Evaluation of Cerebral Blood Flow Autoregulation in the Rat Using Laser Doppler Flowmetry
Summary
This article demonstrates the use of laser Doppler flowmetry to evaluate the ability of the cerebral circulation to autoregulate its blood flow during reductions in arterial blood pressure.
Abstract
When investigating the body's mechanisms for regulating cerebral blood flow, a relative measurement of microcirculatory blood flow can be obtained using laser Doppler flowmetry (LDF). This paper demonstrates a closed skull preparation that allows cerebral blood flow to be assessed without penetrating the skull or installing a chamber or cerebral window. To evaluate autoregulatory mechanisms, a model of controlled blood pressure reduction via graded hemorrhage can be utilized while simultaneously employing LDF. This enables the real time tracking of the relative changes in the blood flow in response to reductions in arterial blood pressure produced by the withdrawal of circulating blood volume. This paradigm is a valuable approach to study cerebral blood flow autoregulation during reductions in arterial blood pressure and, with minor modifications in the protocol, is also valuable as an experimental model of hemorrhagic shock. In addition to evaluating autoregulatory responses, LDF can be used to monitor the cortical blood flow when investigating metabolic, myogenic, endothelial, humoral, or neural mechanisms that regulate cerebral blood flow and the impact of various experimental interventions and pathological conditions on cerebral blood flow.
Introduction
Autoregulatory mechanisms in the cerebral circulation play a crucial role in maintaining homeostasis and normal function in the brain. Autoregulation of the cerebral blood flow is affected by multiple factors including heart rate, blood velocity, perfusion pressure, the diameter of the cerebral resistance arteries, and the microcirculatory resistance, all of which play a role in maintaining the total cerebral blood flow constant in the brain over the physiological range of systemic blood pressures. When arterial pressure increases, these mechanisms constrict arterioles and resistance arteries to prevent dangerous increases in intracranial pressure. When arterial blood pressure decreases, local control mechanisms dilate the arterioles to maintain tissue perfusion and O2 delivery. Various pathological conditions such as hypercapnia, traumatic or global hypoxic brain injury, and diabetic microangiopathy1,2,3,4,5,6 may disrupt the brain's ability to autoregulate its blood flow. For example, chronic hypertension shifts the effective autoregulatory range toward higher pressures7,8,9, and a high salt (HS) diet not only interferes with normal endothelium-dependent dilation in the cerebral microcirculation10, but also impairs the ability of autoregulatory mechanisms in the cerebral circulation to dilate and maintain tissue perfusion when arterial pressure is reduced11. Cerebral autoregulation is also impaired in Dahl salt-sensitive rats when they are fed a HS diet12.
During reductions in arterial pressure, dilation of the cerebral resistance arteries and arterioles initially returns cerebral blood flow to control values despite the reduced perfusion pressure. As arterial pressure is reduced further, cerebral blood flow remains constant at the lower pressure (plateau phase of the autoregulatory response) until the vasculature can no longer dilate to maintain blood flow at the lower pressure. The lowest pressure at which an organ can maintain normal blood flow is termed the lower limit of autoregulation (LLA). At pressures below the LLA, cerebral blood flow decreases significantly from resting values and decreases in a linear fashion with each reduction in arterial perfusion pressure13,14. An upward shift in the LLA, as observed in hypertension7,8,9, may increase the risk and severity of ischemic injury during conditions where the arterial perfusion pressure is reduced (e.g., myocardial infarction, ischemic stroke, or circulatory shock).
LDF has proven to be an extremely valuable approach to evaluate the blood flow in the microcirculation under a variety of circumstances, including autoregulation of the blood flow in the cerebral circulation11,14,15. In addition to evaluating autoregulatory responses, LDF can be used to monitor the cortical blood flow when investigating metabolic, myogenic, endothelial, humoral, or neural mechanisms that regulate the cerebral blood flow and the impact of various experimental interventions and pathological conditions on cerebral blood flow10,16,17,18,19,20,21.
LDF measures the shift in reflected laser light in response to the number and velocity of moving particles–in this case, red blood cells (RBC). For studies of cerebral vascular autoregulation, arterial blood pressure is changed either by the infusion of an alpha-adrenergic agonist to increase arterial pressure (because the cerebral circulation itself is insensitive to alpha-adrenergic vasoconstrictor agonists)12,15 or via controlled blood volume withdrawal to reduce arterial pressure11,14. In the present study, LDF is utilized to demonstrate the effects of graded reductions in blood pressure on cerebral autoregulation in a healthy rat. Although open and closed skull methods have been described in the literature22,23,24,25, the present paper demonstrates a closed skull preparation, allowing cerebral blood flow to be assessed without penetrating the skull or installing a chamber or cerebral window.
Protocol
The Medical College of Wisconsin Institutional Animal Care and Use Committee (IACUC) approved all protocols described in this paper and all procedures are in compliance with the National Institutes of Health (NIH) Office of Laboratory Animal Welfare (OLAW) regulations.
1. Experimental animals and preparation for recording
- Use 8–12-week-old male Sprague-Dawley rats weighing 250–300 g. For these experiments, feed rats a standard diet consisting of 0.4% NaCl, 200 g/kg casein, 3 g/kg DL-methionine, 497.77 g/kg sucrose, 150 g/kg cornstarch, 50 g/kg corn oil, 50 g/kg cellulose, 2 g/kg choline bitartrate, 35 g/kg mineral mix, and 10 g/kg vitamin mix.
- Record arterial blood pressure and LDF readings using data acquisition software or any comparable recording method.
- Attach the arterial pressure transducer to one channel of the recording system and the LDF probe to the other channel on the recording system.
- Prior to the measurement, calibrate the laser Doppler probe to set a motility standard and ensure that the laser Doppler flowmeter is providing a steady output.
- Prepare additional equipment needed for the preparatory surgery and for the experiment: a dissecting microscope, a rodent ventilator, an end tidal CO2 monitor, a stereotaxic instrument to fix the rat's head in position, and a micromanipulator to locate the LDF probe over the pial microcirculation and maintain it in a steady position.
2. Surgical preparation
- Weigh the rat and anesthetize the animal in an induction chamber with 3.5% isoflurane and 30% O2 supplement.
- Remove the animal from the induction chamber and substitute an anesthetic mask delivering 1.5–3% isoflurane with a 30% O2 supplement.
- Place the rat on a circulating water blanket maintained at 37 °C and check reflexes with a toe pinch to ensure that there is a withdrawal reflex. Apply sterile ophthalmic ointment to both eyes to prevent corneal desiccation.
- Shave the top of the cranium, ventral neck area, and femoral triangles. Remove any loose hair from those areas and clean with rubbing alcohol.
- Place the rat in a supine position on a heating pad with a circulating warm water pump to maintain the animal's body temperature at 37 °C and temporarily secure it to the pad using medical tape.
- Install a tracheal cannula (PE240 polyethylene tubing) through a ventral incision in the neck as described elsewhere26.
- Attach the tracheal cannula to an end tidal CO2 monitor and the ventilator delivering 2.5–3.0% isoflurane (depending on the size of the animal) and a 30% O2 inhalation supplement. Make sure the respiratory rate, inspiratory time, and minute ventilatory volume are set and monitored to ensure an expired end tidal CO2 of approximately 35 mmHg throughout the experiment.
NOTE: This is generally achieved with a respiratory rate of approximately 48–60 breaths/min, a tidal volume of 1.70–2.30 mL, and an inspiration time of 0.50–0.60 s for a 250–300 g rat. - Fill two PE50 polyethylene cannulas with 1 U/mL heparin in isotonic NaCl solution to prevent clotting and to maintain patency of the catheters. After filling, bevel the open end of each cannula with surgical scissors to facilitate insertion into the artery.
- Cannulate the right and left femoral arteries as described elsewhere27 to allow continuous monitoring of arterial pressure in one catheter and blood withdrawal from the other catheter.
- After carefully separating the arteries from the surrounding tissue under a dissecting microscope, ligate the distal end of the artery and place two additional sutures around the middle and proximal ends of the artery without tightening the knots.
- Use the proximal suture as a lifting ligature to prevent bleeding from the artery after the incision for cannula insertion (step 2.11).
- Insert a V-shaped wire fashioned from a paper clip under the artery in order to occlude the vessel until the cannula is secured.
- Under a dissecting microscope make a small incision in the femoral artery near the distal ligation using Vannas scissors. Insert the beveled end of the cannula into the incision and advance it into the femoral artery. Tighten the knot on the middle ligature to secure the cannula in place so it is not dislodged by arterial pressure when the lifting ligature or paper clip is removed.
- After the middle ligature is tightened, release the tension on the lifting ligature and/or remove the paper clip, and tighten the proximal ligature.
- Close the incision with fine sutures (3–0 silk) or a surgical staple. Alternatively, place a moist gauze over the incision site, depending on the size of the incision.
3. Skull thinning for LDF measurements
- Immediately after the cannulas are in place, place the animal in a sternal position and secure the head in a stereotaxic device, being careful not to dislodge the catheters or tracheal tube.
- Use surgical scissors to make an elliptical incision in the skin covering the cranium. Use a cotton swab to remove any connective tissue, ensuring that the cranium is clean and dry. Place a small elongated and rolled piece of tissue paper around the incision on the scalp to stop any bleeding.
- Under the dissecting microscope, use a Dremel tool or a dental drill with a 2.15 mm drill bit to thin a small area of bone (approximately 0.5–1 cm depending on the size of the rat) in the parietal area over the left or right somatosensory cortex.
CAUTION: Thin the bone slowly and carefully to avoid penetrating the skull. While performing this step, saline solution should be applied liberally to prevent the area from overheating. - Once the skull has been thinned and the area has a pink appearance and/or blood vessels are visualized, cover the area with mineral oil and use a micromanipulator to position the laser Doppler probe over the exposed cerebral microcirculation so that the tip of the probe is just touching the top of the pool of mineral oil (Figure 1).
NOTE: It is essential to take LDF measurements in an area where there are no external vibrations that would interfere with the laser Doppler readings and that the probe is securely fixed over the same target area throughout the experiment.
4. Assessing cerebral vascular autoregulation
- Once the LDF probe is fixed in position, allow a 30–45 min equilibration period before beginning the experiment. After the equilibration period, measure the mean arterial pressure (MAP) and laser cerebral blood flow (LCBF) every 30 s for 2 min and average the values to obtain the baseline values for the prehemorrhage blood pressure and LCBF.
- To evaluate the cerebral vascular autoregulation in response to arterial pressure reduction, measure the LCBF and MAP following successive withdrawals of 1.5 mL of blood from the femoral artery11. To keep the catheter patent, ensure that a volume of heparin solution (100 U/mL in isotonic saline) approximately equal to the catheter volume is infused after each blood draw.
NOTE: When infusing the heparin solution to maintain catheter patency, it is important to match the volume of the heparin solution to the volume of the catheter as closely as possible to prevent the animal from receiving too much heparin, which could cause unwanted bleeding. - After each blood volume withdrawal, allow the rat to equilibrate for 2 min, after which the MAP and LCBF are recorded every 30 s for 2 min. Repeat the blood volume withdrawals until the animal reaches a MAP of approximately 20 mmHg.
- Determine the effective autoregulatory range by identifying the range of blood pressures from the prehemorrhage MAP to the LLA (steps 4.5 and 5.3, below).
- Determine the LLA by identifying the lowest pressure at which LCBF still returns to within 20% of the prehemorrhage control value following blood volume withdrawal, as previously described11,28 or by identifying the intersection point of the regression lines determined during the plateau phase of autoregulation and below the LLA, where LCBF decreases with each successive blood withdrawal (step 5.3, below).
NOTE: The criteria for defining the LLA and autoregulatory plateau may differ between laboratories (e.g., Takada et al.28 vs. Jones et al.29) as well as procedures for reducing arterial blood pressure (e.g., withdrawal of a specific volume of blood vs. controlled hemorrhage to reach specific arterial pressure levels)11. - At the end of the experiment, euthanize the animal by creating a bilateral pneumothorax while under a surgical plane of anesthesia, as approved by the IACUC.
- LDF values obtained in the tissue after the animal is euthanized will provide the zero baseline flow value for the experimental setup.
5. Statistical analysis
- Perform linear regression analysis to evaluate the correlation between the LDF values and their corresponding arterial pressure. Use the baseline LDF readings obtained after the animal is euthanized to ensure that there was no nonspecific LDF signal affecting the measured flow rates.
- Calculate the LLA using the intersection between the regression lines above and below the autoregulatory plateau. To calculate the LLA using this method, combine the two regression equations and solve the resulting equation for arterial pressure.
- When comparing different experimental groups, use linear regression analysis to calculate the slopes of the LDF vs. arterial pressure relationship above and below the LLA for each animal and summarize them as mean ± SEM for the animals in that experimental group.
Representative Results
Figure 2 summarizes the results of experiments conducted in 10 male Sprague-Dawley rats fed standard laboratory chow. In those experiments, mean LCBF was maintained within 20% of the prehemorrhage value following the first three blood volume withdrawals, until the mean arterial pressure reached the LLA. Subsequent blood volume withdrawals at pressures below the LLA caused a progressive reduction of LCBF, showing that the cerebral circulation was no longer able to produce a sufficient level of vasodilation to maintain cerebral blood flow constant at the lower perfusion pressures.
Figure 3 summarizes the relationship between mean arterial pressure and LCBF in the plateau phase (MAP >65 mmHg) and the decompensatory phase (MAP <65 mmHg) of CBF autoregulation. At pressures at or above the LLA, there was no significant correlation between LCBF and arterial pressure (r2 = 0.0246; p = 0.3534), showing that the LCBF was independent of arterial pressure in the plateau range of the autoregulatory curve. Below the LLA, the LCBF/arterial pressure relationship had a negative slope and LCBF was significantly correlated with arterial pressure (r2 = 0.7907; p = 8.7 x 10–25).
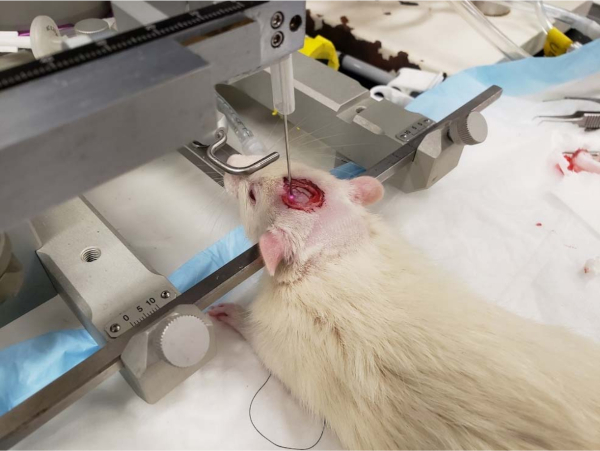
Figure 1: Placement of laser Doppler probe over the thinned skull of an anesthetized rat. Rat in stereotaxic apparatus with an LDF probe positioned over a thinned area of the skull and held in place with a micromanipulator. Please click here to view a larger version of this figure.
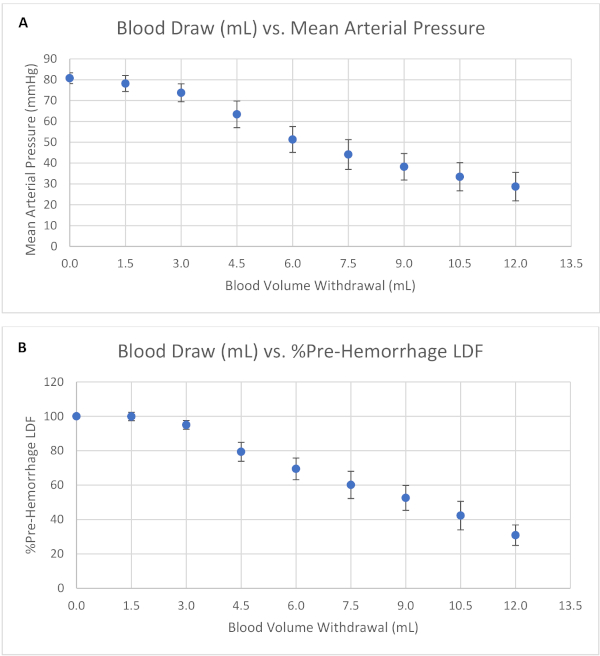
Figure 2: Autoregulation of the cerebral blood flow in response to hemorrhage-induced reductions in arterial blood pressure. Summarized relationship between blood volume withdrawal and (A) mean arterial pressure (MAP) and (B) laser cerebral blood flow (LCBF) in rats fed a standard diet and subjected to sequential blood volume withdrawals. Data shown as mean ± SEM for n = 6–10 after each blood volume withdrawal. Please click here to view a larger version of this figure.
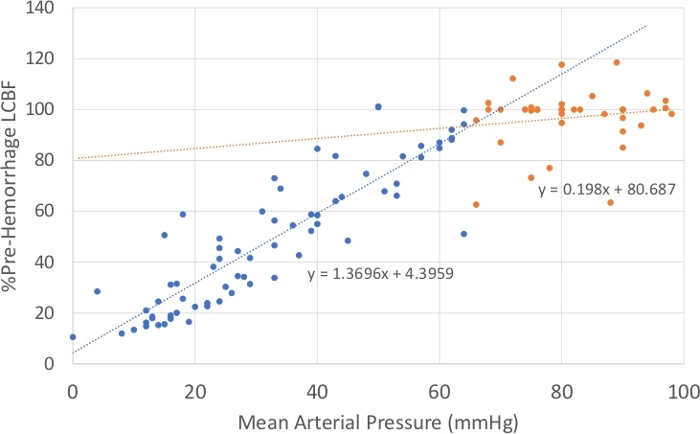
Figure 3: Relationship between the mean arterial pressure and laser cerebral blood flow. Relationship during the plateau phase of the autoregulatory response (n = 37 observations) and in the decompensatory phase of the response (n = 70 observations) are shown, where arterial pressures fell below the LLA (~65 mmHg). LCBF was highly correlated with MAP in the decompensatory phase of autoregulation (r2 = 0.7907; p = 8.7 x 10–25) but not during the plateau phase of autoregulation (r2 = 0.0246; p = 0.3534). Please click here to view a larger version of this figure.
Discussion
Evaluation of Tissue Blood Flow Responses with Laser Doppler Flowmetry (LDF). As noted above, the LDF signal is proportional to the number and velocity of moving particles, in this case RBC, in the microcirculation. LDF readings in different organs are well correlated with whole organ blood flow assessed by established methods such as electromagnetic flow meters and radioactive microspheres30 and are generally consistent with studies evaluating the regulation of active tone in cannulated artery preparations10,31,32,33,34 and in situ microcirculatory preparations35,36.
One consideration when conducting studies of cerebral autoregulation, and possibly autoregulation in other vascular beds, is the potential effect of anesthesia on autoregulatory responses. Although cerebral autoregulation was present in the current study and in an earlier study by our group11 and consistent with the known effects of a HS diet on the vasodilator responses of the cerebral resistance arteries31,32,37, the rat pial arterioles35 and the in situ arterioles of the hamster cheek pouch36, isoflurane anesthesia has been reported to have a strong vasodilator effect38 and to cause cardiovascular suppression39. Isoflurane has also been reported to cause a loss of cerebral vascular autoregulation in mice40,41, so some investigators have used alpha-chloralose anesthesia either alone41 or in combination with urethane42 to study cerebral autoregulation instead.
The numbers and velocities of RBC vary within a microcirculatory bed, between individuals, and within an individual subject over time. Thus, LDF does not provide an absolute value of blood flow within an organ or its microcirculation, between different organs, or in different regions of the microcirculation. Therefore, it is essential to firmly secure the LDF probe so that it remains in the same position and is not subjected to any vibration throughout the experiment. To accurately assess changes in the cerebral blood flow, the rat's head is positioned in a stereotaxic instrument and the LDF probe is held in a micromanipulator over a thinned area of the skull to prevent movement artifacts and to maintain the probe's position relative to the region being studied (Figure 1). Any movement of the probe away from its initial site will produce a signal determined by blood flow in a different area of the tissue, impeding comparisons. Although LDF does not provide a measurement of absolute blood flow, when performed properly it is still a convenient and valuable approach to evaluate the regulation of blood flow at the level of the whole vascular bed30, and the magnitude of the relative increases or decreases in LDF flow relative to a control value can be compared statistically.
Autoregulation of Cerebral Blood Flow. The cerebral circulation can normally tolerate large changes in arterial blood pressure that cause vasoconstriction when arterial pressure is elevated and vasodilation when arterial pressure is reduced via autoregulatory mechanisms. These mechanisms are crucially important to prevent dangerous increases in intracranial pressure when systemic blood pressure increases and to maintain adequate tissue perfusion and oxygen supply when arterial pressure decreases. The present experiments focused on the ability of autoregulatory mechanisms to maintain cerebral blood flow constant as arterial pressure is reduced (rather than the ability of the cerebral circulation to maintain constant blood flow as MAP is increased), although LDF is very valuable and extensively used for the latter studies as well. Another valuable application of this experimental design is to study microvascular blood flow during hemorrhage and in various forms of circulatory shock43,44,45,46.
Autoregulation of LCBF during hemorrhage-induced reductions in arterial pressure is assessed by comparing the LDF flow and MAP measured 2 min after each blood withdrawal with the prehemorrhage control MAP and LCBF measured immediately prior to blood volume withdrawal. At this point, the autoregulatory mechanisms will have acted to dilate the microvasculature to maintain blood flow at the lower perfusion pressure. The LLA is identified as the lowest MAP where autoregulatory mechanisms can still restore blood flow despite the reduction in perfusion pressure. At arterial pressures below the LLA, autoregulatory mechanisms have reached their limit and can no longer dilate the cerebral vasculature enough to prevent further reductions in cerebral blood flow. After the LLA is passed, there is a significant and progressive reduction in LCBF from the prehemorrhage value following each withdrawal of blood to reach the new pressure11. The effectiveness of cerebral vascular autoregulation in response to reductions in arterial blood pressure is evaluated by comparing the slope of the LCBF vs. the arterial pressure relationship before and after the LLA and the width of the plateau phase of autoregulation, defined as the arterial pressure range between prehemorrhage MAP and the LLA. For example, a recent study evaluating the effect of a HS diet on cerebral autoregulation11 found that cerebral blood flow was maintained at a constant level in rats fed with a low salt (LS; 0.4% NaCl) diet during sustained reductions in arterial pressure to values as low as 40–50 mmHg. This finding is consistent with previous estimations of the LLA in healthy rats16,47. However, the plateau phase of cerebral blood flow autoregulation in normotensive Sprague-Dawley rats fed short term (3 days) and chronic (4 weeks) high salt (HS; 4% NaCl) diet decreased progressively with successive reductions in arterial pressure, showing that HS diet eliminates the plateau phase of blood flow regulation that is normally present in healthy normotensive rats and adversely affects the ability of the cerebral circulation to maintain tissue perfusion in the face of reductions in blood pressure11. The finding that autoregulation of cerebral blood flow in response to reduced blood pressure is impaired in rats fed a HS diet is consistent with the results of studies showing that increases in dietary salt impair the relaxation of resistance arteries31,32,33,34,37 and arterioles35,36 of normotensive rats and hamsters.
In addition to providing valuable insights regarding the ability of the microcirculation to autoregulate its blood flow, LDF measurements can be employed in a wide range of applications that provide a dynamic estimation of blood flow control that is unavailable with conventional methods, such as microspheres and electromagnetic flow probes. For example, LDF measurements are extremely valuable in evaluating the response of the microcirculation to vasoactive stimuli such as ACh infusion and administration of other vasoactive agents31,32,33,34,37, elevated arterial pCO210, hypoxia17,48, neurovascular coupling in response to sensory stimuli21,49, functional hyperemia in the brain20, and evaluating tissue responses to hemorrhagic hypotensive stress and various types of circulatory shock43,44,45,46.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors express their sincere thanks to Kaleigh Kozak, Megan Stumpf, and Jack Bullis for their outstanding assistance in completing this study and preparing the manuscript. Grant Support: NIH #R01-HL128242, #R21-OD018309, and #R21-OD024781.
Materials
| 3-0 braided black silk suture | Midwest Vet | 193.73000.2 | |
| Arterial Pressure Transducer | Merit Medical | 041516504A | |
| Automated Data Acquisition Systems (WINDAQ & BIOPAC system) | DATAQ Instruments | ||
| Blood Pressure Display Unit | Stoelting | 50115 | |
| Circulating warm water pump | Gaymar Industries | T-pump | |
| End-tidal CO2 monitor | Stoelting | Capstar-100 | |
| Heparin Sodium | Midwest Vet | 191.46720.3 | |
| Kimwipe | Fisher Scientific | 06-666A | |
| Laser Doppler Flow Meter | Perimed | PeriFlux 5000 LDPM | |
| Laser Doppler Refill Motility Standard | Perimed | PF1001 | |
| Polyethylene Tubing (PE240) (for trachea cannula) | VWR | 63018-828 | |
| Polyethylene Tubing (PE50) (for femoral catheters) | VWR | 63019-048 | |
| Rodent Ventilator | Cwe/Stoelting | SAR-830/P | |
| Saline | Midwest Vet | 193.74504.3 | |
| Sprague-Dawley Outbred Rats | Variable | N/A | Rats were ordered from various companies |
| Standard Rat Chow | Dyets, Inc. | 113755 | |
| Stereotaxic Instrument | Cwe/Stoelting | Clasic Lab Standard |
References
- Aso, Y., Inukai, T., Takemura, Y. Evaluation of microangiopathy of the skin in patients with non-insulin-dependent diabetes mellitus by laser Doppler flowmetry; microvasodilatory responses to beraprost sodium. Diabetes Research and Clinical Practice. 36, 19-26 (1997).
- Golding, E. M., Robertson, C. S., Bryan, R. M. The consequences of traumatic brain injury on cerebral blood flow and autoregulation: a review. Clinical and Experimental Hypertension. 21, 299-332 (1999).
- Grunwald, J. E., DuPont, J., Riva, C. E. Retinal haemodynamics in patients with early diabetes mellitus. British Journal of Ophthalmology. 80, 327-331 (1996).
- Mankovsky, B. N., Piolot, R., Mankovsky, O. L., Ziegler, D. Impairment of cerebral autoregulation in diabetic patients with cardiovascular autonomic neuropathy and orthostatic hypotension. Diabetic Medicine. 20, 119-126 (2003).
- Symon, L., Held, K., Dorsch, N. W. A study of regional autoregulation in the cerebral circulation to increased perfusion pressure in normocapnia and hypercapnia. Stroke. 4, 139-147 (1973).
- Taccone, F. S., et al. Cerebral autoregulation is influenced by carbon dioxide levels in patients with septic shock. Neurocritical Care. 12, 35-42 (2010).
- Barry, D. I., et al. Cerebral blood flow in rats with renal and spontaneous hypertension: resetting of the lower limit of autoregulation. Journal of Cerebral Blood Flow & Metabolism. 2, 347-353 (1982).
- Faraci, F. M., Baumbach, G. L., Heistad, D. D. Cerebral circulation: humoral regulation and effects of chronic hypertension. Journal of the American Society of Nephrology. 1, 53-57 (1990).
- Strandgaard, S. Autoregulation of cerebral blood flow in hypertensive patients. The modifying influence of prolonged antihypertensive treatment on the tolerance to acute, drug-induced hypotension. Circulation. 53, 720-727 (1976).
- McEwen, S. T., Schmidt, J. R., Somberg, L., de la Cruz, L., Lombard, J. H. Time-course and mechanisms of restored vascular relaxation by reduced salt intake and angiotensin II infusion in rats fed a high-salt diet. Microcirculation. 16, 220-234 (2009).
- Allen, L. A., et al. High salt diet impairs cerebral blood flow regulation via salt-induced angiotensin II suppression. Microcirculation. , e12518 (2018).
- Smeda, J. S., Payne, G. W. Alterations in autoregulatory and myogenic function in the cerebrovasculature of Dahl salt-sensitive rats. Stroke. 34, 1484-1490 (2003).
- Greene, N. H., Lee, L. A. Modern and Evolving Understanding of Cerebral Perfusion and Autoregulation. Advances in Anesthesia. 30, 97-129 (2012).
- Merzeau, S., Preckel, M. P., Fromy, B., Leftheriotis, G., Saumet, J. L. Differences between cerebral and cerebellar autoregulation during progressive hypotension in rats. Neuroscience Letters. 280, 103-106 (2000).
- Zagorac, D., Yamaura, K., Zhang, C., Roman, R. J., Harder, D. R. The effect of superoxide anion on autoregulation of cerebral blood flow. Stroke. 36, 2589-2594 (2005).
- Hudetz, A. G., Lee, J. G., Smith, J. J., Bosnjak, Z. J., Kampine, J. P. Effects of volatile anesthetics on cerebrocortical laser Doppler flow: hyperemia, autoregulation, carbon dioxide response, flow oscillations, and role of nitric oxide. Advances in Pharmacology. 31, 577-593 (1994).
- Hudetz, A. G., Shen, H., Kampine, J. P. Nitric oxide from neuronal NOS plays critical role in cerebral capillary flow response to hypoxia. American Journal of Physiology. 274, H982-H989 (1998).
- Okamoto, H., Hudetz, A. G., Roman, R. J., Bosnjak, Z. J., Kampine, J. P. Neuronal NOS-derived NO plays permissive role in cerebral blood flow response to hypercapnia. American Journal of Physiology. 272, H559-H566 (1997).
- Okamoto, H., Roman, R. J., Kampine, J. P., Hudetz, A. G. Endotoxin augments cerebral hyperemic response to halothane by inducing nitric oxide synthase and cyclooxygenase. Anesthesia and Analgesia. 91, 896-903 (2000).
- Schulte, M. L., Hudetz, A. G. Functional hyperemic response in the rat visual cortex under halothane anesthesia. Neuroscience Letters. 394, 63-68 (2006).
- Schulte, M. L., Li, S. J., Hyde, J. S., Hudetz, A. G. Digit tapping model of functional activation in the rat somatosensory cortex. Journal of Neuroscience Methods. 157, 48-53 (2006).
- Alkayed, N. J., et al. Inhibition of brain P-450 arachidonic acid epoxygenase decreases baseline cerebral blood flow. American Journal of Physiology. 271, H1541-H1546 (1996).
- Alonso-Galicia, M., Hudetz, A. G., Shen, H., Harder, D. R., Roman, R. J. Contribution of 20-HETE to vasodilator actions of nitric oxide in the cerebral microcirculation. Stroke. 30, 2727-2734 (1999).
- Kurosawa, M., Messlinger, K., Pawlak, M., Schmidt, R. F. Increase of meningeal blood flow after electrical stimulation of rat dura mater encephali: mediation by calcitonin gene-related peptide. British Journal of Pharmacology. 114, 1397-1402 (1995).
- Mayhan, W. G., Faraci, F. M., Heistad, D. D. Impairment of endothelium-dependent responses of cerebral arterioles in chronic hypertension. American Journal of Physiology. 253, H1435-H1440 (1987).
- Ghali, M. G. Z. Microsurgical technique for tracheostomy in the rat. MethodsX. 5, 61-67 (2018).
- Ghali, M. G. Z. Microsurgical technique for femoral vascular access in the rat. MethodsX. 4, 498-507 (2017).
- Takada, J., et al. Valsartan improves the lower limit of cerebral autoregulation in rats. Hypertension Research. 29, 621-626 (2006).
- Jones, S. C., Radinsky, C. R., Furlan, A. J., Chyatte, D., Perez-Trepichio, A. D. Cortical NOS inhibition raises the lower limit of cerebral blood flow-arterial pressure autoregulation. American Journal of Physiology. 276, H1253-H1262 (1999).
- Smits, G. J., Roman, R. J., Lombard, J. H. Evaluation of laser-Doppler flowmetry as a measure of tissue blood flow. Journal of Applied Physiology (1985). 61, 666-672 (1986).
- Durand, M. J., Raffai, G., Weinberg, B. D., Lombard, J. H. Angiotensin-(1-7) and low-dose angiotensin II infusion reverse salt-induced endothelial dysfunction via different mechanisms in rat middle cerebral arteries. American Journal of Physiology-Heart and Circulatory Physiology. 299, H1024-H1033 (2010).
- Lombard, J. H., Sylvester, F. A., Phillips, S. A., Frisbee, J. C. High-salt diet impairs vascular relaxation mechanisms in rat middle cerebral arteries. American Journal of Physiology-Heart and Circulatory Physiology. 284, H1124-H1133 (2003).
- Weber, D. S., Lombard, J. H. Elevated salt intake impairs dilation of rat skeletal muscle resistance arteries via ANG II suppression. American Journal of Physiology-Heart and Circulatory Physiology. 278, H500-H506 (2000).
- Weber, D. S., Lombard, J. H. Angiotensin II AT1 receptors preserve vasodilator reactivity in skeletal muscle resistance arteries. American Journal of Physiology-Heart and Circulatory Physiology. 280, H2196-H2202 (2001).
- Liu, Y., Rusch, N. J., Lombard, J. H. Loss of endothelium and receptor-mediated dilation in pial arterioles of rats fed a short-term high salt diet. Hypertension. 33, 686-688 (1999).
- Priestley, J. R., et al. Reduced angiotensin II levels cause generalized vascular dysfunction via oxidant stress in hamster cheek pouch arterioles. Microvascular Research. 89, 134-145 (2013).
- McEwen, S. T., Balus, S. F., Durand, M. J., Lombard, J. H. Angiotensin II maintains cerebral vascular relaxation via EGF receptor transactivation and ERK1/2. American Journal of Physiology-Heart and Circulatory Physiology. 297, H1296-H1303 (2009).
- Jensen, N. F., Todd, M. M., Kramer, D. J., Leonard, P. A., Warner, D. S. A comparison of the vasodilating effects of halothane and isoflurane on the isolated rabbit basilar artery with and without intact endothelium. Anesthesiology. 76, 624-634 (1992).
- Avram, M. J., et al. Isoflurane alters the recirculatory pharmacokinetics of physiologic markers. Anesthesiology. 92, 1757-1768 (2000).
- Wang, Z., Schuler, B., Vogel, O., Arras, M., Vogel, J. What is the optimal anesthetic protocol for measurements of cerebral autoregulation in spontaneously breathing mice?. Experimental Brain Research. 207, 249-258 (2010).
- Ayata, C., et al. Pronounced hypoperfusion during spreading depression in mouse cortex. Journal of Cerebral Blood Flow and Metabolism. 24, 1172-1182 (2004).
- Niwa, K., et al. Cerebrovascular autoregulation is profoundly impaired in mice overexpressing amyloid precursor protein. American Journal of Physiology-Heart and Circulatory Physiology. 283, H315-H323 (2002).
- Carreira, S., et al. Diaphragmatic Function Is Preserved during Severe Hemorrhagic Shock in the Rat. Anesthesiology. 120, 425-435 (2014).
- Kerby, J. D., et al. Resuscitation from hemorrhagic shock with HBOC-201 in the setting of traumatic brain injury. Shock. 27, 652-656 (2007).
- Krejci, V., et al. Continuous measurements of microcirculatory blood flow in gastrointestinal organs during acute haemorrhage. British Journal of Anaesthesia. 84, 468-475 (2000).
- Rosengarte, B., Hecht, M., Wolff, S., Kaps, M. Autoregulative function in the brain in an endotoxic rat shock model. Inflammation Research. 57, 542-546 (2008).
- Rozet, I., et al. Cerebral autoregulation and CO2 reactivity in anterior and posterior cerebral circulation during sevoflurane anesthesia. Anesthesia and Analgesia. 102, 560-564 (2006).
- Hudetz, A. G., Biswal, B. B., Feher, G., Kampine, J. P. Effects of hypoxia and hypercapnia on capillary flow velocity in the rat cerebral cortex. Microvascular Research. 54, 35-42 (1997).
- Shi, Y., et al. Interaction of mechanisms involving epoxyeicosatrienoic acids, adenosine receptors, and metabotropic glutamate receptors in neurovascular coupling in rat whisker barrel cortex. Journal of Cerebral Blood Flow and Metabolism. 28, 111-125 (2008).

