Summary
The basophil activation test is a complementary in vitro diagnostic test for the evaluation of IgE-mediated allergic reactions based on the detection of basophil activation in the presence of a specific stimulus through the measure of activation markers by flow cytometry.
Abstract
The basophil activation test (BAT) is a complementary in vitro diagnostic test that can be used in addition to clinical history, skin test (ST), and specific IgE (sIgE) determination in the evaluation of IgE-mediated allergic reactions to food, insect venom, drugs, as well as some forms of chronic urticaria. However, the role of this technique in the diagnostic algorithms is highly variable and not well determined.
BAT is based on the determination of basophil response to allergen/drug cross-linking IgE activation through the measurement of activation markers (such as CD63, CD203c) by flow cytometry. This test can be a useful and complementary tool to avoid controlled challenge tests to confirm allergy diagnosis, especially in subjects experiencing severe life-threatening reactions. In general, the performance of BAT should be considered if i) the allergen/drug produces false positive results in ST; ii) there is no allergen/drug source to use for ST or sIgE determination; iii) there is discordance between patient history and ST or sIgE determination; iv) symptoms suggest that ST may result in systemic response; v) before considering a CCT to confirm the culprit allergen/drug. The main limitations of the test are related to non-optimal sensitivity, especially in drug allergy, the need to perform the test no longer than 24 h after sample extraction, and the lack of standardization between laboratories in terms of procedures, concentrations, and cell markers.
Introduction
IgE-mediated allergy diagnosis is based on clinical history, skin tests (STs), quantification of serum specific IgE (sIgE), and, if it is required and indicated, controlled challenge tests (CCTs)1,2,3,4,5,6. However, clinical history can be unreliable since there may be a lack of accurate information, and STs and CCTs are not risk-free procedures that can be contraindicated in subjects experiencing severe life-threatening reactions1,2,3,4,5,6. These issues, together with the fact that the determination of sIgE by validated and commercial fluoro-enzyme immunoassays is only available for few allergens and drugs, have highlighted the important role of other in vitro functional assays such as basophil activation test (BAT).
Basophils are key effector cells involved in IgE-mediated allergic reactions that are activated upon cross-linking of adjacent sIgE bound on high-affinity receptors (FcεRI) on cell surface after allergen/drug exposure. Basophil activation triggers cell degranulation and release of pre-formed and new synthesized inflammatory mediators contained in intracytoplasmatic secretion granules7,8,9. BAT is an in vitro method that tries to mimic the basophil activation in the presence of a stimulus (allergen or drug) and determines changes in the expression of basophil activation markers by flow cytometry7,10. There are different strategies to identify basophils (IgE+, CCR3+, CRTH2+, CD203c+) and to measure cell activation (mainly upregulation of CD63 and CD203c) using combinations of fluorochrome-labelled antibodies7,10. CD63, the best clinically validated activation marker11,12,13,14, is a membrane protein anchored at the secretory granules containing histamine that, after cell activation and fusion of the granules with the membrane, is expressed on basophil surface15,16,17,18,19,20,21. CD203c is a surface marker that is constitutively expressed on basophils and upregulated after FcεRI stimulation, which has also shown reliable results in BAT15,22,23,24,25. Besides, it appears to co-express with CD6326.
In the last decades, BAT has shown to be useful in the diagnosis of IgE-mediated allergic reactions induced by different triggers like drugs, food, or inhalants, as well as in some forms of chronic urticaria, as described below. However, the position of this technique in the diagnostic algorithms is highly variable and not well determined.
Drug hypersensitivity
BAT has shown to be useful as a complementary test for selected drugs and patients, particularly for those who experience severe reactions due to the fact that diagnostic value of ST is not well established for most drugs, as they are validated and standardized for a limited number of drugs27,28,29,30. In addition, quantification of sIgE is available only for a limited number of drugs, with lower sensitivity than ST27,28,29,30,31,32. Therefore, the diagnosis of drug hypersensitivity usually relies upon drug provocation test, which can be contraindicated in subjects experiencing severe life-threatening reactions33.
Promising results have been reported for the use of BAT in selected patients reporting immediate hypersensitivity reactions to different drugs like betalactams (BLs)20,34,35,36,37,38,39, neuromuscular blocking agents (NMBAs)19,22,40,41,42,43,44,45, fluoroquinolones (FQs)46,47,48,49, pyrazolones50,51,52, radiocontrast media (RCM)53,54,55,56, and platinum compounds57,58,59. BAT has been reported to have a sensitivity and specificity between 51.7-66.9% and 89.2-97.8%, respectively; and positive and negative predictive values are described to range between 93.4% and 66.3%, respectively27,31. Moreover, BAT has been proposed as a predictive biomarker for breakthrough reactions during desensitization with platinum compounds, as CD203c expression is increased compared to CD63 in patients with high risk of adverse reactions during drug desensitization57.
It is of note that BAT is only useful in drug hypersensitivity when the reaction involves basophil degranulation; therefore, it is not useful in reactions resulting from enzymatic inhibition of cyclooxygenase 142.
Food allergy
BAT has emerged as a potential diagnostic tool for food allergy because determination of serum sIgE to the whole allergen extract or single allergens is often ambiguous, requiring oral food challenge to confirm the diagnosis, which, similarly to drug hypersensitivity, is a costly and not risk-free procedure60. Several studies have shown relevant results with cow's milk61,62, egg61,63, wheat64,65,66,67,68, peanut63,69,70,71,72, hazelnut73,74,75,76,77, shellfish78, peach79,80,81, apple21, celery, and carrot82,83.
The main added value of BAT in the diagnosis of food allergy compared to STs and sIgE in serum is that it shows a higher specificity and similar sensitivity. Thus, BAT is a useful tool to differentiate clinically allergic patients from sensitized, but tolerant, subjects that have both high specificity (75-100%) and sensitivity (77-98%)63,69,84. Sensitivity and specificity values depend on the allergen and other factors as phenotypes (e.g., oral allergy syndrome versus anaphylaxis), age, and geography-related sensitization patterns63,85.
BAT using single allergen components can potentially improve diagnostic accuracy for some food allergens61,80. There are studies using seed storage proteins (e.g., Ara h 1, Ara h 2, Ara h 3 and Ara h 6 from peanut)86; lipid transfer proteins (e.g., Pru p 3 from peach and Ara h 9 from peanut)80,86; and Bet v 1 homologues (e.g., Ara h 8 from peanut)87. Other potential utilities are related to the identification of the culprit allergen in cases of pollen-food allergy syndrome21,87,88, allergy to red meat89, or food-dependent exercise-induced anaphylaxis66.
Interestingly, BAT can provide information about the severity and the threshold of allergic reactions since patients with more severe reactions show a greater proportion of activated basophils, as observed in studies of peanut and cow's milk-allergic patients84,90,91; and patients reacting to trace amounts of the allergen show a greater basophil sensitivity84,90,92. These data suggest that BAT may be useful to identify high-risk allergic patients who require closer follow-up and more intensified education93. Moreover, it has been reported that BAT can predict food challenge responses70,91,92,94 and thresholds of reactivity90,95 to help determine when food can be safely (re)introduced84. However, these findings are controversial in some studies63,96 and more research are required.
On the other hand, BAT has been used to monitor resolution of food allergy, either naturally or under immunomodulatory treatments, over time, which until now has been only assessed by oral food challenge, with the associated risks and costs84,97,98,99,100,101,102,103,104,105,106,107,108. Moreover, it has also been used to monitor the effect of omalizumab in food allergy as basophil activation decreases during treatment with omalizumab, but it increases after cessation of treatment109.
Inhalant allergy
BAT is rarely beneficial in inhalant allergy as diagnosis can be routinely established by sIgE quantification and ST. However, in cases of local allergic rhinitis (undetectable levels of sIgE and negative STs with positive nasal provocation tests), BAT has allowed diagnosis in 50% of cases110. It has been further reported a correlation between basophil sensitivity and response to nasal/bronchial provocation tests, as well as between asthma severity and efficacy of treatment with omalizumab111,112.
BAT has also been used to monitor allergen immunotherapy for house dust mite and pollens, as basophil sensitivity decreases during immunotherapy, likely due to interference of blocking IgG antibodies113,114,115,116,117.
Hymenopteravenom allergy
Diagnosis of hymenoptera venom allergy is routinely based on ST and serum sIgE. BAT has shown a high sensitivity (85-100%) and specificity (83-100%) and it has been reported to be useful in cases that yield equivocal results or in patients with a suggestive clinical history of venom allergy but undetectable sIgE and negative ST118,119. However, BAT appears not to be predictive of severity for these reactions120,121.
Up to 60% of patients exhibit sIgE to both wasp and bee venom, and the identification of dominant allergen is crucial for adequate immunotherapy treatment. In these cases, BAT has been reported to be useful in the identification of the dominant allergen119,122,123,124. Although sIgE to the major allergens of bee and wasp venoms may reduce the utility of BAT in patients with double positivity to both venoms, it provides useful information mainly in subjects with negative results in sIgE determinations123.
Some studies suggest that BAT may be useful as a predictive biomarker for side effects during the build-up phase of venom immunotherapy as this treatment option has been reported to decrease basophil sensitivity. However, reactivity does not decrease and this BAT utility is nowadays controversial13,120,125,126,127,128,129,130.
Urticaria and angioedema
A subset of chronic urticaria patients have an autoinmune pathophysiology, due to IgE autoantibodies to autoallergens and IgG autoantibodies that target FcεRI or IgE-FcεRI complexes present on the mast cell surface131,132. In clinical practice, diagnosis of this type of chronic urticaria has relied on positive autologous serum ST, which has risk of accidental infection. BAT has been proposed as an in vitro test for diagnosing and monitoring patients with suspected chronic urticaria. Both CD63 and CD203c expression on the surface of basophils has been reported to be increased following stimulation with sera from chronic urticaria patients, showing the detection of active autoantibodies133,134,135,136,137. Recently, it has been reported that patients with positive BAT often experience the most active disease state, assessed by urticaria activity score, and require higher doses of antihistamines together with third-line treatments (cyclosporine A or omalizumab), compared to those with negative BAT138.
Protocol
The protocol performance was conducted according to the Declaration of Helsinki principles and approved by the local Ethics Committee (Comité de Ética para la Investigación Provincial de Málaga, Spain). All subjects were informed orally about the research study and they signed the corresponding informed consent form.
NOTE: The present protocol details the BAT procedure that the authors use daily. However, this is not a standardized method and differences with procedures published by other authors exist. The main protocol modifications are related to the use of IL-3 in the stimulation buffer, incubation time with the stimulus, method for stopping basophil degranulation, and flow cytometry strategies. Moreover, different commercially available kits for BAT include specific protocols recommended by the manufacturer.
1. Sample preparation
- Collect peripheral blood in 9 mL heparinized tubes and maintain the sample at room temperature (RT) in a rotor until it is required for the experimental protocol.
- Label 5 mL cytometer tubes for negative controls (2 tubes), positive controls (2 tubes) and different concentrations of allergen/drug (1 tube per each allergen/drug concentration tested). Place the tubes in a rack where tubes fit perfectly without slipping.
- Prepare stimulation buffer in double distilled water containing 2% (v/v) HEPES, 78 mg/L NaCl, 3.7 mg/L KCl, 7.8 mg/L CaCl2, 3.3 mg/L MgCl2, 1 g/L HSA. Adjust pH to 7.4 and add IL-3 at 2 ng/mL. Usually prepare 100 mL and divide in 2.5 mL aliquots to be frozen at -20 °C.
- Prepare positive controls in PBS-Tween-20 0.05% (v/v) (PBS-T): Positive control 1, N-Formylmethionyl-leucyl-phenylalanine (fMLP) (4 µM), to confirm quality of basophils; Positive control 2, Anti-IgE (0.05 mg/mL) as a IgE-mediated positive control.
- Prepare the allergen/drug in PBS-T at 2x the desired final concentration.
NOTE: Optimal allergen/drug concentrations to be used must be previously determined by using a wide range of concentrations, by dose-response curves, and by cytotoxicity studies following the same protocol steps139.
2. Staining mix preparation
- Add monoclonal antibodies labelled with fluorochrome to the stimulation buffer following manufacturer´s recommended antibody concentration or by previous antibody titration. In this protocol we add 1 µL of each antibody (CCR3-APC and CD203c-PE for basophil identification; CD63-FITC for basophil activation)140 per 20 µL of stimulation buffer.
NOTE: Protect the staining mix preparation from the light. - Add 23 µL of staining mix to each tube.
3. Blood stimulation
- Add 100 µL of PBS-T to tubes 1 and 2 (negative control), 100 µL of fMLP to tube 3, 100 µL of anti-IgE to tube 4, and 100 µL of the different allergen/drug concentrations to the following tubes. Incubate for 10 min at 37 °C in a thermostatic bath with medium agitation to prewarm reagents.
- Gently add 100 µL of blood to each tube to avoid hemolysis. Gently vortex tubes and incubate for 25 min at 37 °C in a thermostatic bath with medium agitation.
- Stop the degranulation, keeping tubes at 4 °C for at least 5 min.
NOTE: The protocol can be paused here at 4 °C for 30-45 min if required141,142,143.
4. Erythrocytes lysing
- Add 2 mL of 1x lysing buffer to each tube to lyse erythrocytes. Vortex each tube and incubate for 5 min at RT.
NOTE: In this step, cells are fixed due to fixative agents (formaldehyde) contained in the buffer. - Centrifuge at 300 x g at 4 °C for 5 min. Decant the supernatant, overturning the rack into a sink. Cells remain at the bottom of the tubes.
- Add 3 mL of PBS-T to each tube to wash cells. Vortex each tube.
- Centrifuge at 300 x g at 4 °C for 5 min. Decant the supernatant, overturning the rack into a sink.
NOTE: Keep samples at 4 °C, protected from the light until flow cytometer acquisition.
5. Flow cytometry acquisition
- Acquire samples by flow cytometer (e.g., BD FACSCalibur Flow Cytometer). Connect the flow cytometer to the computer software and wait for the cytometer to be ready. Load the template and instrument settings (Table 1).
- Start sample acquisition.
- Use the following cytometer strategies for selection of activated basophils139.
- Gate the lymphocytes from the Side Scatter (SSC) – Forward Scatter (FSC) plot.
- Gate the basophils from the lymphocyte population as CCR3+CD203c+ cells. Acquire at least 500 basophils per tube.
- Show a CCR3 – CD63 plot to analyze activation using CD63 as activation marker. Set the CD63 negative threshold to approximately 2.5% using the negative control tubes.
- Acquire all the samples.
Representative Results
BAT performed with allergens or drugs allows investigation of IgE-dependent hypersensitivity reactions. Basophil reactivity should be measured in at least two optimal concentrations in order to obtain the best results34 and activation is visualized by the upregulation of CD63 on the cell surface. In the case of allergens, moreover, to confirm the basophil reactivity, the basophil sensitivity should be analyzed by measuring the reactivity at multiple decreasing allergen concentrations114. This measure allows the determination of the allergen concentration that induces the response of 50% basophils (EC50), which can be expressed as "CD-sens"141. The measurement of the area under the dose curve (AUC) has been recently proposed to assess both basophil reactivity and basophil sensitivity together58.
The flow cytometry strategy for analyzing BAT results is shown in Figure 1 and Figure2 and includes gating lymphocytes from the SSC-FSC plot (step 1), gating basophils from the lymphocyte population as CCR3+CD203c+ cells (step 2), showing a CCR3 – CD63 plot to analyze activation using CD63 as activation marker (step 3). Figures show representative examples of BAT results obtained for drugs (Figure 1) and allergens (Figure 2).
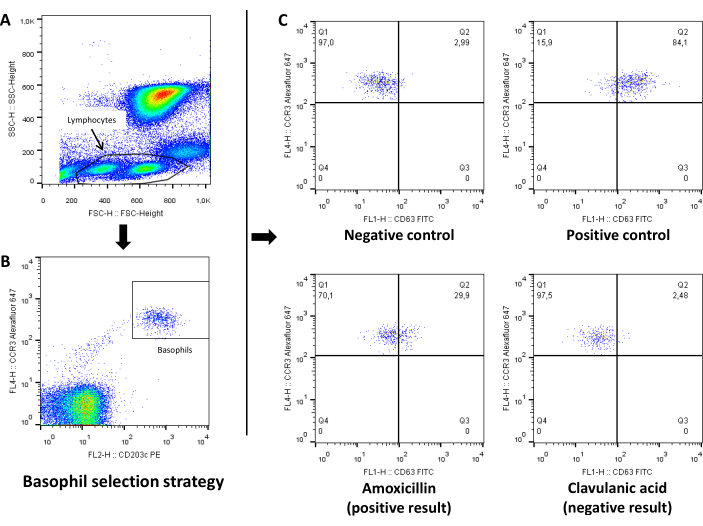
Figure 1: Representative analysis of drug basophil activation test by flow cytometry. (A) SSC-FSC plot to select lymphocyte+basophil population. (B) CCR3-CD203c plot to gate basophils from the lymphocyte population as CCR3+CD203c cells. (C) CCR3-CD63 plot to analyze activation using CD63 as activation marker for negative control, positive control and drug. Values showed in each panel represent percentage of cells. Please click here to view a larger version of this figure.
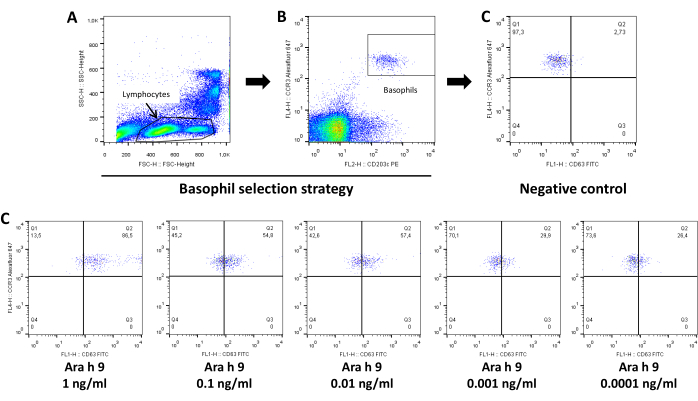
Figure 2: Representative analysis of allergen basophil activation test by flow cytometry. (A) SSC-FSC plot to select lymphocyte+basophil population. (B) CCR3-CD203c plot to gate basophils from the lymphocyte population as CCR3+CD203c+ cells. (C) CCR3-CD63 plot to analyze activation using CD63 as activation marker showing results for negative control and decreasing concentrations of allergen (Ara h 9). Values showed in each panel represent percentage of cells. Please click here to view a larger version of this figure.
| Light Sources (lasers) | 488 nm Coherent SapphireTM air-cooled argon-ion laser; 20 mW; 633-nm JDS UniphaseTM HeNe air-cooled laser; 17 mW |
| Light excitatory wavelength | Blue laser:488 nm; Red laser: 633 nm |
| Light source power at the excitatory wavelength | Blue laser: 20 mW; Red laser: 17 mW |
| Optical filters | SSC: 488/10; FITC: 530/30, PE:585/42, APC: 660/20 |
| Optical detectors | FSC, SSC, FL1-H FITC, FL2-H PE, FL4-H APC |
| Optical detectors tye | air-cooled argon-ion laser |
| Optical paths | BD octagon (488 nm laser line); BD Trigons (633 nm laser line) |
Table 1: Flow cytometer requirements
Discussion
BAT is a complementary in vitro diagnostic test for the evaluation of IgE-mediated allergic reactions that has shown to be useful in the diagnosis of reactions induced by different triggers such as drugs, food, or inhalants, as well as in some forms of chronic urticaria. In general, BAT performance should be considered if i) the allergen/drug produces false positive results in ST; ii) the allergen/drug is not available to be used for ST or sIgE quantification; iii) discordance between clinical history and ST or sIgE determination exists; iv) symptoms suggest that ST may induce a systemic reaction; v) previous to CCT for culprit allergen/drug confirmation10.
Concerning the experimental protocol, there are different important aspects to be considered to get suitable blood samples for the test. Systemic steroids146 and immunosupressors, including oral corticosteroids,147 should be avoided before testing due to decrease of basophil response146 (antihistamines and topical steroids do not affect BAT results)146. The test should not be performed during infection or active chronic inflammatory conditions148. The interval time between the reaction and the test should be no longer than 1 year because of reported negativization of sIgE levels over time42,52,149. The test is performed with fresh whole blood and must be performed no longer than 24 h after blood extraction150,151. Blood has to be collected in heparin-stabilized tubes because basophils do not degranulate if EDTA or acid-citrate dextrose are used as a stabilizer, although it could be used after adding calcium152. On the other hand, the stimulus used in the test should not include any excipients; for this reason, standardized allergen extracts, recombinant or purified allergens, pure active ingredients or intravenous injectable drug preparations are recommended. Moreover, chemical characteristics of the drugs need to be considered. For example, some drugs are unstable in solution and must be freshly prepared before each test, and others are photo labile and must be performed while protecting the assay from the light48. Toxicity and nonspecific activation should be evaluated for each tested allergen/drug and ROC curves with confirmed patients and tolerant controls must be analyzed to determine the cut-off. Finally, the importance of both positive controls should be highlighted in the analysis of BAT. fMLP is a bacterial peptide that induces basophil activation through the G-protein coupled fMLP receptor. For this reason, it is often used as a positive control of non-IgE-mediated activation16. Anti-IgE or alternatively anti-FcεRI are used as positive controls of IgE-mediated basophil activation. No basophil activation in the presence of both non-IgE and IgE-mediated positive controls suggest not enough quality of basophils or mistakes in the experimental protocol. In contrast, basophils activated with fMLP but not with anti-IgE or anti-FcεRI are designated as non-responder basophils, being estimated that 6-17% of the general population is non-responder to stimulation through FcεRI in BAT 63,84,153, although they express normal densities of cells surface IgE. Non-responsiveness could be related to low levels of Syk phosphatase154,155,156 together with increased levels of CD45157. Although studies have shown that non-responder basophils can turn into responders in the presence of IL-3158 in in vitro assays, non-responder basophils can be still detected in BAT and, in these cases, results cannot be considered for evaluation.
Regarding the inclusion of IL-3, a basophil priming cytokine, no general consensus exists. The use of IL-3 has been reported to enhance basophil responsiveness in CD63-based BAT without inducing CD63 upregulation by itself after a short pretreatment7,159,160. However, another study suggests that IL-3 upregulates CD63 expression at baseline161. In contrast, in the case of CD203c-based BAT, studies confirm that IL-3 priming enhances CD203c expression by resting basophils, decreasing differences between unstimulated and stimulated basophils and decreasing BAT sensitivity 159,161.
Different gating strategies can be used to identify basophil population and analyze basophil activation by flow cyotmetry. Basophils are low side scatter cells that can be identified through different selection marker options162,163,164, being a key point that could affect the diagnostic efficiency of BAT165,166. Cell marker selection should be based on specificity to discriminate basophils from other cells populations as well as on cell marker expression on resting and activated cells. The best known and commonly used basophil selection markers are: CD193 (CCR3) (also expressed on mast cells, Th2 lymphocytes162, and eosinophils), CD123 (also expressed on HLA-DR+ plasmacytoid dendritic cells), CD203c (exclusively expressed on basophils and upregulated after basophil activation), and FcεRI (also expressed on pluripotent progenitors of mast cells)139. Based on these cell markers and in combination with SSC, more common selection strategies are SSClowCCR3+, SSClowCCR3+CD203c+ (applied in this protocol), SSClowCD123+HLA-DR−, SSClowCD203c+CD123+HLA-DR−, SSClowFcεRI+HLA-DR− (to exclude antigen presenting cells and monocytes 146,161,162,163), SSClowCD203c+CRTH2+CD3– (to exclude T cells)164, SSClowCD203c+ or SSClowCCR3+CD123+ 10,162,166, SSClowCD123+(CD3–CD14–CD19–CD20–)167,168, and SSClowIgE+169,170, although the latter is not recommended due to limitations in patients with low IgE levels. After accurate selection of basophil population, activation is usually detected through the detection of CD63, located in the membrane of secretory granules16,171 whose expression on the basophil surface is directly correlated with basophil degranulation and histamine release16,172,173. Another option is the analysis of CD203c, although sensitivity is lower due to its upregulation by IL-3159,161, constitutively expressed on resting basophils and upregulated on activated basophils.
Basophil activation is detected by measuring the percentage of CD63 positive cells (CD63-based BAT) or variations in CD203c mean fluorescence intensity (MFI) (CD203c-based BAT) compared to a negative control set as threshold value for each assay. A threshold of 2.5% CD63 positive cells in the negative control (unstimulated cells) is recommended to determine the most accurate BAT results compared to a controlled challenge test. Consideration of positivity depends on the tested stimulus. Basophil activation is considered positive for a stimulus if the percentage of CD63 positive basophils in the presence of the stimulus divided by the percentage of CD63 positive basophils in the negative control is higher than the cut-off calculated by ROC curve analysis of data obtained from confirmed allergic patients and healthy donors.
BAT performance allows to distinguish between IgE-dependent and IgE-independent basophil activation by analyzing the inhibitory effect of wortmannin (WTM)16,174,175, a potent and specific inhibitor of phosphoinositide 3-kinase involved in IgE-mediated basophil activation. The inhibition assay is performed by incubating blood with WTM (1 µM) at 37 °C for 5 min before the incubation with the stimulus. To confirm that BAT inhibition with WTM is correct, inhibition of positive control anti-IgE but not of positive control fMLP must be observed.
Unfortunately, there is no standardization between different laboratories in terms of procedures, concentrations, and markers. Future multicenter studies are required to standardize the method to compare results between centers and to clinically standardize and validate the test.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank Claudia Corazza for her invaluable English language support. This work was supported by Institute of Health ''Carlos III'' (ISCIII) of MINECO (grant cofunded by ERDF: "Una manera de hacer Europa"; Grants Nos. PI20/01715; PI18/00095; PI17/01410; PI17/01318; PI17/01237 and RETIC ARADYAL RD16/0006/0001; Andalusian Regional Ministry of Health (Grant Nos. PI-0127-2020, PIO-0176-2018; PE-0172-2018; PE-0039-2018; PC-0098-2017; PI-0075-2017; PI-0241-2016). ID is a Clinical Investigator (B-0001-2017) and AA holds a Senior Postdoctoral Contract (RH-0099-2020), both supported by Andalusian Regional Ministry of Health (cofunded by ESF: "Andalucía se mueve con Europa").
Materials
| 5 mL Round Bottom Polystyrene Test Tube, without Cap, Nonsterile | Corning | 352008 | |
| APC anti-human CD193 (CCR3) Antibody | BioLegend | 310708 | |
| BD FACSCalibur Flow Cytometer | BD Biosciences | ||
| Calcium chloride | Sigma-Aldrich | C1016 | |
| FITC anti-human CD63 Antibody | BioLegend | 353006 | |
| HEPES (1 M) | Thermo-Fisher | 15630106 | |
| Lysing Solution 10x concentrated | BD Biosciences | 349202 | |
| Magnesium chloride | Sigma-Aldrich | M8266 | |
| N-Formyl-Met-Leu-Phe | Sigma-Aldrich | F3506 | |
| PE anti-human CD203c (E-NPP3) Antibody | BioLegend | 324606 | |
| Potassium chloride | Sigma-Aldrich | P9541 | |
| Purified Mouse Anti-Human IgE | BD Biosciences | 555857 | |
| Recombinant Human IL-3 | R&D Systems | 203-IL | |
| Sheath Fluid | BD Biosciences | 342003 | |
| Sodium chloride | Sigma-Aldrich | S3014 | |
| TUBE 9 mL LH Lithium Heparin | Greiner Bio-One | 455084 | |
| Tween 20 | Sigma-Aldrich | P1379 |
References
- Mayorga, C., et al. In vitro tests for drug hypersensitivity reactions: an ENDA/EAACI Drug Allergy Interest Group position paper. Allergy. 71 (8), 1103-1134 (2016).
- Romano, A., et al. Towards a more precise diagnosis of hypersensitivity to beta-lactams – an EAACI position paper. Allergy. 75 (6), 1300-1315 (2020).
- Garvey, L. H., et al. An EAACI position paper on the investigation of perioperative immediate hypersensitivity reactions. Allergy. 74 (10), 1872-1884 (2019).
- Gomes, E. R., et al. Drug hypersensitivity in children: report from the pediatric task force of the EAACI Drug Allergy Interest Group. Allergy. 71 (2), 149-161 (2016).
- Ansotegui, I. J., et al. IgE allergy diagnostics and other relevant tests in allergy, a World Allergy Organization position paper. World Allergy Organization Journal. 13 (2), 100080 (2020).
- Jeebhay, M. F., et al. Food processing and occupational respiratory allergy- An EAACI position paper. Allergy. 74 (10), 1852-1871 (2019).
- Ebo, D. G., et al. Flow-assisted allergy diagnosis: current applications and future perspectives. Allergy. 61 (9), 1028-1039 (2006).
- Bochner, B. S. Systemic activation of basophils and eosinophils: markers and consequences. Journal of Allergy and Clinical Immunology. 106 (5), 292-302 (2000).
- Ghannadan, M., et al. Detection of novel CD antigens on the surface of human mast cells and basophils. International Archives of Allergy and Immunology. 127 (4), 299-307 (2002).
- Hoffmann, H. J., et al. The clinical utility of basophil activation testing in diagnosis and monitoring of allergic disease. Allergy. 70 (11), 1393-1405 (2015).
- Sainte-Laudy, J., Sabbah, A., Drouet, M., Lauret, M. G., Loiry, M. Diagnosis of venom allergy by flow cytometry. Correlation with clinical history, skin tests, specific IgE, histamine and leukotriene C4 release. Clinical & Experimental Allergy. 30 (8), 1166-1171 (2000).
- Sturm, G. J., et al. The CD63 basophil activation test in Hymenoptera venom allergy: a prospective study. Allergy. 59 (10), 1110-1117 (2004).
- Erdmann, S. M., et al. The basophil activation test in wasp venom allergy: sensitivity, specificity and monitoring specific immunotherapy. Allergy. 59 (10), 1102-1109 (2004).
- De Weck, A. L., et al. Diagnostic tests based on human basophils: more potentials and perspectives than pitfalls. International Archives of Allergy and Immunology. 146 (3), 177-189 (2008).
- Buhring, H. J., Streble, A., Valent, P. The basophil-specific ectoenzyme E-NPP3 (CD203c) as a marker for cell activation and allergy diagnosis. International Archives of Allergy and Immunology. 133 (4), 317-329 (2004).
- Knol, E. F., Mul, F. P., Jansen, H., Calafat, J., Roos, D. Monitoring human basophil activation via CD63 monoclonal antibody 435. Journal of Allergy and Clinical Immunology. 88 (3), 328-338 (1991).
- Fureder, W., Agis, H., Sperr, W. R., Lechner, K., Valent, P. The surface membrane antigen phenotype of human blood basophils. Allergy. 49 (10), 861-865 (1994).
- Sanz, M. L., et al. Allergen-induced basophil activation: CD63 cell expression detected by flow cytometry in patients allergic to Dermatophagoides pteronyssinus and Lolium perenne. Clinical & Experimental Allergy. 31 (7), 1007-1013 (2001).
- Monneret, G., et al. Monitoring of basophil activation using CD63 and CCR3 in allergy to muscle relaxant drugs. Clin Immunol. 102 (2), 192-199 (2002).
- Sanz, M. L., et al. Flow cytometric basophil activation test by detection of CD63 expression in patients with immediate-type reactions to betalactam antibiotics. Clinical & Experimental Allergy. 32 (2), 277-286 (2002).
- Ebo, D. G., et al. Flow cytometric analysis of in vitro activated basophils, specific IgE and skin tests in the diagnosis of pollen-associated food allergy. Cytometry Part B: Clinical Cytometry. 64 (1), 28-33 (2005).
- Sudheer, P. S., Hall, J. E., Read, G. F., Rowbottom, A. W., Williams, P. E. Flow cytometric investigation of peri-anaesthetic anaphylaxis using CD63 and CD203c. Anaesthesia. 60 (3), 251-256 (2005).
- Binder, M., Fierlbeck, G., King, T., Valent, P., Buhring, H. J. Individual hymenoptera venom compounds induce upregulation of the basophil activation marker ectonucleotide pyrophosphatase/phosphodiesterase 3 (CD203c) in sensitized patients. International Archives of Allergy and Immunology. 129 (2), 160-168 (2002).
- Hauswirth, A. W., et al. Recombinant allergens promote expression of CD203c on basophils in sensitized individuals. Journal of Allergy and Clinical Immunology. 110 (1), 102-109 (2002).
- Boumiza, R., et al. Marked improvement of the basophil activation test by detecting CD203c instead of CD63. Clinical & Experimental Allergy. 33 (2), 259-265 (2003).
- Macglashan, D. Expression of CD203c and CD63 in human basophils: relationship to differential regulation of piecemeal and anaphylactic degranulation processes. Clinical & Experimental Allergy. 40 (9), 1365-1377 (2010).
- Mayorga, C., Dona, I., Perez-Inestrosa, E., Fernandez, T. D., Torres, M. J. The Value of In Vitro Tests to DiminishDrug Challenges. International Journal of Molecular Sciences. 18 (6), (2017).
- Brockow, K., et al. General considerations for skin test procedures in the diagnosis of drug hypersensitivity. Allergy. 57 (1), 45-51 (2002).
- Brockow, K., et al. Skin test concentrations for systemically administered drugs — an ENDA/EAACI Drug Allergy Interest Group position paper. Allergy. 68 (6), 702-712 (2013).
- Torres, M. J., et al. Approach to the diagnosis of drug hypersensitivity reactions: similarities and differences between Europe and North America. Clinical and Translational Allergy. 7, 7 (2017).
- Mayorga, C., et al. In vitro tests for drug hypersensitivity reactions: an ENDA/EAACI Drug Allergy Interest Group position paper. Allergy. 71 (8), 1103-1134 (2016).
- Mayorga, C., et al. Controversies in drug allergy: In vitro testing. Journal of Allergy and Clinical Immunology. 143 (1), 56-65 (2019).
- Aberer, W., et al. Drug provocation testing in the diagnosis of drug hypersensitivity reactions: general considerations. Allergy. 58 (9), 854-863 (2003).
- De Week, A. L., et al. Diagnosis of immediate-type beta-lactam allergy in vitro by flow-cytometric basophil activation test and sulfidoleukotriene production: a multicenter study. Journal of Investigational Allergology and Clinical Immunology. 19 (2), 91-109 (2009).
- Abuaf, N., et al. Comparison of two basophil activation markers CD63 and CD203c in the diagnosis of amoxicillin allergy. Clinical & Experimental Allergy. 38 (6), 921-928 (2008).
- Torres, M. J., et al. The diagnostic interpretation of basophil activation test in immediate allergic reactions to betalactams. Clinical & Experimental Allergy. 34 (11), 1768-1775 (2004).
- Torres, M. J., et al. Clavulanic acid can be the component in amoxicillin-clavulanic acid responsible for immediate hypersensitivity reactions. Journal of Allergy and Clinical Immunology. 125 (2), 502-505 (2010).
- Eberlein, B., et al. A new basophil activation test using CD63 and CCR3 in allergy to antibiotics. Clinical & Experimental Allergy. 40 (3), 411-418 (2010).
- Sanchez-Morillas, L., et al. Selective allergic reactions to clavulanic acid: a report of 9 cases. Journal of Allergy and Clinical Immunology. 126 (1), 177-179 (2010).
- Leysen, J., et al. Allergy to rocuronium: from clinical suspicion to correct diagnosis. Allergy. 66 (8), 1014-1019 (2011).
- Ebo, D. G., et al. Flow-assisted diagnostic management of anaphylaxis from rocuronium bromide. Allergy. 61 (8), 935-939 (2006).
- Kvedariene, V., et al. Diagnosis of neuromuscular blocking agent hypersensitivity reactions using cytofluorimetric analysis of basophils. Allergy. 61 (3), 311-315 (2006).
- Hagau, N., Gherman-Ionica, N., Sfichi, M., Petrisor, C. Threshold for basophil activation test positivity in neuromuscular blocking agents hypersensitivity reactions. Allergy Asthma Clin Immunol. 9 (1), 42 (2013).
- Uyttebroek, A. P., et al. Flowcytometric diagnosis of atracurium-induced anaphylaxis. Allergy. 69 (10), 1324-1332 (2014).
- Abuaf, N., et al. Validation of a flow cytometric assay detecting in vitro basophil activation for the diagnosis of muscle relaxant allergy. Journal of Allergy and Clinical Immunology. 104 (2), 411-418 (1999).
- Aranda, A., et al. In vitro evaluation of IgE-mediated hypersensitivity reactions to quinolones. Allergy. 66 (2), 247-254 (2011).
- Fernandez, T. D., et al. Hypersensitivity to fluoroquinolones: The expression of basophil activation markers depends on the clinical entity and the culprit fluoroquinolone. Medicine (Baltimore). 95 (23), 3679 (2016).
- Mayorga, C., et al. Fluoroquinolone photodegradation influences specific basophil activation. International Archives of Allergy and Immunology. 160 (4), 377-382 (2013).
- Rouzaire, P., et al. Negativity of the basophil activation test in quinolone hypersensitivity: a breakthrough for provocation test decision-making. International Archives of Allergy and Immunology. 157 (3), 299-302 (2012).
- Hagau, N., Longrois, D., Petrisor, C. Threshold for positivity and optimal dipyrone concentration in flow cytometry-assisted basophil activation test. Allergy, Asthma & Immunology Research. 5 (6), 383-388 (2013).
- Gamboa, P. M., et al. Use of CD63 expression as a marker of in vitro basophil activation and leukotriene determination in metamizol allergic patients. Allergy. 58 (4), 312-317 (2003).
- Gomez, E., et al. Immunoglobulin E-mediated immediate allergic reactions to dipyrone: value of basophil activation test in the identification of patients. Clinical & Experimental Allergy. 39 (8), 1217-1224 (2009).
- Pinnobphun, P., Buranapraditkun, S., Kampitak, T., Hirankarn, N., Klaewsongkram, J. The diagnostic value of basophil activation test in patients with an immediate hypersensitivity reaction to radiocontrast media. Annals of Allergy, Asthma & Immunology. 106 (5), 387-393 (2011).
- Salas, M., et al. Diagnosis of immediate hypersensitivity reactions to radiocontrast media. Allergy. 68 (9), 1203-1206 (2013).
- Chirumbolo, S. Basophil activation test (BAT) in the diagnosis of immediate hypersensitivity reactions to radiocontrast media. Allergy. 68 (12), 1627-1628 (2013).
- Dona, I., et al. Hypersensitivity Reactions to Multiple Iodinated Contrast Media. Frontiers in Pharmacology. 11, 575437 (2020).
- Giavina-Bianchi, P., Galvao, V. R., Picard, M., Caiado, J., Castells, M. C. Basophil Activation Test is a Relevant Biomarker of the Outcome of Rapid Desensitization in Platinum Compounds-Allergy. Journal of Allergy and Clinical Immunology Practice. 5 (3), 728-736 (2017).
- Iwamoto, T., et al. Evaluation of basophil CD203c as a predictor of carboplatin-related hypersensitivity reaction in patients with gynecologic cancer. Biological and Pharmaceutical Bulletin. 35 (9), 1487-1495 (2012).
- Iwamoto, T., et al. Carboplatin-induced severe hypersensitivity reaction: role of IgE-dependent basophil activation and FcepsilonRI. Cancer Science. 105 (11), 1472-1479 (2014).
- Muraro, A., et al. EAACI food allergy and anaphylaxis guidelines: diagnosis and management of food allergy. Allergy. 69 (8), 1008-1025 (2014).
- Sato, S., et al. Basophil activation marker CD203c is useful in the diagnosis of hen’s egg and cow’s milk allergies in children. International Archives of Allergy and Immunology. 152, 54-61 (2010).
- Ciepiela, O., et al. Basophil activation test based on the expression of CD203c in the diagnostics of cow milk allergy in children. European Journal of Medical Research. 15, 21-26 (2010).
- Ocmant, A., et al. Basophil activation tests for the diagnosis of food allergy in children. Clinical & Experimental Allergy. 39 (8), 1234-1245 (2009).
- Carroccio, A., et al. A comparison between two different in vitro basophil activation tests for gluten- and cow’s milk protein sensitivity in irritable bowel syndrome (IBS)-like patients. Clinical Chemistry and Laboratory Medicine. 51 (6), 1257-1263 (2013).
- Tokuda, R., et al. Antigen-induced expression of CD203c on basophils predicts IgE-mediated wheat allergy. Allergology International. 58 (2), 193-199 (2009).
- Chinuki, Y., et al. CD203c expression-based basophil activation test for diagnosis of wheat-dependent exercise-induced anaphylaxis. Journal of Allergy and Clinical Immunology. 129 (5), 1404-1406 (2012).
- Carroccio, A., et al. Non-celiac wheat sensitivity diagnosed by double-blind placebo-controlled challenge: exploring a new clinical entity. Am J Gastroenterol. 107 (12), 1898-1906 (2012).
- Carroccio, A., et al. A cytologic assay for diagnosis of food hypersensitivity in patients with irritable bowel syndrome. Clin Gastroenterol Hepatol. 8 (3), 254-260 (2010).
- Santos, A. F., et al. Basophil activation test discriminates between allergy and tolerance in peanut-sensitized children. Journal of Allergy and Clinical Immunology. 134 (3), 645-652 (2014).
- Glaumann, S., et al. Basophil allergen threshold sensitivity, CD-sens, IgE-sensitization and DBPCFC in peanut-sensitized children. Allergy. 67 (2), 242-247 (2012).
- Javaloyes, G., et al. Performance of different in vitro techniques in the molecular diagnosis of peanut allergy. Journal of Investigational Allergology and Clinical Immunology. 22 (7), 508-513 (2012).
- Glaumann, S., Nopp, A., Johansson, S. G., Borres, M. P., Nilsson, C. Oral peanut challenge identifies an allergy but the peanut allergen threshold sensitivity is not reproducible. PLoS One. 8 (1), 53465 (2013).
- Elizur, A., et al. NUT Co Reactivity – ACquiring Knowledge for Elimination Recommendations (NUT CRACKER) study. Allergy. 73 (3), 593-601 (2018).
- Cucu, T., De Meulenaer, B., Bridts, C., Devreese, B., Ebo, D. Impact of thermal processing and the Maillard reaction on the basophil activation of hazelnut allergic patients. Food Chem Toxicol. 50 (5), 1722-1728 (2012).
- Worm, M., et al. Impact of native, heat-processed and encapsulated hazelnuts on the allergic response in hazelnut-allergic patients. Clinical & Experimental Allergy. 39 (1), 159-166 (2009).
- Brandstrom, J., et al. Basophil allergen threshold sensitivity and component-resolved diagnostics improve hazelnut allergy diagnosis. Clinical & Experimental Allergy. 45 (9), 1412-1418 (2015).
- Lotzsch, B., Dolle, S., Vieths, S., Worm, M. Exploratory analysis of CD63 and CD203c expression in basophils from hazelnut sensitized and allergic individuals. Clinical and Translational Allergy. 6, 45 (2016).
- Ebo, D. G., Bridts, C. H., Hagendorens, M. M., De Clerck, L. S., Stevens, W. J. Scampi allergy: from fancy name-giving to correct diagnosis. Journal of Investigational Allergology and Clinical Immunology. 18 (3), 228-230 (2008).
- Gamboa, P. M., et al. Component-resolved in vitro diagnosis in peach-allergic patients. Journal of Investigational Allergology and Clinical Immunology. 19 (1), 13-20 (2009).
- Gamboa, P. M., et al. Two different profiles of peach allergy in the north of Spain. Allergy. 62 (4), 408-414 (2007).
- Diaz-Perales, A., et al. Recombinant Pru p 3 and natural Pru p 3, a major peach allergen, show equivalent immunologic reactivity: a new tool for the diagnosis of fruit allergy. Journal of Allergy and Clinical Immunology. 111 (3), 628-633 (2003).
- Erdmann, S. M., Heussen, N., Moll-Slodowy, S., Merk, H. F., Sachs, B. CD63 expression on basophils as a tool for the diagnosis of pollen-associated food allergy: sensitivity and specificity. Clinical & Experimental Allergy. 33 (5), 607-614 (2003).
- Erdmann, S. M., et al. In vitro analysis of birch-pollen-associated food allergy by use of recombinant allergens in the basophil activation test. International Archives of Allergy and Immunology. 136 (3), 230-238 (2005).
- Rubio, A., et al. Benefit of the basophil activation test in deciding when to reintroduce cow’s milk in allergic children. Allergy. 66 (1), 92-100 (2011).
- Decuyper, I. i., et al. Performance of basophil activation test and specific IgG4 as diagnostic tools in nonspecific lipid transfer protein allergy: Antwerp-Barcelona comparison. Allergy. 75 (3), 616-624 (2020).
- Mayorga, C., et al. Basophil response to peanut allergens in Mediterranean peanut-allergic patients. Allergy. 69 (7), 964-968 (2014).
- Glaumann, S., et al. Evaluation of basophil allergen threshold sensitivity (CD-sens) to peanut and Ara h 8 in children IgE-sensitized to Ara h 8. Clinical and Molecular Allergy. 13 (1), 5 (2015).
- Wolbing, F., et al. The clinical relevance of birch pollen profilin cross-reactivity in sensitized patients. Allergy. 72 (4), 562-569 (2017).
- Commins, S. P., et al. Delayed clinical and ex vivo response to mammalian meat in patients with IgE to galactose-alpha-1,3-galactose. Journal of Allergy and Clinical Immunology. 134 (1), 108-115 (2014).
- Santos, A. F., et al. Distinct parameters of the basophil activation test reflect the severity and threshold of allergic reactions to peanut. Journal of Allergy and Clinical Immunology. 135 (1), 179-186 (2015).
- Song, Y., et al. Correlations between basophil activation, allergen-specific IgE with outcome and severity of oral food challenges. Annals of Allergy, Asthma & Immunology. 114 (4), 319-326 (2015).
- Chinthrajah, R. S., et al. Development of a tool predicting severity of allergic reaction during peanut challenge. Annals of Allergy, Asthma & Immunology. 121 (1), 69-76 (2018).
- Santos, A. F., Shreffler, W. G. Road map for the clinical application of the basophil activation test in food allergy. Clinical & Experimental Allergy. 47 (9), 1115-1124 (2017).
- Santos, A. F., et al. Biomarkers of severity and threshold of allergic reactions during oral peanut challenges. Journal of Allergy and Clinical Immunology. 146 (2), 344-355 (2020).
- Reier-Nilsen, T., et al. Predicting reactivity threshold in children with anaphylaxis to peanut. Clinical & Experimental Allergy. 48 (4), 415-423 (2018).
- Chapuis, A., et al. h 2 basophil activation test does not predict clinical reactivity to peanut. Journal of Allergy and Clinical Immunology Practice. 6 (5), 1772-1774 (2018).
- Patil, S. U., et al. Early decrease in basophil sensitivity to Ara h 2 precedes sustained unresponsiveness after peanut oral immunotherapy. Journal of Allergy and Clinical Immunology. 144 (5), 1310-1319 (2019).
- Chinthrajah, R. S., et al. Sustained outcomes in oral immunotherapy for peanut allergy (POISED study): a large, randomised, double-blind, placebo-controlled, phase 2 study. Lancet. 394 (10207), 1437-1449 (2019).
- Kim, E. H., et al. Long-term sublingual immunotherapy for peanut allergy in children: Clinical and immunologic evidence of desensitization. Journal of Allergy and Clinical Immunology. 144 (5), 1320-1326 (2019).
- Tsai, M., Mukai, K., Chinthrajah, R. S., Nadeau, K. C., Galli, S. J. Sustained successful peanut oral immunotherapy associated with low basophil activation and peanut-specific IgE. Journal of Allergy and Clinical Immunology. 145 (3), 885-896 (2020).
- Nachshon, L., et al. Efficacy and Safety of Sesame Oral Immunotherapy-A Real-World, Single-Center Study. Journal of Allergy and Clinical Immunology Practice. 7 (8), 2775-2781 (2019).
- Goldberg, M. R., et al. Efficacy of baked milk oral immunotherapy in baked milk-reactive allergic patients. Journal of Allergy and Clinical Immunology. 136 (6), 1601-1606 (2015).
- Keet, C. A., et al. The safety and efficacy of sublingual and oral immunotherapy for milk allergy. Journal of Allergy and Clinical Immunology. 129 (2), 448-455 (2012).
- Matsui, T., et al. Changes in passively-sensitized basophil activation to alphaS1-casein after oral immunotherapy. Immunity, Inflammation and Disease. 8 (2), 188-197 (2020).
- Giavi, S., et al. Oral immunotherapy with low allergenic hydrolysed egg in egg allergic children. Allergy. 71 (11), 1575-1584 (2016).
- Jones, S. M., et al. Clinical efficacy and immune regulation with peanut oral immunotherapy. Journal of Allergy and Clinical Immunology. 124 (2), 292-300 (2009).
- Burks, A. W., et al. Oral immunotherapy for treatment of egg allergy in children. New England Journal of Medicine. 367 (3), 233-243 (2012).
- Elizur, A., et al. Clinical and laboratory 2-year outcome of oral immunotherapy in patients with cow’s milk allergy. Allergy. 71 (2), 275-278 (2016).
- Gernez, Y., et al. Basophil CD203c levels are increased at baseline and can be used to monitor omalizumab treatment in subjects with nut allergy. International Archives of Allergy and Immunology. 154 (4), 318-327 (2011).
- Gomez, E., et al. Role of the basophil activation test in the diagnosis of local allergic rhinitis. Journal of Allergy and Clinical Immunology. 132 (4), 975-976 (2013).
- Nopp, A., et al. Basophil allergen threshold sensitivity: a useful approach to anti-IgE treatment efficacy evaluation. Allergy. 61 (3), 298-302 (2006).
- Dahlen, B., et al. Basophil allergen threshold sensitivity, CD-sens, is a measure of allergen sensitivity in asthma. Clinical & Experimental Allergy. 41 (8), 1091-1097 (2011).
- Lalek, N., Kosnik, M., Silar, M., Korosec, P. Immunoglobulin G-dependent changes in basophil allergen threshold sensitivity during birch pollen immunotherapy. Clinical & Experimental Allergy. 40 (8), 1186-1193 (2010).
- Schmid, J. M., Wurtzen, P. A., Dahl, R., Hoffmann, H. J. Early improvement in basophil sensitivity predicts symptom relief with grass pollen immunotherapy. Journal of Allergy and Clinical Immunology. 134 (3), 741-744 (2014).
- Sharif, H., et al. Immunologic mechanisms of a short-course of Lolium perenne peptide immunotherapy: A randomized, double-blind, placebo-controlled trial. Journal of Allergy and Clinical Immunology. 144 (3), 738-749 (2019).
- Kim, S. H., et al. Changes in basophil activation during immunotherapy with house dust mite and mugwort in patients with allergic rhinitis. Asia Pacific Allergy. 8 (1), 6 (2018).
- Feng, M., et al. Allergen Immunotherapy-Induced Immunoglobulin G4 Reduces Basophil Activation in House Dust Mite-Allergic Asthma Patients. Frontiers in Cell and Developmental Biology. 8, 30 (2020).
- Korosec, P., et al. Clinical routine utility of basophil activation testing for diagnosis of hymenoptera-allergic patients with emphasis on individuals with negative venom-specific IgE antibodies. International Archives of Allergy and Immunology. 161 (4), 363-368 (2013).
- Ebo, D. G., Hagendorens, M. M., Bridts, C. H., De Clerck, L. S., Stevens, W. J. Hymenoptera venom allergy: taking the sting out of difficult cases. Journal of Investigational Allergology and Clinical Immunology. 17 (6), 357-360 (2007).
- Ebo, D. G., et al. Flow-assisted quantification of in vitro activated basophils in the diagnosis of wasp venom allergy and follow-up of wasp venom immunotherapy. Cytometry Part B: Clinical Cytometry. 72 (3), 196-203 (2007).
- Ott, H., Tenbrock, K., Baron, J., Merk, H., Lehmann, S. Basophil activation test for the diagnosis of hymenoptera venom allergy in childhood: a pilot study. Klin Padiatr. 223 (1), 27-32 (2011).
- Eberlein-Konig, B., Rakoski, J., Behrendt, H., Ring, J. Use of CD63 expression as marker of in vitro basophil activation in identifying the culprit in insect venom allergy. Journal of Investigational Allergology and Clinical Immunology. 14 (1), 10-16 (2004).
- Eberlein, B., Krischan, L., Darsow, U., Ollert, M., Ring, J. Double positivity to bee and wasp venom: improved diagnostic procedure by recombinant allergen-based IgE testing and basophil activation test including data about cross-reactive carbohydrate determinants. Journal of Allergy and Clinical Immunology. 130 (1), 155-161 (2012).
- Sturm, G. J., et al. Inconsistent results of diagnostic tools hamper the differentiation between bee and vespid venom allergy. PLoS One. 6 (6), 20842 (2011).
- Zitnik, S. E., et al. Monitoring honeybee venom immunotherapy in children with the basophil activation test. Pediatric Allergy and Immunology. 23 (2), 166-172 (2012).
- Kosnik, M., Silar, M., Bajrovic, N., Music, E., Korosec, P. High sensitivity of basophils predicts side-effects in venom immunotherapy. Allergy. 60 (11), 1401-1406 (2005).
- Celesnik, N., et al. Short-term venom immunotherapy induces desensitization of FcepsilonRI-mediated basophil response. Allergy. 67 (12), 1594-1600 (2012).
- Nullens, S., et al. Basophilic histamine content and release during venom immunotherapy: insights by flow cytometry. Cytometry Part B: Clinical Cytometry. 84 (3), 173-178 (2013).
- Bidad, K., Nawijn, M. C., Van Oosterhout, A. J., Van Der Heide, S., Elberink, J. N. Basophil activation test in the diagnosis and monitoring of mastocytosis patients with wasp venom allergy on immunotherapy. Cytometry Part B: Clinical Cytometry. 86 (3), 183-190 (2014).
- Eberlein-Konig, B., Schmidt-Leidescher, C., Behrendt, H., Ring, J. Predicting side-effects in venom immunotherapy by basophil activation. Allergy. 61 (7), 897 (2006).
- Kikuchi, Y., Kaplan, A. P. Mechanisms of autoimmune activation of basophils in chronic urticaria. Journal of Allergy and Clinical Immunology. 107 (6), 1056-1062 (2001).
- Huston, D. P., Sabato, V. Decoding the Enigma of Urticaria and Angioedema. Journal of Allergy and Clinical Immunology Practice. 6 (4), 1171-1175 (2018).
- Netchiporouk, E., et al. Positive CD63 Basophil Activation Tests Are Common in Children with Chronic Spontaneous Urticaria and Linked to High Disease Activity. International Archives of Allergy and Immunology. 171 (2), 81-88 (2016).
- Irinyi, B., et al. Extended diagnostic value of autologous serum skin test and basophil CD63 expression assay in chronic urticaria. British Journal of Dermatology. 168 (3), 656-658 (2013).
- Chen, Q., et al. Basophil CD63 expression in chronic spontaneous urticaria: correlation with allergic sensitization, serum autoreactivity and basophil reactivity. Journal of the European Academy of Dermatology and Venereology. 31 (3), 463-468 (2017).
- Wedi, B., Novacovic, V., Koerner, M., Kapp, A. Chronic urticaria serum induces histamine release, leukotriene production, and basophil CD63 surface expression–inhibitory effects ofanti-inflammatory drugs. Journal of Allergy and Clinical Immunology. 105 (3), 552-560 (2000).
- Yasnowsky, K. M., et al. Chronic urticaria sera increase basophil CD203c expression. Journal of Allergy and Clinical Immunology. 117 (6), 1430-1434 (2006).
- Curto-Barredo, L., et al. Basophil Activation Test identifies the patients with Chronic Spontaneous Urticaria suffering the most active disease. Immunity, Inflammation and Disease. 4 (4), 441-445 (2016).
- Santos, A. F., Alpan, O., Hoffmann, H. J. Basophil activation test: Mechanisms and considerations for use in clinical trials and clinical practice. Allergy. , (2021).
- Boumiza, R., Debard, A. L., Monneret, G. The basophil activation test by flow cytometry: recent developments in clinical studies, standardization and emerging perspectives. Clinical and Molecular Allergy. 3, 9 (2005).
- Aljadi, Z., et al. Activation of basophils is a new and sensitive marker of biocompatibility in hemodialysis. Artif Organs. 38 (11), 945-953 (2014).
- Rasmussen, P., Spillner, E., Hoffmann, H. J. Inhibiting phosphatase SHIP-1 enhances suboptimal IgE-mediated activation of human blood basophils but inhibits IgE-mediated activation of cultured human mast cells. Immunology Letters. 210, 40-46 (2019).
- Mueller-Wirth, N., et al. IgE-mediated chlorhexidine allergy-Cross-reactivity with other biguanide disinfectants. Allergy. 75 (12), 3237-3247 (2020).
- Johansson, S. G., et al. Passive IgE-sensitization by blood transfusion. Allergy. 60 (9), 1192-1199 (2005).
- Ariza, A., et al. Basophil activation after nonsteroidal anti-inflammatory drugs stimulation in patients with immediate hypersensitivity reactions to these drugs. Cytometry A. 85 (5), 400-407 (2014).
- Sturm, G. J., et al. The basophil activation test in the diagnosis of allergy: technical issues and critical factors. Allergy. 64 (9), 1319-1326 (2009).
- Iqbal, K., Bhargava, K., Skov, P. S., Falkencrone, S., Grattan, C. E. A positive serum basophil histamine release assay is a marker for ciclosporin-responsiveness in patients with chronic spontaneous urticaria. Clinical and Translational Allergy. 2 (1), 19 (2012).
- Korosec, P., et al. high-affinity IgE receptors, and CCL2 in human anaphylaxis. Journal of Allergy and Clinical Immunology. 140 (3), 750-758 (2017).
- Fernandez, T. D., et al. Negativization rates of IgE radioimmunoassay and basophil activation test in immediate reactions to penicillins. Allergy. 64 (2), 242-248 (2009).
- Kwok, M., Lack, G., Santos, A. F. Improved standardisation of the whole blood basophil activation test to peanut. Clinical and Translational Allergy. 8 (26), 15-16 (2017).
- Mukai, K., et al. Assessing basophil activation by using flow cytometry and mass cytometry in blood stored 24 hours before analysis. Journal of Allergy and Clinical Immunology. 139 (3), 889-899 (2017).
- Sousa, N., Martinez-Aranguren, R., Fernandez-Benitez, M., Ribeiro, F., Sanz, M. L. Comparison of basophil activation test results in blood preserved in acid citrate dextrose and EDTA. Journal of Investigational Allergology and Clinical Immunology. 20 (6), 535-536 (2010).
- Knol, E. F., Koenderman, L., Mul, F. P., Verhoeven, A. J., Roos, D. Differential activation of human basophils by anti-IgE and formyl-methionyl-leucyl-phenylalanine. Indications for protein kinase C-dependent and -independent activation pathways. European Journal of Immunology. 21 (4), 881-885 (1991).
- Macglashan, D. W. Basophil activation testing. Journal of Allergy and Clinical Immunology. 132 (4), 777-787 (2013).
- Macglashan, D., Moore, G., Muchhal, U. Regulation of IgE-mediated signalling in human basophils by CD32b and its role in Syk down-regulation: basic mechanisms in allergic disease. Clinical & Experimental Allergy. 44 (5), 713-723 (2014).
- Macglashan, D. Subthreshold desensitization of human basophils re-capitulates the loss of Syk and FcepsilonRI expression characterized by other methods of desensitization. Clinical & Experimental Allergy. 42 (7), 1060-1070 (2012).
- Grochowy, G., Hermiston, M. L., Kuhny, M., Weiss, A., Huber, M. Requirement for CD45 in fine-tuning mast cell responses mediated by different ligand-receptor systems. Cell Signaling. 21 (8), 1277-1286 (2009).
- Schroeder, J. T., Chichester, K. L., Bieneman, A. P. Human basophils secrete IL-3: evidence of autocrine priming for phenotypic and functional responses in allergic disease. Journal of Immunology. 182 (4), 2432-2438 (2009).
- Ocmant, A., et al. Flow cytometry for basophil activation markers: the measurement of CD203c up-regulation is as reliable as CD63 expression in the diagnosis of cat allergy. Journal of Immunology Methods. 320 (1-2), 40-48 (2007).
- Gentinetta, T., et al. Individual IL-3 priming is crucial for consistent in vitro activation of donor basophils in patients with chronic urticaria. Journal of Allergy and Clinical Immunology. 128 (6), 1227-1234 (2011).
- Sturm, E. M., et al. CD203c-based basophil activation test in allergy diagnosis: characteristics and differences to CD63 upregulation. Cytometry Part B: Clinical Cytometry. 78 (5), 308-318 (2010).
- Hausmann, O. V., et al. Robust expression of CCR3 as a single basophil selection marker in flow cytometry. Allergy. 66 (1), 85-91 (2011).
- Nucera, E., et al. Utility of Basophil Activation Test for monitoring the acquisition of clinical tolerance after oral desensitization to cow’s milk: Pilot study. United European Gastroenterol Journal. 3 (3), 272-276 (2015).
- Imoto, Y., et al. Peripheral basophil reactivity, CD203c expression by Cryj1 stimulation, is useful for diagnosing seasonal allergic rhinitis by Japanese cedar pollen. Immunity, Inflammation and Disease. 3 (3), 300-308 (2015).
- Konstantinou, G. N., et al. EAACI taskforce position paper: evidence for autoimmune urticaria and proposal for defining diagnostic criteria. Allergy. 68 (1), 27-36 (2013).
- Santos, A. F., Becares, N., Stephens, A., Turcanu, V., Lack, G. The expression of CD123 can decrease with basophil activation: implications for the gating strategy of the basophil activation test. Clinical and Translational Allergy. 6, 11 (2016).
- Dijkstra, D., et al. Identification and quantification of basophils in the airways of asthmatics following segmental allergen challenge. Cytometry A. 85 (7), 580-587 (2014).
- Dijkstra, D., Meyer-Bahlburg, A. Human Basophils Modulate Plasma Cell Differentiation and Maturation. Journal of Immunology. 198 (1), 229-238 (2017).
- Sihra, B. S., Kon, O. M., Grant, J. A., Kay, A. B. Expression of high-affinity IgE receptors (Fc epsilon RI) on peripheral blood basophils, monocytes, and eosinophils in atopic and nonatopic subjects: relationship to total serum IgE concentrations. Journal of Allergy and Clinical Immunology. 99 (5), 699-706 (1997).
- Dehlink, E., Baker, A. H., Yen, E., Nurko, S., Fiebiger, E. Relationships between levels of serum IgE, cell-bound IgE, and IgE-receptors on peripheral blood cells in a pediatric population. PLoS One. 5 (8), 12204 (2010).
- Hoffmann, H. J., Frandsen, P. M., Christensen, L. H., Schiotz, P. O., Dahl, R. Cultured human mast cells are heterogeneous for expression of the high-affinity IgE receptor FcepsilonRI. International Archives of Allergy and Immunology. 157 (3), 246-250 (2012).
- Ebo, D. G., et al. Analyzing histamine release by flow cytometry (HistaFlow): a novel instrument to study the degranulation patterns of basophils. Journal of Immunology Methods. 375 (1-2), 30-38 (2012).
- Macglashan, D. Marked differences in the signaling requirements for expression of CD203c and CD11b versus CD63 expression and histamine release in human basophils. International Archives of Allergy and Immunology. 159 (3), 243-252 (2012).
- Torres, M. J., et al. Clavulanic acid can be the component in amoxicillin-clavulanic acid responsible for immediate hypersensitivity reactions. Journal of Allergy and Clinical Immunology. 125 (2), 502-505 (2010).
- Ariza, A., et al. Pyrazolones metabolites are relevant for identifying selective anaphylaxis to metamizole. Scientific Reports. 6, 23845 (2016).

