p.; 1 mL syringe; 26 G needle).
Note: 1 mL of 5% dextrose injection provides energy source and hydration that can increase the survival rate. - During post-care in the incubator (28-30 °C), wipe off excessive secretions such as saliva, tears, and feces.
- At day 1 after SE induction, weigh the mice and keep them in the incubator (28-30 °C) for one extra day. At day 2, after SE induction, weigh the mice and return them to their home cage. Provide moist chow to facilitate their recovery.
Note: In this experiment, the mortality rate for C57BL/6NHsd after SE termination was 8.57% on average (3 out of 35 mice tested). - Measure daily body weight of the animals until 7 days post-pilocarpine and inject the mice with 5% dextrose(i.p.; 1 mL syringe; 26 G needle) when the body weight has not increased. Stop body weight monitoring when the mice start to regain body weight and consume moist chow without difficulty at 2 days after pilocarpine injection.
Note: If the body weight of a mouse did not increase until 7 days post SE, exclude the mouse from the experiment and euthanize by carbon dioxide. Depending on the background of the mouse, death can occasionally occur; however, in the case of C57BL/6NHsd, less than 1% of mice died in these experiments. With survival of more than 7 days after SE induction, the mice rarely die during the latent period.
2. Implantation Surgery for Video-EEG Monitoring
- Prepare 12-week-old mice (4 weeks after SE induction) to perform telemetry implantation for EEG monitoring.
Note: Timing of implantation can be modified depending on the experimental purposes and the mouse strains. - Before surgery, anesthetize the mouse with a mixture (4:0.5) of ketamine (50 mg/mL) and xylazine (23.3 mg/mL) solution dissolved in saline at a dose of 2.5 µL/g body weight (i.p.; 1 mL syringe; 26 G needle). Monitor respiratory rate and respiratory effort of the mouse at regular intervals (15 min interval maximum) and assess the depth of anesthesia by the pedal withdrawal reflex.
- Place the mouse in a stereotaxic frame with ear bars and a bite plate and apply vet ointment on both eyes to avoid blindness.
- To maintain sterile conditions during the surgery, shave surgical sites using a razor blade. Be careful to prevent fur contamination of the surgical field. After shaving, disinfect the skin with 70% ethanol and iodine solution.
Note: Surgery should be performed in an aseptic manner wearing sterile gloves and masks, using sterilized equipment and aseptic techniques. - After confirming the depth of anesthesia, make a midline incision of the skin to expose the skull. Make two burr holes with a drill attached to the stereotaxic apparatus at the coordinates from the Bregma in the anteroposterior axis (AP): +0.1 mm, mediolateral axis (ML): +0.1 mm (Reference), and AP: −0.2 mm, ML: +0.22 mm (Left parietal cortex)13.
- Wipe the skin with PBS-soaked cotton swaps, followed by 70% ethanol and iodine solution. Make a longitudinal incision in the middle of the head with scissors to expose the Lambda (the point where the sagittal and the lambdoid suture meet), the Bregma (the point of intersection of the coronal suture by the sagittal suture), and the target location.
- Identify anatomical landmarks such as Lambda and Bregma. Position the drill bit at Bregma and note the X, Y coordinates for this point.
- Use a brain atlas books or published references to determine the appropriate coordinates of the brain regions for EEG recording. Then, calculate the coordinates for the screws (reference and recording) using the Bregma coordinates measured at step 2.5.2.
- Slowly lower the drill bit to make a burr hole being careful to avoid inserting the drill bit too far at the point where the skull becomes thinner.
- Insert the body of the single channel wireless EEG transmitter behind the nape of the neck subcutaneously and place flexible leads near the skull.
- Place a stainless-steel screw (2 mm length) into each burr hole with tweezers and tighten it using a screw driver by clockwise rotation. Make sure the screw is not inserted too far deep by adjusting the degree of screw rotation in order to prevent damage to the dura or the brain tissues.
- Strip the insulation coat 2 mm from the tip of the leads and stretch the wire long enough to round the screw shaft for a good connection between the wire and the screw. Connect the reference lead to the front screw and the recording lead to the screw in the parietal cortex. Then, apply dental cement to secure the entire assembly in place without exposing the metal parts.
- After verifying that the dental cement has completely dried by visual inspection, suture the skin and apply topical mupirocin ointment. Place the mouse in a 30 °C incubator alone until it recovers from the surgery (about 30 min). Then, return the mouse to the home cage until continuous video-EEG monitoring begins.
Note: After surgery, each mouse is injected (i.p.; 1 mL syringe; 30 G needle) with 5 mg/kg of gentamicin (antibiotics) and 5 mg/kg of ketoprofen (analgesics). Animals should be monitored until they become fully ambulant. Transmitter-implanted mice have a recovery period of approximately 1 week.
3. Video and EEG Monitoring and Analysis
- Place a mouse in an individual cage and place the cage on top of the wireless receiver plates where the Faraday cage is installed to avoid interruption of electrical signals from any sources including a computer, AC lines, and adjacent receivers.
Note: Refer to the manual provided by the manufacturer. Care must be taken to avoid blind spots. - Record the electrical activity using the wireless video-EEG telemetry system.
- Open the software for video-EEG recording. Click "Hardware" and select "Edit configuration". Match the receiver and the transmitter implanted into each mouse. Click "Channel configuration" and confirm all the channels are active. Click "Video configuration" to synchronize the video with the EEG data (resolution of 40 x 480 and frame rate of 30.00).
Note: Install an IP camera with high sensitivity if it is critical to collect good image qualities that can identify subtle behavioral changes of the mice at night. - Click "Setup" and "P3 setup" to input individual animal information, matched camera, and graph setting.
- Click "Acquisition" and "A/D sampling rate" to setup sampling rates. Click "Data set name" to designate each experiment.
Note: Sampling rate for EEG needs to be higher than 500 Hz. Due to the large video and EEG data size, it is recommended that only 24 h of data are handled per session rather than using a continuous 2-week-recording as one file. Data can be separately saved at 8 h or 12 h intervals in case of RAM memory shortage. - Activate the transmitter with a magnetic bar and click "Start acquisition" to collect the video-EEG recording. Continuously monitor the mouse by video-EEG system for at least 2 weeks.
Note: For C57BL/6 mice, SRS tends to occur in clusters that last about 5.2 days and the duration of the seizure-free period is about 6.7 days17.
- Open the software for video-EEG recording. Click "Hardware" and select "Edit configuration". Match the receiver and the transmitter implanted into each mouse. Click "Channel configuration" and confirm all the channels are active. Click "Video configuration" to synchronize the video with the EEG data (resolution of 40 x 480 and frame rate of 30.00).
- Determine the seizure frequency and duration by manual inspection using the analysis software.
- Mark the area showing SRS and export the data for statistical analysis. Be cautious to exclude EEG artifacts that can be generated by scratching the head or chewing; these can be removed by checking the video data.
Note: SRS is defined as a sudden onset of repetitive epileptiform spiking activity (≥ 3 Hz) that persists for more than 10 s. Convulsive behaviors can be frequently accompanied with a burst of spiking activity. Non-convulsive seizures can also demonstrate electrographic spiking activities without convulsive behaviors. Seizure termination is defined as occurring when the firing spikes return to baseline activity. - For analysis of seizure frequency, divide the total number of SRS cases detected during the 2 weeks by the total number of recording days for each individual animal.
- For analysis of seizure duration, measure the time from the onset to the end of the epileptiform spiking activities.
- Mark the area showing SRS and export the data for statistical analysis. Be cautious to exclude EEG artifacts that can be generated by scratching the head or chewing; these can be removed by checking the video data.
- After completion of the video-EEG recording, fix the brain with 4% paraformaldehyde by transcardial perfusion. To anesthetize the mouse, inject a solution (4:0.5) of ketamine (50 mg/mL) and xylazine (23.3 mg/mL) dissolved in saline at a dose of 10 μL/g body weight (i.p.; 1 mL syringe; 26 G needle). Once the mouse is confirmed to be deeply anesthetized, perform transcardial perfusion.
- Before isolating the mouse brain, retrieve the transmitter from the mouse for reuse after cleansing.
- Remove the dental cement around the leads using forceps.
- Soak the tip of the leads with dental cement in 100% acetone until the dental cement is dissolved.
- Remove tissue contaminants around the transmitter by rinsing it with distilled water.
- Rinse the transmitter with 70% ethanol for 3 h and air dry it for storage. Immediately prior to the next use, rinse the screw and transmitter with 70% ethanol, followed by distilled water, and place them in saline.
Note: Care must be taken to avoid cutting the flexible wires when decapitating the mouse since, if the flexible leads are too short, the noise in the next implantation and video-EEG recording may increase.
Representative Results
Successful SE can induce hippocampal cell death and SRS (Figure 1 and Figure 2). We terminated behavioral acute seizures by diazepam injection at 3 h after SE onset and sacrificed the mice 7 days or 6 weeks later.
For video-EEG monitoring, the mice received implant surgery at 4 weeks after SE, and SRS cases were evaluated for 2 weeks from 5-7 weeks after SE onset.
Figure 1 shows cell deaths in the hippocampus assessed by cresyl violet staining. At 7 days after SE, pyknotic cells were detected in the hilar regions (Figure 1Ad) of the dentate gyrus and the pyramidal cell layer of the CA3 subfield of the hippocampus (Figure 1Af), whereas intact hippocampus was observed in the sham animals injected with saline instead of pilocarpine (Figure 1Aa-c). Interestingly, animals who underwent multiple seizures, but failed to enter SE, showed no cell death in either the hilar region or the pyramidal cell layer (Figure 1Ag-i). At 6 weeks after SE, a marked reduction was observed in the number of hilar cells (Figure 1Bm). In addition, neuronal loss was observed in the CA1 (Figure 1Bn) and CA3 (Figure 1Bo) regions.
Figure 2 shows the representative EEG traces in the sham and SE groups (Figure 2A). Compared to the sham group showing physiologic electrical activity, the SE group occasionally showed epileptiform discharges accompanied with convulsive behaviors. The locations of the epidural electrodes are illustrated in Figure 2C. Figure 2B demonstrates an example of the electrical noises from an adjacent monitor and the failure of signal collection. We also demonstrated EEG traces of sham-manipulated animals who showed spontaneous seizures during the 2 weeks of video-EEG recording (Figure 2E) and found cortical injuries at perfusion, possibly derived from the excessively deep placement of the epidural screws (Figure 2D).
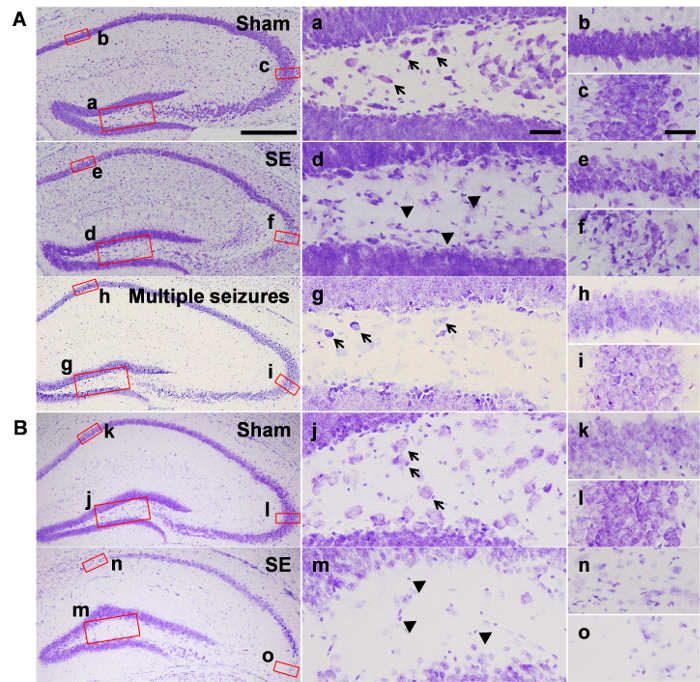
Figure 1: Neuronal death in the hippocampus after pilocarpine-induced SE. Representative images from the groups of (A) 7 days and (B) 6 weeks after pilocarpine or saline injection. Magnified images show the hilus (a, j), CA1 subfield (b, k), and CA3 subfield (c, l) of saline-treated sham group, the hilus (d, m), CA1 subfield (e, n), and the CA3 subfield (f, o) of the SE group, and the hilus (g), CA1 subfield (h), and CA3 subfield (i) of the mice that do not develop SE. Black arrows in a, g, and j indicate some of the representative hilar cells, and arrowheads in d and m indicate pyknotic cells. Scale bar in the bottom left side = 200 µm, valid for the entire left most column, scale bar in a and c = 20 µm, valid for the entire middle column and right column. Abbreviations in this figure: CA1, CA3: Cornu Ammonis hippocampal subfields; DG: Dentate gyrus. Please click here to view a larger version of this figure.
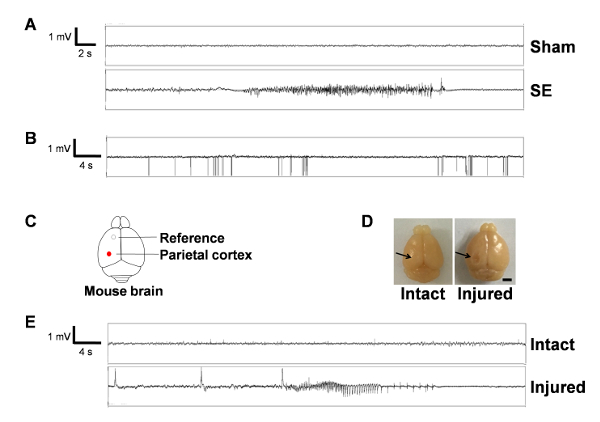
Figure 2: Video and EEG recording shows spontaneous recurrent seizures in the epileptic mouse. (A) Representative EEG traces from sham- (top) and SE-manipulated animals (bottom). Note that SE-subjected animals show epileptiform spiking activity (≥ 3 Hz), not exhibited by sham animals. (B) A representative image of electrical noises and failure of the signal collection. Note the high amplitude unipolar spikes and discontinuation of electrical signals. (C) Schematic drawing of the mouse brain with locations of electrodes. (D) Two images showing the intact and injured brain in the cortex, possibly due to excessively deep insertion of epidural screws. The black arrow in the right image indicates the site of cortical damage. Scale bar = 250 µm. (E) Representative EEG traces of sham-manipulated animals with or without brain injury. Note that sham-manipulated animals with cortical injury generated spiking activity and convulsive SRS. Please click here to view a larger version of this figure.
Discussion
This work describes the experimental procedures for the SE induction and the evaluation of chronic seizures.
Several factors can affect successful SE induction. Accurate behavioral monitoring according to the Racine scale is critical for the development of SRS. Head nodding, forelimb clonus, rearing, and falling are the behavioral hallmarks of acute seizures developing into SE phase4,16. Once the first motor seizure is detected, the length of the interval between the later convulsive seizures decreases before entering SE. Moreover, SE should be sustained for at least 6 h unless behavioral seizures are terminated by diazepam5. It is worth noting that researchers should be aware of the possibility that sustained seizure activities can be detected via electrocorticograms, even though behavioral seizures seem to subside with diazepam injection. Increasing ambient temperature after pilocarpine injection until SE induction can greatly improve the number of animals with SE. It is also important to provide intensive post-seizure care, including sufficient hydration, nutritional support, and weight monitoring for higher survival rates than about 70-80% on average5,18. Depending on the animal’s genetic background, susceptibility to pilocarpine may differ. If animals display severe acute seizures with high mortality, the reduction of pilocarpine dosage, SE duration, or increased scopolamine and terbutaline can be helpful. While severe SE is known to be related to higher incidence of SRS, with higher mortality, it is crucial to consider these factors. Hippocampal neuronal deaths are consistently observed after SE. At 6 h post-SE, extensive loss of hilar interneurons has been reported19. At 7 days after SE, pyramidal neurons in the CA3 subfield typically die, and are continuously observed at 6 weeks after SE induction, although the extent of neuronal damage in the pyramidal cell layer is variable in mice. Neuronal loss in the CA1 subfield of the hippocampus can also be frequently observed. Interestingly, animals with multiple seizures who do not enter SE showed almost intact hippocampus similar to the sham group, indicating histologic cell death can be a good marker of sufficiently severe acute seizures.
At approximately 4 weeks after SE, all pilocarpine-treated C57BL/6NHsd mice became epileptic and showed SRS, demonstrating one of the major merits using this model. Since chronic seizures are clustered, the video-EEG should be recorded continuously for at least 2 weeks rather than intermittently, even though the recording period can be modified depending on the mouse strains or experimental purposes17. Moreover, spontaneous seizures in TLE have diurnal variations demonstrating high SRS frequency in the late afternoon20. Practically, for accurate video-EEG recording, any sources including AC lines, computers, cameras, or transmitters implanted in the mouse in the adjacent receiver need to be blocked. The installation of the Faraday cage and cameras with high sensitivity during the day and night can diminish electrical noise and help to identify generalized chronic seizures, respectively. Finally, it is extremely important to avoid brain damage when the screws are placed on the surface of the dura mater as it can generate misleading EEG data. Although the SRS frequency was not high, any mechanical cortical injuries by forced screw insertion could generate convulsive seizures in only one week.
In conclusion, despite the time constraints for modeling and the requirement of in-depth training to perform implant surgeries and EEG analysis, the pilocarpine-induced SE is one of the best models for studying chronic epilepsy among currently available animal models of TLE. Here, we describe a method of inducing SE after pilocarpine injection and assessing chronic seizures using a wireless telemetry video-EEG system. We believe this protocol can provide detailed information for suitable animal models for chronic epilepsy and epileptogenesis.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean Government (NRF-2014R1A1A3049456) and a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (HI15C2854).
Materials
| C57BL/6 | Envigo | C57BL/6NHsd | |
| Scopolamine methyl nitrate | Sigma | S2250 | Make 10X stock |
| Terbutaline hemisulfate salt | Sigma | T2528 | Make 10X stock |
| Pilocarpine hydrochloride | Sigma | P6503 | |
| Intensive care unit | Daejong instrument industry Co., Ltd. | 28~30℃ | |
| Ketamine hydrochloride | Yuhan corporation | ||
| Xylazine hydrochloride | Bayer Korea | ||
| Diazepam | SAMJIN | ||
| Castor oil (Kolliphor EL) | Sigma | C5135 | Polyoxyl 35 hydrogenated castor oil |
| Saline | Daihan pharm. Co. | ||
| 5% Dextrose | Daihan pharm. Co. | ||
| Iodine solution (Povidin) | Firson | ||
| vet ointment (Terramycin) | Pfizer | ||
| Blue Nylon | AILEE | NB617 | |
| Mupirocin (Bearoban) | Daewoong Pharmaceutical Co., Ltd | ||
| Ketoprofen | Samchundang Pharm. Co., Ltd | 5 mg/kg | |
| Gentamicin | Huons, Ltd. | 5 mg/kg | |
| 1 mL syringe | Sung shim medial Co., Ltd. | ||
| 26 guage needle | Sung shim medial Co., Ltd. | 26 G * 13 mm (1/2") | |
| 30 guage needle | Sung shim medial Co., Ltd. | 30 G * 13 mm (1/2") | |
| Razor blade | Dorco | ||
| Drill | Saeshin precision Co., Ltd. | 207A, 35K (speed) | |
| Telemetry video/EEG system | Data sciences International. Inc. | Version 5.20-SP6 | |
| Implantable transmitter | Data sciences International. Inc. | ETA-F10 | |
| Screw | Sungho Steel | M1.4, 2 mm length stainless steel | |
| Vertex dental material | Dentimex | ||
| Acetone | Duksan pure chemicals Co., Ltd. | CAS 67-64-1 | |
| Paraformaldehyde (PFA) | millipore | 1.04005.1000 | 4 % |
| Sucrose | Sigma | S9378 | 30 % solution in 0.01 M PBS |
| Cresyl violet acetate | Sigma | C5042 | |
| Ethanol | EMD Millipore Co. | UN1170 | |
| xylene | Duksan pure chemicals Co., Ltd. | UN1307 | |
| Acetic acid glacial | Junsei chemical | 31010-0350 | |
| FSC33 Clear | Leica biosystems | OCT compound for tissue freezing | |
| DPX Mounting for histology | Sigma | 6522 | |
| Forceps | Fine science tools | 11002-12 | |
| Scissors | Solco biomedical | 02-2445 | |
| Stereotaxic frame | David Kopf Instruments | E51070012 |
References
- Chang, B. S., Lowenstein, D. H. Epilepsy. N Engl J Med. 349 (13), 1257-1266 (2003).
- Scharfman, H. E. The neurobiology of epilepsy. Curr Neurol Neurosci Rep. 7 (4), 348-354 (2007).
- Rakhade, S. N., Jensen, F. E. Epileptogenesis in the immature brain: emerging mechanisms. Nat Rev Neurol. 5 (7), 380-391 (2009).
- Cavalheiro, E. A. The pilocarpine model of epilepsy. Ital J Neurol Sci. 16 (1-2), 33-37 (1995).
- Curia, G., Longo, D., Biagini, G., Jones, R. S., Avoli, M. The pilocarpine model of temporal lobe epilepsy. J Neurosci Methods. 172 (2), 143-157 (2008).
- Morimoto, K., Fahnestock, M., Racine, R. J. Kindling and status epilepticus models of epilepsy: rewiring the brain. Prog Neurobiol. 73 (1), 1-60 (2004).
- Levesque, M., Avoli, M. The kainic acid model of temporal lobe epilepsy. Neuroscience and Biobehavioral Reviews. 37 (10), 2887-2899 (2013).
- Sharma, A. K., et al. Mesial temporal lobe epilepsy: pathogenesis, induced rodent models and lesions. Toxicol Pathol. 35 (7), 984-999 (2007).
- Hellier, J. L., Dudek, F. E. Chemoconvulsant model of chronic spontaneous seizures. Curr Protoc Neurosci. 9, 19 (2005).
- Pitkanen, A., Lukasiuk, K. Molecular and cellular basis of epileptogenesis in symptomatic epilepsy. Epilepsy Behav. 14, 16-25 (2009).
- Mathern, G. W., Adelson, P. D., Cahan, L. D., Leite, J. P. Hippocampal neuron damage in human epilepsy: Meyer’s hypothesis revisited. Prog Brain Res. 135, 237-251 (2002).
- Turski, W. A., et al. Limbic seizures produced by pilocarpine in rats: behavioural, electroencephalographic and neuropathological study. Behav Brain Res. 9 (3), 315-335 (1983).
- Brulet, R., Zhu, J., Aktar, M., Hsieh, J., Cho, K. O. Mice with conditional NeuroD1 knockout display reduced aberrant hippocampal neurogenesis but no change in epileptic seizures. Exp Neurol. 293, 190-198 (2017).
- Cho, K. O., et al. Aberrant hippocampal neurogenesis contributes to epilepsy and associated cognitive decline. Nat Commun. 6, 6606 (2015).
- Cavalheiro, E. A., Santos, N. F., Priel, M. R. The pilocarpine model of epilepsy in mice. Epilepsia. 37 (10), 1015-1019 (1996).
- Racine, R. J. Modification of seizure activity by electrical stimulation. II. Motor seizure. Electroencephalogr Clin Neurophysiol. 32 (3), 281-294 (1972).
- Hester, M. S., Danzer, S. C. Accumulation of abnormal adult-generated hippocampal granule cells predicts seizure frequency and severity. J Neurosci. 33 (21), 8926-8936 (2013).
- Shibley, H., Smith, B. N. Pilocarpine-induced status epilepticus results in mossy fiber sprouting and spontaneous seizures in C57BL/6 and CD-1 mice. Epilepsy Research. 49 (2), 109-120 (2002).
- Borges, K., et al. Neuronal and glial pathological changes during epileptogenesis in the mouse pilocarpine model. Exp Neurol. 182 (1), 21-34 (2003).
- Pavlova, M. K., Shea, S. A., Bromfield, E. B. Day/night patterns of focal seizures. Epilepsy Behav. 5 (1), 44-49 (2004).

