Surfactant Depletion Combined with Injurious Ventilation Results in a Reproducible Model of the Acute Respiratory Distress Syndrome (ARDS)
Summary
A combination of surfactant washout using 0.9% saline (35 mL/kg body weight, 37 °C) and high tidal volume ventilation with low PEEP to cause moderate ventilator induced lung injury (VILI) results in experimental acute respiratory distress syndrome (ARDS). This method provides a model of lung injury with low/limited recruitability to study the effect of various ventilation strategies for extended periods.
Abstract
Various animal models exist to study the complex pathomechanisms of the acute respiratory distress syndrome (ARDS). These models include pulmo-arterial infusion of oleic acid, infusion of endotoxins or bacteria, cecal ligation and puncture, various pneumonia models, lung ischemia/reperfusion models and, of course, surfactant depletion models, among others. Surfactant depletion produces a rapid, reproducible deterioration of pulmonary gas exchange and hemodynamics and can be induced in anesthetized pigs using repeated lung lavages with 0.9% saline (35 mL/kg body weight, 37 °C). The surfactant depletion model supports investigations with standard respiratory and hemodynamic monitoring with clinically applied devices. But the model suffers from a relatively high recruitability and ventilation with high airway pressures can immediately reduce the severity of the injury by reopening atelectatic lung areas. Thus, this model is not suitable for investigations of ventilator regimes that use high airway pressures. A combination of surfactant depletion and injurious ventilation with high tidal volume/low positive end-expiratory pressure (high Tv/low PEEP) to cause ventilator induced lung injury (VILI) will reduce the recruitability of the resulting lung injury. The advantages of a timely induction and the possibility to perform experimental research in a setting comparable to an intensive care unit are preserved.
Introduction
The mortality of the acute respiratory distress syndrome (ARDS) remains high with values above 40%1 despite intensive research since its first description by Ashbough and Petty in 19672. Naturally, the investigation of novel therapeutic approaches is limited in the clinic due to ethical concerns and the lack of standardization of the underlying pathologies, ambient conditions, and co-medications, whereas animal models enable systematic research under standardized conditions.
Thus, experimental ARDS has been induced in either large animals (e.g., pigs) or small animals (e.g., rodents) using various methods such as pulmo-arterial infusion of oleic acid, intravenous (i.v.) infusion of bacteria and endotoxins, or cecal ligation and puncture (CLP) models causing sepsis-induced ARDS. In addition, direct lung injuries caused by burns and smoke inhalation or lung ischemia/reperfusion (I/R) are used3. One frequently used model of direct lung injury is surfactant depletion with lung lavages as first described by Lachmann et al. in guinea pigs4.
Surfactant depletion is a highly reproducible method that results rapidly in compromises in gas exchange and hemodynamics5. A major advantage is the possibility to apply surfactant depletion in large species which enable support research with clinically used mechanical ventilators, catheters, and monitors. However, a major disadvantage of the surfactant depletion model is the instant recruitment of atelectatic lung areas whenever high airway pressures or recruiting maneuvers, such as prone positioning, are applied. Thus, the model is not suitable to investigate, e.g., automated ventilation with high PEEP levels for prolonged times6. Yoshida et al. described a combination of surfactant depletion and ventilation with high inspiratory airway pressures to induce experimental ARDS7, but their model requires an elaborate maintenance of partial pressure of oxygen (PaO2) in a predefined corridor via repeated blood gas sampling and adjustment of the driving pressure according to a sliding table of inspiratory pressure and PEEP.
Overall, a model with an overly aggressive injurious ventilation or a laborious, repeated adjustment of the ventilation regime can result in structural damage of the lungs, which is too severe and results in subsequent multiple organ failure. Thus, this article provides a detailed description of an easily feasible model of surfactant depletion plus injurious ventilation with high Tv/low PEEP for induction of experimental ARDS, which supports research with clinically used ventilation parameters for prolonged periods.
Protocol
The experiments were conducted at the Department of Experimental Medicine, Charité – University Medicine, Berlin, Germany (certified according to the EN DIN ISO 9001:2000) and were approved by the federal authorities for animal research in Berlin, Germany, prior to the experiments (G0229/18). The principles of laboratory animal care were used in all experiments and are in accordance with the guidelines of the European and German Society of Laboratory Animal Sciences.
1. Laboratory animals and animal welfare
- Conduct all the experiments in deeply anesthetized male pigs (German Landrace × Large White) of 3-4 months of age with a body weight (bw) of 30-40 kg.
2. Anesthesia, intubation, and mechanical ventilation
- Do not provide dry food for 12 h prior to anesthesia to avoid a full stomach of the pigs. Allow free access to water and straw/hay to minimize stress.
- Premedicate with an intramuscular injection of a combination of azaperone (3 mg/kg bw), atropine (0.03 mg/kg bw), ketamine (25 mg/kg bw), and xylazine (3.5 mg/kg bw) into the neck musculature of the pig, while the animals are still kept in their housing facility to minimize stress.
NOTE: Daily training of petting the animal's neck while feeding a few sugar cubes prior to the experiment and applying the injection while feeding sugar cubes in the trained fashion will facilitate a smooth premedication and reduce stress further.- Place the animal onto a stretcher and cover the eyes with a cloth for transportation once an adequate level of anesthesia is reached.
- Transfer the pig to the surgical theater and always ensure sufficient spontaneous breathing.
- Take an oxygen cylinder, fitting tubing, and mask to provide supplemental oxygen while transporting the pigs, if the housing facilities are not adjacent to the laboratory.
- Place the pig in the prone position and preoxygenate with a mask that fits the animal's snout using a high flow of oxygen (e.g., 10 L/min).
- Use a peripheral vein catheter (usually 18 or 20 G) to gain venous access. Place the peripheral vein catheter into one of the ear veins after a wipe-down procedure with alcohol swaps.
- Start an infusion with a balanced crystalloid solution and ensure the correct placement of the catheter for subsequent infusion of anesthetics.
- Infuse 500 mL of a balanced crystalloid solution as bolus i.v. followed by continuous infusion of 4 mL/kg/h for fluid support.
- Start monitoring the peripheral oxygen saturation (SpO2) by securing the SpO2-Sensor at one of the ears or the tail.
- Induce anesthesia by injecting propofol (about 5-10 mg/kg – the exact dose depends on the effect of the premedication and differs from animal to animal) for orotracheal intubation.
NOTE: Prior injection of an opioid will facilitate intubation further but requires ample experience to avoid a premature apnea of the animal. An injection of 100 µg of fentanyl (fentanyl citrate, 100 µg/mL) may be repeated until the spontaneous respiratory rate slows down to about 20/min before injecting propofol. - Intubate the animal with a cuffed endotracheal tube (7.5 – 8.0 mm ID) and a laryngoscope designed for large animals (straight blade of about 25 cm length).
NOTE: Intubation is easiest in the prone position as described in detail by Theisen et al.8.- Verify the placement of the endotracheal tube by observing the typical waveform of CO2 during expiration on the CO2-monitor (capnograph).
- Use auscultation to check for equal bilateral breath sounds.
NOTE: The pigs can be mechanically ventilated with manual compression of the rib cage from both sides while supplying oxygen with a high flow in case of failed or delayed intubation.
- Set the fraction of inspired oxygen (FIO2) to 1.0, respirator frequency to 15-20/min, tidal volume to 8-9 mL/kg bw, inspiration to expiration ratio (I:E) to 1:1.5, and apply a positive end-expiratory pressure (PEEP) of 5 cmH2O to start mechanical ventilation. Adjust the settings to target an end-expiratory partial pressure of carbon dioxide (PetCO2) of 35-40 mmHg and a SpO2 above 95%.
- Use a continuous i.v. infusion of thiopentone (20 mg/kg/h) and fentanyl (7 µg/kg/h) to maintain anesthesia.
NOTE: The necessary dosage may vary from animal to animal and between experimental settings. It is essential to maintain a sufficient depth of anesthesia during the course of the experiment for animal welfare and scientific reasons. - Monitor the animal closely for stress/pain reactions (such as an increase in heart rate, blood pressure, or respiratory rate) during instrumentation.
NOTE: Instrumentation should be possible without administering a muscle relaxant if the depth of anesthesia is sufficient. - Administer a muscle relaxant, e.g., pancuronium bromide (0.15 mg/kg bw i.v. bolus, followed by a continuous infusion of 0.15 mg/kg bw/h or repeated bolus injections), if muscle relaxation is necessary for the experiment (e.g., before a surfactant depletion, before injurious ventilaion lung compliance measurements).
- Use a continuous i.v. infusion of thiopentone (20 mg/kg/h) and fentanyl (7 µg/kg/h) to maintain anesthesia.
- Instrumentation techniques
- Turn the animal into the supine position.
- Secure the endotracheal tube and i.v. line while turning the animal.
- Retract the legs using bandages to stretch the skin above the planned incision sites.
- Sterilize the operating areas with an appropriate skin disinfectant such as an alcohol and iodine 1% solution.
- Cannulate the external jugular vein with a central venous catheter and, in addition, introduce the introducer sheath of the pulmonary arterial catheter (PAC) into the same vein.
- Perform a 10 cm skin incision on the line connecting the mandible and the sternum (left or right side possible).
- Always reassess the depth of anesthesia and adjust the dosage, if necessary.
- Separate the subcutaneous tissue and the platysma with tissue forceps and surgical scissors until the brachiocephalic and the sternocephalic muscles are visible.
- Continue with a blunt cut down procedure to separate the fascia between the muscles until the external jugular vein is visible.
- Use the Seldinger technique9 to cannulate the external jugular vein with the central venous catheter and the introducer sheath for later insertion of the PAC.
NOTE: Do not dilate the vein with a dilator as it is done in case of a percutaneous approach. This would tear the vein. Close with standard sutures. The sizes of the sheath depend on the size of the chosen PAC. A 6F introducer sheath (10 cm length) and a 5F PAC of 75 cm length in pigs of 30-40 kg body weight are typically used.
- Cannulate the femoral artery for invasive blood pressure monitoring.
- Identify the fold between the gracilis and sartorius muscle of the hind leg (left or right is possible) to place an arterial line.
NOTE: The pulsation of the femoral artery should be easily palpable. - Cannulate the artery percutaneously with the Seldinger technique9.
- Use a direct approach if the artery is not easily palpated.
- Cut through the skin with a 5 cm long incision and separate the subcutaneous tissue with tissue forceps and surgical scissors.
- Use a blunt cut down procedure separating the fascia between the muscles to the level of the femoral artery.
NOTE :Do not injure the saphenous vessels by performing the cut down procedure cranial of them. - Loop a ligature around the femoral artery so that the vessel can be closed in case of bleeding at the site of puncture. Avoid this step whenever possible, as it compromises blood flow to the hind leg.
- Cannulate the artery with the Seldinger technique9.
- Identify the fold between the gracilis and sartorius muscle of the hind leg (left or right is possible) to place an arterial line.
- Calibrate the transducers against the atmosphere (zero) and either 200 mmHg (arterial line) or 50 mmHg (central venous line) and connect them to the arterial catheter and the central venous line to start monitoring.
- Place the pressure transducers about half the height of the thorax at the estimated position of the right atrium.
- Perform a small (4-5 cm) incision cutting through the skin above the bladder for catherization of the urinary bladder.
- Separate the subcutaneous tissue using blunt instruments.
- Place a purse-string suture (1-2 cm in diameter) in the wall of the bladder.
NOTE: The sutures should not penetrate through all layers of the bladder wall, which would result in the loss of urine through the punctures. - Perform a small incision in the middle of the suture and introduce the urinary catheter.
- Immediately, block the balloon with 10 mL of distilled water and pull the catheter toward the bladder wall until a light resistance is felt.
- Close the purse-string suture around the catheter. Close the skin using standard sutures.
3. Introduction of the pulmonary artery catheter (PAC)
- Check the patency of the balloon of the PAC with 0.5-1 mL of air depending on the size of the catheter and deflate the balloon again.
- Connect the PAC to the pressure transducer system and calibrate the transducer against the atmosphere (zero) and 100 mmHg.
- Introduce the PAC through the introducer sheath with a deflated balloon for 10-15 cm (depending on the sheath length).
- Inflate the balloon after it has left the sheath and advance the PAC further while monitoring the pressure and the typical wave forms on the pressure monitor.
- Push the PAC forward while the waveforms typical of the right atrium, right ventricle, and the pulmonary artery appear and stop advancing the PAC when the pulmonary capillary wedge pressure (PCWP) waveform is seen.
- Record the PCWP at end-expiration and deflate the balloon (see Figure 1 for the respective curves).
NOTE: After deflation of the balloon, the PCWP-waveform must disappear, and the pulmonary arterial pressure waveform must be visible. If the pulmonary arterial pressure waveform cannot be seen, the catheter is most likely inserted too far into a pulmonary artery and has reached an auto-wedge position. This results in a permanent occlusion of a pulmonary vessel and must be corrected by pulling the catheter back until the pulmonary arterial pressure waveform reappears thereby avoiding complications, e.g., rupture of the pulmonary vessel10. The PAC catheters are often accidentally advanced into liver veins via the inferior caval vein in pigs. Thus, if the right ventricle pressure signal is not reached after about 30 – 50 cm, pull the catheter back and start all over again.
4. Pulmonary artery thermodilution technique for hemodynamic measurements
- Measure cardiac output (CO) with the thermodilution technique11.
- Connect the thermistor and a flow through housing to the respective lumen of the PAC.
- Next, connect the hemodynamic monitor with the distal temperature port of the PAC (red cap).
- Adjust the hemodynamic monitor to the necessary mode compensating for catheter size, catheter length, injected volume, and temperature of the injected saline solution.
- Inject the appropriate volume of 0.9% saline as quickly as possible (usually 5 or 10 mL of 0.9% saline with a temperature of 4 °C).
- Wait until the measurement is completed.
- Randomize five measurements in quick succession over the respiratory cycle of the ventilator.
- Delete the highest and the lowest values and use the remaining three values to calculate the mean.
- Note this mean value as the cardiac output.
- Measure the PCWP afterwards by inflating the catheter balloon, and deflate it after the measurement.
- Use the mean arterial pressure (MAP), pulmonary arterial pressure (PAP), central venous pressure (CVP), PCWP, and the CO for all further hemodynamic calculations.
NOTE: The volume of saline as well as the temperature have to be entered into the monitor before the measurements. The normal saline has to be kept at the same temperature (usually <5 °C) for correct measurements. The size and length of the catheter have to be entered as well. Some monitors require the entry of a correction factor. - For studies involving exact measurements of electrolyte balance, use 5% glucose solution instead of 0.9% saline.
- Ensure to record all the parameters. Take simultaneous arterial and mixed venous blood samples shortly before or after CO measurements to enable calculation of the intra-pulmonary right-to-left shunt.
- Record all needed respiratory settings and measurements to complete the data set, e.g. peak, plateau and end-expiratory pressure.
NOTE: The induction of anesthesia, intubation, and full instrumentation may require 1.5 h depending on the experience and number of the investigators.
- Record all needed respiratory settings and measurements to complete the data set, e.g. peak, plateau and end-expiratory pressure.
5. Surfactant depletion
- Ventilate the animal with a FIO2 of 1.0.
- Disconnect the animal from the ventilator.
- Fill the lungs with prewarmed 0.9% saline (37 °C, 35 mL/kg) with a funnel connected to the endotracheal tube.
- For this, raise the funnel about 1 m above the animal.
NOTE: The hydrostatic pressure will allocate the saline into all pulmonary sections. - Immediately stop filling when the MAP decreases below <50 mmHg.
- For this, raise the funnel about 1 m above the animal.
- Lower the funnel to ground level to drain the lavage fluid. Reconnect the animal to the ventilator for oxygenation.
- Wait until the animal recuperates and repeat the lavage a soon as possible, if required.
NOTE: The necessity for a further lavage is defined by the PaO2/FIO2 ratio.- Take an arterial blood gas sample after 5 min following each lavage.
- Repeat lavages until the PaO2/FIO2 ratio (Horowitz index) decreases below 100 mmHg for at least 5 min at FIO2 1.0 and PEEP > 5 cmH2O.
NOTE: The respiratory rate must be adjusted during the period of lavages to keep the arterial pH above 7.25 in order to prevent hemodynamic decompensation.
- Be aware that this animal model is based on a combination of surfactant depletion and VILI.
NOTE: The lavages will be stopped after the PaO2/FIO2 ratio remains below 100 for 5 min NOT after 60 min as published previously for a model of surfactant washout without VILI5.- Commence with high Tv/low PEEP ventilation after the targeted PaO2/FIO2 has been reached.
NOTE: Otherwise, an overly aggressive surfactant depletion combined with VILI will result in multiple organ failure and compromise the experiment. The duration of surfactant depletion varies between animals, since a defined PaO2/FIO2 is targeted. It may take 45 min to 1.5 h.
- Commence with high Tv/low PEEP ventilation after the targeted PaO2/FIO2 has been reached.
6. Injurious ventilation with high tidal volume/low PEEP (high Tv/low PEEP)
- Keep an FIO2 of 1.0.
- Set the ventilator on a volume guaranteed, pressure-controlled ventilation mode.
- Increase the alarm threshold for peak inspiratory pressure to 60 mbar.
NOTE: The ventilator should apply an inspiratory pressure up to 60 mbar, but not higher. - Lower the respiratory rate to 12/min and set the inspiration to expiration (I:E) ratio to 1:1.5 (resulting in an inspiration time of 2 s and expiration time of 3 s).
- Increase the tidal volume slowly up to 17 mL/kg bw over at least 2 min.
- Do not increase the tidal volume further if an inspiratory pressure of 60 mbar is reached.
NOTE: The limited inspiratory pressure can result in tidal volume below 17 mL/kg body weight depending on the lung injury after surfactant washout. A sudden increase in tidal volume may result in barotrauma or hemodynamic decompensation. Therefore, it is of utmost importance to increase tidal volumes slowly over several minutes.
- Do not increase the tidal volume further if an inspiratory pressure of 60 mbar is reached.
- Reduce the PEEP to 2 mbar.
- Ventilate the animal for up to 2 h (see Figure 2 for the ventilator settings and the flow curve).
NOTE: Ventilation with high tidal volumes will result in good oxygenation of the animal, but the cyclic near-complete inflation and deflation results in structural injury of the lungs. Structural damage cannot be reversed with recruitment maneuvers, prone positioning, high PEEP, etc. The resulting injury has to be tolerated throughout the investigation. A shorter high Tv/low PEEP ventilation time may be required depending on the following experiment and duration of the investigation.
7. End of experiment and euthanasia
- Ensure that all measurements of the experimental protocol, which will follow the induction of lung injury, are performed.
- Inject fentanyl (at least 0.5 mg) additionally to the continuous anesthesia and wait at least 5 minutes. Inject thiopental (at least 1000 mg) quickly followed by at least 60 mmol of potassium using the central line.
Representative Results
The PaO2/FIO2-ratio decreased during surfactant washout in all animals (Figure 3). The resulting hypoxemia, hypercapnia, and atelectasis caused an increase in pulmonary artery pressure. The details of the lung lavages are already described elsewhere6.
The surfactant depletion was repeated until the PaO2/FIO2 ratio remained below 100 mmHg despite mechanical ventilation with a PEEP of 5 mbar for at least 5 min. Afterwards, ventilation with high tidal volumes, low PEEP, and nearly complete inflation/deflation was commenced for 2 h to cause VILI. Of note, parameters of gas exchange (oxygen saturation, PaO2) can improve during ventilation with high tidal volumes due to the cyclic recruitment while mPAP usually remains elevated due to high intrathoracic pressures and hypercapnia (Figure 3B). On average, induction of anesthesia, instrumentation, surfactant depletion, and injurious ventilation require about 5 h depending on experience of the investigator and the number of lavages required to achieve the targeted PaO2/FIO2 ratio.
The recruitability of the lungs was tested after each experimental step with a recruitment maneuver (inspiratory pressure of 50 mbar and PEEP 24 mbar for five breaths). An arterial blood gas sample was taken 5 min after the recruitment maneuver while ventilation was commenced with a tidal volume of 6 mL/kg bw, a PEEP of 15 mbar and an FIO2 of 1.0. This recruitment maneuver resulted in a notable increase in the oxygenation in all the animals after surfactant washout (Figure 3a), whereas 2 h of injurious ventilation diminished lung recruitability with respect to gas exchange and mPAP (Figure 3, Table 1). The lung injury induced with the protocol was not prone to recruitment even when ventilation was performed according to the ARDS-Network high PEEP table for 3 h after an additional recruitment maneuver.
Computer tomographic (CT) imaging of one animal showed atelectasis of the dependent areas of the lung during ventilation with a PEEP of 6 mbar, which resolved largely when ventilation was escalated to a PEEP of 15 mbar (Figure 4), whereas the substantial ubiquitous ground glass opacities did not resolve. Furthermore, some CT findings such as alveolar opacities indicated structural damage of the lungs corresponding with post-mortem examination of the lungs (Figure 4).
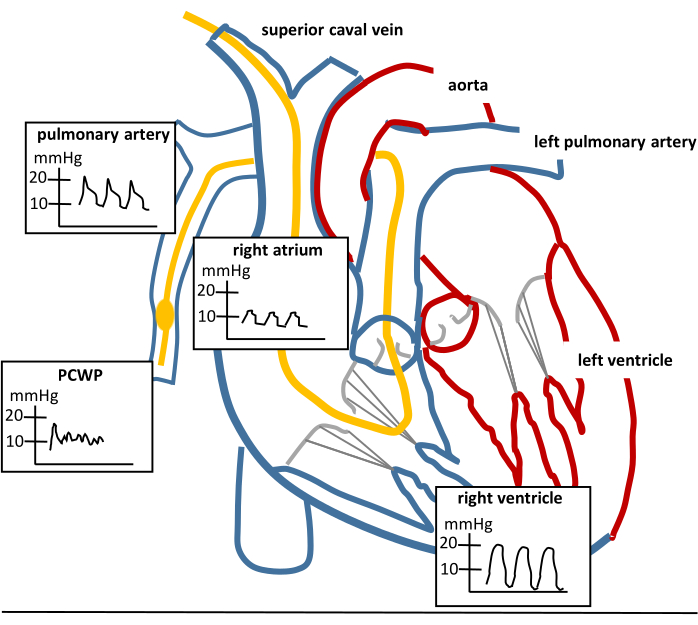
Figure 1: Pulmonary artery catheter placement. Sketch of the heart, a properly placed pulmonary artery catheter (PAC; yellow catheter) and the respective waveforms that can be seen while advancing a PAC. PCWP means pulmonary capillary wedge pressure. The PCWP waveform can only be seen in wedge position while the balloon is inflated. The PCWP curve should disappear and the pulmonary artery curve should be visible if the balloon is deflated and the PAC is placed properly. Please click here to view a larger version of this figure.
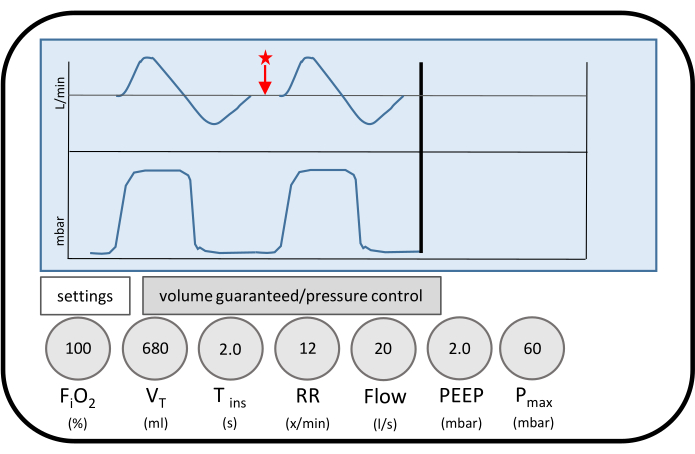
Figure 2: Ventilator settings of injurious ventilation. Displayed are the ventilator settings during ventilation to provoke ventilator-induced lung injury (VILI). The tidal volume corresponds to 17 mL/kg body weight in the respective animal. The flow pattern decreases to zero flow on expiration (red star). Zero flow is maintained for a relevant period of the respiratory cycle. Thus, almost complete inflation and deflation of the lungs is achieved to promote baro- and atelectrauma. Please click here to view a larger version of this figure.
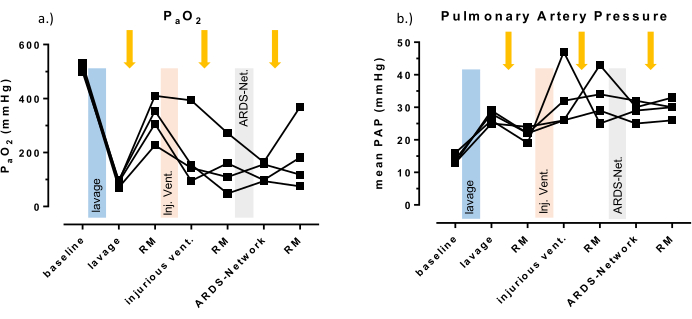
Figure 3: Systemic oxygenation and pulmonary artery pressure. (A) Individual results of the partial arterial pressure of oxygen. (B) Mean pulmonary arterial pressure of four animals are displayed as representative values for the induced lung injury. Test for statistical significance were not performed due to the small number of animals (n = 4). A recruitment maneuver was performed after each intervention (yellow arrows) to test for recruitability of the model. Note that PaO2 increases after lung lavage and after recruitment by at least 150 mmHg, but not after injurious ventilation. Please click here to view a larger version of this figure.
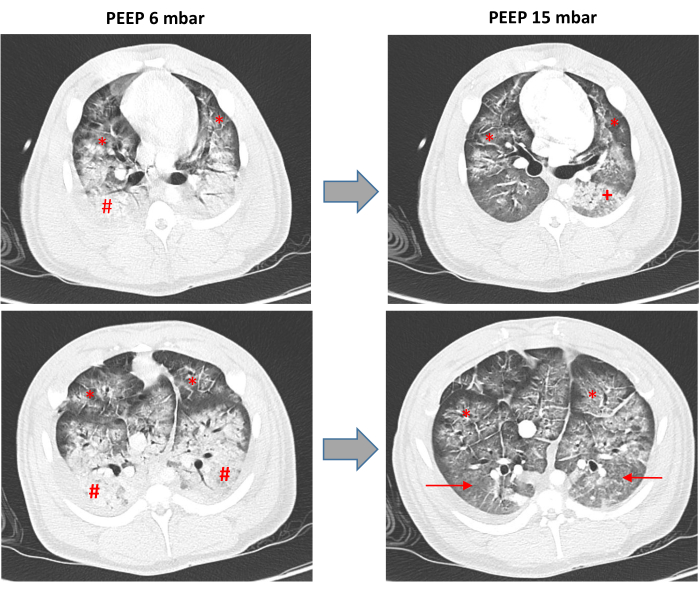
Figure 4: Computed tomography of the lungs. Representative computer tomographic scans (CT) of one animal after surfactant washout and mechanical ventilation with high tidal volumes and low PEEP to cause ventilator-induced lung injury (VILI). The scans were taken during ventilation with high positive end expiratory pressure of 15 mbar (PEEP 15 mbar) and low PEEP of 6 mbar (PEEP 6 mbar) with a tidal volume of 6 mL/kg body weight. The upper panels show the same apical region of the lungs. The lower panels show the same region of the lung at height of the heart. The # marks the dependent lung areas with basal atelectasis; the → marks the dependent lung areas/former atelectasis, which are recruited under ventilation with a PEEP of 15 mbar; the * marks extensive ground glass opacities with superimposed inter- and intralobular septeal thickening, which are not resolved during ventilation with a PEEP of 15 mbar, the + marks diffuse alveolar opacifications, which indicate alveolar hemorrhage and are not visible during ventilation with a PEEP of 6 mbar due to the extensive atelectasis. Please click here to view a larger version of this figure.
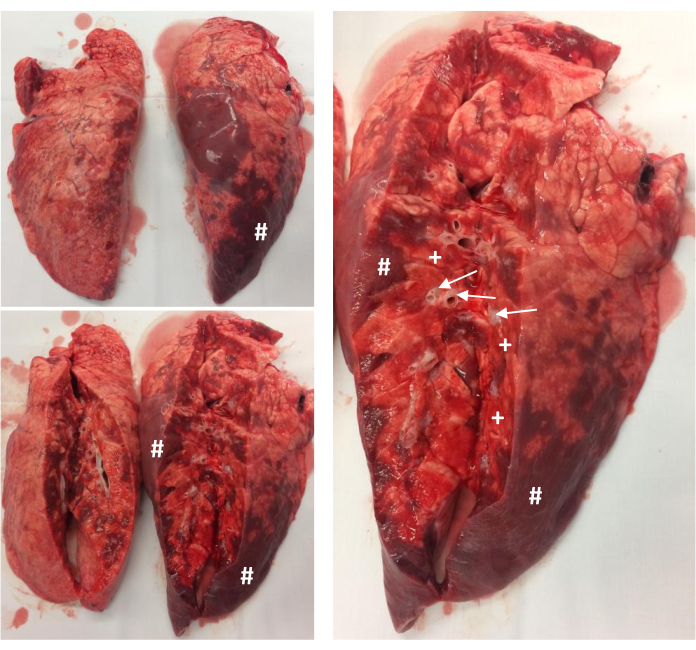
Figure 5: Postmortem examination of the lungs. Representative pathology of the unfixed lungs of one animal right after the experiment. The basal area of the lungs faces toward the reader. The # marks atelectasis; the + marks diffuse alveolar hemorrhage; the → marks distended, edematous peribronchial spaces. Please click here to view a larger version of this figure.
| baseline | after lavage | RM | after injuriuos ventilation | RM | after ARDS-Net | RM | |
| PaO2 (mmHg) |
514 ±13 |
87 ±12 |
324 ±78 |
197 ±134 |
147 ±95 |
128 ±37 |
185 ±129 |
| PaCO2 (mmHg) |
48 ±6 |
86 ±10 |
82 ±12 |
66 ±5 |
96 ±4 |
92 ±5 |
123 ±10 |
| pH | 7.39 ±0.09 |
7.14 ±0.05 |
7.17 ±0.08 |
7.26 ±0.06 |
7.11 ±0.04 |
7.14 ±0.04 |
7.04 ±0.03 |
| lactate (mg/dL) |
4 ±3.9 |
6 ±5.0 |
6 ±5.9 |
4 ±3.6 |
4 ±3.5 |
4 ±3.6 |
6 ±5.3 |
| heart rate (beats/min) |
86 ±8 |
90 ±11 |
92 ±12 |
104 ±18 |
129 ±30 |
147 ±13 |
149 ±5 |
| CO (L/min) | 4 ±0.8 |
3.7 ±1.4 |
3.6 ±0.8 |
5.2 ±0.8 |
5.1 ±0.8 |
6.9 ±1.0 |
|
| mAP (mmHg) |
93 ±4 |
101 ±21 |
108 ±31 |
78 ±8 |
96 ±31 |
65 ±12 |
72 ±9 |
| SVR (dyn.sec.cm-5) |
1856 ±302 |
2552 ±777 |
1624 ±468 |
1179 ±237 |
903 ±292 |
711 ±166 |
|
| mPAP (mmHg) |
14 ±1 |
27 ±2 |
22 ±2 |
33 ±10 |
33 ±8 |
29 ±3 |
30 ±3 |
| PVR (dyn.sec.cm-5) |
106 ±170 |
267 ±442 |
170 ±258 |
92 ±126 |
108 ±160 |
66 ±88 |
|
| PCWP | 6 ±2 |
10 ±2 |
8 ±2 |
9 ±1 |
10 ±4 |
11 ±5 |
|
| Cdyn (mL/mbar) |
33 ±4 |
12 ±2 |
21 ±4 |
23 ±8 |
20 ±2 |
26 ±8 |
24 ±5 |
Table 1: Arterial blood gases, hemodynamic data and lung compliance. The table presents the respective arterial blood gases and hemodynamic data. RM: recruitment maneuver, PaO2: arterial partial pressure of oxygen, PaCO2: arterial partial pressure of carbon dioxide, CO: cardiac output, MAP: mean arterial pressure, SRV: systemic vascular resistance, mPAP: mean pulmonary arterial pressure, PVR: pulmonary vascular resistance, PCWP: pulmonary capillary wedge pressure. Data presented as mean ± SD.
| baseline | after lavage | RM | ||||||
| I | PaO2 (mmHg) | 540 | 81.3 | 270 | 21.9 | -the recruitment maneuver after surfactant depletion was preformed without prior injection of a muscle relaxant -the recruitment maneuver (RM) resulted in a tension pneumothorax with rapid cardiopulmonary deterioration (grey background) despite immediate chest drain insertion – following animals received a bolus injection of a muscle relaxant prior to a RM and the problem was not observed again |
||
| PaCO2 (mmHg) | 42.6 | 69.4 | 84.9 | 93.9 | ||||
| pH | 7.44 | 7.17 | 7.01 | 6.99 | ||||
| Lactate (mmol/L) | 11 | 17 | 67 | 56 | ||||
| heart rate (beats/min) | 138 | 155 | 141 | 221 | ||||
| CO (L/min) | 7.7 | 3.6 | 1.6 | |||||
| mAP (mmHg) | 82 | 60 | 143 | 53 | ||||
| mPAP (mmHg) | 26 | 18 | 22 | 22 | ||||
| PCWP (mmHg) | 10 | 12 | 12 | 17 | ||||
| Cdyn (mbar/mL) | 35 | 11 | 19 | 13 | ||||
| PCWP (mmHg) |
10 | 12 | 12 | 17 | ||||
| Cdyn (mbar/mL) | 35 | 11 | 19 | 13 | ||||
| baseline | after lavage | RM | after injurious ventilation | RM | ||||
| II | PaO2 (mmHg) | 638 | 60 | 84 | 83.2 | 61.4 | 82.7 | -injurous ventilation was performed with tidal a volume of 17 ml/kg body weight for 3 hours -after injurious ventilation the animal deteriorated rapidly and could not be stabilized with e.g. bolus injections of epinephrine -the last blood gas analysis was obtained under ventilation with PEEP: 20 mbar. Ppeak: 35 mbar. resulting in a tidal volume of only 187 ml (4ml/kg body weight) – reduction of the injurious ventilation period was necessary in following experiments |
| PaCO2 (mmHg) | 41 | 78 | 77 | 85.1 | 120 | 183 | ||
| pH | 7.37 | 7.17 | 7.16 | 7.13 | 7.02 | 6.81 | ||
| Lactate (mg/dL) | 16 | 18 | 20 | 17 | 30 | 65 | ||
| heart rate (beats/min) | 86 | 64 | 109 | 133 | 150 | 185 | ||
| CO (L/min) | 4.3 | 3.3 | 3.7 | 5.6 | 2.4 | |||
| mAP (mmHg) | 77 | 82 | 61 | 53 | 77 | 40 | ||
| mPAP (mmHg) | 15 | 30 | 24 | 35 | 35 | 32 | ||
| PCWP (mmHg) | 7 | 8 | 9 | 8 | 9 | |||
| Cdyn (mbar/mL) | 34 | 9 | 12 | 17 | 14 | 13 | ||
Table 2: Arterial blood gases and hemodynamic data during implementation of the protocol. The table presents the respective arterial blood gases and hemodynamic data of two animals, which died prematurely during the implementation of the protocol. Gray background highlights the last results before death. RM: recruitment maneuver, PaO2: arterial partial pressure of oxygen, PaCO2: arterial partial pressure of carbon dioxide, CO: cardiac output, mAP: mean arterial pressure, mPAP: mean pulmonary arterial pressure, PCWP: pulmonary capillary wedge pressure, PEEP: positive end-expiratory pressure, Ppeak: peak inspiratory pressure.
Discussion
This article describes the induction of experimental ARDS in pigs combining surfactant depletion by repeated lung lavages and ventilation with high tidal volumes, low PEEP, and complete inflation/deflation of the lungs. This combination causes a reproducible and comparable deterioration in gas exchange and the resulting hemodynamic compromise but limits the recruitability of the lungs. Thus, this model mimics clinical ARDS with low recruitability and allows the investigation of new ventilation regimes.
There are a few limitations of the protocol. First, repeated lavages result in some of the histopathological properties of clinical (human) ARDS, including the formation of major atelectasis, perivascular edema formation, and an increase of the alveolar-capillary membrane thickness. High Tv/low PEEP ventilation adds some properties such as diffuse alveolar hemorrhage, which are not vulnerable to recruitment. Nevertheless, important features of human ARDS such as the formation of hyaline membranes cannot be induced within hours and are therefore missing in this model2,3. Second, the structural damage of the lungs is irreversible for hours or possibly days. But care must be taken to avoid an excessive baro-, volu-, and atelectrauma of the lungs, which would render the following experiment impossible. Using the ventilator settings described in the article, the protocol started with initially 3 h of VILI to test automated ventilation modes, which integrate recent clinical evidence regarding ventilation of ARDS patients. Unfortunately, some animals deteriorated during the course of the experiment and one case of a severe pneumothorax (Table 2) was observed. Reducing the VILI period to 2 h was suitable for the experimental design, but this time period may be adapted in other experimental settings. Third, lung lavages can result in abrupt right heart failure and death of the animal. About 10%-15% of the animals may die during the induction period. This number can be reduced following the recommendations published previously5. Finally, the study only presented the results of four animals and two further animals, which died prematurely during the implementation of the model. Strict local animal protection laws do not support experiments in further animals once the model is sufficiently implemented, but two-hit models consisting of surfactant depletion and injurious ventilation have been used by other research groups7.
Of importance, the aggravation of lung injury by ventilation with high Tv/low PEEP ventilation can result in uncontrollable structural damage of the lungs or hemodynamic decompensation. Hence, the tidal volumes have to be increased in steps over several minutes and an upper threshold for peak inspiratory pressure has to be set to avoid pneumothorax and hemodynamic instability. It was found that an upper threshold of 60 mbar was most suitable to cause VILI without losing animals prematurely.
The cyclic recruitment with high tidal volumes will result in sufficient oxygenation despite low PEEP. After the lung lavages, the PEEP was decreased to 2 mbar in a stepwise fashion parallel to increasing tidal volume in order to avoid intolerable hypoxemia.
Some investigators use higher respiratory rates to generate VILI7 due to a faster onset of VILI, but high respiratory rates can result in air trapping if the flow curve of the ventilator is not monitored closely. Air trapping may reduce VILI due to incomplete deflation of the lungs for one thing, while it also promotes hemodynamic instability caused by sustained high intrathoracic pressures. Thus, a slower respiratory rate was used with an ensured deflation of the lungs and longer VILI period in the described model.
Of note, markers of pulmonary inflammation such as interleukin 8 in the brochoalveolar fluid were not measured since prolonged ventilation in a reproducible model of low recruitability is the main application of the model. For research concerning specific inflammatory patterns (such as the hyper-inflammatory subphenotype of ARDS) a multiple hit model combining an inflammatory first hit such as i.v. lipopolysaccharide infusion with injurious ventilation could be favourable12.
The combination of surfactant washout and high Tv/low PEEP ventilation results in a time-efficient and reproducible model of human ARDS with respect to gas exchange and hemodynamic changes. The lung injury induced in this model presents low recruitability and permits the experimental investigation of therapeutic strategies, including mechanical ventilation.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We gratefully acknowledge the excellent technical assistance of Birgit Brandt. This study was supported by a grant of the German Federal Ministry of Education and Research (FKZ 13GW0240A-D).
Materials
| Evita Infinity V500 | Dräger | intensive care ventilator | |
| Flow through chamber thermistor | Baxter | 93-505 | for measuring cardiac output |
| Leader Cath Set | Vygon | 1,15,805 | arterial catheter |
| Mallinckrodt Tracheal Tube Cuffed | Covidien | 107-80 | 8.0 mm ID |
| MultiCath3 | Vygon | 1,57,300 | 3 lumen central venous catheter, 20 cm length |
| Percutaneus Sheath Introducer Set | Arrow | SI-09600 | introducer sheath for pulmonary artery catheter of 4-6 Fr., 10 cm length |
| Swan-Ganz True Size Thermodilution Catheter | Edwards | 132F5 | pulmonary artery catheter, 75 cm length |
| urinary catheter | no specific model requiered | ||
| Vasofix Braunüle 20G | B Braun | 4268113B | peripheral vein catheter |
| Vigilance I | Edwards | monitor |
References
- Bellani, G., et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA. 315 (8), 788-800 (2016).
- Ashbaugh, D. G., Bigelow, D. B., Petty, T. L., Levine, B. E. Acute respiratory distress in adults. Lancet. 2 (7511), 319-323 (1967).
- Ballard-Croft, C., Wang, D., Sumpter, L. R., Zhou, X., Zwischenberger, J. B. Large-animal models of acute respiratory distress syndrome. The Annals of Thoracic Surgery. 93 (4), 1331-1339 (2012).
- Lachmann, B., Robertson, B., Vogel, J. In vivo lung lavage as an experimental model of the respiratory distress syndrome. Acta Anaesthesiologica Scandinavica. 24 (3), 231-236 (1980).
- Russ, M., et al. Lavage-induced surfactant depletion in pigs as a model of the acute respiratory distress syndrome (ARDS). Journal of Visualized Experiments: JoVE. (115), e53610 (2016).
- Pomprapa, A., et al. Artificial intelligence for closed-loop ventilation therapy with hemodynamic control using the open lung concept. International Journal of Intelligent Computing and Cybernetics. 8 (1), 50-68 (2015).
- Yoshida, T., et al. Continuous negative abdominal pressure reduces ventilator-induced lung Injury in a porcine model. Anesthesiology. 129 (1), 163-172 (2018).
- Theisen, M. M., et al. Ventral recumbency is crucial for fast and safe orotracheal intubation in laboratory swine. Laboratory Animals. 43 (1), 96-101 (2009).
- Seldinger, S. I. Catheter replacement of the needle in percutaneous arteriography: A new technique. Acta Radiologica. 39 (5), 368-376 (1953).
- Kelly, C. R., Rabbani, L. E. Videos in clinical medicine. Pulmonary-artery catheterization. The New England Journal of Medicine. 369 (25), 35 (2013).
- Forrester, J. S., et al. Thermodilution cardiac output determination with a single flow-directed catheter. American Heart Journal. 83 (3), 306-311 (1972).
- Dos Santos Rocha, A., et al. Physiologically variable ventilation reduces regional lung inflammation in a pediatric model of acute respiratory distress syndrome. Respiratory Research. 21 (1), 288 (2020).

