Studying Murine Small Bowel Mechanosensing of Luminal Particulates
Summary
To study how the small bowel handles particulates of varying sizes, we have modified an established in vivo method to determine small bowel transit.
Abstract
Gastrointestinal (GI) motility is critical for normal digestion and absorption. In the small bowel, which absorbs nutrients, motility optimizes digestion and absorption. For this reason, some of the motility patterns in the small bowel include segmentation for mixing of luminal contents and peristalsis for their propulsion. Physical properties of luminal contents modulate the patterns of small bowel motility. The mechanical stimulation of GI mechanosensory circuits by transiting luminal contents and underlying gut motility initiate and modulate complex GI motor patterns. Yet, the mechanosensory mechanisms that drive this process remain poorly understood. This is primarily due to a lack of tools to dissect how the small bowel handles materials of different physical properties. To study how the small bowel handles particulates of varying sizes, we have modified an established in vivo method to determine small bowel transit. We gavage live mice with fluorescent liquid or tiny fluorescent beads. After 30 minutes, we dissect out the bowels to image the distribution of fluorescent contents across the entirety of the GI tract. In addition to high-resolution measurements of the geometric center, we use variable size binning and spectral analysis to determine how different materials affect small bowel transit. We have explored how a recently discovered “gut touch” mechanism affects small bowel motility using this approach.
Introduction
The human gastrointestinal (GI) tract is a multiple-foot-long organ system, roughly approximated as a tube of varying dimensions and physical properties1. As the contents move through its length, the GI tract's primary function is to absorb substances critical for life. The small intestine is specifically responsible for nutrient absorption. The small intestine's transit is tightly regulated to match the digestion and absorption functions, resulting in various motility patterns. Bayliss and Starling described the "law of the intestine"2 in 1899, showing the contractile propulsion program in the intestine known today as the peristaltic reflex; the segment proximal to the food bolus contracts to propel it forward, and the distal segment relaxes to receive it. In theory, this pattern alone could be sufficient to transport material aborally, but over a century of research has painted a more complex picture of contractile activity in the GI tract. Three small intestine motility periods are recognized in humans: the migrating motor complex (MMC), the fasting period, and the postprandial period3. The same patterns have been reported in mice4,5. The MMC is a cyclic motor pattern conserved across most mammals6,7. The MMC has a characteristic four-phase pattern that serves as a useful clinical marker in functional GI disorders7. The four phases, in order of occurrence, are (I) quiescence, (II) irregular, low amplitude contractions, (III) regular high amplitude contractions, and (IV) ramp-down period of declining activity7. The MMC marks the major motor pattern of the fasting period3. MMCs of the fasting period clear up the contents of the small intestine in preparation for the next meal.
The motor patterns of the postprandial period are optimized for the digestive and absorptive functions3. Regardless of caloric composition, initial transit is quick along the small intestine, contents are spread along the length of the bowel, and transit subsequently slows down8. Absorption is optimized by increasing contact surface area and slowing it down to increase residence time. Once the nutrients are inside the lumen, the dominant pattern consists of close (<2 cm apart) uncoordinated contractions (segmenting contractions), with a few superimposed large-amplitude contractions spanning the whole length of the small bowel (peristaltic contractions)9. Segmenting contractions mix the intraluminal contents in place. The occasional large peristaltic contractions propel the contents towards the colon.
The timing of this transition back to MMCs depends on food volume and caloric composition10. Thus, the small bowel samples luminal cues to regulate when to transition between motility periods. Mechanical cues, such as physical properties of luminal contents11, luminal volume, and wall tension, engage mechanoreceptor cells in the GI wall12,13,14,15,16. Indeed, increasing the solid component of a meal leads to an increase in small bowel transit17. We speculate that physical properties, such as the liquid or solid state of intraluminal contents, must engage different mechanoreceptors due to the various forces they generate on the GI wall18.
The gold standard for measuring in vivo GI transit in humans, as in mice, is the use of radioactive tracers measured by scintigraphy as they exit the stomach or transit along the colon19,20. In mammals, the small bowel loops in unpredictable ways making the small bowel difficult to image in vivo reliably, but progress is being made21. Further, there is currently a lack of tools to quantify how the small bowel handles particulates of varying properties and sizes. The starting point here was a gold-standard technique that standardizes the study of small bowel transit22,23,24 and barrier function22. It consists of gavaging mice with a fluorescent material, waiting for GI motility to transport the material, excising the GI tract, segmenting it into several sections from stomach to colon, sectioning, and homogenizing intraluminal contents for fluorescence quantification. We made two improvements. First, we altered the makeup of the gavaged contents to include fluorescent microscopic beads to determine how the small bowel distributes physical particulates. Second, we improved the spatial resolution by imaging the whole GI tract from stomach to colon ex vivo and used variable size binning to standardize our analysis across animals. We postulate that this reveals novel insights into the balance of propulsive versus segmenting contractions during the postprandial phase.
Protocol
All methods described here have been approved by the Institutional Animal Care and Use Committee (IACUC) of Mayo Clinic.
1. Setup
- Fast 8- to 10-week-old mice for 4 h. Provide mice with access to water.
NOTE: We use wild-type male C57BL/6J mice for all experiments presented here, but they can be performed on mice of any strain, gender and genotype. - Cool 15 mL of distilled water in a 50 mL conical tube in a 4 °C refrigerator.
- Heat another 15 mL of distilled water in a beaker using a hot plate with a magnetic stir bar to approximately 80-90 °C.
- Measure 0.5% methylcellulose for a total of 30 mL (0.15 g).
- Gently add methylcellulose to 15 mL of hot water so that it does not clump. The methylcellulose solution will be cloudy during this process.
- Once dissolved, remove the beaker from the hot plate and add the 15 mL of chilled water to the hot beaker.
- Place in a 4 °C fridge until the solution is clear, approximately 15-20 min.
- When clear, weigh 3 mg of rhodamine isothiocyanate (RITC)-dextran per 5 mL of 0.5% methylcellulose solution and mix to combine. This is the liquid condition in the Representative Results section.
- Alternatively, weigh 25 mg of fluorescent polyethylene microspheres and mix with 200 µL of the methylcellulose solution until well incorporated. Thorough incorporation of microspheres is ensured by vortex prior to gavage. The experimenter must visualize homogeneous distribution of suspended microspheres in the mixture.
- As the RITC-dextran solution is light sensitive, refrigerate the solution. Prepare the RITC-dextran solution and the microshpere mixture in advance and use within 2 months. Resuspend the microsphere mixture before use.
NOTE: Microspheres of varying sizes are used to study gastrointestinal handling of different materials. In the Representative Results section, we demonstrate the results of using small (diameter: 75-90 µm) and larger (diameter: 180-212 µm) microspheres.
2. Intraluminal content gavage
- Prepare the gavage by attaching an 18 Ga x 50 mm feeding tube to a 1 mL syringe and drawing 200 µL of RITC solution or microsphere solution.
- Manually restrain the fasted animal using the one-handed restraint technique25. Refer to the institution's information on proper murine restraint techniques.
- Gently insert the feeding tube through the mouth and esophagus of the mouse until it enters the stomach.
NOTE: The gavage step is critical for a successful experiment. It requires experienced hands that can consistently insert the tube roughly the same distance in each mouse. This step needs to be standardized by experimenters carrying out the protocol. The same experimenter should gavage all mice cohorts that will be compared to each other. - Slowly expel the syringe contents into the stomach and carefully remove the tube from the mouse.
- Following the gavage, return the mouse to the cage.
- Dispose of the gavage tube.
- Repeat the process as needed for the desired number of animals.
3. Bowel dissection
- Before beginning the dissections, turn on the in vivo imaging instrument to allow it to come to temperature.
- Sacrifice the mouse 30 min after the gavage. Use carbon dioxide inhalation to sacrifice the mouse, followed by cervical dislocation to ensure successful euthanasia.
- After confirming successful euthanasia, place the mouse in supine position on a dissection stage and pin its four appendages on the stage to have access to the abdomen. Wet the surface of the abdomen with 70% ethanol to wet the abdominal hairs.
- Use micro-dissection forceps (#5) to pull the skin and acquire sharp surgical scissors to make a transverse incision 1 cm above the anus. To expose the intraperitoneal cavity, continue the incision vertically up the abdomen until the rib cage.
- Gently move the cecum to the left with the micro-dissection forceps to expose the distal colon.
- Cut the distal colon with micro-dissection scissors just proximal to the rectum.
- Gently unravel the colon, cecum, and small intestine by pulling slowly in the opposite direction.
NOTE: Maintaining the dissected bowels in one continuous segment will create the most ease for the investigator. Rips and tears along the segment will cause subsequent steps to be more difficult. - Use micro-dissection scissors to cut proximal to the stomach.
- Use forceps to transfer the dissected bowels to the measurement sheet (Figure 1). Place the stomach at 0 mm and arrange the bowels along the ruler until 200 mm. Use micro-dissection scissors to cut at 200 mm. Repeat this process of aligning the bowels from 0 mm to 200 mm along the rulers while remaining confident that the orientation of the bowels does not get confused.
- Be careful to avoid stretching the bowels while aligning to the rulers.
- Once the tissue is arranged on the ruler, arrange the cecum so that it is parallel with the tissue but not in direct contact with it (if any fluorescence is found within that region, then a clear difference distinction between the cecum and the intestines will be needed).
- Place the measurement sheet with the dissected tissue in a dark area so the fluorescence can be preserved until it is time to image.
4. Ex vivo imaging
- Open the in vivo imaging software and log in.
- Initialize the imaging instrument so it is ready for image acquisition.
- Set the 'Excitation' and 'Emission' fields to the corresponding color used for the bead or RITC gavage. Red (liquid): excitation 535 nm /emission 600 nm. Green (microspheres): excitation 465 nm /emission 520 nm.
- Set the exposure to 'Auto'.
- Select the field of view.
- Before placing the measurement sheet into the instrument, ensure the intestines have not shifted during transport.
- Place the measurement sheet into the instrument within the field of view.
- Securely shut the door to the instrument and select Snapshot to photograph the field of view.
- Save collected pictures to a flash drive for analysis. Save individual captures of the fluorescent and photograph images. An overlay of the photograph and fluorescence indicates the location of fluorescent material in the GI tract (Figure 1).
NOTE: We recommend saving the files in the Tag Image File (.tif) format for downstream processing.
5. Analysis
- Open the fluorescent and photograph image files in a picture editing software.
- Adjust the pixel size of both images such that they have the exact same dimensions (our convention was to set both images to 1280 pixels by 850 pixels [height x width]).
- Close the photograph file. The following steps involve only the fluorescent image.
- Use an eraser tool to remove the background and make it transparent. An eraser may be needed to remove patches of the remaining background.
- Create a new layer. Make it an entirely black background to the fluorescent signal. This can be achieved by choosing a black fill to the layer and dragging the layer to lie below the layer with the fluorescent image.
- Save the new fluorescent image, containing only the fluorescent signal on a black background, as a new .tif file.
- Open the new fluorescent and photograph images in ImageJ.
- Turn each image into a 32-bit image by choosing Image > Type > 32-bit.
- Create a merged image of both by choosing Image > Color > Merge Channels. In the dialog box that opens, select the photograph file for the gray channel and the fluorescent file under any colored channels.
- Turn off the scale of the merged image by selecting Analyze > Set Scale > Click to Remove Scale.
- Select the "Rectangle" tool in ImageJ.
- Draw a rectangle around a section of the small bowel. Pay close attention to the width of this region of interest (ROI), as it should remain constant between all ROIs. The nominal value is not essential, just that it is kept consistent across all ROIs drawn on this image [since the small bowel takes over several rows, individual ROIs will be drawn over each section of the small bowel in a different row (Figure 1)]. Figure 2 shows the location of the width value in the ImageJ toolbar.
- Duplicate the ROI by selecting Image > Duplicate. Select only the channel corresponding to the colored channel.
- Use the rectangle tool again to draw an ROI over the whole new image.
- Retrieve a profile of the fluorescence by selecting Analyze > Plot Profile. The resulting graph plots on the y-axis the average intensity for each pixel along the length of the ROI. This was selected in step 5.12 to be the length of the analyzed section of the small bowel
- Open the list of values and copy them to a spreadsheet software.
- Repeat steps 5.12-5.16 for each section of the small bowel on a different ruler row. Keep pasting the values on the same spreadsheet file immediately under each previous set of values, such that all continuous rows in that column contain the average intensity value for the entire length of the small bowel.
- In the spreadsheet software, multiply each average intensity value by the constant width of the ROI rectangle created in step 5.12. This will yield the real intensity value along the small bowel at each point.
NOTE: Different animals will have slight variations on small bowel length. To prevent the length differences from affecting the outcome of downstream data analysis, the string of intensity values needs to be binned into a consistent number of bins across all experimental samples (Figure 3, Figure 4, and Figure 5 display results for three bin sizes). - Divide the number of intensity values by the number of bins desired. The resulting quotient S determines the number of intensity values being included in each bin.
- Determine the bin where each raw intensity value will go by using a roundup formula. This step places each raw intensity value into a bin. Each raw intensity value is chronologically indexed with the integer N. The quotient N/S determines the bin number assigned to each raw value. The roundup formula should round this quotient to a whole number with no decimals.
- Generate the value for each bin by using an averageif formula. The objective is to average the raw values assigned to a specific bin in Step 5.20. Hence, the arguments in the formula should be: (1) assigned bins generated in Step 5.20, (2) bin number, (3) raw intensity values.
NOTE: The first analysis we can perform on the binned data is a geometric center analysis, which quantifies how far we observe the highest fluorescent intensity along the small intestine (Figure 4). - Normalize each intensity value over the total fluorescent intensity. In other words, divide each intensity value by the sum of all intensities.
- Multiply each normalized value by the bin number. The product reflects the relative weight of each bin, contributing to the total fluorescent intensity.
- Adding all values generated in Step 5.23 yields the bin number at the center of the fluorescent signal. Divide by the number of bins to express the geometric center as the fraction of small bowel traveled by the fluorescent center.
NOTE: To reflect the spatial distribution of the signal, the next step is to generate the power spectrum of the binned intensity data set (Figure 5). - Open a Fast Fourier Transform (FFT) wizard in the spreadsheet software. Select the input as the binned data set and the output as an empty set of the same number of rows as bins.
- Extract the real value component of the FFT.
- Raise each real value of the FFT to the second power. This yields a data set of power spectra.
- Select the first half of the power spectra data set and move it so that it lies below the second half. This has the effect of centering the power spectra around the center frequency when it is plotted in the next step.
- Plot the power spectra on the y-axis of an x-y graph where the x-axis values are the range from -1 x (number of bins/2) to (number of bins/2) -1. In the case of 1000 bins, the axis values would range from -500 to 499.
- Power spectra for each animal should be compared on the basis of the spread of non-zero peaks and the heights of these non-zero peaks.
Representative Results
We show representative outcomes from Step 3 onwards. Figure 1 shows the intact explanted bowels, with fluorescent measurements overlaid. The stomach (purple) is laid along the same axis as the small intestine (orange), but we prefer moving the cecum (blue) to the side to prevent overlap with the large intestine (orange). As evidenced in the left panel, this is not always possible due to organ size. We cut the small bowel at ~200 mm to maximize the coverage of continuous segments, but this is not always possible due to mesentery limiting the ability to unfold the intestines, and our preference not to cut through structures like fluorescent pellets or the cecum. The red intensity in the left panel of Figure 1 and the green intensities of the middle and right panels are sorted into different sized bins to generate Figure 3. Only the intensities within the orange ROIs (small bowel) are extracted for analysis. The full length of the small bowel (every orange ROI in each panel) is extracted for analysis. Orange ROIs are the same width, as displayed in ImageJ (Figure 2). They are stitched together by placing all intensities in sequence prior to binning. Figure 3 shows the average fluorescent trace ± SEM per cohort; see figure legend for specifics.
The last two figures demonstrate the results of the analyses: geometric center and power spectrum. The geometric center measures the average location of the fluorescent signals in Figure 3, weighing the average by the signal strength of each bin. Hence, higher peaks in the trace pull the average closer to the position of that peak. The geometric center fails to completely characterize the spatial distribution of intraluminal contents. For example, the same geometric center can be obtained from a bolus concentrated at a single point and a spread-out signal centered around the same point. This limitation of the geometric center measurement is evident in the comparison between liquids and larger beads (Figure 3 and Figure 4). The majority of the liquid fluorescent trace is centered around two early peaks, whereas the larger beads show multiple peaks along the full length of the small bowel (Figure 3, left and right panels). The fluorescent trace of the larger beads is more distributed, but it averages out to a similar point as that of the liquid, regardless of binning granularity, which highlights the limitation of solely focusing on the geometric center (Figure 4). To account for the distributive nature of small bowel contractions, we have incorporated a power spectral analysis into the protocol. The power spectrum analysis works by deconstructing the fluorescence distribution in space into multiple sinusoid curves of different spatial frequencies. Each spatial frequency reflects the distance between two randomly selected points in the fluorescence trace. Some of these distances occur more frequently than others, so each is attributed a strength (power). The power correlates with how frequently two random points are separated by that distance. By plotting the power spectrum (Figure 5), we can demonstrate how many spatial frequencies contribute to the measured signal (spread of the spectrum) and compare the relative strength of those contributions (height of the spectrum). This allows us to quantitatively describe the spread of fluorescence along the GI tract.
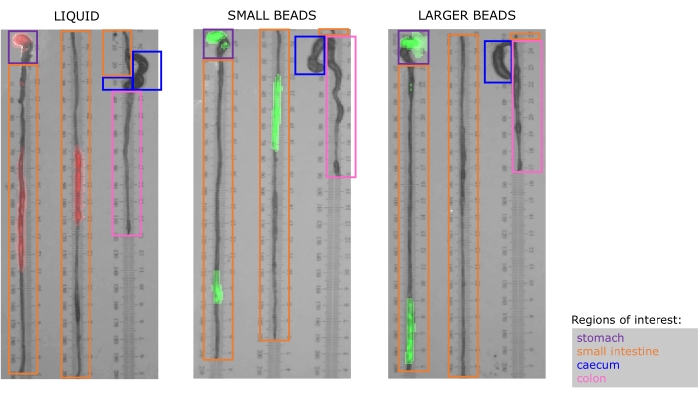
Figure 1. The dissected bowels are explanted and placed on laminated ruler paper prior to photography and fluorescence measurement. Fluorescence intensity is overlayed and pseudocolored to match the native fluorescence of gavaged material. Left: liquid rhodamine isothiocyanate (RITC) fluoresces in the red spectrum; Middle: small (diameter: 75-90 µm), and Right: larger (diameter: 180-212 µm) beads both fluoresce in the green spectrum. Purple, orange, blue, and pink boxes surround regions of interest (ROIs) in the stomach, small intestine, cecum, and colon in each specimen, respectively. The width of each ROI is kept consistent across rows to avoid confusion when calculating the raw fluorescence intensity during downstream analysis. Please click here to view a larger version of this figure.
Figure 2. Location of the region of interest width (in pixels) in the ImageJ toolbar (yellow underline). To reiterate, the width is displayed in pixels only if the scale has been removed (Step 5.10). Please click here to view a larger version of this figure.
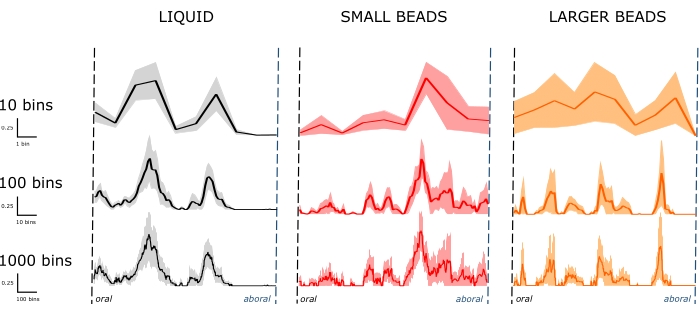
Figure 3. Fluorescence traces along the length of the small bowel. Line denotes the average fluorescence at a given bin. The shaded area denotes the standard error of the mean (SEM). The distribution of fluorescent material varies according to the material properties of the intraluminal contents (columns). Left: liquid (n = 7 mice) distribution across a few small bowel segments 30 min after gavage. Middle and right: small (n = 6 mice) and larger (n = 5 mice) beads distribute more broadly along the small bowel 30 min after gavage. Increasing the number of bins used to sort the same raw data sets reveals granular trace features unresolvable with fewer bins (rows). Smaller bins decrease measurement uncertainty, increase spatial resolution and better reflect the distributive component of small bowel contractions. Please click here to view a larger version of this figure.
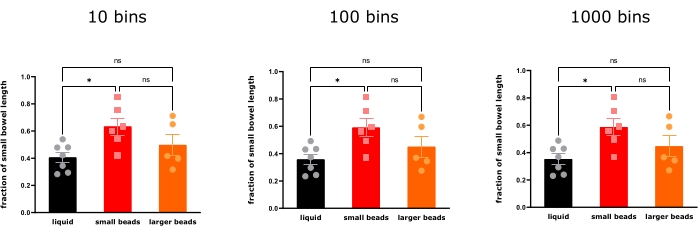
Figure 4. The geometric center of the fluorescence distribution in small bowel 30 min after gavage with liquid RITC, small beads (75-90 µm) and larger beads (180-212 µm) (mean ± SEM, n = 7, n = 6, n = 5, one-way ANOVA with Bonferroni correction). Bin size does not affect the transit interpretation that small beads are transported more distally than liquids 30 min after gavage, but the larger beads distribute so widely that there are no significant differences in their geometric center compared to both liquids and small beads (*P < 0.05. ns = not statistically significant). Please click here to view a larger version of this figure.
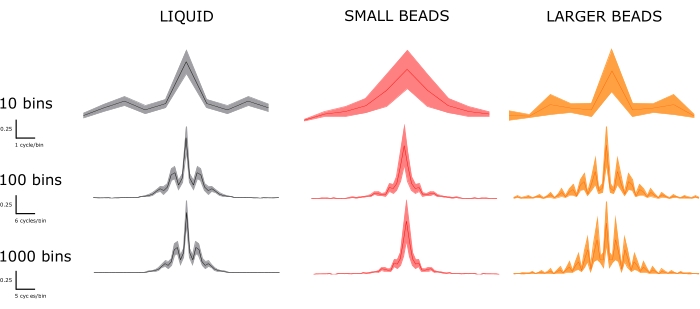
Figure 5. Power spectra are categorized by material property (columns) and bin size (rows). The improvements in spatial resolution and smaller binning are evident. Top row: few differences can be distinguished in the widely binned spectra. Bottom row: as bin sizes shrink, we can appreciate significant dominant frequencies present in the larger beads' spectrum but not in that of the small beads. Some, but not all, of those additional dominant frequencies, are present in the liquid spectrum. Using the bottom row spectra, we can compare to the fluorescent traces in Figure 3. In Figure 3, the liquid fluorescent trace displays two prominent peaks, which correlates with a few dominant peaks in the power spectrum. The small beads trace in Figure 3 displays a single dominant peak, which correlates with a single dominant peak in the power spectrum. The larger beads trace in Figure 3 displays more dominant peaks. Accordingly, the power spectrum shows a greater number of dominant frequencies. Please click here to view a larger version of this figure.
Discussion
The GI tract, like other tubular organs, such as blood vessels, requires mechanical sensors and effectors to maintain homeostasis26,27,28. However, the GI tract is unique in that the physical properties of the materials that traverse it are not constant across meals. Intraluminal contents of various physical properties (solid, liquid, and gas) transit the gut, generating different mechanical inputs to the GI mechanoreceptors. Indeed, different mechanical stimuli in the colon are sensed and relayed by distinct pathways29.
This protocol is technically accessible to anyone who performs routine mouse work, but a few critical steps require close attention. We find fasting mice is important before starting the study, as it removes the contribution from prior meals diluting the fluorescent signal. Moreover, mice fed a standard diet exhibit intraluminal fluorescence from diet components, specifically the chlorophyll component30. By fasting and fluorophore selection, we prevent chow fluorescence from interfering with the controlled conditions of the experiments outlined in this protocol. We recommend experimenters independently confirm that the selected fasting period is sufficient to eliminate non-specific fluorescence in the small bowel, as this may vary by strain, age, or gender. The experimenter should visually assess the homogeneous distribution of the gavaged contents. Otherwise, there will be uncertainty in their delivery and the study results. Gavages should be consistently performed by the same experimenter, preferably someone who has developed experience with the technique. The experimenter should aim for consistent delivery of contents to the stomach. Finding fluorescence in the esophagus or exclusively in the stomach are grounds to exclude an animal from the analysis, as it is uncertain if the small intestine had the same access to the gavaged contents. After gavaging, wait times can be shortened to 15 minutes or extended to 90 minutes, depending on whether early or late transit phases are of interest22. The original protocol using liquid gavage demonstrated that the majority of the material resides in the small bowel after 30 minutes. Moreover, while it is critical to work with haste during the dissection step, care must be taken not to disturb the location of the intraluminal contents and not rupture the GI tract.
This protocol evolved from previously published approaches that physically binned the small intestine by cutting it into five to 10 segments and homogenized intraluminal contents prior to fluorescence/radioactivity measurement22,23,24. The previous approach was significant because it standardized the measurement of small bowel transit. The small bowel notoriously loops in complicated and unpredictable patterns inside the abdominal cavity. Imaging-based transit studies are challenging regardless of species31,32. While our protocol remains a terminal experiment that cannot be expanded to humans, we significantly improve the spatial resolution, and seize upon that improvement to resolve regional transits and translate them into an unbiased measurement of spread within the small bowel (power spectrum).
In this protocol, spatial resolution is limited by the camera's resolution in the imaging system. Since binning normalizes small bowel length across animals, we can directly compare the average position of the intraluminal contents. Figure 3 clearly demonstrates the drastic smoothing, increased uncertainty, and decreased resolution resulting from large bin sizing. The geometric center quantifies the fluorescence center along the GI tract (Figure 4). As expected, this measurement is not significantly affected by the bin size. The power spectrum is an unbiased and complementary method that standardizes the analysis of peaks and spreads in the fluorescent traces of Figure 3. Improving the resolution improves the calculated spectra, making it possible to demonstrate visible differences between fluorescent traces (Figure 5). At low resolution (10 bins), it would be difficult to distinguish between liquids and larger beads, either by the geometric center or spectrum assessments, even though it is clear from looking at the fluorescent traces that they are not distributed similarly in space. At higher resolution (1000 bins), the geometric centers are still similar (owing to the fact that the geometric center is an average measurement), but the power spectrum can be reliably used to differentiate between both cohorts. It is worth noting the fluorescent traces are amenable to other types of analyses, such as leading-edge measurements. Anticipated changes in the geometric center can be used during sample size calculations prior to embarking on the experiments. Knowing that liquids have a geometric center of approximately 0.4-0.518, we used at least five mice in each group to statistically resolve changes of up to 45%.
We expanded the scope and used this protocol to study the transit of solids within the small bowel. As with liquids, we gavaged the solid microspheres into the stomach. The pyloric sphincter is located at the stomach-small bowel transition. This sphincter works as a size exclusion filter. In mice, the size cutoff is at ~300 µm, with particulates <300 µm encountering no resistance in entering the small bowel33, but this approach also works with slightly larger particulates we used in a recent study18. We chose our microsphere sizes here to provide a range of sizes that complement the recent study. We utilized two commercial fluorescent bead preparations that have diameters of 75-90 µm and 180-212 µm. Figure 1 shows similar fluorescence density and profiles in the stomach across cohorts, suggesting that neither liquids nor particulates are differentially retained by the stomach. The original data presented here demonstrates how the small bowel of wild-type mice handles liquids and microscopic solids differently. So far, we have used beads of one size range per independent experiment. Future studies can focus on how mixtures of different size particles are handled, and mixing fluorescent fluids of various physical properties with fluorescent beads to determine how more realistic chyme mixtures are handled by the small bowel.
The distribution differences reported here suggest a difference in the motility patterns activated by materials of different properties. We postulate that the balance of segmenting versus propulsive contractions determines how far and how homogeneously materials transit the small bowel. The geometric center for small beads is farther along the small bowel compared to liquids, suggesting that small solid particulates engage propulsive contractions. On the other hand, while the geometric center for larger beads is similar to a liquid, spectral analysis shows significant differences in spatial distribution, which could be a result of more segmenting contractions in the larger bead setting. We hypothesize that the small bowel utilizes size discrimination, a type of mechanosensory circuit, as one of the inputs that feed into deciding the balance between contraction types. For example, Dogiel type II neurons are mechanosensory neurons speculated to play a role in directing the switch from propulsive to segmenting contractions16,34. These intriguing speculations require further research to determine the mechanisms by which the GI tract senses physical properties and transduces them to physiological outputs such as contractions.
We believe this protocol will be helpful as an additional tool to bridge the gap from molecules to cells to organ function. We recognized that GI epithelial mechanoreceptors share developmental and structural similarities with skin mechanoreceptors35. Using the protocol outlined above, we helped demonstrate the sensory role of "gut touch" where these GI epithelial mechanoreceptors sense light mechanical stimuli and participate in intrinsic tactile sensitivity18. We anticipate that this protocol will prove similarly helpful to study how other sensory circuits contribute to small bowel transit.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank Mrs. Lyndsay Busby for administrative assistance and Mr. Joel Pino for media support. NIH grants supported this work: DK123549, AT010875, DK052766, DK128913, and Mayo Clinic Center for Cell Signaling in Gastroenterology (DK084567).
Materials
| C57BL/6J mice | Jackson Laboratory | 664 | other mice can be used with this protocol |
| Dissection tools | n/a | n/a | |
| Excel software | Microsoft | n/a | used for spreadsheet analysis |
| Fluorescent Green Polyethylene Microspheres 1.00g/cc 75-90um – 10g | Cospheric | UVPMS-BG-1.00 75-90um – 10g | "smaller beads" in the manuscript |
| Fluorescent Green Polyethylene Microspheres 1.00g/cc 180-212um – 10g | Cospheric | UVPMS-BG-1.00 180-212um – 10g | "larger beads" in the manuscript |
| Gavage needles | Instech | FTP-18-50-50 | |
| ImageJ software | n/a | n/a | used to extract fluorescence profile |
| Laminated ruler paper (prepared in-house) | n/a | n/a | |
| Methyl cellulose (viscosity: 400 cP) | Sigma | M0262 | |
| Photoshop software | Adobe | n/a | used for image processing |
| Rhodamine B isothiocyanate-Dextran | Sigma | r8881-100mg | "liquid" condition in the manuscript |
| Xenogen IVIS 200 | Perkin Elmer | 124262 | In vivo imaging system |
References
- Stevens, C. E., Hume, I. D. . Comparative Physiology of the Vertebrate Digestive System. 2nd ed. , (2004).
- Bayliss, W. M., Starling, E. H. The movements and innervation of the small intestine. The Journal of Physiology. 24 (2), 99-143 (1899).
- Husebye, E. The patterns of small bowel motility: physiology and implications in organic disease and functional disorders. Neurogastroenterology and Motility. (11), 141-161 (1999).
- Bush, T. G., et al. Effects of alosetron on spontaneous migrating motor complexes in murine small and large bowel in vitro. American Journal of Physiology-Gastrointestinal and Liver Physiology. 281 (4), 974-983 (2001).
- Der-Silaphet, T., et al. Interstitial cells of cajal direct normal propulsive contractile activity in the mouse small intestine. Gastroenterology. 114 (4), 724-736 (1998).
- Szurszewski, J. H. A migrating electric complex of the canine small intestine. American Journal of Physiology. 217 (6), 1757-1763 (1969).
- Deloose, E., et al. The migrating motor complex: control mechanisms and its role in health and disease. Nature Reviews Gastroenterology and Hepatology. 9 (5), 271-285 (2012).
- Johansoon, C., Ekelund, K. Relation between body weight and the gastric and intestinal handling of an oral caloric load. Gut. 17, 456-462 (1976).
- Sarna, S. K., et al. Spatial and temporal patterns of human jejunal contractions. American Journal of Physiology. 257 (1), 423-432 (1989).
- Hall, K. E., El-Sharkawy, T. Y., Diamant, N. E. Vagal control ofcanine postprandial upper gastrointestinal motility. American Journal of Physiology. 250, 501-510 (1986).
- Mayer, E. A. Gut feelings: the emerging biology of gut-brain communication. Nature Reviews Neuroscience. 12 (8), 453-466 (2011).
- Alcaino, C., et al. A population of gut epithelial enterochromaffin cells is mechanosensitive and requires Piezo2 to convert force into serotonin release. Proceedings of the National Academy of Sciences of the United States of America Sciences. 115 (32), 7632-7641 (2018).
- Kugler, E. M., et al. Mechanical stress activates neurites and somata of myenteric neurons. Frontiers in Cellular Neuroscience. 9, 342 (2015).
- Mazzuoli, G., Schemann, M. Mechanosensitive enteric neurons in the myenteric plexus of the mouse intestine. PloS One. 7 (7), 39887 (2012).
- Won, K. J., Sanders, K. M., Ward, S. M. Interstitial cells of Cajal mediate mechanosensitive responses in the stomach. Proceedings of the National Academy of Sciences of the United States of America. 102 (41), 14913-14918 (2005).
- Mao, Y., Wang, B., Kunze, W. Characterization of myenteric sensory neurons in the mouse small intestine. Journal of Neurophysiology. 96 (3), 998-1010 (2006).
- McIntyre, A., et al. Effect of bran, ispaghula, and inert plastic particles on gastric emptying and small bowel transit in humans: the role of physical factors. Gut. 40 (2), 223-227 (1997).
- Treichel, A. J., et al. Specialized mechanosensory epithelial cells in mouse gut intrinsic tactile sensitivity. Gastroenterology. 162 (2), 535-547 (2022).
- Bharucha, A. E., Anderson, B., Bouchoucha, M. More movement with evaluating colonic transit in humans. Neurogastroenterology and Motility. 31 (2), 13541 (2019).
- Camilleri, M., et al. Human gastric emptying and colonic filling of solids characterized by a new method. American Journal of Physiology. 257 (2), 284-290 (1989).
- Wang, D., et al. Trans-illumination intestine projection imaging of intestinal motility in mice. Nature Communications. 12 (1), 1682 (2021).
- Woting, A., Blaut, M. Small intestinal permeability and gut-transit time determined with low and high molecular weight fluorescein isothiocyanate-dextrans in C3H mice. Nutrients. 10 (6), 685 (2018).
- Miller, M. S., Galligan, J. J., Burks, T. F. Accurate measurement of intestinal transit in the rat. The Journal of Pharmacologial and Toxicological Methods. 6 (3), 211-217 (1981).
- Moore, B. A., et al. Inhaled carbon monoxide suppresses the development of postoperative ileus in the murine small intestine. Gastroenterology. 124 (2), 377-391 (2003).
- Machholz, E., et al. Manual restraint and common compound administration routes in mice and rats. Journal of Visualized Experiments. (67), e2771 (2012).
- Baeyens, N., Schwartz, M. A. Biomechanics of vascular mechanosensation and remodeling. Molecular Biology of the Cell. 27 (1), 7-11 (2016).
- Ye, G. J., Nesmith, A. P., Parker, K. K. The role of mechanotransduction on vascular smooth muscle myocytes’ cytoskeleton and contractile function. The Anatomical Record (Hoboken). 297 (9), 1758-1769 (2014).
- Mercado-Perez, A., Beyder, A. Gut feelings: mechanosensing in the gastrointestinal tract. Nature Reviews Gastroenterology & Hepatology. , 1-14 (2022).
- Brierley, S. M., et al. Splanchnic and pelvic mechanosensory afferents signal different qualities of colonic stimuli in mice. Gastroenterology. 127 (1), 166-178 (2004).
- Inoue, Y., et al. Diet and abdominal autofluorescence detected by in vivo fluorescence imaging of living mice. Molecular Imaging. 7 (1), 21-27 (2008).
- Szarka, L. A., Camilleri, M. Methods for the assessment of small-bowel and colonic transit. Seminars in Nuclear Medicine. 42 (2), 113-123 (2012).
- Padmanabhan, P., et al. Gastrointestinal transit measurements in mice with 99mTc-DTPA-labeled activated charcoal using NanoSPECT-CT. European Journal of Nuclear Medicine and Molecular Imaging. 3 (1), 1-8 (2013).
- Jang, S. F., et al. Size discrimination in rat and mouse gastric emptying. Biopharmaceutics and Drug Disposition. 34 (2), 107-124 (2013).
- Zhu, Y. F., et al. Enteric sensory neurons communicate with interstitial cells of Cajal to affect pacemaker activity in the small intestine. Pflügers Archiv: European Journal of Physiology. 446 (7), 1467-1475 (2014).
- Treichel, A. J., Farrugia, G., Beyder, A. The touchy business of gastrointestinal (GI) mechanosensitivity. Brain Research. 1693, 197-200 (2018).

