Protocol to Create Chronic Wounds in Diabetic Mice
Summary
Chronic wounds are developed from acute wounds on a diabetic mouse model by inducing high levels of oxidative stress after a full-thickness cutaneous wound. The wound is treated with inhibitors for catalase and glutathione peroxidase, resulting in impaired healing and biofilm development by bacteria present in the skin microbiome.
Abstract
Chronic wounds develop as a result of defective regulation in one or more complex cellular and molecular processes involved in proper healing. They impact ~6.5M people and cost ~$40B/year in the US alone. Although a significant effort has been invested in understanding how chronic wounds develop in humans, fundamental questions remain unanswered. Recently, we developed a novel mouse model for diabetic chronic wounds that have many characteristics of human chronic wounds. Using db/db-/- mice, we can generate chronic wounds by inducing high levels of oxidative stress (OS) in the wound tissue immediately after wounding, using a one-time treatment with inhibitors specific to the antioxidant enzymes catalase and glutathione peroxidase. These wounds have high levels of OS, develop biofilm naturally, become fully chronic within 20 days after treatment and can remain open more for more than 60 days. This novel model has many features of diabetic chronic wounds in humans and therefore can contribute significantly to advancing fundamental understanding of how wounds become chronic. This is a major breakthrough because chronic wounds in humans cause significant pain and distress to patients and result in amputation if unresolved. Moreover, these wounds are very expensive and time-consuming to treat, and lead to significant loss of personal income to patients. Advancements in this field of study through the use of our chronic wound model can significantly improve health care for millions who suffer under this debilitating condition. In this protocol, we describe in great detail the procedure to cause acute wounds to become chronic, which has not been done before.
Introduction
Wound healing involves complex cellular and molecular processes that are temporally and spatially regulated, organized in sequential and overlapping stages that involve many different cell types including but not limited to the immune response and the vascular system1. Immediately after the skin sustains an injury, factors and blood cells aggregate to the wound site and initiate the coagulation cascade to form a clot. After homeostasis is achieved, the blood vessels dilate to let into the wound site oxygen, nutrients, enzymes, antibodies and chemotactic factors that chemoattract polymorphonucleocytes to clear the wound bed of foreign debris and secrete proteolytic enzymes2. Activated platelets secrete a variety of growth factors to stimulate the keratinocytes at the wound edge to re-epithelialize the wounded area. Monocytes recruited to the wound site differentiate into macrophages which phagocytose bacteria and dead neutrophils and secrete additional factors to maintain keratinocyte proliferative and pro-migratory signals. In the proliferation phase, while re-epithelialization continues, new granulation tissue composed of fibroblasts, monocytes/macrophages, lymphocytes, and endothelial cells continue the rebuilding process2. Angiogenesis is stimulated by promoting endothelial cell proliferation and migration, resulting in new vessel development. Epithelialization and remodeling of the extracellular matrix construct a barrier against the environment. As the wound heals and granulation tissue evolves into a scar, apoptosis eliminates inflammatory cells, fibroblasts, and endothelial cells without causing additional tissue damage. The tensile strength of the tissue is enhanced by fibroblasts remodeling various components of the extracellular matrix, like collagen, so that the newly formed tissue is almost as strong and flexible as unwounded skin2.
Any deviation from this highly concerted progression towards wound closure leads to impaired and/or chronic wounds3. Chronic wounds are characterized by increased oxidative stress, chronic inflammation, damaged microvasculature, and abnormal collagen matrix in the wound4. Oxidative stress, especially in the wound, can delay wound closure2,5. When, in the first stage of wound healing, the inflammatory phase becomes unregulated, the host tissue assumes extensive damage due to a continuous influx of inflammatory cells5 that release cytotoxic enzymes, an increase in free oxygen radicals, and unregulated inflammatory mediators, resulting in cell death6,7.
In this destructive microenvironment, biofilm-forming bacteria take advantage of host nutrients and contribute to the damage of the host tissue2. These biofilms are difficult to control and remove because the hydrated extracellular polymeric substances composed of proteins, DNA, RNA, and polysaccharides allows bacteria harbored within to be tolerant to conventional antibiotic therapies and evade the host's innate and adaptive immune response2,8,9.
Studying chronic wounds is crucial because they impact ~6.5 million people and cost ~$40 billion per year in the US alone10. Patients with diabetes have increased risks for developing chronic wounds that require amputation in order to contain the spread of infection. These patients have a 50% mortality risk within 5 years of amputation that is attributed to the pathophysiology mechanism of diabetes11. The relationship between the host's immune system and the microbiome in wound healing is a vital topic of ongoing research because consequences of chronic wounds, if unresolved, include amputation and death12.
Although a significant effort has been invested in understanding how chronic wounds develop in humans, it is still unclear how and why chronic wounds form. Experiments to study the mechanisms of impaired healing is difficult to conduct in humans, and wound healing specialists only see patients with chronic wounds that have already reached chronicity for weeks to months. Thus, specialists are unable to study what processes went wrong that lead the wound to develop to become chronic2. There is a lack of animal models that recapitulate the complexity of human chronic wounds. Until our model was developed, no model for chronic wound studies existed.
The chronic wound model was developed in mice that have a mutation in the leptin receptor (db/db-/-)13. These mice are obese, diabetic, and have impaired healing but do not develop chronic wounds14. Blood glucose levels average around 200 mg/dL, but can be as high as 400 mg/dL15. When high levels of oxidative stress (OS) in the wound tissue are induced immediately after wounding, the wound becomes chronic16. The db/db-/- wounds are considered chronic by 20 days and remain open for 60 days or more. Biofilm produced by bacteria can be seen developing beginning three days after wounding; a mature biofilm can be seen 20 days after wounding and persists until either wound closure. The biofilm-forming bacteria we find in these mice are also found in human diabetic chronic wounds.
Oxidative stress is induced by treating the wounds with two inhibitors of antioxidant enzymes, catalase and glutathione peroxidase, two enzymes with the capacity to break down hydrogen peroxide. Hydrogen peroxide is a reactive oxygen species and can cause cellular damage through the oxidation of proteins, lipids, and DNA. Catalase catalyzes the decomposition of hydrogen peroxide into less harmful chemicals oxygen and water. 3-Amino-1,2,4-triazole (ATZ) inhibits catalase by binding specifically and covalently to the active center of the enzyme, inactivating it17,18,19. ATZ has been used to study the effects of oxidative stress both in vitro and in vivo through the inhibition of catalase20,21,22,23,24. Glutathione peroxidase catalyzes the reduction of hydrogen peroxide through the antioxidant, glutathione, and is an important enzyme that protects the cell against oxidative stress25. Mercaptosuccinic acid (MSA) inhibits glutathione peroxidase by binding to the selenocysteine active site of the enzyme with thiol, inactivating it26. MSA has been used to study the effects of oxidative stress in vitro and in vivo as well20,27,28.
This novel model of chronic wounds is a powerful model to study because it shares many of the same features observed in human diabetic chronic wounds, including prolonged inflammation from increased OS and natural biofilm formation from skin microbiome. The wounds have impaired dermal-epidermal interaction, abnormal matrix deposition, poor angiogenesis and damaged vasculature. Chronic wounds will develop in both male and female mice, so both sexes can be used to study chronic wounds. Therefore, the chronic wound model can contribute significantly to advance fundamental understanding of how such wounds begin. Using this chronic wound model can provide answers to fundamental questions about how chronicity is initiated/achieved through contributions from the physiology of impaired wound healing and the microbiome of the host.
Protocol
All experiments were completed in accordance and compliance with federal regulations and University of California policy and procedures have been approved by the University of California, Riverside IACUC.
1. Animal
- Use diabetic and obese B6.BKS(D)-Leprdb/J mice for the chronic wound model. Purchasing options include either heterozygotes for breeding or homozygotes directly for experiments.
- Breed heterozygote males and females to produce offspring. Only a quarter of the litter, statistically, will grow to become diabetic and obese (db/db-/-).
- Wean and house db/db-/- mice together with their littermates 3 weeks after birth. Separate db/db-/- mice from their littermates 5 weeks after birth and house with other db/db-/- mice until they are 5-6 months old and can be used for the chronic wound model. During this time, a mature and complex skin microbiome can develop.
NOTE: The db/db-/- mice are easily identified visually from littermates between 3-5 weeks of age. The db/db-/- mice are obese, are diabetic, and will be significantly larger and rounder than the wild type and heterozygous littermates. Their abdomen may appear slightly pinker and their hips larger. Increased weight should be confirmed prior to surgery. Also, it is possible to measure high levels of glucose in the blood and genotype the mouse to confirm the mutation in the leptin receptor.
2. Vivarium and Husbandry
- House db/db-/- mice in a conventional vivarium (not a barrier/specific pathogen free facility) so that a microflora can be established on the skin of db/db-/- mice. To specifically model humans who suffer from chronic wounds, do not take special precautions to prevent exposure to pathogens.
- Protect the cages with micro isolator tops to minimize spread of infection within the vivarium. Change cages twice a week with new bedding and feed the mice with regular vivarium chow. Do not autoclave either the bedding or the food.
- Set room temperature to range between 21 and 24 °C, with minor fluctuations depending on the time of year. The humidity, reflective of the climate and location, will range between 19 and 70%.
3. Requirements for the Development of Chronic Wounds
- Use only male and female mice that are phenotypically obese, diabetic, and at least 5-6 months of age for the development of chronic wounds. The weight of these mice should vary between 40-80 g, averaging around 60 g.
- Do not use mice that are considered obese but weigh less than 50 g.
NOTE: All mice referred to in this protocol pass the qualifications described here unless otherwise noted.
4. Shaving and Application of Depilatory Lotion
NOTE: Remove unwanted hair on the dorsum of the mouse before wounding. The following procedure is done on live db/db-/- mice that are not under anesthesia the day before surgery. Take precautions to prevent stress and harm to the animal.
- Shaving
- Place the mouse on a clean surface and grip the base of the tail of the mouse with the thumb and second finger to secure the position of the mouse. The mouse may jump or make sudden movements; if it does, quickly respond and pull back the clippers to prevent injury.
- Shave the hair of the mouse with a hair clipper (Figures 1A, 1B). Position the blade parallel to the skin of the mouse and shave the whole back from the neck to the tail, including all around the tail, to allow for a large enough surface area to place a transparent film dressing (see Table of Materials). Lightly run the blade against the direction of hair growth for the most efficient cut. (Figure 1B).
- Do not press the blade deeply into the skin, since it may damage the skin by bruising or cutting.
- Application of depilatory lotion
NOTE: Use depilatory lotion to obtain a very smooth skin so that the transparent dressing can adhere firmly.- Soak the skin of the mouse with water to prevent chemical burns by lightly pressing a drenched paper wipe against the shaved skin with enough pressure to wet the already cut hair down against the skin.
- While the skin is wet, lightly rub the skin of the mouse with a small dollop of depilatory lotion (see Table of Materials) for 15-20 s (Figures 1C, 1D). Spread the lotion completely over wherever the hair has been cut short. Use more lotion if the mouse is larger.
- Do not apply lotion on the ears of the mouse, tail, or anywhere near the face. If the lotion gets on the ears or tail, simply wipe with a wet paper wipe until rinsing. If the lotion gets on the face of the mouse, immediately wash the mouse under running tap or deionized water to prevent damage to the eyes, nose, and mouth.
- Leave the lotion to react with the hair for an additional 20-45 s after application.
- Before rinsing, check the completion of the depilatory reaction by lightly wiping away the lotion from the skin at various locations (Figure 1E) with a gloved finger or a thin metal spatula. The reaction is complete if the skin is pink without the presence of black hair. It is best to quickly check that the hair has actually been removed before rinsing than rinsing prematurely and then having to apply lotion again.
- Rinsing
NOTE: Once the completion of the hair removal reaction has been verified, wash the mouse with running tap or deionized water to remove the depilatory lotion and prevent chemical burns.- Place the mouse on the left gloved hand and press the base of the tail against the palm with the left thumb to prevent the mouse from moving around. Close and straighten the rest of the left fingers to prevent the mouse from biting.
- Position the mouse so the face/head region is away from the stream of water and the stream of lukewarm water can fall behind the head and only on the back. Quickly but gently rub the back of the mouse with the right gloved hand to wash away the lotion.
- Once the skin of the mouse is free of the lotion, quickly wipe the mouse with a paper towel to absorb most of the water (Figure 1F).
- Care after shaving and hair removal
- Check and clean any residual depilatory lotion that may remain on the ears and tail with a wet paper wipe.
- Place the mouse back in its individual cage and place the cage on a heating pad (40-45 °C) for about 30 min. The mouse should return to normal behavior, scurry around, and groom itself within a few min.
- House each mouse in a separate cage throughout the duration of the experiment. The skin of the mice is no longer protected and can be easily scratched and bitten by other mice.
- Since the depilatory lotion can have slight irritating effects that may interfere with or alter the wound healing process, wait 18-24 h before surgery to allow the mouse skin to calm down and the mouse to adjust to the lack of hair on its back.
- Removing Hair from Skin with Dark Pigmentation
NOTE: Some db/db-/- mice will potentially have dark patches in the skin that will have hair growing back faster and stronger than skin without these patches. (Figure 2A). These dark areas of the skin appear darker in color due to mid-late anagen stage hair follicles and/or pigment incontinence29,30.- If dark patches are noticed, apply depilatory lotion again, but only in these areas, and repeat step 4.2 and 4.3 (Figures 2B, 2C).
NOTE: The dark patches of skin will grow hair faster throughout the duration of the experiment, so clip the hair short every 3-5 days, when necessary. A patch or two away from the desired location of the wound is acceptable. If the back of the mouse is significantly covered by these dark patches, do not use this mouse for the chronic wound model.
- If dark patches are noticed, apply depilatory lotion again, but only in these areas, and repeat step 4.2 and 4.3 (Figures 2B, 2C).
5. Reagent Setup
NOTE: The development of chronic wounds in db/db-/- mice is accomplished by treatment with specific inhibitors for catalase and glutathione peroxidase, 3-amino-1,2,4-triazole (ATZ) and mercaptosuccinic acid (MSA), respectively16. The following procedure details the dose and administration of the analgesia and inhibitors based on the weight of the mouse.
- Inject the pain reliever Buprenex intraperitoneally at 0.05 mg/kg mouse in sterile PBS. Inject a volume of 120 µL for a 60 g mouse approximately 30 min before surgery. Administer another dose 6 h after surgery. An extra dose may be given as needed.
- Inject ATZ intraperitoneally at 1 g/kg mouse in sterile PBS. Inject a volume of 480 µL for a 60 g mouse approximately 20 min before surgery. Inject half of the volume on the left side of the abdomen and the other half on the right side.
- Deposit MSA topically onto the wound between the transparent dressing and the wound tissue at 150 mg/kg mouse in sterile PBS. Administer a volume of 60 µL for a 60 g mouse within 10 min after surgery.
6. Surgery
NOTE: The success of the chronic wound model relies on non-sterile conditions. These mice are not germ-free and are housed in a conventional vivarium. The bacteria microbiome that resides in the skin is crucial for the subsequent initiation and development of chronic wounds upon treatment with inhibitors of anti-oxidant enzymes. Therefore, traditional pre-surgical preparation of the site is contra-indicated.
- Intraperitoneal Injection
- Secure the mouse on slanted portion of the cage rack and hold the tail with the mouse comfortably standing with the head lower than the body of the mouse. This ensures that we attempt to move the internal organs as far away from the injection point possible.
- Insert the needle in the lower right quadrant of the abdomen to avoid injection into the bladder, or other abdominal organs.
- Aspirate the needle before injection.
- Treatment and Anesthesia
- Administer Buprenex 30 min and ATZ 20 min before surgery, as described in Steps 5.1 and 5.2.
- Place the mouse in a small plastic container on top of a warm heating pad as shown in Figure 3A for approximately 15-20 min. Providing heat support prior to the procedure protects against potential mortality.
- Place a paper wipe or paper towel over the top of the small container to better contain the heat. The mouse should calm down as it warms. Figure 3B shows materials necessary for surgery.
- Place the mouse in an enclosed container that is hooked up to the isoflurane vaporizer in the chemical hood if using an open system.
- In an open system, administer 5% isoflurane for 1-2 min at a flow rate of 2-3.5 L/min. Continuously monitor the status of the mouse.
- Once the mouse is unconscious or no longer moving, place the mouse on a white surgical pad and fit the head with a nose cone that is secured to the vaporizer to allow continuous administration of isoflurane during surgery.
- In an open system, administer 2-3% isoflurane at the same flow rate during surgery and adjust to the flow of isoflurane to maintain depth of anesthesia.
- Minimize both the concentration and duration of isoflurane administered during surgery. db/db-/- mice are very sensitive to anesthesia so it is best to keep isoflurane exposure to a minimum. If the mouse is still responsive after 2 min at 5% isoflurane, secure the nose cone and administer 3-5% isoflurane for 15-30 s before starting surgery.
- Confirm proper anesthetization before wounding. Depth of anesthesia is confirmed by lack of response to physical stimuli such as strong toe pinch. The length of time of the mouse under inhaled anesthesia is under 5 min so no vet ointment is applied to the eyes.
- Wounding
- Spray a paper wipe with 70% ethanol or a clinical ethanol towelette and wipe the back of the mouse to clean the area of the wound site. Clean the surface of the skin so the transparent dressing can adhere firmly, since dust from the bedding, food, or skin can prevent the dressing from sticking properly (Figure 4A). Do not wipe excessively, or there will be a risk of killing the bacteria present on the skin.
- Determine the position of the wound site. The best place to perform the wound is on the dorsal side of the mouse, centered and away from patches of skin with higher pigmentation.
NOTE: We have determined by experience that these mice can only withstand the burden of one wound. - Create a wound within 30-45 s using a 7 mm skin biopsy punch, tweezers and surgical scissors. Lightly press the biopsy punch onto the desired wound site and twist the punch around just deep enough to leave a slight impression of the punch (Figure 4B). Excise the outlined skin by pulling up the center of the punch with tweezers and cutting along the outline with surgical scissors (Figures 4C-4E).
- Cover the wound firmly with half of a piece of transparent film dressing (6 cm x 3.5 cm).
- Stop administration of 2% isoflurane to the mouse (Figure 4F).
7. Post-Surgery Treatment and Recovery
- Administer MSA treatment after surgery is complete and the transparent dressing is applied as described in Step 5.3. Deposit MSA onto the wound area under the dressing.
- Place the mouse back in the small container on the heating pad for 30 min to help with recovery. Once the mouse has warmed up, put the mouse back in its cage. The effect of the isoflurane is temporary, and the mouse should be moving around shortly thereafter.
- Do not leave mice unattended nor return them to the vivarium until the mice have regained sufficient consciousness to maintain sternal recumbency.
NOTE: The anesthesia of choice when working with these mice is isoflurane precisely due to its quick induction and subsequent emergence from anesthesia.- House mice that have undergone surgery individually to avoid one mouse interfering with the chronic wound of another. As stated above, they do not return to the vivarium until fully recovered.
- Administer the second dose of Buprenex 6 h after surgery.
- Observe the mice carefully for the first 48 h after surgery.
NOTE: The surgery, coupled with the inhibitors to create the chronic wound, is very stressful on the animal that is already both diabetic and obese. Mice that survive the first couple days after surgery will usually survive the duration of the experiment.
8. Data Collection, Survival Strategies, Handling the Mice After Wounding, and Additional Tips
- Data collection
- Take pictures as early as immediately after surgery. Biofilms are observed anywhere between 5-10 days after wounding, and as early as 3 days.
- If bacteria are the focus of analysis, roll a sterile swap with light pressure around the wound for 10-15 s. Store the swab, in an appropriate freezer media for culturing or dry without any media for culture-independent sequencing analysis, at -80 °C. Collect extracellular polymeric substances via a sterile metal spatula into a microcentrifuge tube and store at -80 °C before analysis.
- Do not administer anesthesia to the mouse during handling for biofilm collection or picture taking. During these procedures, place a piece of food in front of the mouse to calm the mouse down and prevent it from running off. Most mice will climb on top of the food, sit on it, and not move.
- As secondary infections and unintended chronic wounds or ulcers can develop if the mouse is not properly moving or if the dressing is not applied correctly, periodically check the activity of the mouse and the ventral side for sores. Friction between the skin and wet bedding (db/db-/- mice are polyuric) can disturb the skin if the cages are not changed frequently.
NOTE: Fluid buildup under the dressing can cause the adhesive to lose its stickiness and allow the fluid to leak. Dead skin cell, bedding, and fecal matter can then stick onto the skin and harden. These dried patches and aggregates on the skin should be cleared promptly to prevent a secondary infection. - If the mice stand or sit on their hind legs, move these mice to a cage where access to food and water is much lower. If the position of food and water in the cage is high, the mouse may stand or sit on its hind legs to reach it. Most mice will not have a problem eating and drinking if they could do so before surgery, though there is a possibility that some mice might flip over onto their backs. These "flippers" may have great difficulty turning over so they will need assistance and further monitoring.
- Leave the transparent dressing on the skin for up to 20 days if the skin is clear of debris, flakey skin and hair. If any of these occur on the skin, remove the old dressing and apply a new piece.
- To take off the dressing, lightly pinch the skin directly behind the head, then pull the dressing away from the head in one smooth motion.
- To place a piece of dressing on securely, have the mouse stay as still as possible. It is important that the skin is clean and free from flaky dead skin cells, dust from the bedding, and pieces of food. Place and then press the dressing into the skin around the wound to secure.
- If placing a piece of food in front of the mouse does not limit its movements, then place the mouse on top of the wire cage. Once the mouse has gripped a rail on the cage, hold the tail as close to the body as possible and pull with minimal force. As the mouse pulls forward the back will stretch and straighten for easy application; place the dressing securely at this point.
- Never re-use an old dressing. Always apply new dressing to the back for the greatest adhesion.
Representative Results
Figure 5 depicts an example of a wound without treatment of inhibitors progressing towards wound closure and a wound with treatment of inhibitors progressing towards chronicity. The transparent dressing has been left in place on the chronic wound so that biofilm and fluid accumulation can be seen.
Chronic wound initiation takes place in less than 6 hours and the wound margin is visibly altered from oxidative stress. Histological evidence of this wound margin reveals that the tissue is necrotic and will not participate in wound healing. The biofilm-forming bacteria in the wound can later use this necrotic tissue as a source of nutrients and structural components to produce biofilm. A chronic wound is a wound that remains open, enlarged in comparison to the initial wound, contains biofilm (EPS plus pathogenic bacterial found in human chronic wounds) and takes month or years to heal depending on the amount and content of the biofilm that prevents the wound from resolving normally. In the chronic wound model, full chronicity is set to > 20 days after surgery because the wounds not treated with the inhibitors will close by this time (Figure 5). Healing typically takes > 60 days and the time depends on the primary pathogenic bacteria present in the wound. Sometimes the mice may succumb to infection when more aggressive biofilm-forming bacteria, such as Pseudomonas, are predominate in the wound. Thus, a chronic wound is defined as a wound that will not close within 20 days, takes more than 60 days to heal, and has biofilm present on the wound.
Sex differences have been found in various diabetes models, including the db/db-/- mouse model32,33. While such differences exist, we have observed sex to not be a significant factor in the development of chronic wounds. Chronic wounds in male and female mice develop to a similar extent, so both sexes can be used to study chronic wounds. Thus, utilizing this model is advantageous since human chronic wounds can be found on both male and female diabetic patients.
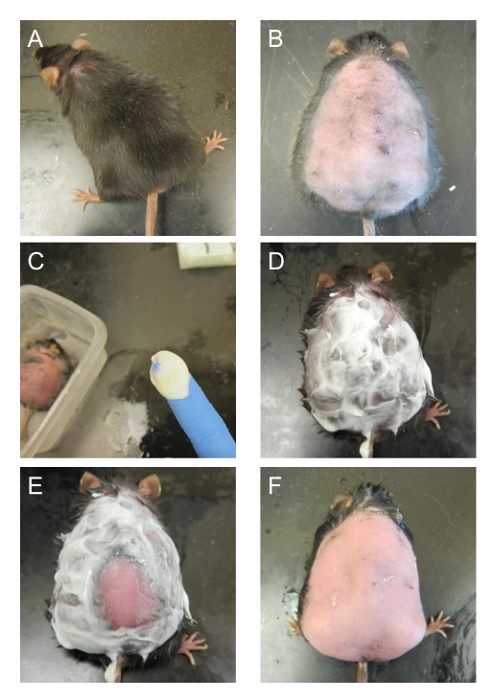
Figure 1. Shaving and depilatory process. (A) The mouse before shaving. (B) The skin of the mouse is shaved to remove most of the hair on the back. (C) A dollop of depilatory lotion on the tip of a finger. More is used if the mouse is bigger. (D) The back of the mouse is covered with depilatory lotion and left to react. (E) A spatula is used to scrape off some of the lotion to see if the hair has been removed. Bright pink skin without any presence of hair is indicative that the hair removal is complete. (F) The lotion on the back is removed with running water. The skin of the mouse should be slightly pink. This figure has been modified from Kim and Martins-Green31. Please click here to view a larger version of this figure.
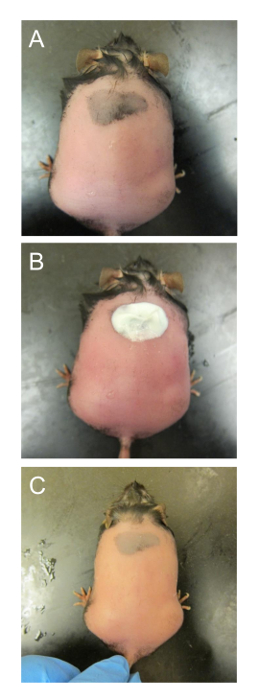
Figure 2. Removing hair from smaller patches of dark skin. (A) The mouse has already been treated once and the skin is wet again to prevent burns. (B) Depilatory lotion is only applied onto the patch of skin that is dark and has dense hair. (C) The lotion is washed off after the reaction to reveal the dark patch of skin without hair. This figure has been modified from Kim and Martins-Green31. Please click here to view a larger version of this figure.
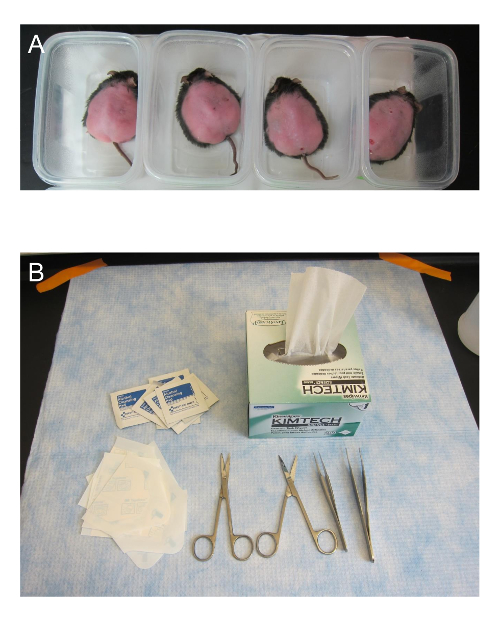
Figure 3. Pre-surgery set up. (A) The mice that are to be wounded are placed in small plastic containers on top of a heating pad. (B) Some of the materials used in surgery are shown. The surgical scissors need to be sharp to ensure that the skin is not crushed when cut. The transparent dressing is cut in half. This figure has been modified from Kim and Martins-Green31. Please click here to view a larger version of this figure.
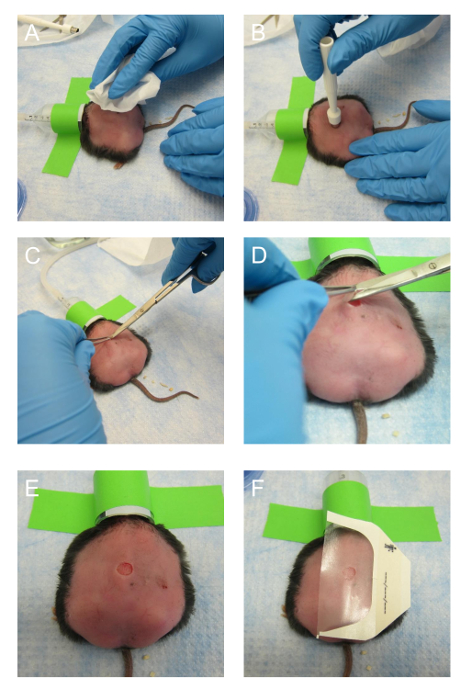
Figure 4. Making the excision wound. (A) After the mouse is under anesthesia, the back of the mouse is wiped with 70% ethanol once. (B) The skin biopsy punch is placed on the back of the mouse and pressed hard enough to leave an impression. The biopsy punch can be rotated to make a shallow incision. (C) The middle of the outlined area is pinched with tweezers and a sharp surgical scissor is used to make the initial incision. (D) The surgical scissors are maneuvered to cut along the outline made by the biopsy punch. (E) A region of skin outlined by the biopsy pump is successfully excised. (F) The transparent dressing is positioned on the back of the mouse and secured. This figure has been modified from Kim and Martins-Green31. Please click here to view a larger version of this figure.
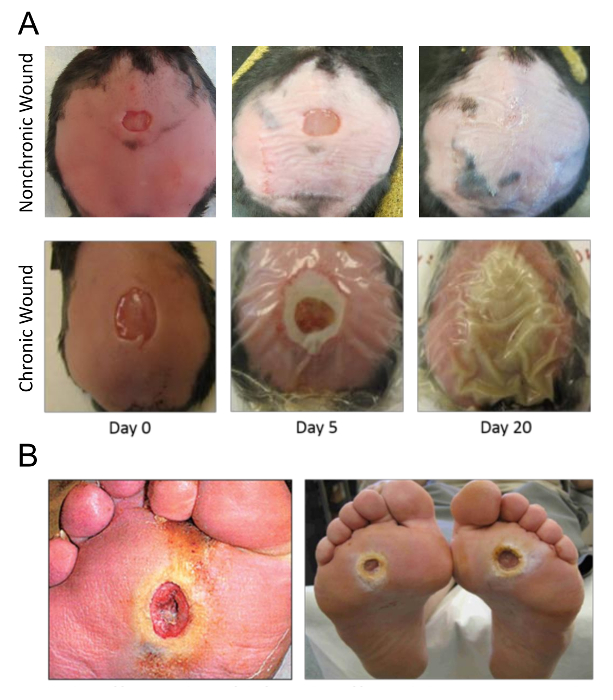
Figure 5. Pictures of wounds. (A) The wound on a mouse at successive times after surgery as it progresses into chronicity starting on the day of surgery. Biofilm can be seen as early as day 5 and detected as early as day 3. The wound is fully chronic with strong biofilm on Day 20. (B) Examples of human chronic wounds, specifically diabetic foot ulcers. This figure has been modified from Kim and Martins-Green31. Please click here to view a larger version of this figure.
Discussion
Once chronic wounds are created on the mice, the model can be used to study impaired wound healing processes involved in the initiation of chronicity. The model can also be used to test the efficacy of a wide range of chemicals and drugs that can reverse chronic wound development and impaired healing and lead to wound closure and healing. Different time points after the onset of chronicity can be studied: e.g., days 1-5 after wounding for early onset of chronicity and days 20 and beyond for full strength chronic wounds.
The chronic wound model is also a powerful model to study various aspects of wound healing and complications such as bioburden and cachexia. Bioburden is just one of many facets of chronic wounds that can be studied in this model, as it also affects human chronic wounds. Our procedures and Animal Use Protocol clearly identify symptomology to be monitored, along with a defined monitoring schedule. Animals identified as morbid, based on IACUC approved criteria, are euthanized in order to avoid significant suffering. In addition, the Campus Veterinarian and Animal Health Technician are consulted when certain symptomologies arise and provide supportive guidance in assessment of criteria.
Critical steps within the protocol include housing the db/db-/- mice in a conventional vivarium, removing hair with depilatory lotion, and heating before isoflurane administration. The development of chronic wounds in the chronic wound model relies on non-sterile conditions and practices. These mice are not germ-free and do not grow in very clean vivariums. The microflora that resides in the skin is crucial for the subsequent initiation and development of chronic wounds upon treatment with inhibitors for anti-oxidant enzymes. These db/db-/- mice must be exposed to an environment that contains bacteria, both commensal and pathogenic. If the skin is cleaned and dis-infected with iodine or other antiseptic methods prior to surgery in order to "sterilize" the skin, the wound may not become chronic. The bacteria in the skin microbiome are necessary for the formation and development of biofilm, and delay healing and wound closure. In the clinic, the presence of biofilm in a wound further complicates the wound healing processes and increases the risk of amputation in humans if the infection in the wound is not controlled.
Removing hair with depilatory lotion is an important step to remove excess hair and allow a smooth and clean surface for the transparent dressing to adhere firmly. The depilatory lotion used in this protocol is a chemical depilatory with added aloe for minimizing burns. This product is minimal in altering the morphology of the skin as well as preserving the skin microbiome. The product listed in the Table of Materials is recommended over other hair removing products, including physical and mechanical depilatories (waxes and commercial epilators) that can further burn and tug at the skin and/or kill the skin microbiome. Even though this chemical depilatory is physically gentle, it can still irritate the skin slightly, an effect that could alter the wound healing processes. Thus, it is most effective to wait 18-24 hours before surgery to allow the skin to recover from the procedure and ensure the skin and wound are not affected by it.
These mice are extremely docile and non-responsive to stressors. They are very easy to handle without anesthesia. They are calm enough to place on a palm or on top of a bench without running away. If the base of the tail of the mouse is secured with the thumb and second finger, the mouse will not be able to run away or turn back to bite due to their large belly size. Importantly, in our experience, these mice are extremely sensitive to anesthesia, especially as it relates to loss of homeostasis. Thus, in consultation with our IACUC, it was determined that the better option is to limit anesthetic administration.
The main purpose of this model is to create a large unhealing wound on its back, so once a wound is made, holding a mouse conventionally to make IP injections of additional buprenex or other chemical treatments periodically is impossible. Holding the mouse by the dorsal skin causes the mouse a lot of discomfort and quite possibly pain if the mouse is held in such a traditional manner; thus, all injection with the mouse right side up while it is standing on all four feet. Other experiments utilizing the db/db-/- strain use these mice when they are younger and do not weigh as much, therefore the traditional way of performing an IP injection may used. Since the chronic wound model utilizes mice up to 6 months of age and these mice can weigh up to 80 g, the conventional method is not optimal and can potentially hurt the mouse. We have utilized the method described above for a mouse this obese and have not observed any adverse outcomes when injected with this method with successful aspiration.
Previously, injectable anesthesia such as ketamine and xylazine, were used; however, they proved difficult to use with db/db-/- mice. With a total operation time of less than 5 minutes, the long induction and recovery time was not necessary for the purposes of the experiment. Isoflurane was determined to be the better choice of anesthesia for this procedure due to easy administration and titration, rapid onset and recovery, and adequate anesthetic depth. Also, isoflurane causes minimal cardiac depression and maintains BP very well34. So, a major change in the procedure for the chronic wound model was to use isoflurane as the preferred anesthesia.
The heating before surgery is important for preventing mortality in the 2-3 days following surgery. These mice have significantly lower core body temperature35 and are not able to control their core body temperature effectively due to their genetic manipulation, so an external heating source is provided before the surgery to protect against the additional drop in core body temperature induced by anesthesia. We have assessed the need for a heating pad before, during, and after surgery. It is important to note that db/db-/- mice have unusual physiological responses. Empirically, we have found these mice to be most effectively protected with pre- and post-surgical heat support. By substituting isoflurane as the anesthetic, we measured and determined that the core body temperature did not drop significantly during the less than five minutes of surgery. While we have found the pre- and post- surgical heat support to be critical for these mice, we have not found surgical heat support to have an effect. It should be noted that the IACUC Chair provided guidance and monitored our tests related to temperature and anesthetic effects. As we are communicating this method, we find it important to indicate what is necessary for the success of the procedure.
A limitation of utilizing this method to study biofilm development is the fact that the bacteria present on the wound and producing the biofilm are not controlled. If a specific biofilm-forming bacterium is to be studied, this model may be useful if the native microbiome can be abolished before wounding either through iodine or other antiseptic methods. In response to excessive levels of oxidative stress, key pathogenic bacteria in the skin microbiome are stimulated to initiate biofilm formation. In our chronic wounds, biofilm-forming bacteria include, but are not limited to, Pseudomonas aeruginosa36,37,38,39, Enterobacter cloacae37,38,39, and various Staphylococcus40,41 and Corynebacterium species41,42,43,44,45, all of which can be found in human chronic wounds. Several chronic wound microbiome studies have been conducted on human chronic wounds for the bacteria39,40,41,43,44,45, and fungi46,47 community, including longitudinal surveys associated with poor healing48,49. Longitudinal studies of this nature can also be followed through with the chronic wound model.
It is important to acknowledge that different results may be obtained due to differences in vivaria conditions, supplies, and equipment, vendors, and source colonies for db/db-/- mice. To minimize such differences, provided in the protocol is the exact mice variety and source that is used for the chronic wound experiment. For the husbandry of these mice, the exact bedding and food brands have been provided in the Table of Materials to limit variability. In our experiments, we find that high oxidative stress is necessary and sufficient to create chronic wounds in these mice, as long as the mice are housed in a conventional vivarium and exposed to bacteria. Bacteria populations and communities may differ with vivaria; however, as long as a germ-free facility is not used to house the mice, they should have enough bacteria, both commensal and pathogenic, to reside on the hair and skin.
This method of creating chronic wounds in this protocol is significant to study chronic wounds and impaired wound healing because only oxidative stress levels are significantly altered experimentally. Oxidative stress is required for normal wound healing processes2. It is important for timely regulation and a crucial component in the functionality of cells necessary for wound healing5. However, when the levels of oxidative stress are not controlled, reactive oxygen species can damage endothelia cells, inhibit keratinocyte functionality, and delay wound closure2,5. Human chronic wounds have high levels of oxidative stress50. The mouse model has high blood glucose and already increased levels of oxidative stress due to its morbidity. These characteristics are shared with humans living with diabetes and provide a microenvironment conducive for chronicity after sustaining injury50. Humans also host diverse and complex microbiota in many locations on the body, including the skin, so a complex, but natural, microbiome is allowed to develop on the skin of the mouse.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors have no acknowledgements.
Materials
| B6.BKS(D)-Leprdb/J | The Jackson Laboratory | 00697 | Homozygotes and heterozygotes available |
| Nair Hair Remover Lotion with Soothing Aloe and Lanolin | Nair | a chemical depilatory | |
| Buprenex (buprenorphine HCl) | Henry Stein Animal Health | 059122 | 0.3 mg/ml, Class 3 |
| 3-Amino-1,2,4-triazole (ATZ) | TCI | A0432 | |
| Mercaptosuccinic acid (MSA) | Aldrich | 88460 | |
| Phosphate buffer solution (PBS) | autoclave steriled | ||
| Isoflurane | Henry Schein Animal Health | 029405 | NDC 11695-6776-2 |
| Oxygen | Tank must be compatible with vaporizing system | ||
| Isoflurane vaporizer | JA Baulch & Associates | ||
| Wahl hair clipper | Wahl | Lithium Ion Pro | |
| Acu Punch 7mm skin biopsy punches | Acuderm Inc. | P750 | |
| Tegaderm | 3M | Ref: 1624W | Transparent film dressing (6 cm x 7 cm) |
| Heating pad | Conair | Moist Dry Heating Pad | |
| Insulin syringes | BD | 329461 | 0.35 mm (28G) x 12.7 mm (1/2") |
| 70% ethanol | |||
| Kimwipes | |||
| Tweezers | |||
| Sharp surgical scissors | |||
| Thin metal spatula | |||
| Tubing | |||
| Mouse nose cone | |||
| Gloves | |||
| small plastic containers |
References
- Singer, A. J., Clark, R. A. F. Cutaneous wound healing. New England Journal of Medicine. 341 (10), 738-746 (1999).
- Nouvong, A., Ambrus, A. M., Zhang, E. R., Hultman, L., Coller, H. A. Reactive oxygen species and bacterial biofilms in diabetic wound healing. Physiological Genomics. 48 (12), 889-896 (2016).
- MacLeod, A. S., Mansbridge, J. N. The innate immune system in acute and chronic wounds. Advanced Wound Care. 5 (2), 65-78 (2016).
- Zhao, G., et al. Biofilms and Inflammation in Chronic Wounds. Advanced Wound Care. 2 (7), 389-399 (2013).
- Wlaschek, M., Scharffetter-Kochanek, K. Oxidative stress in chronic venous leg ulcers. Wound Repair and Regeneration. 13 (5), 452-461 (2005).
- Stadelmann, W. K., Digenis, A. G., Tobin, G. R. Physiology and healing dynamics of chronic cutaneous wounds. American Journal of Surgery. 176 (2), 26-38 (1998).
- Loots, M. A., Lamme, E. N., Zeegelaar, J., Mekkes, J. R., Bos, J. D., Middelkoop, E. Differences in cellular infiltrate and extracellular matrix of chronic diabetic and venous ulcers versus acute wounds. Journal of Investigative Dermatology. 111 (5), 850-857 (1998).
- Costerton, W., Veeh, R., Shirtliff, M., Pasmore, M., Post, C., Ehrlich, G. The application of biofilm science to the study and control of chronic bacterial infections. Journal of Clinical Investigation. 112 (10), 1466-1477 (2003).
- Fux, C. A., Costerton, J. W., Stewart, P. S., Stoodley, P. Survival strategies of infectious biofilms. Trends in Microbiology. 13 (1), 34-40 (2005).
- Sen, C. K., et al. Human skin wounds: A major and snowballing threat to public health and the economy. Wound Repair and Regeneration. 17 (6), 763-771 (2009).
- Armstrong, D. G., Wrobel, J., Robbins, J. M. Are diabetes-related wounds and amputations worse than cancer. International Wound Journal. 4 (4), 286-287 (2007).
- James, G. A., et al. Biofilms in chronic wounds. Wound Repair and Regeneration. 16 (1), 37-44 (2008).
- Chen, H., et al. Evidence that the diabetes gene encodes the leptin receptor: Identification of a mutation in the leptin receptor gene in db/db mice. Cell. 84 (3), 491-495 (1996).
- Coleman, D. L. Obese and diabetes: Two mutant genes causing diabetes-obesity syndromes in mice. Diabetologia. 14 (3), 141-148 (1978).
- Garris, D. R., Garris, B. L. Genomic modulation of diabetes (db/db) and obese (ob/ob) mutation-induced hypercytolipidemia: cytochemical basis of female reproductive tract involution. Cell Tissue Research. 316 (2), 233-241 (2014).
- Dhall, S., et al. Generating and reversing chronic wounds in diabetic mice by manipulating wound redox parameters. Journal of Diabetes Research. , (2014).
- Feinstein, R. N., Berliner, S., Green, F. O. Mechanism of inhibition of catalase by 3-amino-1,2,4-triazole. Archives of Biochemistry and Biophysics. 76 (1), 32-44 (1958).
- Margoliash, E., Novogrodsky, A. A study of the inhibition of catalase by 3-amino-1:2:4:-triazole. Biochemical Journal. 68 (3), 468-475 (1958).
- Margoliash, E., Novogrodsky, A., Schejter, A. Irreversible reaction of 3-amino-1:2:4-triazole and related inhibitors with the protein of catalase. Biochemical Journal. 74 (2), 339-348 (1960).
- Shiba, D., Shimamoto, N. Attenuation of endogenous oxidative stress-induced cell death by cytochrome P450 inhibitors in primary cultures of rat hepatocytes. Free Radical Biology and Medicine. 27 (9-10), 1019-1026 (1999).
- Ishihara, Y., Shimamoto, N. Critical role of exposure time to endogenous oxidative stress in hepatocyte apoptosis. Redox Report. 12 (6), 275-281 (2007).
- Valenti, V. E., de Abreu, L. C., Sato, M. A., Ferreira, C. ATZ (3-amino-1,2,4-triazole) injected into the fourth cerebral ventricle influences the Bezold-Jarisch reflex in conscious rats. Clinics. 65 (12), 1339-1343 (2010).
- Welker, A. F., Campos, E. G., Cardoso, L. A., Hermes-Lima, M. Role of catalase on the hypoxia/reoxygenation stress in the hypoxia-tolerant Nile tilapia. American Journal of Physiology. Regulatory, Integrative and Comparative Physiology. 302 (9), 1111-1118 (2012).
- Bagnyukova, T. V., Vasylkiv, O. Y., Storey, K. B., Lushchak, V. I. Catalase inhibition by amino triazole induces oxidative stress in goldfish brain. Brain Research. 1052 (2), 180-186 (2005).
- Falck, E., Karlsson, S., Carlsson, J., Helenius, G., Karlsson, M., Klinga-Levan, K. Loss of glutathione peroxidase 3 expression is correlated with epigenetic mechanisms in endometrial adenocarcinoma. Cancer Cell International. 10 (46), (2010).
- Chaudiere, J., Wilhelmsen, E. C., Tappel, A. L. Mechanism of selenium-glutathione peroxidase and its inhibition by mercaptocarboxylic acids and other mercaptans. Journal of Biological Chemistry. 259 (2), 1043-1050 (1984).
- Dunning, S., et al. Glutathione and antioxidant enzymes serve complementary roles in protecting activated hepatic stellate cells against hydrogen peroxide-induced cell death. Biochimica et Biophysica Acta. 1832 (12), 2027-2034 (2013).
- Franco, J. L., et al. Methylmercury neurotoxicity is associated with inhibition of the antioxidant enzyme glutathione peroxidase. Free Radical Biology and Medicine. 47 (4), 449-457 (2009).
- Sundberg, J. P., Silva, K. A. What color is the skin of a mouse. Veterinary Pathology. 49 (1), 142-145 (2012).
- Curtis, A., Calabro, K., Galarneau, J. R., Bigio, I. J., Krucker, T. Temporal variations of skin pigmentation in C57BL/6 mice affect optical bioluminescence quantitation. Molecular Imaging & Biology. 13 (6), 1114-1123 (2011).
- Kim, J. H., Martins-Green, M. Protocol to create chronic wounds in diabetic mice. Nature Protocols Exchange. , (2016).
- Aasum, E., Hafstad, A. D., Severson, D. L., Larsen, T. S. Age-dependent changes in metabolism, contractile function, and ischemic sensitivity in hearts from db/db mice. Diabetes. 52 (2), 434-441 (2003).
- Vannucci, S. J., et al. Experimental stroke in the female diabetic, db/db, mouse. Journal of Cerebral Blood Flow & Metabolism. 21 (1), 52-60 (2001).
- Janssen, B. J., et al. Effects of anesthetics on systemic hemodynamics in mice. American Journal of Physiology-Heart and Circulatory Physiology. 287 (4), 1618-1624 (2004).
- Osborn, O., et al. Metabolic characterization of a mouse deficient in all known leptin receptor isoforms. Cellular and Molecular Neurobiology. 30 (1), 23 (2010).
- Scales, B. S., Huffnagle, G. B. The microbiome in wound repair and tissue fibrosis. Journal of Pathology. 229 (2), 323-331 (2013).
- Gjødsbøl, K., et al. No need for biopsies: Comparison of three sample techniques for wound microbiota determination. International Wound Journal. 9 (3), 295-302 (2012).
- Wolcott, R. D., et al. Analysis of the chronic wound microbiota of 2,963 patients by 16S rDNA pyrosequencing. Wound Repair Regeneration. 24 (1), 163-174 (2016).
- Gjødsbøl, K., Christensen, J. J., Karlsmark, T., Jørgensen, B., Klein, B. M., Krogfelt, K. A. Multiple bacterial species reside in chronic wounds: a longitudinal study. International Wound Journal. 3 (3), 225-231 (2006).
- Dowd, S. E., et al. Survey of bacterial diversity in chronic wounds using Pyrosequencing, DGGE, and full ribosome shotgun sequencing. BMC Microbiology. 8 (43), (2008).
- Price, L. B., et al. Community analysis of chronic wound bacteria using 16S rrna gene-based pyrosequencing: Impact of diabetes and antibiotics on chronic wound microbiota. PLoS One. 4 (7), 6462 (2009).
- Scales, B. S., Huffnagle, G. B. The microbiome in wound repair and tissue fibrosis. Journal of Pathology. 229 (2), 323-331 (2013).
- Dowd, S. E., et al. Polymicrobial nature of chronic diabetic foot ulcer biofilm infections determined using bacterial tag encoded FLX amplicon pyrosequencing (bTEFAP). PLoS One. 3 (10), 3326 (2008).
- Price, L. B., et al. Macroscale spatial variation in chronic wound microbiota: A cross-sectional study. Wound Repair and Regeneration. 19 (1), 80-88 (2011).
- Gontcharova, V., Youn, E., Sun, Y., Wolcott, R. D., Dowd, S. E. Comparison of bacterial composition in diabetic ulcers and contralateral intact skin. Open Microbiology Journal. 4, 8-19 (2010).
- Smith, K., et al. One step closer to understanding the role of bacteria in diabetic foot ulcers: characterising the microbiome of ulcers. BMC Microbiologyogy. 16 (54), (2016).
- Gardner, S. E., Hillis, S. L., Heilmann, K., Segre, J. A., Grice, E. A. The Neuropathic diabetic foot ulcer microbiome is associated with clinical factors. Diabetes. 62 (3), 923-930 (2013).
- Loesche, M., et al. Temporal stability in chronic wound microbiota is associated with poor healing. Journal of Investigative Dermatology. 137 (1), 237-244 (2017).
- Kalan, L., et al. Redefining the chronic-wound microbiome: Fungal communities are prevalent, dynamic, and associated with delayed healing. MBio. 7 (5), 01058-01116 (2016).
- Blakytny, R., Jude, E. The molecular biology of chronic wounds and delayed healing in diabetes. Diabetic Medicine. 23 (6), 594-608 (2006).

