Isolation of Conditionally Immortalized Mouse Glomerular Endothelial Cells with Fluorescent Mitochondria
Summary
The article describes the method for isolating conditionally immortalized glomerular endothelial cells from the kidneys of transgenic mice expressing the thermolabile simian virus 40 and photo-activatable mitochondria, PhAMexcised. We describe the procedure for glomeruli isolation from whole kidneys using beads, digestion steps, seeding, and culturing of GECs-CD31 positive.
Abstract
Glomerular endothelial cell (GEC) dysfunction can initiate and contribute to glomerular filtration barrier breakdown. Increased mitochondrial oxidative stress has been suggested as a mechanism resulting in GEC dysfunction in the pathogenesis of some glomerular diseases. Historically the isolation of GECs from in vivo models has been notoriously challenging due to difficulties in isolating pure cultures from glomeruli. GECs have complex growth requirements in vitro and a very limited lifespan. Here, we describe the procedure for isolating and culturing conditionally immortalized GECs with fluorescent mitochondria, enabling the tracking of mitochondrial fission and fusion events. GECs were isolated from the kidneys of a double transgenic mouse expressing the thermolabile SV40 TAg (from the Immortomouse), conditionally promoting proliferation and suppressing cell differentiation, and a photo-convertible fluorescent protein (Dendra2) in all mitochondria (from the photo-activatable mitochondria [PhAMexcised] mouse). The stable cell line generated allows for cell differentiation after inactivation of the immortalizing SV40 TAg gene and photo-activation of a subset of mitochondria causing a switch in fluorescence from green to red. The use of mitoDendra2-GECs allows for live imaging of fluorescent mitochondria's distribution, fusion, and fission events without staining the cells.
Introduction
The glomerulus is critical for blood filtration by restricting the passage of large molecules through the glomerular filtration barrier1,2. The glomerulus contains four cell types: parietal epithelial cells, podocytes (visceral epithelial cells), glomerular endothelial cells (GEC), and mesangial cells3. The glomerular endothelium is characterized by a unique vascular structure, as per the presence of fenestrae required for large filtration volumes4. The apical surface of the glomerular endothelium is covered with a negatively charged glycocalyx layer and a coat called the endothelial surface layer that creates a space between the endothelium and blood. This structure provides high charge selectivity restricting the passage of negatively charged molecules such as albumin and preventing leukocyte and platelet adhesion5.
GECs are very sensitive to metabolic changes, such as the hyperglycemia associated with the diabetic milieu. Indeed, diabetes leads to increased circulation of noxious substances, the saturation of glucose metabolism pathways, and disturbed cellular redox balance3,6. Moreover, the increase in reactive oxygen species induces mitochondrial dysfunction, which affects endothelial function7.
The overall goal of the current protocol is to isolate immortalized glomerular endothelial cells with fluorescent mitochondrial features. Indeed, the cell culture of primary GECs has a limited proliferative cycle and early senescence8. In addition, the presence of fluorescent mitochondria helps examine fission and fusion events in response to hyperglycemia or any other treatment. As an alternative method, other labs used h-TERT to immortalize cells in vitro9.
The method described here allows for the isolation of conditionally immortalized mitoDendra2 glomerular endothelial cells from 4-6-week-old animals (Figure 1). This detailed protocol describes the use of transgenic mice (H-2Kb-tsA58) harboring the simian virus 40 large tumor antigen (SV40 TAg) gene10,11 for generating thermolabile conditionally immortalized cells. The tsA58 TAg gene product is functional at the permissive temperature of 33 °C under the control of the inducible 5' flanking promoter of the mouse H-2Kb gene, which is increased above basal levels upon exposure to interferon gamma (IFNγ), therefore maintaining the conditional proliferation phenotype12. H-2Kb is rapidly degraded at the non-permissive temperature of 37 °C in the absence of IFNγ, removing the immortalizing function of the tsA58Tag in cells and allowing the cells to develop a more differentiated phenotype13,14,15. Optional crossing of H-2Kb-tsA58 transgenic mice with PhAM mice, which express a mitochondria-specific (subunit VIII of cytochrome c oxidase) Dendra2-green, allows the live detection of fluorescent mitochondria16. Dendra2 green fluorescence switches to red fluorescence after exposure to a 405 nm laser16. When mitochondria fuse after photo-switching, they form elongated shapes that appear yellow from the exchange of green and yellow material or appear red when they undergo fission7,17. The mitoDendra2-GECs are a great tool for studying the cellular responses of GEC mitochondria to different stimuli.
Protocol
All animal procedures described here were approved by the IACUC at Icahn School of Medicine at Mount Sinai. We used three male mice (H-2Kb-tsA58 transgenic mice with photo-activatable mitochondria [PhAM]) purchased from Jackson lab and kept on a normal chow diet.
1. Working conditions and preparations
- Clean the working space inside the laminar flow hood using 70% ethanol and UV-C light.
- Prepare the bead's washing buffers. Make buffer-1 using 0.1 M sodium phosphate at pH 7.4-8.0, buffer-2 using 1x phosphate-buffered saline (PBS) without calcium and magnesium supplemented with 0.1% bovine serum antigen (BSA), 3 mM EDTA, pH 7.4, and buffer-3 using 0.2 M Tris with 0.1% BSA, pH 8.5.
2. Magnetic particle concentrator
- Preparation of magnetic beads
- In a 1.5 mL microcentrifuge tube, gently mix 200 µL of magnetic beads (4 x 108 beads) with 200 µL of buffer-1.
NOTE: Prepare 200 µL of magnetic beads per mouse 1 day before tissue harvesting. The beads will be used to harvest the glomeruli. - Place the tube in a magnetic particle concentrator for 1 min, then carefully aspirate the supernatant. Remove the tube from the magnet and resuspend the washed beads in 200 µL of buffer-1.
- Place the tube containing the magnetic beads in the magnetic particle concentrator for 1 min and gently aspirate the supernatant.
- Inactivate the free tosyl groups by washing the magnetic beads 2x, 5 min each, using 200 µL of buffer-2 by placing the tube in the magnetic particle concentrator and discarding the buffer.
- Incubate the magnetic beads in 200 µL of buffer-3 for 24 h at 4 °C, or for 4 h at 37 °C, to inactivate the remaining free tosyl groups. Place the tube in the magnetic concentrator for 1 min, gently aspirate the supernatant, and discard it.
- Wash the magnetic beads 1x with 200 µL of buffer-2 for 5 min at 4 °C. Place the tube back in the magnetic concentrator for 1 min and gently aspirate the supernatant. Remove the tube from the magnetic concentrator and resuspend the magnetic beads in 200 µL of buffer-2 to obtain 4 x 108 beads/mL.
NOTE: At this stage, it is possible to leave the beads at 4 °C for up to 15 days.
- In a 1.5 mL microcentrifuge tube, gently mix 200 µL of magnetic beads (4 x 108 beads) with 200 µL of buffer-1.
3. Isolation of mouse kidney glomerular cells
- Prepare the perfusion material: 30 mL syringe, 25 G butterfly needle. Prepare the isolation material: sterile 10 cm dish, scalpel, magnetic particle concentrator, 40 µm cell strainer, 100 µm cell strainer.
- Prepare 100 mL of 1x Hank's Balanced salt solution (HBSS) supplemented with 1% antibiotics and adjust the pH to 7.35. Filter the solution with a 0.2 µm filter and keep it under the hood to avoid contamination.
- Mix 200 µL of magnetic beads from step 2.1.6 with 20 mL of 1x HBSS.
NOTE: For each mouse, 200 µL of beads diluted in 20 mL of HBSS 1x will be needed. - Prepare a 1 mL aliquot of collagenase type-II (125 CDU/mg) at 1 mg/mL in 1x HBSS solution, pH 7.35. Pre-warm the RPMI with 10% FCS medium in a water bath at 37 °C. Pre-warm the endothelial cell culture medium at 37 °C (see Table of Materials for details).
NOTE: The RPMI will be used to neutralize the collagenase. - Pre-coat two wells of a 6-well plate with 2 mL of 10 µg/mL collagen IV (1 mL/well) for 45 min at room temperature. Wash the plate 3x with 1x PBS to remove the acetic acid used to dilute the collagen. Use the coated plates immediately or store them at 2-8 °C for up to 4 weeks.
NOTE: Sealing the plate(s) with a transparent film is preferred to prevent drying out. The collagen coating facilitates the attachment of cells to the plate. - Clean and sterilize all the surgical instruments in Figure 2 by using 70% ethanol and subsequently autoclaving them for 30 min. Clean the surgical workspace with 70% ethanol.
- Anesthetize the mice with an intraperitoneal injection of ketamine/xylazine (10 mg/mL ketamine and 1 mg/mL xylazine). To confirm that the animal is fully anesthetized, check for the toe pinch reflex.
- Fix the paws of the mouse with 19 G needles to hold it in place on a dissection platform. Clean the chest and the abdomen with 70% ethanol, and open the abdomen along the chest with scissors a (Figure 2).
- Insert a butterfly needle into the left ventricle (Figure 3A) and inject about 100 μL of the bead solution (from step 1.2) to expand the blood circulation.
- Carefully break the inferior vena cava below the kidneys with a 19 G needle (Figure 3A). Continue the perfusion initiated in step 3.9.
NOTE: Alternatively, a mini pump can be used at speed: 18.0+, time: 5 min. Good perfusion is important to harvest a great number of glomeruli bearing magnetic beads. This is a non-survival/ terminal procedure. - Gently remove the fat tissue with tweezers and excise both kidneys with thin surgical scissors c (Figure 2) and put them in a plate with 1 mL of 1x HBSS on ice. Mince the kidney with scissors b (Figure 2) then with a sterile scalpel to 1 mm small pieces as shown in Figure 3B.
- Next, incubate the minced kidney with 1 mL of collagenase type II from step 3.4 for 35 min at 37 °C with gentle tilting using a horizontal shaker. The tissue should be completely digested after this step, as shown in Figure 3C.
- Add 4 mL of RPMI with 10% FBS (from step 3.4) to neutralize the collagenase. Filter the digested tissue through a sterile 100 µm cell strainer into a 50 mL tube. Use the bottom of a sterile 1.5 mL microcentrifuge tube to stir the digested tissue against the strainer to allow the glomeruli to go through.
- Rinse the cell strainer with 15 mL of 1x HBSS and then centrifuge the flow-through at 200 x g for 5 min at room temperature. At this point, the pellet contains three layers; the beads with glomeruli are visible in the middle layer of the tube, the upper layer consists of smaller structures such as tubules, and the bottom layer is debris (Figure 3D).
- Carefully aspirate the supernatant and the top white layer. The remaining pellet contains the beads bearing the glomeruli and debris (Figure 3D). Resuspend the pellet with 1.5 mL of 1x HBSS and transfer into a 2 mL tube.
- Place the tube in the magnetic concentrator (Figure 3E), aspirate the supernatant, and then wash the beads 2x with 1 mL of 1x HBSS; at this stage, put 20 µL of the cell suspension on a slide to verify the presence of glomeruli under the microscope (Figure 3F; a glomerulus [black arrow], and some beads [yellow arrows] in the suspension).
- Carefully aspirate the supernatant, resuspend the pellet in endothelial cell growth medium (Table of Materials), and transfer it to a clean 2 mL tube. Add 2 µL of IFNγ (100 U/mL) to the tube and plate the cells on to one well of a 6-well plate. Incubate the cells at 33 °C.
- After 3 days of culture, carefully change the medium by pipetting out 1 mL of the medium and replacing it with 1 mL of fresh media. At this stage, the cells are heterogeneous with a mixture of glomerular cells (Figure 4G).
NOTE: In the outgrowing culture, there will be a mix of glomerular cells and some magnetic beads (Figure 4G). Notice that all the cells should show green, fluorescent mitochondria. - After 7 days of culture, completely aspirate the medium and replace it with a fresh 2 mL of endothelial cell growth medium supplemented with IFNγ (100 U/mL). At this stage, the cells start proliferating but are still heterogeneous (Figure 4A).
- After 10 days of culture when the cells reach ~80% confluency, passage the cells using trypsin as described in step 5.1 and then transfer them into a T25 flask coated with collagen IV.
- After 1 week (total ~21 days of culture), passage the cells to a T75 flask coated with collagen IV.
4. Preparation of beads for endothelial cell isolation
- Start by coating the sheep anti-rat beads with a rat anti-mouse CD31 antibody3,18,19. Use 10 µL of beads for each T75 flask.
- Wash 10 µL of beads 3x with 200 µL of 1x PBS supplemented with 0.1% BSA, 2 mM EDTA, 1% penicillin/streptomycin. Place the tube in the concentrator magnet to pipette the washing buffer each time.
- Resuspend the beads in 200 µL of PBS, 1% BSA, 2 mM EDTA, 1% penicillin/streptomycin. Add 4.8 µL of rat anti-mouse CD31 antibody7,18,19, place the tube on a horizontal shaker and gently mix for 1 h at room temperature, and then keep the tube at 4 °C overnight.
5. Isolating the glomerular endothelial cells with CD31-coated beads
- Add 3 mL of 0.25% trypsin to a T75 flask and incubate the cells at 37 °C for 5 min. Neutralize the trypsin with 3 mL of endothelial growth medium, transfer the cells to a 15 mL tube, and then centrifuge at 200 x g for 5 min.
- Aspirate the supernatant and wash the cells in 1 mL of PBS containing 1% BSA, 2 mM EDTA, 1% penicillin/streptomycin. Centrifuge at 200 x g for 5 min.
- Resuspend the pellet in 200 µL of coated beads prepared in step 4, and add 800 µL of PBS containing 1% BSA, 2 mM EDTA, 1% penicillin/streptomycin. Incubate the cells for 45 min with continuous shaking at 37 °C, 5% CO2.
- Place the tube in the magnetic concentrator and wash the cells 4x with 1x PBS containing 1% BSA, 2 mM EDTA, 1% penicillin/streptomycin. Resuspend the cells in 3 mL of endothelial growth medium supplemented with 100 U/mL IFNγ.
- Plate 1.5 mL of cells/well in collagen IV-coated wells on a 6-well plate. Culture the cells under permissive conditions at 33 °C.
NOTE: The isolated cells express simian virus 40 (SV40) large T antigen tsA58, enabling the activation of continuous proliferation at 33 °C (permissive condition). - After 4 days in culture, remove 750 µL of the medium and replace it with the same volume of fresh medium supplemented with 100 U/mL IFNγ.
- After 10 days to 14 days of culture, check for growing CD31 positive GECs colonies (Figure 4C) that express fluorescent mitochondria using an epifluorescence microscope with a 488 nm filter.
- After 21 days of culture, the cells may have reached 80%-90% confluency; transfer them into a T25 flask. After reaching confluency, cells can be maintained with RPMI growth medium and 10% FCS.
NOTE: Before reaching confluence, the cells may look heterogeneous (Figure 4B). However, once they are confluent, they acquire a cobble-stone morphology. - At this stage, the cells could be cryopreserved. Upon reaching 80% confluency, add 3 mL of 0.25% trypsin to the cells and incubate them at 37 °C for 5 min. Neutralize the trypsin with an equal volume of growth medium supplemented with 10% FBS.
- Centrifuge at 200 x g for 5 min. Discard the supernatant and resuspend the cells in 3 mL of freezing medium (RPMI, 20% FBS, 10% DMSO). Aliquot the cells in cryo-tubes and store them in liquid nitrogen vapor temperature.
6. Cultivating the mitoDendra2-GECs
- Thaw the cells from the cryoaliquot, from step 5.10, in a water bath at 37 °C and transfer them into a 15 mL tube with 10 mL of pre-warmed endothelial growth medium.
- Centrifuge the suspension at 200 x g for 5 min at room temperature. Remove the medium, and transfer the pellet to the collagen IV-coated T25 cm2 cell culture flask with pre-warmed endothelial cell growth medium supplemented with 100 U/mL IFNγ and incubate at 33 °C.
- Change the medium the next day. When the cells reach 70%-80% confluency, usually after 5 days, transfer them to a T75 cm2 cell culture flask.
- To differentiate the cells to endothelial phenotype, transfer the cells to non-permissive conditions at 37 °C in growth media without IFNγ.
7. Passaging of mitoDendra2-GECs
- Remove the medium, wash the cells with PBS, and add 3 mL of warm 0.25% trypsin-EDTA solution (T75 cm2 flask), incubate at 37 °C for 5 min.
- When the cells are detached, add 5 mL of growth medium and pipette up and down. Centrifuge the suspension at 200 x g for 5 min at room temperature and transfer the cells to a new collagen IV-coated flask or plate.
NOTE: Passaging should be done once a week at a ratio of 1:4.
8. Characterization of mitoDendra2-GECs
- Perform immunofluorescence staining for CD31 on differentiated cells7,18,19,20 (Figure 4D). Use other markers such as vWF and Isolectin-B4 as well20.
- To confirm mitochondrial specificity of Dendra2, label cells with mitochondrial-specific markers by incubating the cells in growth medium containing 100 nM of mitochondrial tracker for 30 min at 37 °C, 5% CO2.
- Remove the medium and wash the cells gently with pre-warmed medium without phenol red to perform live imaging.
NOTE: Alternatively, cells may be fixed using formaldehyde fixatives (4%) for 30 min followed by washing several times in PBS. Fixed cells can be stored at 4 °C in 1x PBS until use. A confirmation with flow cytometry is also possible: label cells with anti-CD31, anti-PECAM, anti-CD45. GECs should stain negative for podocyte markers (synaptopodin, Nphs1, Podxl), mesangial cell markers (PDGFRβ, angiotensinogen), and tubular cell markers (Aquaporin, Aminopeptidase-N). These markers serve as negative controls. Due to the mitochondrial-specific Dendra2 expression of these cells, avoid using the FITC channel.
9. Live cell imaging of mitochondria structural changes in response to oxidative stress or high glucose
- Plate 1 x 105 GECs on a 35 mm glass-bottom dish coated with collagen IV using endothelial cell growth medium (Table of Materials). After 24 h, change to medium without phenol red. Cells have green, fluorescent mitochondria.
- Set the environmental chamber at 37 °C, 5% humidity, and 5% CO2 1 h prior to the start of the experiment.
- Under a confocal microscope, use a 40x/1.2 W objective to adjust the focus and select the region of interest. Incubate the cells with 2mL of 5 mM or 25 mM glucose for 60 min.
- Expose the cells to a 405 nm laser (4% laser power) to photo-convert a subpopulation of mitochondria from green to red (Figure 5A-B, D-E).
- Use the 561 nm laser to excite Dendra2 in the unconverted state to visualize the red fluorescence. See the detailed photo-switching protocol as previously described16. The fusion events appear as yellow fluorescence after several minutes because of green matrix and red matrix fusion (Figure 4C, F).
Representative Results
In this article, a detailed protocol for the isolation of conditionally immortalized glomerular endothelial cells with stable fluorescent mitochondria (mitoDendra2-GECs) is described (Figure 1). The use of young 6-10-week-old mice is essential to obtain a substantial number of healthy cells. After 3 days of culture, cells start growing slowly from the isolated glomeruli, as shown in Figure 3G. After 7 days, cells are heterogeneous, showing other glomerular cell types, such as podocytes, parietal epithelial cells, and mesangial cells (Figure 4A). After reaching 70% to 80% confluency, other glomerular cells were removed using a positive selection of endothelial cells with magnetic CD31 labeled beads (Figure 4B–C). The mitoDendra2-GECs expressed CD31 (Figure 4D) and were negative for podocyte marker as shown by synaptopodin negative staining (Figure 4E-F).
The fluorescent mitochondrial feature in the mitoDendra2-GECs allows for studying mitochondria morphology under various conditions in vitro. Here, we tested the effect of high glucose (25mM) on the mitochondria structure of mitoDendra2-GECs (Figure 5D-F). Compared to the elongated mitochondria visible in cells under normal glucose (5mM; Figure 5A-C), high glucose-induced fragmentation or fission of mitochondria as observed by prominent spheroid-shaped mitochondria (Figure 5E). Furthermore, we used a 405 nm laser to photo-convert a selected subpopulation of mitochondria in a single live mitoDendra2-GEC (Figure 5A,D). In Figure 5B and Figure 5E, successful photo-switching of mitochondria from green (488nm) to red (561 nm) in the selected area is presented. Importantly, it allowed for witnessing the fusion events in mitoDendra2-GECs grown under normal glucose, as can be observed by the yellow merged green and red fluorescence as a result of mitochondria matrix fusion (Figure 5C). In contrast, in the high glucose-treated mitoDendra2-GEC, the mitochondria were fragmented and were mainly red (Figure 5F), suggesting that, in the time frame tested, the fission/fusion events were delayed or inhibited due to the damage caused by the high glucose treatment, and this is consistent with previous reports18,19.
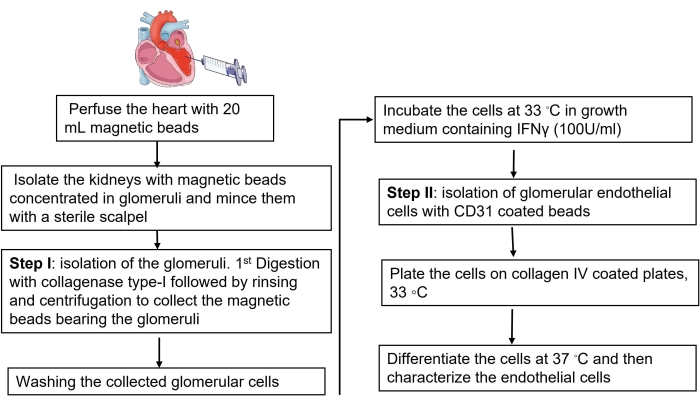
Figure 1: Representative description of murine GEC isolation. The diagram represents the significant steps of GEC isolation, starting from perfusing mice heart with magnetic beads to isolate the kidney glomeruli, the digestion of collagenase, the culture of glomerular cells, and finally, the purification of GEC using CD31-coated beads. Please click here to view a larger version of this figure.
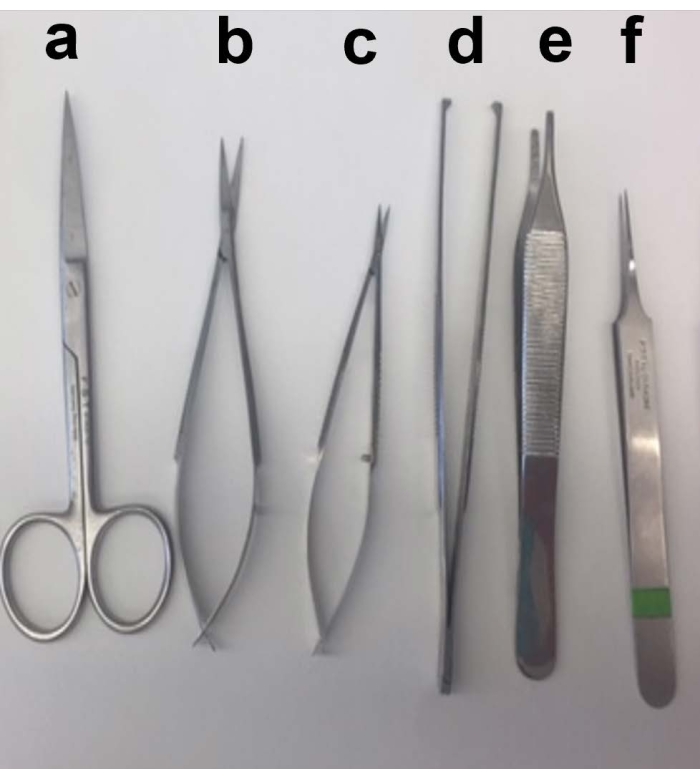
Figure 2: Description of kidney dissection. Representative image of all the sterile surgical instruments needed for the dissection. Scissors (a) are needed to open the skin of the mouse, and a separate pair of scissors (b,c) are needed to isolate the kidney. Tweezers (d) and (e) are needed to hold the skin. Tweezer (f) is needed to collect the dissected kidney. Please click here to view a larger version of this figure.
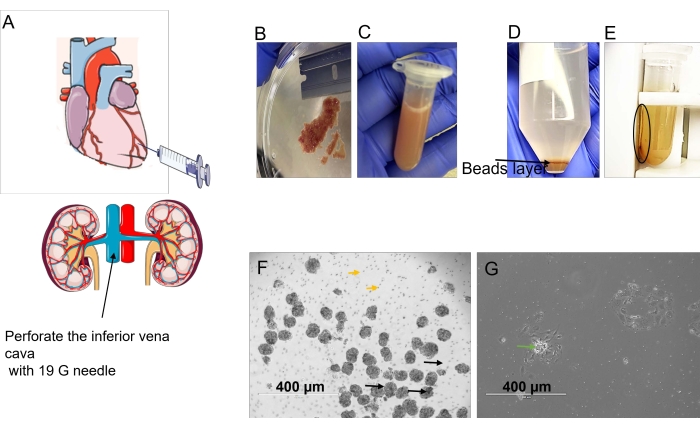
Figure 3: Description of glomeruli harvesting. (A) Perfuse the left ventricle with the HBSS-beads solution using a 20 mL syringe. Perforate the inferior vena cava with a 19 G needle. (B) After perfusion, remove the fat from the kidney and cut it into 1 mm pieces. (C) Digest the tissue with collagenase type II. (D) After digestion and centrifugation, three layers are formed; the beads bearing the glomeruli are in the middle layer, as indicated by the black arrow. (E) Separate the isolated beads bearing the glomeruli using the magnetic concentrator. (F) Representative image of freshly isolated glomeruli. (G) Growing cells from isolated glomeruli after 3 days. Please click here to view a larger version of this figure.
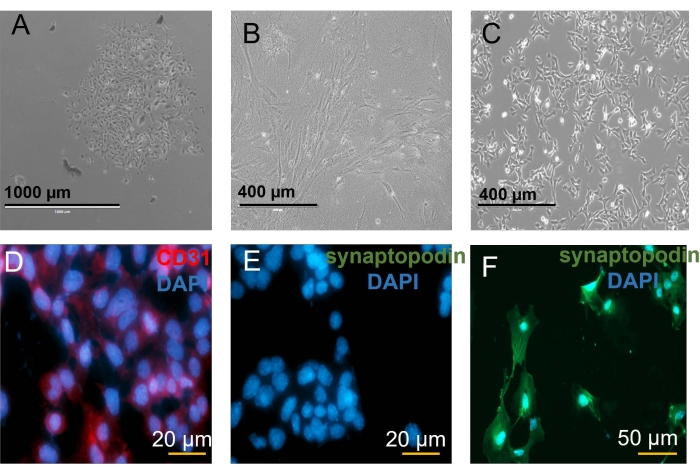
Figure 4: Mitochondrial assay in vitro. (A) Glomerular cells after 3 days of culture. (B) GEC after 7 days. (D) Purification of GEC with CD31-labeled beads. Microscopic imaging of CD31 staining. (E) GECs are negative for synaptopodin. (F) Podocytes stained with synaptopodin (488 nm, green fluorescence) as a negative control. Please click here to view a larger version of this figure.
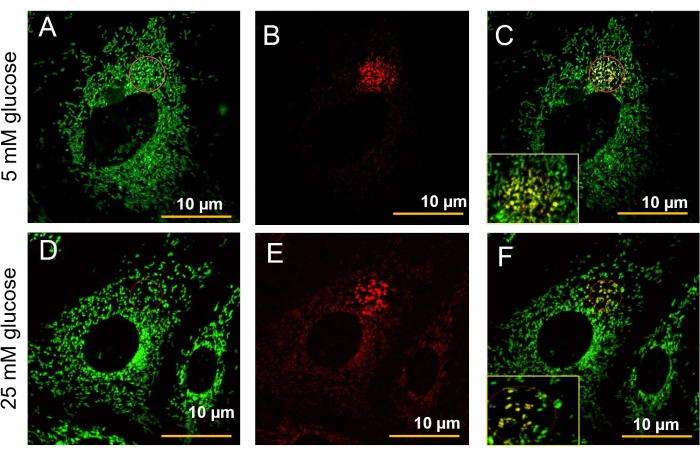
Figure 5: Microscopic imaging of fluorescent mitochondrial fusion in vitro. Images were acquired with a confocal microscope using a 40x water objective. (A–B) Selected area (red circle) was illuminated with a 405 nm line (4% laser power) for 300 bleaching iterations at a speed of 6.3-12.61/pixel, as previously described16. (A–C) Cells in growth media containing 5 mM glucose; green fluorescence (488 nm) shows healthy mitochondria. (B,E) Mitochondria were photo-switched into red (567 nm). (C) Yellow fluorescence detects an active fusion event of GEC treated with 5mM glucose. (D) Cells were treated with high glucose 25 mM for 30 min. Green fluorescence shows fragmented mitochondria and (E) a portion that was photo-switched into red. (F) The figure shows a decreased fusion event as shown by reduced yellow fluorescence. Please click here to view a larger version of this figure.
Discussion
Mitochondria are critical for cellular metabolism, homeostasis, and stress responses, and their dysfunction is linked to many diseases, including kidney disease. Mitochondria have a role in the pathologic generation of excessive reactive oxygen species (ROS), the regulation of intracellular calcium levels, cell death pathways, and cytoskeletal dynamics21,22,23.
The isolation of murine GECs is challenging because of their small number, size, matrix factors, and interdependent nature with other glomerular cells that are essential for their survival. The current article presents a detailed protocol for mouse GEC isolation. Specifically, we used transgenic mice as a tool to obtain conditionally immortalized cell lines with high proliferation potential. The transgenic mice, expressing simian virus 40 (SV40) large T antigen tsA58, enable the activation of continuous proliferation under permissive conditions at 33 °C and were here crossed with the PHAMexcised mice expressing mitochondrial-specific fluorescence. The double transgenic mice offered an excellent tool for studying mitochondria structure. Indeed, PHAMexcised mice bear mitochondrial-specific (subunit VIII of cytochrome c oxidase) Dendra2 green, allowing the live detection of fluorescent mitochondria and the observation of fission and fusion events through green-red photo-switching16.
The protocol consists of three significant steps: extracting glomeruli using beads, the proliferation of glomerular cells, and the subsequent purification of GEC using CD31 beads once the cells are in culture. The use of only one digestion with collagenase is sufficient to isolate glomerular cells. Other protocols make use of a second digestion with DNAse and proteinase24. However, we have noticed better cell yields with only one digestion step. The DNAse may remain active and slow down the proliferation of the cells. Furthermore, GEC could be isolated using differential sieving as previously described20, which is not always easy when using mice kidneys.
It is crucial to autoclave all the surgical tools before starting the isolation of the kidneys and the glomeruli to avoid contamination. Moreover, it is important to filter and supplement the solutions with antibiotics to eliminate any source of contaminants. Indeed, mycoplasma infections may cause cytopathology that consequently interferes with every parameter measured in cell culture25.
The isolation of glomeruli relies on the intra-cardiac perfusion of beads. Certainly, slow perfusion with 20 mL of 1x HBSS-containing beads is essential to harvest a high number of glomeruli. The beads will be trapped in the glomeruli, which eases their purification using a magnetic concentrator. The beads might be prepared and stored at 4 °C for up to 15 days before using them to ease the experimental procedure.
Furthermore, digesting glomeruli with continuous shaking is crucial for dissociating a good number of glomeruli from the kidney. On the other hand, the isolation of murine GECs is challenging because of their small size and the presence of other glomerular cells. Therefore, the stepwise isolation of glomeruli, the growth of all glomerular cells in culture, and the subsequent use of CD31-coated beads to purify endothelial cells are vital, specifically for obtaining more significant numbers of viable and conditionally proliferative GECs. Nevertheless, validating the purity of cells with immunostaining and flow cytometry is needed.
In addition, the protocol describes the characterization of the GEC phenotype and the mitochondria fusion/fission assay in vitro. The mitoDendra2-GECs are an excellent tool for studying the cellular responses of GEC mitochondria to different stimuli in vitro without the need for transfection or staining.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors thank Professor Cijiang He and Dr. Fu Jia for their insights in mice endothelial cell isolation and thank Professor Mone Zaidi for providing the PhAMexcised mice and valuable discussions. The authors would also like to acknowledge the Microscopy CORE at the Icahn School of Medicine at Mount Sinai and staff for the guidance we received. This work was supported by grants from National Institutes of Health grant R01DK097253 and Department of Defense CDMRP grant E01 W81XWH2010836 to I.S.D.
Materials
| 100 µm cell strainer | Fisher | 22-363-549 | |
| 1ml Insulin Syringes | BD | 329424 | |
| 25G butterfly | BD | 367298 | |
| 3 mm cutting edge scissors | F.S.T | 15000-00 | |
| 30ml syringe | BD Biooscience | 309650 | |
| 40 µm cell strainer | Fisher | 22-363-547 | |
| 40 µm nylon mesh | |||
| Bonn Scissors | F.S.T | 14184-09 | |
| Bovine serum albumin | Fisher | BP1600-100 | |
| CD31 | abcam | ab7388 | |
| Collagenase type I | Corning | 354236 | |
| Collagenase type II | SIGMA | C6885 | 125CDU/mg |
| Collagene type IV | SIGMA | C5533-5M | |
| Dnase-I | Qiagen | 79254 | |
| Dynabeads 450 | Thermofisher Scientific | 14013 | |
| endothelial cells growth medium | Lonza | cc-3156 | |
| Extra fine graefe forceps | F.S.T | 11150-10 | |
| FBS | Gemini | 100-106 | Heat inactivated |
| Fibronectin | Thermofisher | 33016015 | |
| Fine forceps | F.S.T Dumont | E6511 | |
| HBSS | GIBCO | 14065-056 | |
| IFNg | Cell Science | CRI001B | |
| Immortomouse | Jackson laboratory | 32619 | Tg(H2-K1-tsA58)6Kio/LicrmJ |
| L-Glutamine 100x | Thermofisher Scientific | 25030081 | |
| Magnetic particle concentrator | Thermofisher Scientific | 12320D | |
| mitotracker | Thermofisher Scientific | M7512 | |
| PBS 1X | Corning | 46-013-CM | |
| penecillin streptomycin 100x | Thermofisher Scientific | 10378016 | |
| PhaM mice | Jackson laboratory | 18397 | B6;129S-Gt(ROSA)26Sortm1.1(CAG-COX8A/Dendra2)Dcc/J |
| Protease (10 mg/ml) | SIGMA | P6911 | |
| RPMI | GIBCO | 3945 | |
| Sodium Pyruvate 100mM | Thermofisher Scientific | 11360070 | |
| Standard pattern forceps | F.S.T | 11000-12 | |
| Surgical Scissors – Sharp-Blunt | F.S.T | 14008-14 | |
| synaptopodin | Santa Cruz | sc-515842 | |
| Trypsin 0.05% | Thermofisher Scientific | 25300054 |
References
- Daehn, I. S., Duffield, J. S. The glomerular filtration barrier: A structural target for novel kidney therapies. Nature Reviews. Drug Discovery. 20 (10), 770-788 (2021).
- Fu, J., Lee, K., Chuang, P. Y., Liu, Z., He, J. C. Glomerular endothelial cell injury and cross talk in diabetic kidney disease. American Journal of Physiology. Renal Physiology. 308 (4), 287-297 (2015).
- Lassen, E., Daehn, I. S. Molecular mechanisms in early diabetic kidney disease: Glomerular endothelial cell dysfunction. International Journal of Molecular Sciences. 21 (24), 9456 (2020).
- Haraldsson, B., Jeansson, M. Glomerular filtration barrier. Current Opinion in Nephrology and Hypertension. 18 (4), 331-335 (2009).
- Yilmaz, O., Afsar, B., Ortiz, A., Kanbay, M. The role of endothelial glycocalyx in health and disease. Clinical Kidney Journal. 12 (5), 611-619 (2019).
- Giacco, F., Brownlee, M. Oxidative stress and diabetic complications. Circulation Research. 107 (9), 1058-1070 (2010).
- Daehn, I. S. Glomerular endothelial cell stress and cross-talk With podocytes in early [corrected] diabetic kidney disease. Frontiers in Medicine. 5, 76 (2018).
- Satchell, S. C., et al. Conditionally immortalized human glomerular endothelial cells expressing fenestrations in response to VEGF. Kidney International. 69 (9), 1633-1640 (2006).
- Wieser, M., et al. hTERT alone immortalizes epithelial cells of renal proximal tubules without changing their functional characteristics. American Journal of Physiology. Renal Physiology. 295 (5), 1365-1375 (2008).
- Jat, P. S., et al. Direct derivation of conditionally immortal cell lines from an H-2Kb- tsA58 transgenic mouse. Proceedings of the National Academy of Sciences of the United States of America. 88 (12), 5096-5100 (1991).
- Jat, P. S., Sharp, P. A. Cell lines established by a temperature-sensitive simian virus 40 large- T-antigen gene are growth restricted at the nonpermissive temperature. Molecular and Cellular Biology. 9 (4), 1672-1681 (1989).
- Israel, A., Kimura, A., Fournier, A., Fellous, M., Kourilsky, P. Interferon response sequence potentiates activity of an enhancer in the promoter region of a mouse H-2 gene. Nature. 322 (6081), 743-746 (1986).
- Mundel, P., et al. Rearrangements of the cytoskeleton and cell contacts induce process formation during differentiation of conditionally immortalized mouse podocyte cell lines. Experimental Cell Research. 236 (1), 248-258 (1997).
- Ohse, T., et al. Establishment of conditionally immortalized mouse glomerular parietal epithelial cells in culture. Journal of the American Society of Nephrology. 19 (10), 1879-1890 (2008).
- Rops, A. L., et al. Isolation and characterization of conditionally immortalized mouse glomerular endothelial cell lines. Kidney International. 66 (6), 2193-2201 (2004).
- Pham, A. H., McCaffery, J. M., Chan, D. C. Mouse lines with photo-activatable mitochondria to study mitochondrial dynamics. Genesis. 50 (11), 833-843 (2012).
- Archer, S. L. Mitochondrial dynamics–Mitochondrial fission and fusion in human diseases. The New England Journal of Medicine. 369 (23), 2236-2251 (2013).
- Casalena, G. A., et al. The diabetic microenvironment causes mitochondrial oxidative stress in glomerular endothelial cells and pathological crosstalk with podocytes. Cell Communication and Signaling. 18 (1), 105 (2020).
- Qi, H., et al. Glomerular endothelial mitochondrial dysfunction is essential and characteristic of diabetic kidney disease susceptibility. Diabetes. 66 (3), 763-778 (2017).
- Akis, N., Madaio, M. P. Isolation, culture, and characterization of endothelial cells from mouse glomeruli. Kidney International. 65 (6), 2223-2227 (2004).
- Schuler, M. H., et al. Miro1-mediated mitochondrial positioning shapes intracellular energy gradients required for cell migration. Molecular Biology of the Cell. 28 (16), 2159-2169 (2017).
- Tang, C., et al. Mitochondrial quality control in kidney injury and repair. Nature Reviews. Nephrology. 17 (5), 299-318 (2020).
- Daniel, R., Mengeta, A., Bilodeau, P., Lee, J. M. Mitochondria tether to Focal Adhesions during cell migration and regulate their size. bioRxiv. , 827998 (2019).
- Dylewski, J. F., et al. Isolation, purification, and conditional immortalization of murine glomerular endothelial cells of microvascular phenotype. MethodsX. 7, 101048 (2020).
- Drexler, H. G., Uphoff, C. C. Mycoplasma contamination of cell cultures: Incidence, sources, effects, detection, elimination, prevention. Cytotechnology. 39 (2), 75-90 (2002).

