Differentiation Capacity of Human Aortic Perivascular Adipose Progenitor Cells
Summary
The goal of this protocol is to test the ability of progenitor cells derived from human perivascular adipose tissue to differentiate into multiple cell lineages. Differentiation was compared to mesenchymal stem cells derived from human bone marrow, which is known to differentiate into adipocyte, osteocyte, and chondrocyte lineages.
Abstract
Adipose tissue is a rich source of multi-potent mesenchymal stem cells (MSC) capable of differentiating into osteogenic, adipogenic, and chondrogenic lineages. Adipogenic differentiation of progenitor cells is a major mechanism driving adipose tissue expansion and dysfunction in response to obesity. Understanding changes to perivascular adipose tissue (PVAT) is thus clinically relevant in metabolic disease. However, previous studies have been predominately performed in the mouse and other animal models. This protocol uses human thoracic PVAT samples collected from patients undergoing coronary artery bypass graft surgery. Adipose tissue from the ascending aorta was collected and used for explantation of the stromal vascular fraction. We previously confirmed the presence of adipose progenitor cells in human PVAT with the capacity to differentiate into lipid-containing adipocytes. In this study, we further analyzed the differentiation potential of cells from the stromal vascular fraction, presumably containing multi-potent progenitor cells. We compared PVAT-derived cells to human bone marrow MSC for differentiation into adipogenic, osteogenic, and chondrogenic lineages. Following 14 days of differentiation, specific stains were utilized to detect lipid accumulation in adipocytes (Oil red O), calcific deposits in osteogenic cells (Alizarin Red), or glycosaminoglycans and collagen in chondrogenic cells (Masson’s Trichrome). While bone marrow MSC efficiently differentiated into all three lineages, PVAT-derived cells had adipogenic and chondrogenic potential, but lacked robust osteogenic potential.
Introduction
Adipose tissue is a rich source of multi-potent mesenchymal stem cells (MSC) capable of differentiating into osteogenic, adipogenic, and chondrogenic lineages1. This tissue expands through hypertrophy of mature adipocytes and de novo differentiation of resident MSC to adipocytes. Perivascular adipose tissue (PVAT) surrounds blood vessels and regulates vascular function2,3. Obesity-induced PVAT expansion exacerbates cardiovascular pathologies. While the multipotent potential of MSC from human subcutaneous adipose depots have been well studied4,5, no studies have explanted and evaluated the differentiation capacity of human PVAT-derived progenitor cells, likely due to the invasiveness of procurement. Thus, the goal of this work is to provide a methodology to explant and propagate progenitor cells from human aortic PVAT from patients with cardiovascular disease and to test their propensity to differentiate to osteogenic, chondrogenic, and adipogenic lineages. Our source of PVAT is from the site of anastomosis of the bypass graft on ascending aorta of obese patients undergoing coronary artery bypass graft surgery. Freshly-isolated PVAT is enzymatically-dissociated and the stromal vascular fraction is isolated and propagated in vitro, enabling us to test for the first time the differentiation capacity of human PVAT-derived progenitor cells.
Using primary cultured human PVAT stromal vascular fraction, we tested three assays designed to induce stem/progenitor cells to differentiate toward adipogenic, osteogenic, or chondrogenic lineages. Our prior study identified a population of CD73+, CD105+, and PDGFRa+ (CD140a) cells that can robustly differentiate into adipocytes6, although their multipotency was not tested. PVAT directly regulates vascular tone and inflammation7. The rationale for testing the differentiation potential of this novel cell population is to begin to understand the specialized influence of PVAT on vascular function, and mechanisms of PVAT expansion during obesity. This methodology enhances our understanding of the functions of adipose-tissue derived progenitor cells and enables us to identify and compare similarities and differences of progenitor cells from different tissue sources. We build upon established and validated approaches for isolating and differentiating MSC towards different lineages and optimize procedures to maximize the viability of human PVAT-derived progenitor cells. These techniques have broad applications in the fields of stem and progenitor cell research and adipose tissue development.
Protocol
The use of human tissues in this study was evaluated and approved by the Institutional Review Board of Maine Medical Center, and all personnel received appropriate training prior to experimentation.
1. Preparations
- Make dissociation buffer by reconstituting 50 mg animal-free collagenase/dispase blend I solution with 1 mL of nanopure H2O. Prepare 1 mg/mL working solution by adding 49 mL of high glucose DMEM containing 1% w/v BSA to the reconstituted collagenase/dispase solution. Store 5 mL working aliquots at -20 °C and warm to 37 °C using a water bath prior to use.
- Make antibiotic solution by adding 1 mL of 100x antibiotic/antimycotic solution (final concentration is 200 units/mL penicillin, 200 units/mL streptomycin and 0.5 µg/mL fungizone) to 49 mL of HBSS, and store on ice.
- Prepare gelatin solution (0.2% w/v) by dissolving 200 mg gelatin from bovine skin in 100 mL of nanopure H2O. Autoclave the solution for 20 min and store at 4 °C.
- Prepare 500 mL of PVAT growth media: high glucose DMEM/F12 with glutamine supplement, sodium pyruvate, and sodium bicarbonate, supplemented with 10% FBS and 100 µg/mL antimicrobial agent (see Table of Materials).
- Prepare 50 mL adipogenic induction media: 40.8 mL high glucose DMEM, supplemented with 7.5 mL PVAT growth media, 500 µL of 100x antibiotic/antimycotic solution (final concentration is 100 units/mL penicillin, 100 units/mL streptomycin and 0.25 µg/mL fungizone), 0.5 mM IBMX (500 mM stock in 0.1 M NaOH), 1 µM dexamethasone (1 mM stock in ethanol), 5 µM rosiglitazone (5 mM stock in DMSO), 33 µM biotin (66 mM stock in DMSO), 100 nM insulin (200 µM stock in 0.1% acetic acid), and 20 µM pantothenic acid (100 mM stock in H2O).
- Prepare 50 mL of control induction media for the adipogenic lineage: 40.8 mL high glucose DMEM supplemented with 7.5 mL PVAT growth media and 500 µL of pen-strep (100x stock).
- Prepare 50 mL adipogenic maintenance media: 41.5 mL high glucose DMEM supplemented with 7.5 mL PVAT growth media from step 1.3, 500 µL pen-strep (100x stock), 1 µM dexamethasone (1 mM stock in EtOH), 33 µM biotin (66 mM stock in DMSO), 100 nM insulin (200 µM stock in 0.1% acetic acid), and 20 µM pantothenic acid (100 mM stock in H2O).
- Prepare 500 mL of growth media for the osteogenic and chondrogenic lineages: high glucose αMEM supplemented with 10% FBS, 1x glutamine supplement (see Table of Materials), and 5 mL of pen-strep (100x stock).
- Prepare 50 mL of induction media for the osteogenic lineage: 50 mL of the bone marrow MSC growth media supplemented with 10 nM dexamethasone and 10 mM β-glycerophosphate.
- Prepare 50 mL of induction media for the chondrogenic lineage: 50 mL of the bone marrow MSC growth media supplemented with 100 nM dexamethasone, 50 µg/mL sodium ascorbate-2-phosphate, and 10 ng/mL TGFβ1. For the osteogenic and chondrogenic conditions, the basal bone marrow MSC growth media will serve as the non-induction media.
2. Protocol 1: Culture Human PVAT Cells from the Stromal Vascular Fraction
NOTE: PVAT is resected from the site of graft anastomosis on the ascending aorta of anesthetized patients undergoing coronary artery bypass graft procedures. Aortic PVAT is placed in a 15 mL conical containing 10 mL ice cold high glucose DMEM F12 and transferred from the operating room to the laboratory within 2 h of resection. Aortic PVAT is discarded tissue during bypass procedure and has been deemed as non-human subjects research by Maine Medical Center’s Internal Review Board.
- This protocol is for a ~500 mg piece of human PVAT (approximately 3 x 1 x 0.5 cm3). Transfer fresh human PVAT from DMEM to a 50 mL conical tube containing 25 mL antibiotic solution. Incubate with rocking for 20 min at 4 °C. While the PVAT is in antibiotic solution, thaw an aliquot of dissociation buffer at 37 °C.
- Add 50 µL of 100x antibiotic/antimycotic solution to 5 mL dissociation buffer and sterilize using a 0.22 µm syringe filter. Add 1 mL of gelatin solution to 1 well of a 24-well plate. In a laminar flow hood, use sterile forceps and scissors to transfer PVAT from the antibiotic solution to a sterile Petri dish. Add 1 mL of pre-warmed dissociation buffer to the tissue and finely mince the entire tissue into a slurry (no pieces larger than ~2 x 2 mm2) using sterile forceps and dissection scissors.
- Transfer the 1 mL slurry to 4 mL dissociation buffer and incubate the tube on its side in a pre-warmed 37 °C orbital shaker at 200 rpm for 1 h. After 1 h, no visible tissue pieces will be present, and the solution will appear as a cloudy cell suspension.
- Filter the solution through a 70 µm cell strainer set atop a 50 mL conical tube. Rinse the strainer with an additional 10 mL antibiotic solution to capture as many cells as possible. Do not squeeze the strainer.
- Pellet the cells for 12 min at 300 x g in a swinging bucket centrifuge.
NOTE: After centrifugation, the tube will be separated into a fatty top layer of adipocytes, an interphase, and a pellet. The pellet is the stromal vascular fraction containing endothelial cells, immune cells, blood cells, and progenitor cells. - Resuspend the pellet in 10 mL of HBSS and centrifuge for 5 min at 300 x g. Repeat this step for a total of 2 washes in HBSS. After the final wash, do not lyse the red blood cells. Repeated attempts with several commercially-available buffers and incubation times have led to marked reductions in progenitor cell attachment and viability.
- Aspirate gelatin from the 24-well plate. Gently wash the well 1x with HBSS to remove unbound gelatin.
- Resuspend stromal vascular fraction pellet with intact red blood cells in 1 mL of growth media and seed onto the gelatin-coated well. Add human FGF2 (resuspended in PBS supplemented with 0.01% BSA w/v) to a final concentration of 25 ng/mL in culture medium. Incubate for 24 h at 37 °C with 5% CO2.
- On the next day, remove growth media and wash wells 5x with HBSS to remove red blood cells and dead cells. Add in 1 mL of fresh growth media supplemented with 25 ng/mL FGF2.
- Change the media every 48 h, making sure to supplement with 25 ng/mL fresh FGF2 each time.
- Cells typically reach 100% confluence 7-10 days after explant; passage cells then:
- Aspirate growth media and wash monolayer 2x in 1 mL HBSS. Aspirate all HBSS from the wells and add a few drops of cell dissociation solution.
- Tap and swirl the plate several times and incubate at 37 °C with 5% CO2 for 5–7 min to lift the cells. Add ~1 mL of fresh culture medium to the detached cells and distribute 500 µL to 2 wells of a 24-well plate, each containing 500 µL growth media and 25 ng FGF2.
- Continue to expand human PVAT-derived cells as indicated in the previous step. Each passage should be no greater than a 1:2 split. Cells are passaged 5–7 times before allocating for differentiation assays.
3. Protocol 2: Culture Human Bone Marrow MSC Colonies
NOTE: Human bone marrow MSC are isolated as described8 and stored as early passage frozen stocks in freeze media (70% FBS 20% basal DMEM and 10% DMSO) at ~100,000 cell/mL in liquid N2.
- Rapidly thaw a vial of bone marrow MSC from the liquid N2 in a 37 °C water bath and plate to one well of a 6-well culture plate containing 3 mL of MSC growth media and incubate overnight at 37°C and 5% CO2.
- On the next day, aspirate MSC culture media and wash the cells 3x in 2 mL of HBSS. At 100% confluence, aspirate growth media and wash the cells 3x in 2 mL of HBSS. Add 500 µL/well cell detachment solution and incubate at 37 °C and 5% CO2 for 5 min. Tap the plate to ensure all cells are dislodged and evenly distribute contents to 2 wells of a 6-well plate containing 2 mL MSC growth media.
- Expand the human bone marrow and PVAT-derived MSC in parallel for approximately 5–7 passages or until sufficient quantities for testing has been achieved.
4. Protocol 3: Plate and Induce Adipogenic, Osteogenic, and Chondrogenic Lineages
- Plate appropriate numbers of bone marrow and PVAT-derived cells per well of a 12-well plate and appropriate replicates for each experimental condition. For adipogenic and osteogenic conditions, ~200,000–225,000 cells/well are needed, for chondrogenic conditions, 150,000–175,000 cells/well are needed.
NOTE: We ran a minimum of N = 3 replicate wells for control and induced conditions for each experimental lineage for a total of 2 independent runs (N = 6 replicates total). - Disassociate cells from both the human PVAT progenitor cell population and the human bone marrow MSC population using cell detachment solution and incubation at 37 °C and 5% CO2 for 5 min. Pool populations into separate 15 mL conical vials. Spin the vial down at 500 x g for 7 min to pellet the cells. Resuspend in 1 mL of PBS and use a hemocytometer to estimate the cell number.
- Plate the cells in 12-well dishes as indicated in step 4.1. Provide separate dishes for induced and non-induced adipogenic, osteogenic, and conditions so that the non-induced condition can be fixed at an earlier time point without disrupting continuing culture of the induced condition.
- Add 1.5 mL of adipogenic and osteogenic induction media to each well of the induced condition. Add 1.5 mL of adipogenic and osteogenic non-induction media to each well of the non-induced condition. Begin incubation of the adipogenic and osteogenic induced and non-induced cell populations at 37 °C and 5% CO2.
- Spin down the remaining volume of human PVAT progenitor cells and human bone marrow MSCs for 7 min at 500 x g.
- Determine the volume needed to resuspend remaining bone marrow and PVAT-derived cell pellets to achieve a density of 100,000 cells/10 µL (106 cells/mL). Resuspend pellets in the calculated volume of MSC growth media for chondrogenic lineage induction. Gently move the volume of cells up and down using a pipet to ensure a homogenous distribution.
- Pipette a 10 µL droplet of the concentrated cell solution into the center of each well to form a micromass of 100,000 cells. Place 1 mL of sterile H2O in the adjacent well to prevent evaporation. Incubate the micromass cultures for 2 h at 37 °C and 5% CO2 to allow the micromass to aggregate.
- After 2 hours, carefully add chondrogenic differentiation media spiked with 10 ng/mL human TGFβ1 to each of the induced-condition wells. Carefully add 1.5 mL of the non-induction media (bone marrow MSC growth media) to the non-induced condition wells. Use separate 12-well plates for the induced and non-induced conditions so that the non-induced condition can be fixed at an earlier time point.
5. Protocol 4: Culture Adipogenic, Osteogenic, and Chondrogenic Lineages for 14 Days
- Culture the induced and non-induced conditions of all three lineages for 4 days at 37 °C and 5% CO2, refreshing media every 2 days. On day 4, change the induced adipogenic lineage condition from induction media to maintenance media for the remainder of the assay.
- Fix all of the non-induced conditions in 10% formalin for 12 h. Dispose of formalin and wash all fixed wells 2x in PBS to remove all traces of formalin. Store plates in PBS at 4 °C until processing.
- Culture the induced conditions for a total of 14 days, refreshing media every 2 days. Add fresh TGFβ1 at 10 ng/mL final concentration with each refresh of the chondrogenic induction media. Continue to culture the adipogenic lineage in the adipogenic maintenance media and osteogenic lineage induction media, refreshing every 2 days. At day 14, fix all induced conditions for 12 h in 10% formalin for staining.
- Scrape or pour the micromass in the induced chondrogenic condition into a cassette for embedding. Dehydrate the micromass in a series of increasingly concentrated alcohol baths for 5 min, beginning at 70%, then 80%, twice in 95%, and twice more in absolute alcohol. Place the cassette in a dealcoholization agent and then finally embed the cassette in paraffin wax for sectioning and staining.
6. Protocol 5: Staining Adipogenic Condition with Oil Red O
- Prepare Oil Red O stock solution by dissolving 350 mg of Oil Red O in 100 mL of 100% isopropanol. Mix for 2 h with a stir bar and vacuum through a 0.2 µm filter. Stock solution can be stored in the dark at room temperature for 1 year.
- Prepare Oil Red O working solution by mixing 3 parts stock solution to 2 parts diH20 (e.g. 60 mL of stock solution to 40 mL diH20). The final concentration of Oil Red O in the working solution is 2.1 mg/mL.
- Remove all fluid from wells. Wash each well 2x with 60% isopropanol, making sure to completely remove all water from the sides of the wells. Aspirate 60% isopropanol and quickly add Oil Red O working solution without touching the walls of the wells to cover the bottom. It is critical that this step is done quickly so the wells do not dry.
- Once the Oil Red O is added, incubate for 10 min at room temperature with gentle rocking.
- Remove all Oil Red O and immediately add distilled H2O. Wash with distilled H2O for 10m to begin to remove unbound Oil Red O. After 10 min, aspirate Oil Red O and repeat the washing procedure 3x.
- Once washing is complete, add PBS and image the cells. Store stained cells in PBS at 4 °C.
7. Protocol 6: Staining Osteogenic Condition with Alizarin Red
- Remove all fluid from each well. Add appropriate volume of 2% Alizarin Red stain solution to each well (1.5 mL per well for a 12-well plate) and gently tilt the plate side-to-side until the solution completely covers the bottom of the well.
- Incubate for 15 min at room temperature. Remove Alizarin Red from the wells. Gently rinse each well four times with distilled H2O, taking caution to not dislodge calcium crystals. Allow to dry.
8. Protocol 7: Staining Chondrogenic Condition with Masson’s Trichrome
- Bake slides in a 60 °C dry oven for 30 min. Deparaffinize and hydrate sections to distilled water.
- To rehydrate, place slides in three 5 min incubations with dealcoholization agents, followed by 5 min incubations in descending alcohol concentrations as follows: two at 100% (absolute ethanol), two at 95% alcohol, two at 80%, two at 70%, and finally two in distilled H2O. Slides can remain in H2O while Bouin’s fixative is prepared.
- Place 40 mL of Bouin’s fixative in a plastic microwavable coplin jar (uncovered). Microwave on high to bring temp to 55 °C. Place slide in heated solution for 20 min; let stand on the counter covered. Wash in running tap water for 10 min or until all of the yellow color disappears.
- Stain sections in Weigert’s hematoxylin for 10 min. Wash in running tap water for 10 min.
- Stain sections in Beibrich’s scarlet acid fuchsin solution for 10 min. Wash 3x 10 min in tap water. Mordant slides in phosphotungstic/phosphomolybdic acid solution for 10 min. Do not rinse.
- Place slides directly in aniline blue solution for 10 min. Rinse with two brief dips in tap water.
- Place slides in 1% acetic acid solution for 3 to 5 min. Wash in tap water for 1 min. Place in 95% ethanol for 1 min. Dehydrate as indicated in step 5.4 and mount with synthetic resin.
Representative Results
Isolation of stromal vascular fraction from human PVAT
Figure 1A shows a schematic of the anatomical region where the PVAT overlying the ascending aorta was obtained. We previously described the patient populations undergoing coronary artery bypass grafting from which these samples were derived6. Figure 1B shows an example of the human PVAT obtained following surgery. Figure 1C shows a representative PVAT tissue section stained with Masson’s Trichrome stain. Figure 1D is a phase contract micrograph showing a population of PVAT-derived stromal cells during the expansion phase prior to differentiation. Cells can be expanded and frozen for future use. Cells are typically frozen at a density of 2.5 x 105 cells/mL in a media comprised of 70% FBS, 20% basal (antibiotic free) DMEM F12 and 10% DMSO in an isopropanol cryochamber at -80 °C for 24 h and moved to the liquid phase of a liquid N2 freezer for long-term storage.
Adipogenic differentiation
Studies were performed in parallel with human bone marrow-derived MSC (Figure 2A,B) and PVAT-derived progenitor cells (Figure 2C,D). The left panels of Figure 2 show the non-induced condition, where no lipid accumulation is evident. The right panels show cells following adipocyte differentiation and staining of neutral lipids with Oil Red O. While the degree of differentiation in the human aortic PVAT-derived cells is more robust, both human cell sources exhibited the ability to differentiate towards the adipogenic lineage.
Osteogenic differentiation
The osteogenic differentiation protocol was used for human bone marrow-derived MSC (Figure 3A–C) and PVAT-derived cells (Figure 3D–F). Non-induced cells (Figure 3A,D) did not stain with Alizarin Red. After the osteogenic differentiation protocol, the human MSC developed calcified nodules that stained with Alizarin Red (Figure 3B–C), while human aortic PVAT-derived cells did not (Figure 3E–F). These data indicate that our preparation of cells from the stromal vascular fraction of human PVAT lack significant number of progenitors with the ability to undergo osteogenesis. Depending on the study and time course of differentiation, it is advisable to follow up standard stains with detection of molecular markers that define osteogenic lineage commitment (e.g. RUNX2, osterix, alkaline phosphatase) or osteoblasts (osteopontin, osteocalcin, alkaline phosphatase, BAP1).
Chondrogenic differentiation
Cells derived from both human bone marrow MSC (Figure 4A) and human PVAT (Figure 4B) display features characteristic of chondrogenic differentiation, with abundant collagen accumulation in the micromass. Micromasses formed from human bone marrow MSC and aortic PVAT-derived cells also exhibited abundant accumulation of glycosaminoglycans (blue) as indicated by Alcian blue staining (Figure 4C–D, respectively). Morphologically, structures similar to lacunae were detected with cells sitting in cavities surrounding by collagen deposition (Figure 4, arrows). Depending on the study and time course of differentiation, the detection of specific chondrogenic markers (aggrecan, collagen type II, osteonectin, Sox9) is useful.
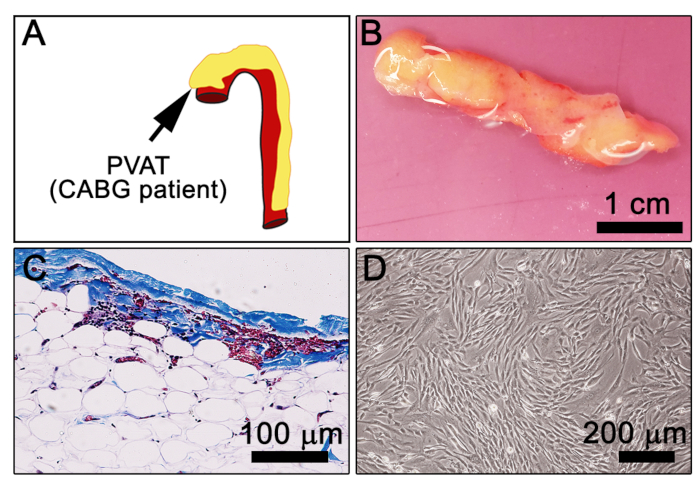
Figure 1: Morphological characteristics of human PVAT. (A) Cartoon depiction of the human aorta (red) with surrounding PVAT (yellow). Arrow indicates ascending aorta. (B) A 480 mg piece of human aortic PVAT from a CABG patient in DMEM prior to dissociation. (C) Masson’s trichrome staining of formalin-fixed paraffin-embedded human PVAT (dark brown/black = nuclei, blue/purple = connective tissue, pink = cytoplasm; Note, RBC appear reddish-pink. (D) Representative image of explanted PVAT-stromal cells at 7 days in culture. Please click here to view a larger version of this figure.
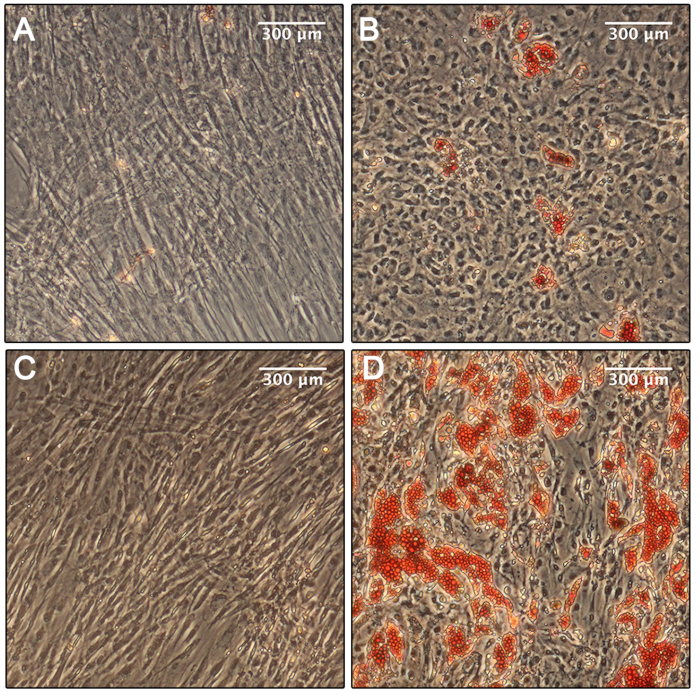
Figure 2: Adipogenic differentiation. (A) Phase microscopy of the human bone marrow MSC in the non-induced condition shows no evidence of differentiation. (B) Phase microscopy of the human bone marrow MSC in the induced adipogenic condition shows some differentiation and immobilization of neutral lipids, stained with Oil Red O after 14 days. (C) Phase microscopy of the human aortic PVAT-derived cells in the non-induced adipogenic condition shows no evidence of differentiation. (D) Phase microscopy of the human aortic PVAT-derived cells in the induced adipogenic condition shows robust differentiation and immobilization of neutral lipids, stained with Oil Red O after 14 days. Please click here to view a larger version of this figure.
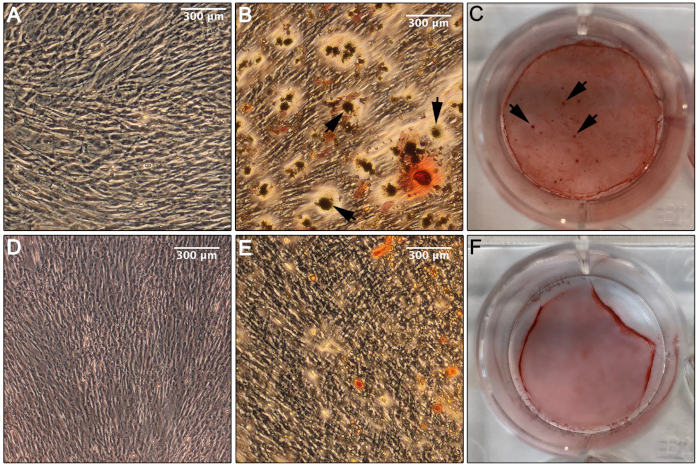
Figure 3: Osteogenic differentiation. (A) Phase microscopy of the human bone marrow MSC in the non-induced osteogenic condition shows no evidence of differentiation toward an osteogenic lineage. (B) Phase microscopy of the human bone marrow MSC in the induced condition after 14 days in culture shows evidence of differentiation and formation of calcium deposits stained with Alizarin Red (arrows). (C) An image of the well containing the human bone marrow MSC in the induced osteogenic condition following staining with Alizarin Red indicates abundant calcium deposition, suggesting successful differentiation toward an osteogenic lineage (arrows, calcium deposition). (D) Phase microscopy of the aortic human PVAT-derived cells in the non-induced osteogenic condition exhibits no indication of differentiation toward an osteogenic lineage. (E) Phase microscopy of the aortic human PVAT-derived cells in the induced osteogenic condition after 14 days in culture shows no evidence of differentiation toward an osteogenic lineage or deposition of calcium when stained with Alizarin Red, only non-specific staining. (F) An image of the well containing the human aortic PVAT-derived cells in the induced osteogenic condition following staining with Alizarin Red shows only non-specific staining. Please click here to view a larger version of this figure.
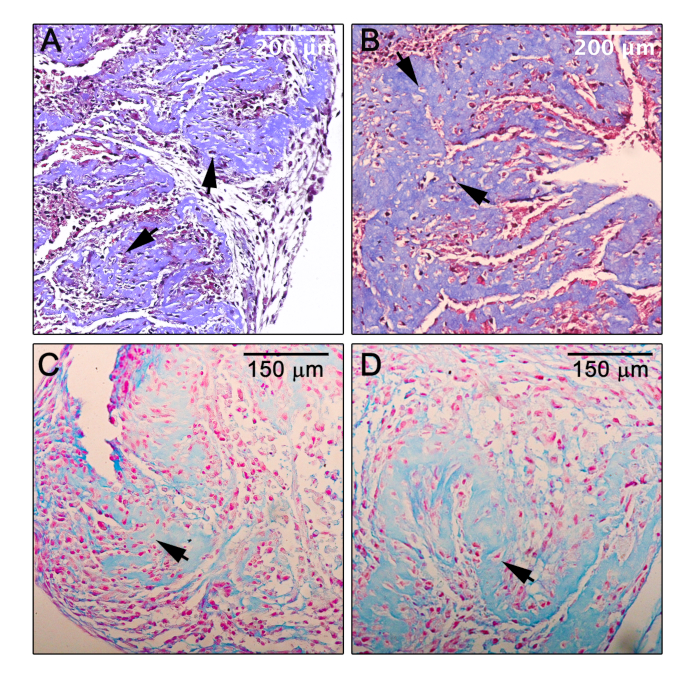
Figure 4: Chondrogenic differentiation. (A,B) Light microscopy of a section of the micromass formed by human bone marrow MSC (A) or human aortic PVAT progenitor cells (B) in the induced chondrogenic condition after 14 days in culture and subsequently stained with Masson’s Trichrome, which indicates significant deposition of collagen (blue) and suggests successful differentiation toward a chondrogenic lineage. (C,D) Light microscopy of a section of the micromass formed by human bone marrow MSC (C) or human aortic PVAT progenitor cells (D) in the induced chondrogenic condition after 14 days in culture and subsequently stained with Alcian blue, which indicates deposition of acidic proteoglycans (e.g. glycosaminoglycans) as are typically found in cartilage. Counterstain, nuclear fast red; arrows, lacunae-like structures. Please click here to view a larger version of this figure.
Discussion
Adipose progenitor cells from different depots vary widely in phenotype and differentiation potential9. Culturing PVAT-derived progenitors from a single patient donor in simultaneous induction down three different lineages, adipogenic, osteogenic, and chondrogenic, allows for a well-controlled investigation of the pluripotent capacity of this novel population of progenitor cells. The methodology described in this report can be used to test the differentiation capacity of progenitor cells from human PVAT and to understand their function in regulating PVAT pathologies and vascular tone. Some benefits of this technique are its simplicity and use of primary human tissue from coronary artery bypass patients, thus enabling the investigation of PVAT progenitor cell function from patients with severe cardiovascular disease. However, explants from a 500 mg piece of tissue typically yield <1000 adherent cells and thus require 5–7 passages to obtain adequate numbers, highlighting one important caveat to this approach. Critical for success of the tri-lineage differentiation experiments is the quality of the initial cell explant from donor PVAT. It is essential to finely mince the tissue thoroughly prior to enzymatic dissociation. Additionally, unlike other explant protocols, we found a dramatic loss of cell number, viability and overall quality when red blood cell lysis buffer was used prior to plating. Instead, it is strongly recommended to plate the entire stromal vascular fraction (including red blood cells). After 24 h, adherent cells will have attached and non-lysed red blood cells can be removed through repeated washes with HBSS. Passaging of the PVAT-derived stromal cells should be no greater than 1:2 splits. We observed a reduction in growth rate when the cells were split 1:3 or greater.
The most difficult component of the trilineage differentiation assay is the chondrogenic condition, as it requires a less common method of culturing cells in a “micromass” and histological embedding and sectioning. Care must be taken to properly plate the 10 µL droplet of 100,000 cells and not disturb the mass on adding culture medium. We found variation between assays as to whether or not the micromass remained adhered to the plate or suspended in the medium. Even when the mass did not remain adhered, the micromass structure and cell viability remained consistent.
Stem or progenitor cells within the vascular microenvironment are responsible for tissue repair in response to injury or disease. Most well characterized are populations that derive from pericyte/adventitial compartments, although there is still controversy about whether there is a standardized molecular identification of these populations in humans10,11. In this protocol, we study a progenitor cell population specifically derived from human PVAT to test their propensity to differentiate to an osteogenic, chondrogenic or adipogenic lineage. Using staining techniques to define cellular commitment to each lineage, we demonstrate that unlike MSC from human bone marrow, progenitor cells from human PVAT were not able to differentiate towards an osteogenic lineage (Figure 3) but exhibit robust adipogenic differentiation. The chondrogenic differentiation capacity is comparable between bone marrow and PVAT-derived cells. This suggests that PVAT-derived progenitor cells might be more lineage restricted than bone marrow MSC or that the PVAT progenitors do not readily differentiate into osteogenic lineages. Future studies should include an investigation of gene and protein expression of markers for each lineage to more quantitatively evaluate the differentiation capacity of PVAT-derived cells.
Adipose-derived stem cells have been a focus for regenerative medicine applications, due to easy access and the high number of cells that can be derived from adipose tissue12,13. Within the stromal vascular fraction of adipose tissue, there are vascular cells, inflammatory cells, fibroblasts, preadipocytes, and adipose-derived stem cells. There are differences in the stem cell population based on anatomical location of the adipose depot and adipocyte characteristics14. Most importantly, the adipose-derived stem cell appears to have the capacity not only to differentiate into mesenchymal lineages15, but also potential to adopt neuronal16,17 and epidermal18 fates.
Numerous studies have focused on stem/progenitor cells derived from human subcutaneous white adipose tissues, but very few studies have addressed characteristics of the progenitor populations in PVAT. Recent studies have isolated adipocyte progenitor cells from PVAT surrounding mesenteric vessels or thoracic aorta of the rat. These CD34+/CD140a+ populations differentiated into adipocytes, although the capacity to derive other lineages was not tested19.
We recently characterized adipose progenitor cells derived from the stromal vascular fraction of human PVAT from patients with advanced coronary artery disease6. These adipose progenitor cells were CD73+, CD105+, and CD140a+, and efficiently differentiated into adipocytes with thermogenic characteristics (UCP1 expression)6. In the current work, we found a limited lineage potency of the PVAT-derived cells compared to bone marrow-derived MSC, suggesting either a lack or minimal population of adipose-derived stem cells that have been characterized from other human adipose tissues. A consideration for this protocol is the expected variability between human specimens, which may affect the number of stem/progenitor cells within the PVAT sample and their capacity to undergo differentiation to distinct lineages. Age, gender, BMI, medications and other clinical parameters likely affect the phenotype of progenitor cells within PVAT. Thus, expected future modifications of this protocol await a more clearly defined progenitor population within human PVAT, and possibly using cell sorting to isolate specific sub-populations for lineage studies.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We acknowledge the assistance of Research Navigation at Maine Medical Center for assisting with the procurement of clinical tissue, and the Histopathology and Histomorphometry Core (supported by 1P20GM121301, L. Liaw PI) at Maine Medical Center Research Institute for sectioning and staining. This work was supported by NIH grant R01 HL141149 (L. Liaw).
Materials
| animal-free collagenase/dispase blend I | Millipore-Sigma | SCR139 | 50mg |
| Alcian Blue | NewComerSupply | 1003A | 1% Aqueous solution pH 2.5 |
| Alizarin Red | Amresco | 9436-25G | |
| alpha-MEM | ThermoFisher | 12561056 | |
| Aniline Blue | NewComerSupply | 10073C | |
| antibiotic/antimycotic | ThermoFisher | 15240062 | |
| Beibrich's scarlet acid fuchsin | Millipore-Sigma | A3908-25G | |
| b-glycerophosphate | Millipore-Sigma | G9422-10G | |
| Biebrich Scarlet | EKI | 2248-25G | |
| biotin | Millipore-Sigma | B4501-100MG | |
| Bouin's fixative | NewComerSupply | 1020A | |
| bovine serum albumin | Calbiochem | 12659 | stored at 4C |
| Cell detachment solution | Accutase | AT104 | |
| cell strainer (70mm) | Corning | 352350 | |
| dexamethasone | Millipore-Sigma | D4902-100MG | |
| DMEM | Corning | 10-013-CV | 4.5g/L glucose, L-glut and pyruvate |
| DMEM/F12 medium | ThermoFisher | 10565-042 | high glucose, glutamax, sodium bicarbinate |
| DMSO | Millipore-Sigma | D2650 | |
| fetal bovine serum | Atlanta Biologicals | S11550 | |
| FGF2 | Peprotech | 100-18B | |
| formalin | NewComerSupply | 1090 | |
| gelatin, bovine skin | Millipore-Sigma | G9391-500G | |
| glutamax | ThermoFisher | 35050061 | glutamine supplement |
| HBSS | Lonza | 10-547F | |
| IBMX | Millipore-Sigma | I5879-250MG | |
| insulin solution | Millipore-Sigma | I9278-5ML | |
| Oil red O | Millipore-Sigma | O0625-100G | |
| pantothenic acid | Millipore-Sigma | P5155-100G | |
| penicillin-streptomycin solution | ThermoFisher | 15240062 | 100ml |
| permount | Fisher | SP15-500 | |
| phosphotungstic/phosphomoybdic acid solution | Millipore-Sigma | P4006-100G/221856-100G | |
| primocin | Invivogen | ant-pm-1 | Antimicrobial reagent for culture media. |
| rosiglitazone | Millipore-Sigma | R2408-10MG | |
| TGFb1 | Peprotech | 100-21 | |
| Weigert's hematoxylin | EKI | 4880-100G |
References
- Minteer, D., Marra, K. G., Rubin, J. P. Adipose-derived mesenchymal stem cells: biology and potential applications. Advances in Biochemical Engineering/Biotechnology. 129, 59-71 (2013).
- Akoumianakis, I., Tarun, A., Antoniades, C. Perivascular adipose tissue as a regulator of vascular disease pathogenesis: identifying novel therapeutic targets. British Journal of Pharmacology. 174 (20), 3411-3424 (2017).
- Brown, N. K., et al. Perivascular adipose tissue in vascular function and disease: a review of current research and animal models. Arteriosclerosis, Thrombosis, and Vascular Biology. 34 (8), 1621-1630 (2014).
- Bunnell, B. A., Estes, B. T., Guilak, F., Gimble, J. M. Differentiation of adipose stem cells. Methods in Molecular Biology. , 155-171 (2008).
- Scott, M. A., Nguyen, V. T., Levi, B., James, A. W. Current methods of adipogenic differentiation of mesenchymal stem cells. Stem Cells and Development. 20 (10), 1793-1804 (2011).
- Boucher, J. M., et al. Rab27a Regulates Human Perivascular Adipose Progenitor Cell Differentiation. Cardiovascular Drugs and Therapy. 32 (5), 519-530 (2018).
- Nosalski, R., Guzik, T. J. Perivascular adipose tissue inflammation in vascular disease. British Journal of Pharmacology. 174 (20), 3496-3513 (2017).
- Nadri, S., et al. An efficient method for isolation of murine bone marrow mesenchymal stem cells. The International Journal of Developmental Biology. 51 (8), 723-729 (2007).
- Cleal, L., Aldea, T., Chau, Y. Y. Fifty shades of white: Understanding heterogeneity in white adipose stem cells. Adipocyte. 6 (3), 205-216 (2017).
- de Souza, L. E., Malta, T. M., Kashima Haddad, S., Covas, D. T. Mesenchymal Stem Cells and Pericytes: To What Extent Are They Related. Stem Cells and Development. 25 (24), 1843-1852 (2016).
- Majesky, M. W. Adventitia and perivascular cells. Arteriosclerosis, Thrombosis, and Vascular Biology. 35 (8), e31-e35 (2015).
- Miana, V. V., Gonzalez, E. A. P. Adipose tissue stem cells in regenerative medicine. Ecancermedicalscience. 12, 822 (2018).
- Frese, L., Dijkman, P. E., Hoerstrup, S. P. Adipose Tissue-Derived Stem Cells in Regenerative Medicine. Transfusion Medicine and Hemotherapy. 43 (4), 268-274 (2016).
- Mizuno, H., Tobita, M., Uysal, A. C. Concise review: Adipose-derived stem cells as a novel tool for future regenerative medicine. Stem Cells. 30 (5), 804-810 (2012).
- Zuk, P. A., et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Engineering. 7 (2), 211-228 (2001).
- Safford, K. M., et al. Neurogenic differentiation of murine and human adipose-derived stromal cells. Biochemical and Biophysical Research Communications. 294 (2), 371-379 (2002).
- Ashjian, P. H., et al. In vitro differentiation of human processed lipoaspirate cells into early neural progenitors. Plastic and Reconstructive Surgery. 111 (6), 1922-1931 (2003).
- Trottier, V., Marceau-Fortier, G., Germain, L., Vincent, C., Fradette, J. IFATS collection: Using human adipose-derived stem/stromal cells for the production of new skin substitutes. Stem Cells. 26 (10), 2713-2723 (2008).
- Thelen, K., Ayala-Lopez, N., Watts, S. W., Contreras, G. A. Expansion and Adipogenesis Induction of Adipocyte Progenitors from Perivascular Adipose Tissue Isolated by Magnetic Activated Cell Sorting. Journal of Visualized Experiments. (124), (2017).

