Isolation, Transfection, and Long-Term Culture of Adult Mouse and Rat Cardiomyocytes
Summary
Here, we present a protocol for the isolation, transfection, and long-term culture of adult mouse and rat cardiomyocytes.
Abstract
Ex vivo culture of the adult mammalian cardiomyocytes (CMs) presents the most relevant experimental system for the in vitro study of cardiac biology. Adult mammalian CMs are terminally differentiated cells with minimal proliferative capacity. The post-mitotic state of adult CMs not only restricts cardiomyocyte cell cycle progression but also limits the efficient culture of CMs. Moreover, the long-term culture of adult CMs is necessary for many studies, such as CM proliferation and analysis of gene expression.
The mouse and the rat are the two most preferred laboratory animals to be used for cardiomyocyte isolation. While the long-term culture of rat CMs is possible, adult mouse CMs are susceptible to death and cannot be cultured more than five days under normal conditions. Therefore, there is a critical need to optimize the cell isolation and long-term culture protocol for adult murine CMs. With this modified protocol, it is possible to successfully isolate and culture both adult mouse and rat CMs for more than 20 days. Moreover, the siRNA transfection efficiency of isolated CM is significantly increased compared to previous reports. For adult mouse CM isolation, the Langendorff perfusion method is utilized with an optimal enzyme solution and sufficient time for complete extracellular matrix dissociation. In order to obtain pure ventricular CMs, both atria were dissected and discarded before proceeding with the disassociation and plating. Cells were dispersed on a laminin coated plate, which allowed for efficient and rapid attachment. CMs were allowed to settle for 4-6 h before siRNA transfection. Culture media was refreshed every 24 h for 20 days, and subsequently, CMs were fixed and stained for cardiac-specific markers such as Troponin and markers of cell cycle such as KI67.
Introduction
Cardiac diseases are one of the leading causes of death worldwide. Almost all types of cardiac injury result in a significant loss of adult cardiomyocytes (CMs). Adult mammalian hearts are unable to repair their cardiac injury due to the senescent nature of the adult CM1. Thus, any insult to the adult mammalian heart results in a permanent loss of CMs, leading to reduced cardiac function and heart failure. Unlike adult mammals, small animals like zebrafish and newt hearts can regenerate their cardiac injury through existing CM proliferation2,3,4. A worldwide effort is ongoing to find a novel therapeutic intervention for cardiac injury via both proliferative and non-proliferative approaches. In past decades, various types of genetic mouse models have been developed to study cardiac injury and repair. However, using in vivo animal models continues to be an expensive approach with the additional complexity to decipher a cell-autonomous mechanism from secondary effects. Besides, in vivo systems are challenging to analyze CM specific effects of a pharmacological intervention that induces cardioprotective signaling from the CM.
Moreover, the long-term culture of adult CMs is necessary to perform CM proliferation analyses. CM proliferation assays require a minimum of 4-5 days for cells to be induced into the cell cycle and to obtain accurate data after that. Additionally, studies that utilize isolated CMs for electrophysiological studies, drug screening, toxicity studies, and Ca++ homeostasis studies are all in need of an improved culture system5,6,7. Furthermore, recent studies show the cardioprotective significance of cytokines secreted from CMs (cardiokines)8,9. In order to investigate the therapeutic role and molecular mechanism of these cardiokines during heart repair and regeneration, a prolonged culture is required.
Adult rat CMs are robust enough for single-cell isolation and long-term culture in an in vitro system10,11,12. However, adult mouse CMs are of great interest for in vitro assays, due to the availability of a variety of genetically modified mouse models, which allows for the design and execution of various innovative analyses that are not possible with rat CM13. In contrast to adult rat CM isolation, it is quite challenging to obtain a single-cell suspension from adult mouse hearts, and the long-term culture of adult mouse CMs in culture is even more challenging.
Adult CM isolation from mouse and rat hearts using a Langendorff system has previously been established to study CM function5,14,15. Here, we have described in detail the protocols for adult CM isolation from both rats and mice, as well as a modified long-term culture, transfection, and CM proliferation of isolated cells.
Protocol
All experiments should be performed in accordance with the guidelines of the Guide for the Care and Use of Laboratory Animals published by the U.S. National Institute of Health (NIH). All protocols displayed in the video were approved by the Institutional Animal Care and Use Committee (IACUC) of the University of Cincinnati, College of Medicine.
1. Preparation before heart extraction from adult mice (and rats)
- Prepare the corresponding perfusion, enzyme, and stop solutions, according to the recipes given in Table 1 and Table 2, for rat and mouse isolation, respectively. Filter the solutions through a 0.22 µm filter to remove any contamination or undissolved particles.
- Clean and sterilize the surgical instruments by soaking them in 70% alcohol for 15 min, and subsequently wash with double distilled water. Leave the tools to air dry on a clean paper towel.
- Pre-heat the water bath to 37 ºC. Check the water level in the water bath to ensure an uninterrupted circulation of warm water through the perfusion apparatus during the procedure.
- Clean the perfusion apparatus by running with 70% alcohol for 5 min. Repeat.
NOTE: Increase the flow rate, which helps to clean the tubing. - After the alcohol flowthrough, clean the remnants of alcohol by running double distilled water for 10 min.
- Set up 3 medium-size disposable polystyrene weighing dishes to clean the heart after excision. Fill the dishes with 20 mL of myocyte buffer.
- Add 2-3 drops of heparin to each dish with the help of Pasteur pipette and mix well by pipetting multiple times.
- Take a 10 mL syringe with a blunt needle cannula.
NOTE: A blunt needle cannula can be prepared by cutting the tip of a clean, sterile needle. A 21 G needle and 14 G needle were used to make the cannula for the mouse and rat, respectively. - Fill the syringe with perfusion buffer (Table 1). Remove any air bubbles from the syringe and place it in the third dish at an angled position. Secure it with tape.
NOTE: For rats, Solution A (Table 2) was used instead of myocyte perfusion buffer. - Prepare a loose knot with a surgical suture and place it around the needle.
- Inject the mouse with heparin (100 USP units/mouse) by intraperitoneal injection, 20 min before the anesthesia injection.
- Circulate the myocyte perfusion buffer (Table 1) through the perfusion apparatus. Decrease the flow rate to 3 mL/min.
NOTE: For rats, Solution A (Table 2) was used instead of myocyte perfusion buffer. - After 5 min, replace the perfusion buffer with the enzyme solution (Table 1). Saturate the enzyme solution with oxygen during the process. Use a flow rate of 3 mL/min.
NOTE: For rat CM isolation, use solution E (Table 2) instead. - Anesthetize the mouse by intraperitoneal injection of anesthesia. Use the appropriate dose of anesthesia, recommended by IACUC, for the terminal surgery.
- Confirm sufficient depth of anesthesia by the lack of a response to a toe pinch. Then, put the mouse on the surgical platform.
2. Extraction of heart from adult mice (and rats)
- Sterilize the skin by wiping with 70% alcohol.
- Carefully open the chest.
- Excise the heart. Avoid non-heart tissues during excision.
- Put the heart on the first dish, filled with perfusion buffer (Table 1).
NOTE: Use solution A for the rat (Table 2). - Clear the blood from the heart by gently squeezing.
- Transfer the heart to the second dish.
- Clean the blood from the heart and remove non-cardiac tissues. Subsequently, transfer the heart to the third dish.
- Trim off lungs and other surrounding tissue and cannulate via the ascending aorta. This procedure should be performed as rapidly as possible (<5 min) to improve CM quality and quantity.
3. Digestion of the heart
- Digestion of the mouse heart
NOTE: All the solutions/buffers which circulate through the mouse heart should be oxygenated throughout the procedure.- Carefully remove the cannulating needle from the syringe and connect it to the Langendorff perfusion apparatus.
- Avoid any air bubbles going into the heart, which may affect the flow-through of the enzyme solution and digestion.
- Move the water jacket up to provide a homogenous environment to the heart. Allow the enzyme solution to flow through the heart at a speed of 2-3 mL/min.
NOTE: The speed of the passing solution is critical and has a significant impact on the outcome of CM quantity and quality. - Let the enzyme solution flow through the heart for 2 min.
- At 2 min, add 40 µL of 100 µM CaCl2 solution to the enzyme solution and mix. Let the enzyme solution pass through the heart for another 10-15 min.
NOTE: Once the flow becomes smooth, the heart becomes brownish and soft, indicating the even distribution of the collagenase enzyme and proper digestion of heart.
- Digestion of the rat heart
NOTE: All the solutions/buffers which circulate through the rat heart should be oxygenated throughout the procedure.- Carefully remove the cannulating needle from the syringe and connect it to the Langendorff perfusion apparatus.
- Avoid any air bubbles going into the heart, which may affect the flow-through of the enzyme solution and digestion.
- Move the water jacket up to provide a homogenous environment to the heart.
- Start the flow of solution A at a speed of 2-3 mL/min for 3-5 min to remove any blood from the heart.
NOTE: The speed of the passing solution is critical and has a significant impact on the outcome of CM quality. - Once the blood is cleared from the heart, switch from solution A to solution E (Table 2). Titrate solution E with CaCl2 as indicated below for a final concentration of 0.1 mM in solution:
- After 10 min of perfusion, add 12.5 µL of 0.1 M CaCl2.
- After 15 min of perfusion, add 25 µL of 0.1 M CaCl2.
- Let the enzyme solution pass through the heart for another 30-40 min, until the flow becomes rapid and the heart is pliable.
4. Preparation of CM single-cell suspension
- Preparation of the CM single-cell suspension (mouse)
- Remove the heart from the Langendorff perfusion system. Move it to a 60 mm Petri dish filled with 5 mL of enzyme solution and transfer the dish to a biosafety hood.
NOTE: Ensure sufficient heart digestion before removing the heart from the Langendorff perfusion system. The digestion time depends on the enzyme activity, the flow of solution, and the size of the heart. - Perform any further mechanical disaggregation in a biosafety hood to ensure sterility and avoid contamination.
- Carefully remove the atria (1/4th of the basal heart portion) and extra fat tissues.
- Mince the heart with forceps into fine pieces.
- Take a sterile Pasteur pipette and cut its tip at a 45º angle.
- Use this Pasteur pipette to dispense the heart tissue in a single-cell suspension by gentle pipetting. Optimal digestion provides a suspension of single-cell cardiomyocytes.
NOTE: Remove the narrow, bottom portion of the transfer pipet to reduce CM damage due to mechanical searing. - Then, add 5 mL of stop solution to stop the enzyme activity, which avoids the possibility of overdigestion.
- Take a fresh 50 mL conical and place a sterile 100 µm cell strainer on it.
- Pass the cardiomyocyte suspension through the cell strainer to remove the tissue chunk. Wash the Petri dish and strainer with another 5 mL of stop solution (Table 1) to collect any cardiomyocytes that remain attached to the strainer.
- Remove the heart from the Langendorff perfusion system. Move it to a 60 mm Petri dish filled with 5 mL of enzyme solution and transfer the dish to a biosafety hood.
- Preparation of the CM single-cell suspension (rat)
- Remove the heart from the Langendorff perfusion system. Move the heart to a 100 mm Petri dish filled with 5 mL of solution A and move the dish to a biosafety hood.
NOTE: Ensure sufficient heart digestion before removing the heart from the Langendorff perfusion system. The digestion time depends on the enzyme activity, the flow of solution, and the size of heart. - Perform any further mechanical disaggregation in a biosafety hood to ensure sterility and avoid contamination.
- Carefully remove the atria (1/4th of the basal heart portion) and extra fat tissues.
- Mince the heart with forceps into fine pieces.
- Take a sterile Pasteur pipette and cut its tip at a 45º angle.
- Use this Pasteur pipette to dispense the heart tissue in single-cell suspension by gentle pipetting. Optimal digestion provides a suspension of single-cell cardiomyocytes.
NOTE: Remove the narrow, bottom portion of the transfer pipet to reduce CM damage due to mechanical searing. - Add 5 mL of solution B (Table 2) to the CM suspension and pass the solution through a 100 µm cell strainer to remove any remaining pieces of fat or other non-digested tissue.
- Collect filtrate in a fresh 50 mL conical vial.
- Use another 5 mL of solution B to wash the Petri dish and any remaining CM through the cell strainer.
- Remove the heart from the Langendorff perfusion system. Move the heart to a 100 mm Petri dish filled with 5 mL of solution A and move the dish to a biosafety hood.
5. Removal of non-CMs
- Removal of non-CMs from the adult single-cell suspension (mouse)
- Centrifuge the cell suspension at 20 x g for 3 min.
- Discard the supernatant and resuspend the cells in 10 mL of stop solution (Table 1).
NOTE: Increasing the concentration of BSA in the stop solution will increase its viscosity and reduce the sedimentation rate of non-CMs. - Resuspend the CMs by gentle inversion of the tube 3-5 times.
- At 3 min intervals, add 10 µL of 100 µM CaCl2 solution and mix. Repeat thrice.
- After the fourth addition, centrifuge the cardiomyocyte suspension at 20 x g for 3 min.
- Discard the supernatant.
- Resuspend CMs in pre-warmed (37 ºC), adult mouse CM plating media (Table 3).
- Removal of non-CMs from the adult single-cell suspensions (rat)
- Pellet cells at 20 x g for 3 min at 25 ºC.
- Discard the supernatant and resuspend the cells into 25 mL of solution B (Table 2).
- Mix the cells by gentle inversion and place it in a tube stand to allow the CM to settle under gravity.
- Carefully discard the supernatant.
- Resuspend the cells in fresh 25 mL of solution B and titrate it to 1.0 mM CaCl2 by stepwise addition of 50 µL, 75 µL, and 100 µL of 0.1 M CaCl2 at 3-5 min intervals.
NOTE: A higher concentration of BSA in solution B could be used at this step. Increasing the concentration of BSA in solution B increases the viscosity and thus, reduces the sedimentation rate of non-CMs. - Pellet cells at 20 x g for 3 min at 25 ºC, aspirate the supernatant, and add the desired volume of adult rat cell culture media (Table 4).
- Seed CMs on a laminin coated plate.
NOTE: Pre-plating of CMs on a non-coated culture plate can be used to minimize the contamination of non-CM cells.
6. Adult CM plating
- Pre-plating
- Resuspend the cardiomyocytes into culture media.
- Pre-plate the cells into a 60 mm or 100 mm dish for mouse or rat CM, respectively.
- Incubate CMs for 2 h in an incubator supplemented with 5% CO2 at 37 ºC.
- During the pre-plate incubation, coat the cell culture plates with laminin (10 µg/mL in PBS) for long term CM culture.
- After 2 h of pre-plating, collect CMs in a 50 mL conical vial.
- Collect the cells from the dish and re-plate them into a laminin coated 24 well culture plate for transfection experiments.
- Culture cells in an incubator at 37 ºC supplemented with 5% with CO2. 4-6 hours is sufficient to adhere to the cardiomyocytes with the surface, and thus, transfection can be performed 6 hours post-plating.
NOTE: The plating medium containing FBS is commonly used and shows better compatibility with proliferation studies. However, a serum-free medium can be used for experiments where secretory factors are being analyzed.
7. Transfection
- Incubate the CMs to the cell culture plates coated with laminin for 4-6 hours.
- Four hours after CM seeding on the laminin coated plate, transfect cells with siRNAs (50 nM) of interest using a transfection reagent (e.g., Lipofectamine RNAiMAX) according to the manufacturer’s protocol.
- Change media 24 hours following transfection and every 24 hours after that for up to 20 days.
- After 20 days, fix cells with 4% PFA in PBS for downstream applications, including immunocytochemistry for cardiac-specific markers such as Troponin to ensure live cardiomyocytes were successfully cultured long-term.
Representative Results
The current modified protocol allows efficient isolation and culture of rat and mice CMs in vitro. For rat CM isolation, a total of 3 adult (12-week-old) male Fischer 344 rats were used in the procedure. Figure 1 shows the surgical apparatus and isolation setups that are required in the procedure; each part has been marked and described in the figure legend. Collagenase type 2 was used for digestion, which yields a high quantity of high quality CMs from successful isolation (Figure 2A). Twenty-four hours post-isolation, these cells were transfected with cell cycle inducing specific siRNAs against Rb1 and Meis2, whereas, cel-miR-67 was used as transfection control in the experiment11. CMs were maintained in culture for either seven days (Figure 2B) or up to twenty days (Figure 2C) to observe the morphological changes. To score for cell cycle activity, CMs were fixed on day 7 and stained for cardiac-specific marker Troponin-I, mitotic marker KI67, and nucleus was visualized through DAPI (Figure 2D-F). A significant increase in KI67 positive cardiomyocytes was observed with siRb1+siMeis2 treatment when compared to control11. Moreover, the rat cardiomyocytes were beating (contractile function) in culture on day seven post-plating, which is a hallmark characteristic of healthy cardiomyocytes11.
For CM isolation in mice, a total of 3 male and 3 female adult (12-week-old) C57BL/6 mice were used in the procedure. Similar to the rat study, collagenase type 2 was used to digest the collagen and the extracellular matrix of the adult mice heart. Adult mice CM is comparatively fragile for isolation and culture in vitro; thus, this protocol uses blebbistatin to improve the viability of the adult mouse CM. Successful isolation yields >70-80% healthy rod shape CMs, which can be cultured for up to 20 days (Figure 3A-C) and maintain their cardiac Troponin-I staining and contractility (Figure 3C). In separate experiments, four hours post isolation, mouse CMs were transfected with a cell cycle inducing siRNA cocktail as described for the rat CM. Briefly, mice CM was simultaneously transfected with the specific siRNAs against Rb1 and Meis2, and control (cel-miR-67). Post-transfection CMs were subject to time course imaging for 10 days showing the morphological changes (Figure 4) which is followed by a proliferation assay (Figure 5). On day 10, immune-fluorescent staining for Troponin-I, KI67, and DAPI was performed as previously described. A significant increase in KI67 positive cardiomyocytes was observed with siRb1+siMeis2 treatment when compared to control (Figure 5A-C).
The present protocol demonstrates that adult rats and mice CMs can be cultured long-term in vitro for up to 20 days and potentially longer. After 20 days, CMs maintain their Troponin-I staining as well as contractility if the inhibitors are removed from the media.
| Myocyte buffer composition, pH 7.4 (Prepare and can be stored at 4 °C ) | |||
| Reagent | Concentration, mM | Molecular wt. | For 1 Liter |
| NaCl | 113 | 58.44 | 6.6037 g |
| KCl | 4.7 | 74.5513 | 350.3911 mg |
| MgSO4 | 1.2 | 120.366 | 144.4392 mg |
| KH2PO4 | 0.6 | 136.086 | 81.6516 mg |
| NaH2PO4 | 0.6 | 119.98 | 71.988 mg |
| Perfusion buffer, pH 7.4 (Prepare fresh for each isolation) | |||
| Concentration, mM | Molecular wt. | Amount required | |
| Myocyte buffer | 250 mL | ||
| HEPES | 10 | 238.3012 | 595.75 mg |
| 2,3-butanedione monoximine | 10 | 101.105 | 252.775 mg |
| NaHCO3 Fresh | 1.6 | 84.007 | 33.6028 mg |
| Taurine Fresh | 30 | 125.15 | 938.625 mg |
| Glucose Fresh | 20 | 180.156 | 900.775 mg |
| Enzyme solution | |||
| Stock Conc. | Working Conc. | Required | |
| Myocyte buffer | 50 mL | 50 mL | |
| Collagenase type 2 | 330 U/mg | 620 U/mL | 93 mg |
| Protease XIV | >3.5 U/mg | 0.104 U/mL | 1.48 mg |
| DNase I grade 2 | 0.015 mg/mL | ||
| Stop Buffer A | |||
| Stock Conc. | Working Conc. | Required | |
| Myocyte buffer | 30 mL | ||
| BSA | 2.50% | 0.75 g | |
| CaCl2 | 100 mM | 0.1 mM | 30 µL |
| Stop Buffer B | |||
| Stock Conc. | Working Conc. | Required | |
| Myocyte buffer | 30 mL | ||
| BSA | 5.00% | 1.5 g | |
| CaCl2 | 100 mM | 0.1 mM | 30 µL |
Table 1: Solutions required for adult mouse cardiomyocyte isolation.
| 10x KHB Stock Solution (Total volume= 1L) |
Reagent | Molarity (mM) | Amount (g) |
| NaCl | 1180 | 68.9 g | |
| KCl | 48 | 3.5 g | |
| HEPES | 250 | 59.7 g | |
| MgSO4 | 12.5 | 1.4 g | |
| K2HPO4 | 12.5 | 2.1 g | |
| Adjust pH to 7.4 with 4 M NaOH (~20 mL), store at 4 °C | |||
| KHB Solution, 500 mL | Reagent | Amount | |
| 10x KHB | 50 mL | ||
| Glucose | 0.99 g | ||
| Taurine | 0.31 g | ||
| Add H2O to bring volume to 500 mL, and pH should be ~7.35 | |||
| Solution A | Reagent | Amount | |
| KHB solution | 500 mL (10 mM) | ||
| BDM | 0.5 g | ||
| Oxygenate with 100% O2 and warm to 37 °C | |||
| Solution B, 50 mL | Reagent | Amount | |
| Solution A | 50 mL | ||
| BSA | 0.5 g | ||
| 0.1 M CaCl2 (Ca++=0.1 mM) | 50 µL | ||
| Solution E, 50mL | Reagent | Amount | |
| Solution A | 50 mL | ||
| BSA | 0.05 g | ||
| Collagenase type II (263 units/mg) | 35 mg | ||
| Hyaluronidase (Type I-S) | 10 mg | ||
| 0.1 M CaCl2 stock | 12.5 µL | ||
| Mix well | |||
| CaCl2 Stock, 0.1M | Reagent | Amount | |
| CaCl2 | 7.35 g | ||
| H2O | 500 mL | ||
| Store at 4 °C | |||
Table 2: Solutions required for adult rat cardiomyocyte isolation.
| Plating media composition, pH 7.4 | |||
| Working Concentration | Molecular Wt. | Required amount | |
| Culture media without Blebbistatin | 50 mL | ||
| BDM | 10 mM | 101.105 g/mol | 50.55 mg |
| FBS | 5% | 2.5 g | |
| Culture media composition, pH 7.4 | |||
| Reagent | Working Concentration | Molecular Wt. | Required amount |
| DMEM | 1X | 250 mL | |
| Insulin | 1 µg/mL | 0.25 mg | |
| Transferrin | 0.55 µg/mL | 0.138 mg | |
| Selenium | 0.5 ng/mL | 0.125 µg | |
| Penicillin (U/mL)-streptomycin (g/mL) | 100-100 | 2.5 mL | |
| HEPES | 10 mM | 238.3012 g/mol | 595.753 mg |
| *FBS | 10% | 25 mL | |
| *BSA | 0.20% | 0.5 g | |
| #Blebbistatin | 25 µM | 292.338 g/mol | 1.8271 mg |
| *Use either of them, as per the experimental requirement. # Aliquot the culture media to prepare plating media before adding Blebbistatin. NOTE: Prepare 200 mL of culture media. NOTE: Prepare 50 mL of plating media. |
|||
Table 3: Media composition for adult mouse cardiomyocyte plating and culture.
| Culture media composition, pH 7.4 | ||
| Reagent | Working Concentration | Required amount |
| DMEM | 1x | 250 mL |
| Penicillin (U/mL)-streptomycin (g/mL) | 100-100 | 2.5 mL |
| *FBS | 10% | 25 mL |
Table 4: Media composition for adult rat cardiomyocyte plating and culture.
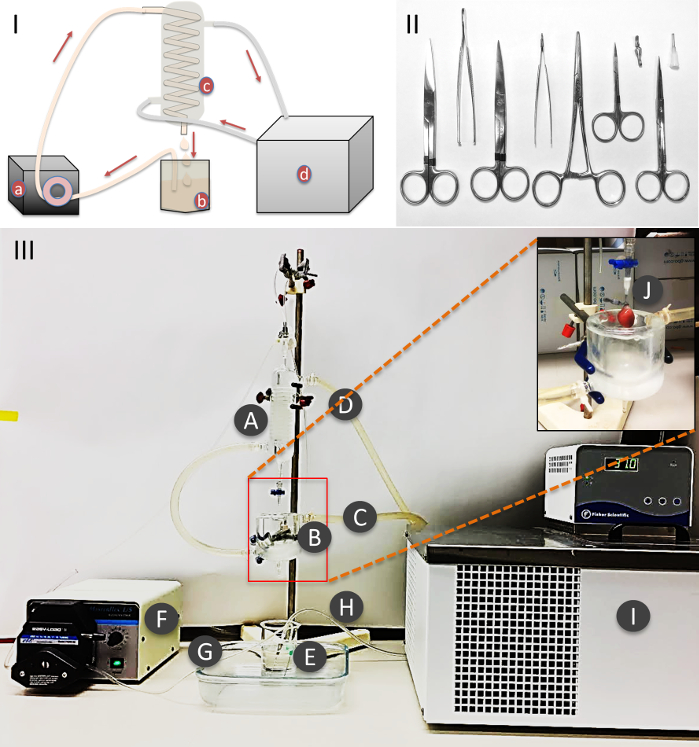
Figure 1: Procedure setup and equipment. (I) Schematic representation of the perfusion. (II) Surgical instruments and cannulation needle. (III) Heart perfusion assembly: A) Heating jacket. B) Double wall water jacket vessel. C) Circulating heated water inlet. D) Circulating heated water outlet. E) Heart perfusion solution. F) Circulating pump G) Perfusion solution tube. H) Oxygen supply tube. I) Circulating water bath. J) Cannulation needle with heart, attached to the perfusion outlet port. Please click here to view a larger version of this figure.
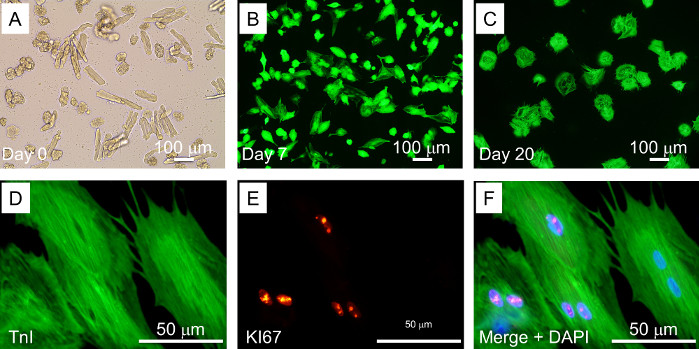
Figure 2: Adult rat CM isolation, transfection, and long-term culture. A) Adult rat CM, immediately after isolation. B) Adult rat CM on Day 7 after isolation. C) Adult rat CM on day 20. D-F) KI67 positive rat CM on day 7 after siRb1+siMeis2 transfection. Troponin-I = green; DAPI = blue; KI67 = red. All the experiments were performed in duplicate and repeated at least three times. Please click here to view a larger version of this figure.
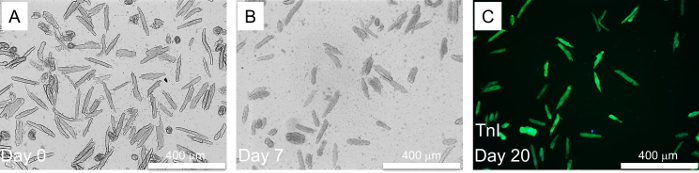
Figure 3: Adult mouse CM isolation and long term culture. A) Adult mice CM, immediately after isolation. B) Adult mice CM on Day 7 after isolation. C) Adult mice CM stained with Cardiac Troponin-I = green on day 20. Please click here to view a larger version of this figure.
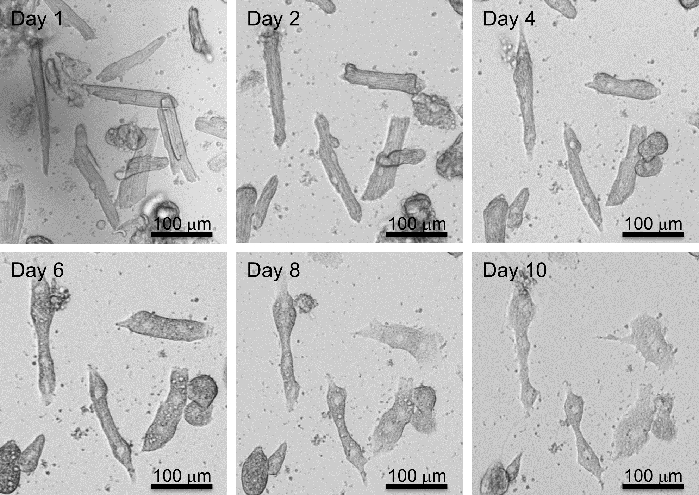
Figure 4: Adult mouse CM de-differentiation. Adult mice CM showing morphological changes during long-term culture (day 0 to Day 10) in DMEM-HG media, supplemented with 10%FBS. Please click here to view a larger version of this figure.
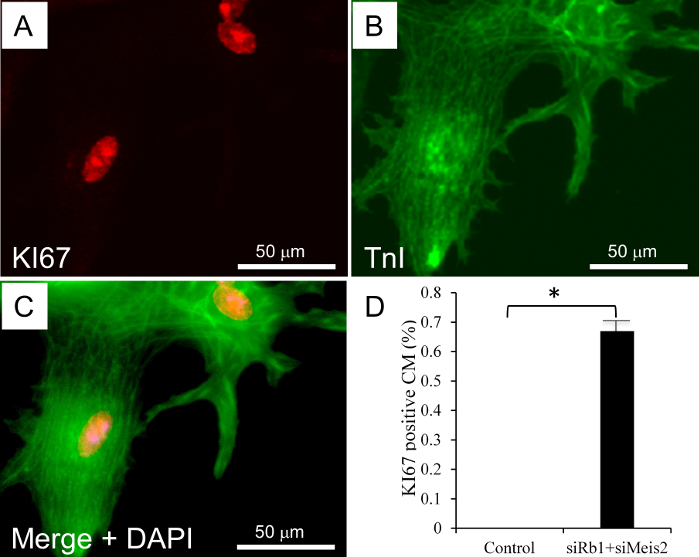
Figure 5: Adult mouse CM transfection and proliferation. A-C) KI67 positive mouse CM on day 10 after siRb1+siMeis2 transfection. Troponin-I = green; DAPI = blue; KI67 = red. D) Bar graph shows a significant increase in KI67 positive adult mouse CMs in the siRNA-cocktail transfected group versus control. Results are presented as mean±SEM; * = p-value ≤0.05. p-value ≤0.05 was considered statistically significant. All the experiments were performed in triplicate (n=3 Male,3 Female). Please click here to view a larger version of this figure.
Discussion
There is a critical need to establish a protocol for adult cardiomyocyte isolation and long-term culture to perform cell-specific mechanistic studies. There are only a few reports discussing adult CM isolation protocols, and even fewer of them are used for long-term culture of adult mice CM15,16,17. It is been shown that the adult rat CM has a higher tolerance to in vitro culture than the adult mice CM10,11,12. In this report, we establish a protocol for adult rat and mice CM isolation, siRNA transfection, long term culture, and downstream analysis of induced proliferation, with minimal modifications in the regularly used protocols.
Available protocols for adult CM isolation are based on the use of two well-known enzymes, collagenase and liberase, for the extracellular matrix (ECM) digestion18,19. Collagenase is a commonly used enzyme in the adult CM isolation protocols. Collagenase digests the collagen fibers in the ECM, which results in the dissociation of the heart tissue in the single-cell CM. Similarly, liberase digests the ECM. Liberase is the purified alternative for collagenase, which shows higher activity and thus requires higher precision during CM isolation to achieve successful isolation in comparison to collagenase. Moreover, in the procedure, we noticed that the CM isolated with liberase does not survive in the long-term culture. However, CM isolated with collagenase improves the longevity of CM in the culture conditions.
Additionally, we used a specific myosin II inhibitor called blebbistatin in the culture media for the long-term culture of adult mouse CM20,21. Previously, 2,3-butanedione monoximine (BDM) has been used for the adult CM culture. BDM is an ATPase inhibitor that inhibits the contractile function through a poorly defined mechanism that includes inhibition of ATPase and impaired Ca++ transition22. In comparison to the BDM, blebbistatin is a specific inhibitor of myosin II and shows more significant inhibition of contractile function at lower concentrations than the BDM. A pre-plating of the adult mouse CM in a culture media, supplemented with 10 mM of BDM and subsequent cultures of the CM in the 25 µM blebbistatin-supplemented and low serum media improves the survivability and rod shape structure of CM in long-term culture. However, a direct plating of the adult mouse CM in the media, supplemented with 25 µM blebbistatin and 10% FBS is better for proliferative studies. Similar to the adult rat CM, which shows a great extent of de-differentiation in the in vitro culture, we also observed de-differentiation in the adult mouse CM to some extent (Figure 5). Morphological changes are the necessary step for the proliferation. Thus, providing an environment to adult CM that facilitates their de-differentiation is a critical factor to consider in such studies. Even though at this time, it is not clear which of the exact steps or components used in this protocol is responsible for the improved longevity of adult CMs, we believe that it is a combination effect of the reagents used, the modifications made in the isolation procedure, and maybe most importantly the trained hands.
Similar to the previous reports showing the viral transduction of the adult CM in blebbistatin-supplemented media, we also observed an efficient transfection of these cells with siRNAs. We used specific siRNAs against two senescence-associated genes, Rb1 and Meis2, to induce the CM proliferation. For the rat CM, we performed KI67 analysis to assess the proliferation on day seven after transfection; however, the proliferation in mouse adult CM was analyzed on day ten after transfection. A species-specific biological variability could be a possible reason for the observed time difference in the induced cell cycle re-entry of adult rat and mouse CM.
Overall, the protocol described here provide an improved, reliable, and comparative procedure to isolate adult rat and mouse CM, and accordingly culture them as per experimental need. Moreover, this protocol allows a long-term culture of the adult CM, which provide an in vitro system to perform various long-term analysis such as proliferation, paracrine factor, stress response, etc.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by funding from the Department of Pathology and Laboratory Medicine, University of Cincinnati, College of Medicine, to Dr. Onur Kanisicak; a grant from the National Institutes of Health (R01HL148598) to Dr. Onur Kanisicak. Dr. Onur Kanisicak is supported by the American Heart Association Career Development Award (18CDA34110117). Dr. Perwez Alam is supported by the American Heart Association postdoctoral grant (AHA_20POST35200267). Dr Malina J. Ivey is supported by an NIH T32 grant (HL 125204-06A1).
Materials
| 2,3-Butane Dione monoxime | Sigma-Aldrich | B-0753 | |
| Blebbistatin | APExBIO | B1387 | |
| Bovine serum albumin | Sigma-Aldrich | A3059 | |
| CaCl2 | Sigma-Aldrich | 449709 | |
| Cell culture plate | Corning Costar | 3526 | |
| Cell strainer | BD Biosciences | 352360 | |
| Cel-miR-67 | Dharmacon | CN-001000-01-50 | |
| Collagenase type2 | Worthington | LS004177 | |
| Disposable Graduated Transfer Pipettes | Fisherbrand | 13-711-20 | |
| Disposable polystyrene weighing dishes | Sigma-Aldrich | Z154881-500EA | |
| Dulbecco's Modified Eagle's medium | Thermo Scientific | SH30022.01 | |
| EdU | Life Technologies | C10337 | |
| Fetal bovine serum | Corning | 35-015-CV | |
| Fine Point High Precision Forceps | Fisherbrand | 22-327379 | |
| Glucose | Sigma-Aldrich | G-5400 | |
| Hemocytometer | Hausser Scientific | 1483 | |
| Heparin | Sagent Pharmaceuticals | PSLAB-018285-02 | |
| HEPES | Sigma-Aldrich | H3375 | |
| High Precision Straight Broad Strong Point Tweezers/Forceps | Fisherbrand | 12-000-128 | |
| Hyaluronidase | Sigma | H3506 | |
| Insulin | Sigma-Aldrich | I0516-5ML | |
| K2HPO4 | Sigma-Aldrich | P-8281 | |
| KCl | Sigma-Aldrich | 746436 | |
| Light Microscope | Nikon | ||
| Lipofectamine RNAiMAX | Life Technologies | 13778-150 | |
| MgSO4 | Sigma-Aldrich | M-2643 | |
| NaCl | Sigma-Aldrich | S9888 | |
| NaOH | Fisher Scientific | S318-500 | |
| Natural Mouse Laminin | Invitrogen | 23017-015 | |
| Penicillin/Streptomycin | Corning | 30-002-CI | |
| Pentobarbital | Henry Schein | 24352 | |
| Phosphate buffered saline | Life Technologies | 20012-027 | |
| Protease XIV | Sigma-Aldrich | P5147-1G | |
| Selenium | Sigma-Aldrich | 229865+5G | |
| siMeis2 | Dharmacon | s161030 | |
| siRb1 | Dharmacon | s128325 | |
| Straight Blunt/SharpDissecting Scissors | Fisher Scientific | 28252 | |
| Straight Very Fine Precision Tip Forceps | Fisherbrand | 16-100-120 | |
| Taurine | Sigma-Aldrich | T0625 | |
| Transferrin | Sigma-Aldrich | T8158-100MG | |
| Ultra-smooth, beveled-edge finish scissor | Fisherbrand | 22-079-747 | |
| Water Bath | Fisher Scientific | 3006S |
References
- van Amerongen, M. J., Engel, F. B. Features of cardiomyocyte proliferation and its potential for cardiac regeneration. Journal of Cellular and Molecular Medicine. 12, 2233-2244 (2008).
- Parente, V., et al. Hypoxia/reoxygenation cardiac injury and regeneration in zebrafish adult heart. PLoS One. 8, 53748 (2013).
- Wang, J., et al. The regenerative capacity of zebrafish reverses cardiac failure caused by genetic cardiomyocyte depletion. Development. 138, 3421-3430 (2011).
- Gonzalez-Rosa, J. M., Martin, V., Peralta, M., Torres, M., Mercader, N. Extensive scar formation and regression during heart regeneration after cryoinjury in zebrafish. Development. 138, 1663-1674 (2011).
- Graham, E. L., et al. Isolation, culture, and functional characterization of adult mouse cardiomyoctyes. Journal of Visualized Experiments. , e50289 (2013).
- Brette, F., Orchard, C. T-tubule function in mammalian cardiac myocytes. Circulation Research. 92, 1182-1192 (2003).
- Müller, J. G., et al. Differential regulation of the cardiac sodium calcium exchanger promoter in adult and neonatal cardiomyocytes by Nkx2.5 and serum response factor. Journal of Molecular and Cellular Cardiology. 34, 807-821 (2002).
- Zhou, H., et al. Exosomes in ischemic heart disease: novel carriers for bioinformation. Biomedicine & Pharmacotherapy. 120, 109451 (2019).
- Wu, Y. S., Zhu, B., Luo, A. L., Yang, L., Yang, C. The Role of Cardiokines in Heart Diseases: Beneficial or Detrimental. BioMed Research International. 2018, 8207058 (2018).
- Eppenberger, H. M., Hertig, C., Eppenberger-Eberhardt, M. Adult rat cardiomyocytes in culture A model system to study the plasticity of the differentiated cardiac phenotype at the molecular and cellular levels. Trends in Cardiovascular Medicine. 4, 187-193 (1994).
- Alam, P., et al. Inhibition of Senescence-Associated Genes Rb1 and Meis2 in Adult Cardiomyocytes Results in Cell Cycle Reentry and Cardiac Repair Post-Myocardial Infarction. Journal of the American Heart Association. 8, 012089 (2019).
- Arif, M., et al. MicroRNA-210-mediated proliferation, survival, and angiogenesis promote cardiac repair post myocardial infarction in rodents. Journal of Molecular Medicine. 95, 1369-1385 (2017).
- Rosenthal, N., Brown, S. The mouse ascending: perspectives for human-disease models. Nature Cell Biology. 9, 993-999 (2007).
- Nippert, F., Schreckenberg, R., Schlüter, K. D. Isolation and Cultivation of Adult Rat Cardiomyocytes. Journal of Visualized Experiments. , e56634 (2017).
- Judd, J., Lovas, J., Huang, G. N. Isolation, Culture and Transduction of Adult Mouse Cardiomyocytes. Journal of Visualized Experiments. , e54012 (2016).
- Ackers-Johnson, M., et al. A Simplified, Langendorff-Free Method for Concomitant Isolation of Viable Cardiac Myocytes and Nonmyocytes From the Adult Mouse Heart. Circulation Research. 119, 909-920 (2016).
- Li, D., Wu, J., Bai, Y., Zhao, X., Liu, L. Isolation and culture of adult mouse cardiomyocytes for cell signaling and in vitro cardiac hypertrophy. Journal of Visualized Experiments. , e51357 (2014).
- Pinz, I., Zhu, M., Mende, U., Ingwall, J. S. An improved isolation procedure for adult mouse cardiomyocytes. Cell Biochemistry and Biophysics. 61, 93-101 (2011).
- O’Connell, T. D., Rodrigo, M. C., Simpson, P. C. Isolation and culture of adult mouse cardiac myocytes. Methods in Molecular Biology. 357, 271-296 (2007).
- Dou, Y., Arlock, P., Arner, A. Blebbistatin specifically inhibits actin-myosin interaction in mouse cardiac muscle. American Journal of Physiology: Cell Physiology. 293, 1148-1153 (2007).
- Kabaeva, Z., Zhao, M., Michele, D. E. Blebbistatin extends culture life of adult mouse cardiac myocytes and allows efficient and stable transgene expression. American Journal of Physiology: Heart and Circulatory Physiology. 294, 1667-1674 (2008).
- Sellin, L. C., McArdle, J. J. Multiple effects of 2,3-butanedione monoxime. Pharmacology & Toxicology. 74, 305-313 (1994).

