Measuring Skeletal Muscle Thermogenesis in Mice and Rats
Summary
Mice and rats are surgically implanted with remote temperature transponders and then habituated to the testing environment and procedure. Changes in muscle temperature are measured in response to pharmacological or contextual stimuli in the home cage or during prescribed physical activity (i.e., treadmill walking at a constant speed).
Abstract
Skeletal muscle thermogenesis provides a potential avenue for better understanding metabolic homeostasis and the mechanisms underlying energy expenditure. Surprisingly little evidence is available to link the neural, myocellular, and molecular mechanisms of thermogenesis directly to measurable changes in muscle temperature. This paper describes a method in which temperature transponders are utilized to retrieve direct measurements of mouse and rat skeletal muscle temperature.
Remote transponders are surgically implanted within the muscle of mice and rats, and the animals are given time to recover. Mice and rats must then be repeatedly habituated to the testing environment and procedure. Changes in muscle temperature are measured in response to pharmacological or contextual stimuli in the home cage. Muscle temperature can also be measured during prescribed physical activity (i.e., treadmill walking at a constant speed) to factor out changes in activity as contributors to the changes in muscle temperature induced by these stimuli.
This method has been successfully used to elucidate mechanisms underlying muscle thermogenic control at the level of the brain, sympathetic nervous system, and skeletal muscle. Provided are demonstrations of this success using predator odor (PO; ferret odor) as a contextual stimulus and injections of oxytocin (Oxt) as a pharmacological stimulus, where predator odor induces muscle thermogenesis, and Oxt suppresses muscle temperature. Thus, these datasets display the efficacy of this method in detecting rapid changes in muscle temperature.
Introduction
Within metabolic research, the examination of skeletal muscle thermogenesis is a promising new avenue for probing body weight homeostasis. The published literature supports the idea that the thermogenic responses of one of the body's largest organ systems—the skeletal muscle—provide an avenue for increasing energy expenditure and other metabolic effects, thereby effectively rebalancing systems within diseases such as obesity1,2,3. If the muscle can be considered a thermogenic organ, studies must utilize a practical methodology to study thermogenic changes within this organ. The desire to understand the endothermic impact of skeletal muscles and the utility of this methodology for studying non-shivering muscle thermogenesis are not specific to metabolic studies. Disciplines including evolution4, comparative physiology5, and ecophysiology6,7 have shown a vested interest in understanding the ways in which muscle thermogenesis may contribute to endothermy and how this mechanism adapts to the environment. The presented protocol provides the critical methods necessary to address these questions.
The provided method can be utilized in the assessment of both contextual and pharmacological stimuli modulation of muscle temperature, including the unique technique of providing predator odor (PO) to shift the context to replicate predator threat. Prior reports have demonstrated the ability of PO to rapidly induce a sizable increase in muscle thermogenesis8. Moreover, pharmacological stimuli can also alter muscle temperature. This has been demonstrated in the context of PO-induced muscle thermogenesis, where pharmacological blockade of peripheral β-adrenergic receptors, using nadolol, inhibited the ability of PO to induce muscle thermogenesis without significantly affecting contractile thermogenesis during treadmill walking8. Central administration of melanocortin receptor agonists in rats has also been used to discern brain mechanisms altering thermogenesis9,10.
Provided here is a preliminary investigation of the ability of the neurohormone oxytocin (Oxt) to alter muscle thermogenesis in mice. Similar to predator threat, social encounters with a same-sex conspecific increase body temperature, a phenomenon referred to as social hyperthermia11. Given the relevance of Oxt to social behavior12, it has been speculated that Oxt is a mediator of social hyperthermia in mice. Indeed, an oxytocin receptor antagonist decreases social hyperthermia in mice11, and mouse pups lacking Oxt show deficits in behavioral and physiological aspects of thermoregulation, including thermogenesis13. Given that Harshaw et al. (2021) did not find evidence supporting β3 adrenergic receptor-dependent brown adipose tissue (BAT) thermogenesis with social hyperthermia11, it has been posited that social hyperthermia may be driven by Oxt's induction of muscle thermogenesis.
To measure skeletal muscle thermogenesis, the following protocol uses the implantation of preprogrammed IPTT-300 transponders adjacent to the muscle of interest within a mouse or rat8,10,14,15. These transponders are glass-encapsulated microchips that are read using corresponding transponder readers. Little to no research has utilized this technology in this capacity, though studies have suggested a need for the specificity provided by this method16,17. Previous investigations have shown the reliability of this method and a variety of ways in which temperature transponders can be used in comparison with other temperature-testing methods18 or in conjunction with surgical methods (e.g., cannulation19). However, studies of this nature rely on different strategic placements to measure overall body temperature20,21,22 or specified tissues such as BAT23,24,25.
Rather than measuring temperature from these locations or while using ear or rectal thermometers26, the method described here provides specificity for the muscle of interest. The ability to target a site by directly implanting transponders adjacent to the muscles of interest is more effective for probing muscle thermogenesis specifically. It provides a new avenue in addition to those provided by surface infrared thermometry27,28 or cutaneous temperature measurements via thermocouple29. Furthermore, the data provided through this method offer a range of avenues of research, avoiding the need for large, expensive, high-tech equipment and software such as infrared thermography30,31,32.
This method has been successfully used to measure temperature in the quadriceps and gastrocnemius, either unilaterally or bilaterally. This method has also been effective in conjunction with stereotaxic surgery14,15. Within ~7-10 cm of the transponder limb, portable transponder readers (DAS-8027/DAS-7007R) are used to scan, measure, and display the temperature. This distance has been critical and valuable to prior investigations8,9,10 because it minimizes potential stressors and temperature-altering variables such as animal handling during the testing procedures. Using timers, measurements can then be recorded and collected over a period of time without direct interaction with the animals.
To further minimize the disturbance of mice during testing, this method describes the assembly and use of risers made of PVC piping to give the experimenter access to the bottom of the home cages during testing. Using the risers in tandem with the digital reader, temperature measurements of the transponder limb can be made without any animal interaction after the stimulus is placed. At a minimal cost, this method can be used in conjunction with pharmacological and contextual stimuli, making it quite accessible for researchers. Additionally, this method can be employed with a substantial number of subjects (~16 mice or ~12 rats) at a time, saving time in increasing the overall throughput for any research project.
Introduced in this method is a crafted mechanism for presenting odors to mice using stainless steel mesh tea infuser balls, from now on referred to as "tea balls". Though these tea balls are ideal for containing any odor material, in these studies, towels that served as in-cage bedding over 2-3 weeks for ferrets, a natural predator of mice and rats, are placed within each treatment tea ball. Each towel is cut into 5 cm x 5 cm squares. This aliquoting is also repeated with otherwise identical odorless control towels. Presenting these odors without a barrier (i.e., tea ball) led to mice shredding the fibers within their cages, increasing physical activity. This behavior was not as salient in rats. Tea balls provide a ventilated casing to the towel, giving full access to the odor while staying protected for the entirety of the experimental trial. These tea balls can be sanitized in accordance with animal use protocols, prepared, and introduced directly after surgery to begin habituating the animals to the structure along with the control stimulus. Mice can then live with the additional enrichment, decreasing the salience of the acute stimulus presentation.
Habituation to the presence of the tea ball is only one aspect of habituation that is critical to this method. The described habituation protocol also consists of repeated exposure to the testing procedure to normalize the testing environment (i.e., personnel, transportation and movement to the testing location, exposure to stimulus). This extended habituation minimizes nuanced responses from the animals and focuses measurements on the desired dependent variables (e.g., pharmacological or contextual stimuli). Previous assessment of this protocol has identified four trials as the minimum number of habituations necessary before temperature testing within home cages in rats8. If testing is separated by long periods (more than 2-3 weeks), the animals must be habituated again. For repeated habituation, a minimum of one to two trials are sufficient. However, if temperature tests are separated by more prolonged bouts of time, repeating more trials may be necessary.
In the continued effort to accustom mice and rats to the testing procedure, an acclimation period before stimulus presentation should be included in every experimental trial. This acclimation time is critical to rebalance temperature and activity after being shifted to the testing location. Rodents tend to have sharp temperature increases due to translocation. Acclimation should consist of a minimum of 1 h without interaction from the experimenter on the day of testing before any addition of a pharmacological agent or contextual stimuli. This is necessary each day of testing.
In the outlined home-cage temperature tests, mice have the free range of their home cage to roam in response to the tested stimulus. This can cause variable shifts in activity, impacting the accuracy of temperature readings and, therefore, the analysis of the thermogenic effects of the independent variable (e.g., pharmacological or contextual stimulus). In recognition of the potential changes in temperature due to activity level, a protocol is included below describing the use of temperature during treadmill walking. The published literature describes the successful use of this procedure in rats, and it is currently being employed with mice8,10,14,15. Treadmill walking maintains a constant speed of activity for the testing subject. For this study, treadmills are strictly used to control activity level and, therefore, are set to the lowest available speed on the treadmill to promote walking for mice and a similarly low setting for rats.
The following procedure is outlined for the temperature measurement of unilateral gastrocnemius in mice and predator odor presentation. The design can be used in conjunction with pharmacological agents and is transferrable to rats and other skeletal muscle groups (i.e., quadriceps) in mice. For rats, transponders can be placed in the gastrocnemius bilaterally and in brown adipose tissue. Due to size and distance limitations, only one transponder can be used per mouse. Minor modifications (e.g., the removal of contextual stimuli) can be made to assess thermogenic responses to pharmacological agents.
Protocol
These methods can be applied to both rat and mouse models and were performed with institutional approval (Kent State University, IACUC Approval #359 and #340 CN 12-04). Prior to the implementation of the protocol, animals should be housed in conformance with the Guide for the Care and Use of Laboratory Animals.
1. Preparing the transponder reader
NOTE: Prior to use, the transponder reader must be set; the following steps only include the setting changes necessary for this study. This portion of the protocol is directly associated with the DAS-8027-IUS portable readers; other reader models should follow instructions provided by the manual to achieve programming results.
- Set Audio Beep 세스 OFF.
- Turn on the device by pressing the SCAN button and wait for the lighting to appear on the OLED display. Press and hold the BACK/MENU button to get to the menu screen.
- Using the NEXT/ENTER button, scroll through the options until OPERATIONAL SETUP. Here, toggle the up or down arrows to turn YES and open the operational submenu.
- Using the NEXT/ENTER button, scroll to AUDIO BEEP. As the default setting is ON, toggle the up or down arrows and change the setting to OFF.
- Press the NEXT/ENTER button to save this setting change.
- Set Vibrate Upon Read 세스 ON.
- Follow step 1.1 through step 1.2 or complete the next step directly after step 1.4.
- Using the NEXT/ENTER button, scroll to VIBRATE UPON READ. As the default setting is OFF, toggle the up and down arrows and change the setting to ON to feel, via vibration, when the reading has been completed regardless of being able to view the screen.
2. Program transponders
NOTE: Each implanted transponder should first be programmed with an animal identification (animal ID or transponder ID). This nomenclature can be used as secondary identification for the test subject (e.g., four digits for mouse strain abbreviation, location of transponder, and an additional three to four digits to indicate animal number). Programming can be completed days in advance of surgery while keeping the transponders sterile prior to surgery.
- Enter the ID Code on the transponder.
- Apply a booster coil to the reader head—a specific accessory for model DAS 8027-IUS, which helps in the programming procedure.
- Using a gloved hand, place the transponder (within the applicator) into the booster coil.
- Turn on the device by pressing the SCAN button and wait for the OLED display to light up. Press and hold the BACK/MENU button to get to the menu screen.
- Using the NEXT/ENTER button, scroll through the options until WRITE TRANSPONDER ID. Here, toggle the up or down arrows to turn YES.
- Using the NEXT/ENTER button, toggle to ENTER ID CODE.
- Use the up and down arrow keys to scroll through numbers and letters. Press NEXT/ENTER after each character selection to move to the following character.
- When the ID code is complete, press SCAN to write the transponder.
- Remove the transponder from the booster coil and repeat as necessary. Check that the transponder reads temperature changes by warming the enclosed transponders between gloved hands and measuring using the temperature scanner.
NOTE: AUTO MULTI WRITE and SEQUENTIAL COUNT settings can be set to ON to allow for multiple or sequential transponder programming during a session. Each transponder should be tested during programming.
3. Prepare "home cage balls"
- Place 5 cm x 5 cm odorless/control towel into a tea ball.
- Place these home cage balls in new home cages after surgery to begin habituating the animal to the method in which the contextual stimuli will be presented during testing. Replace these home cage balls every 2 weeks.
4. Surgery and postoperative care
- Weigh and record the subjects' pre-surgery body weight. Using an induction chamber, provide anesthesia (e.g., 2-5% isoflurane) to the animal.
- Using electric clippers, completely shave the hind limb. Administer analgesia (e.g., 5 mg/kg of ketoprofen, s.c.) in compliance with institutional guidelines.
NOTE: Additional analgesia may be required if this procedure is combined with other surgical methods. - Clean the area with 70% alcohol (or commercially available sterile alcohol wipe) and povidone-iodine wash (or commercially available sterile, individually wrapped betadine swabs) alternating at least three times, ending with povidone-iodine.
- Return the animal to the induction chamber, and anesthetize the animal to surgical levels. Then, set up the mouse in a face mask for continued exposure to anesthesia. Apply neomycin ophthalmic ointment to the eyes of the animal to prevent dryness while under anesthesia.
NOTE: The procedure should not start until the mouse shows no evidence of pain reception (i.e., corneal reflex, tail pinch response, toe pinch reflex). - Using only surgical scissors, make a shallow cut through the skin on the right hind limb.
- Moving parallel to the gastrocnemius, place the sharp edge of a preprogrammed and uncapped sterile transponder into the incision. Ensure that the green plunger faces up and is visible. Continue pushing the transponder applicator into the incision until the opening of the transponder applicator is no longer visible.
NOTE: Do not accidentally press the green plunger on the transponder applicator during step 4.6. Premature discharge of the transponder will lead to improper placement. - Turn the applicator 180°, resulting in the green plunger facing down toward the mouse's limb, no longer visible to the experimenter. Push the transponder applicator into the final location. Once in ideal placement, adjacent to or partially enclosed in the gastrocnemius, push the green plunger, allowing the applicator's pressure to guide the investigator's hand back away from the mouse.
- Using forceps, hold together the opened skin and place a wound clip with a sterile autoclip or sterile suture. If needed, use absorbable sutures prior to the sterile autoclip to close the fascia layer. Using the transponder-reader, check the temperature of the mouse muscle.
- Remove the mouse from anesthesia and place it in a clean home cage placed atop a water-circulating heating pad set to low for recovery. Ensure that the home cage includes a tea ball with an odorless towel to begin habituation.
NOTE: The mouse should awaken from surgery within 15 min. Food can be placed at the bottom of the cage for easy access during recovery days. - Postoperative care
- Record mouse weights and temperatures daily using a transponder-reader for at least 2 days after surgery or until mice regain or stabilize body weight.
- Administer non-narcotic analgesia (e.g., 5 mg/kg of ketoprofen, s.c.) once daily to the mice for at least 2 days post surgery, with additional doses provided as needed.
NOTE: Mice and rats should fully recover within 5-8 days of surgery and can undergo habituation and testing procedures.
5. Testing preparation – home cage
- Constructing risers
NOTE: The below step is based on 194 mm x 181 mm x 398 mm mouse filter-topped cages. To fit larger cages (e.g., a rat home cage), the width will need to be adjusted.- Cut the PVC pipe with a ratcheting PVC cutter into eight sections and assemble following Figure 1C. This will give an open tabletop structure that can hold approximately four cages. Make the desired number of risers.
- Room setup
- Assign a location to each riser within the testing room. Separate the risers set to receive different contextual stimuli (i.e., odors) by a minimum of 2 m to avoid confounding variables.
NOTE: Each mouse should have an assigned testing location within the testing room and on the physical risers as much as is feasible to avoid developing associations between different locations and thermogenic stimuli. - Using magnetic strips, attach surgical sheets or gowns across the risers, creating a visual barrier between the researcher and the test subjects. Set this barrier to minimize temperature changes resulting from mouse activity when viewing experimenters moving toward the cage or around the testing room.
- (Optional) Place mirrors on the surface below the risers to ease viewing of the cage bottom during testing.
NOTE: Risers can be sanitized through a cage wash system. Cloth or surgical sheets should be laundered prior to habituation and testing.
- Assign a location to each riser within the testing room. Separate the risers set to receive different contextual stimuli (i.e., odors) by a minimum of 2 m to avoid confounding variables.
- Tea ball preparation
- Prepare tea balls with control and PO towels (approximately 5 cm x 5 cm). To avoid cross-contamination, prepare control-odor tea balls first.
NOTE: Predator-odor towels should be pathogen-tested prior to use. These towels should also be contained, and materials that interact with them should be immediately sanitized (i.e., cage wash), preventing exposure of the odor to other animals.
- Prepare tea balls with control and PO towels (approximately 5 cm x 5 cm). To avoid cross-contamination, prepare control-odor tea balls first.
6. Temperature testing – home cage
NOTE: Animals need to be habituated to the entire testing procedure, excluding experimental contextual, or pharmacological stimuli. This should be completed a minimum of 4x before testing.
- Transfer the animals to the prepared testing room. Place the animals in a preassigned location on the riser. This location should be the same throughout all habituation and testing procedures.
- Remove the "home cage ball" from the mouse home cage and re-cover the cages with a cloth or surgical sheet. Allow the mice to acclimate to the testing space for 1-2 h.
- After acclimation is complete, use the scanner to measure and record the baseline temperature of each subject. Avoid manipulating the cloth coverings during measurements.
NOTE: Pharmacological agents can be applied here. Wait time post injection or application can be added as needed before testing. Recording a secondary baseline directly before testing is recommended after the addition of a pharmacological agent to monitor the response to pharmacological stimuli. If odor response is not being tested, temperature measurements of the mice can begin directly after injection. Randomization should be employed when providing any stimuli. - Uncover the cage and place the tea ball (control or PO) onto the floor of the home cage. Replace the cage lid and cloth covering.
- Begin the stopwatch. Measure the temperatures of the test subjects in the same order of tea ball placement. Record temperatures and clock time of measurements following the desired time points.
- When the experiment is complete, remove the treatment ball. Place the mice that received PO in a new home cage with the original "home cage ball". Return the "home cage ball" to the cage of the mice that received control odor. Transfer the mice to the housing location.
NOTE: The above procedure can be translated to rat models within appropriately sized cages. Adjustments to the measurements suggested in Figure 1C may be required to allow for better access to the bottom of the home cage.
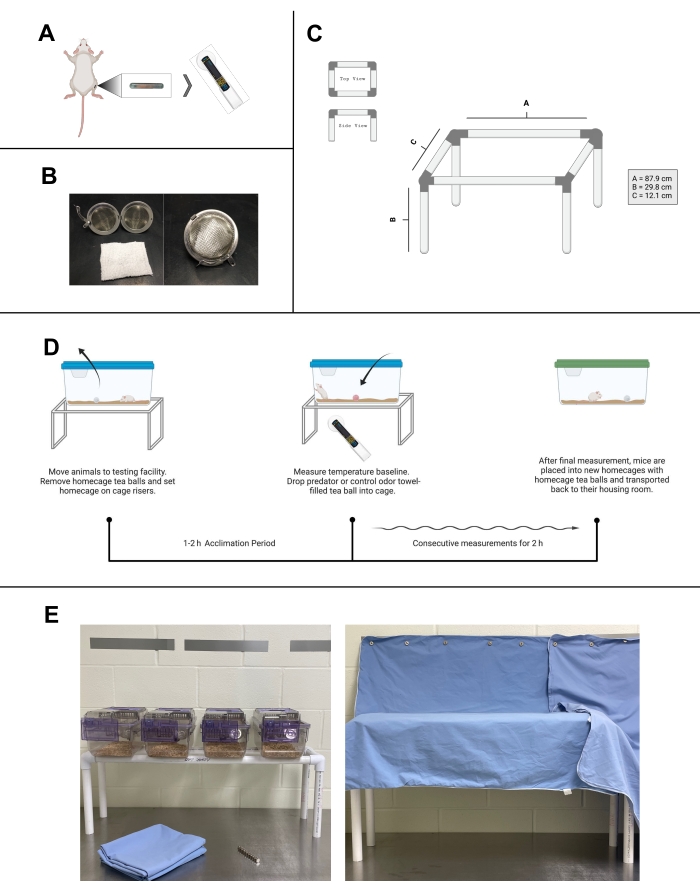
Figure 1: Transponders and home cage temperature testing. (A) Diagram of unilateral transponder placement for testing temperature in a mouse gastrocnemius. Once programmed and placed, the transponder-reader (DAS-8027-IUS, shown) can be used to measure temperature. (B) Left, photo of an open mesh stainless steel tea ball and a 5 cm x 5 cm towel. Right, enclosed tea ball, used to hold habituation and odor towels in home cage testing. (C) Schematic of risers constructed with PVC piping for home cage testing. (D) Workflow of home cage testing protocol. (E) Facility images of home cage testing area. Left, four mouse cages atop a riser. Magnetic strips are located on the adjacent wall, and magnets and surgical cloth are on the table. Right, covered mouse cages on risers. (A), (C), and (D) were created with Biorender.com. Please click here to view a larger version of this figure.
7. Temperature testing – treadmill walking
- Assign each animal a treadmill as their assigned location for habituation and testing procedures.
- Prepare the treadmills for testing, ensuring that the shockers are functional.
NOTE: For treadmill walking, treadmills should be set at the lowest available pace that promotes continuous movement but not running for both habituation and testing. For the 1012M-2 Modular Enclosed Metabolic Treadmill, this is 5.2 m/min for mice and 7 m/min for rats. This pace may need to be adjusted based on the obesity of the subject. Shockers should be set to an intensity and repetition rate of 5.0. - Habituation
- Move the mice to the testing room. Allow mice 1-2 h to acclimate to the room transfer in their home cages.
- After acclimation, guide the mice to the opening of their assigned treadmill and close the treadmill. Start the belt, shocker, and stopwatch.
- Allow the mice to walk on the treadmills for 15 min, using shock stimulus as motivation for movement. Stop the test immediately if an animal remains on an active shocker for an extended period.
- After the test, remove the mice and return them to the home cages.
- Clean the treadmills using liquid detergent and water.
- Testing
- Move the mice to the testing room. Allow the mice 1-2 h to acclimate to the room transfer in their home cages.
- Measure and record baseline temperature prior to moving the mouse to the treadmill.
NOTE: For tests including pharmacological agents, apply or inject them here, following the schematic shown in Figure 2A. Wait time after injection can be added as needed before the mice are placed on the treadmill. Randomization should be employed when providing any stimuli. - Place 5 cm x 5 cm squares of control or PO towels within the treadmill closest to the front of the treadmill. Adhere the towels to the ceiling of the treadmill or underneath for easy placement and removal.
- Guide the mice into the assigned treadmill. Turn on the treadmill belt and shocker.
- Start the stopwatch. Take measurements of the test subjects in the same order in which the mice were set up in the treadmills. Record the temperatures and clock time of the measurements following the desired time points.
NOTE: Temperature can reliably be measured from outside the treadmill while a mouse is inside an enclosed treadmill during walking activity. For rats, the treadmill size and transponder-reader distance limitations may require an experimenter to keep the back of the treadmill open to insert the reader inside the treadmill, closer to the subject. - When the test is complete, turn off the shockers and treadmills; return the mice to their home cages. Transfer the mice to the housing location.
- Clean the treadmills using liquid detergent and water, paying specific attention to remove any residual PO.
- When experiments are complete, euthanize the animals (e.g., using CO2 inhalation), and visually confirm the transponder location.
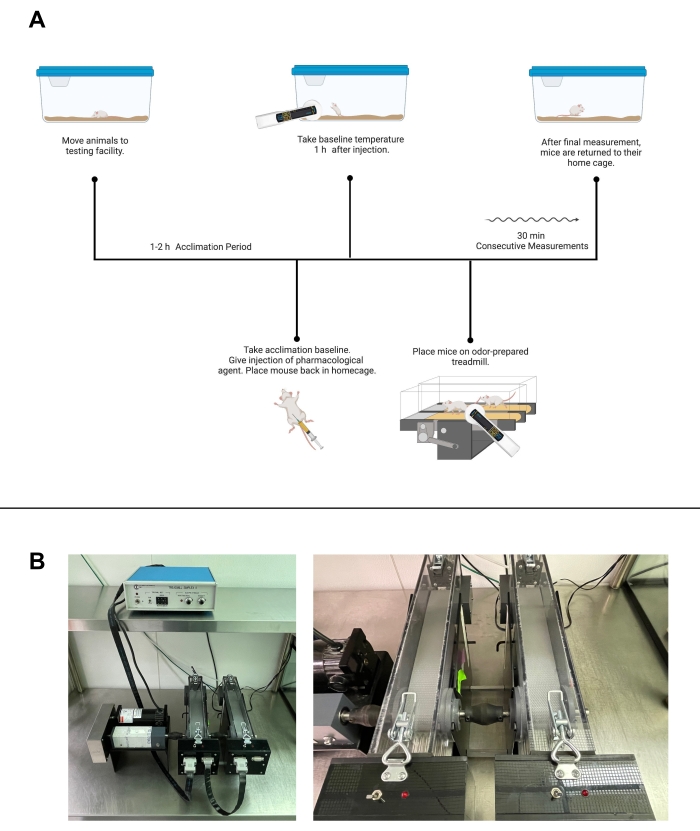
Figure 2: Activity-controlled temperature testing. (A) Workflow of activity-controlled temperature testing with a pharmacological agent using treadmill walking. (B) Facility images of treadmills. Left, an image of full equipment setup. Right, a closer image of individual treadmills and shockers. (A) was created with Biorender.com. Please click here to view a larger version of this figure.
Representative Results
Transponders were unilaterally implanted into the right gastrocnemius of ten 4-6-month-old, wild-type (WT) mice bred from the SF1-Cre strain (Tg(Nr5a1-cre)7Lowl/J, Strain #012462, C57BL/6J and FVB backgrounds; female N = 5; male N = 5). After recovery, the mice were habituated to a home cage temperature-testing procedure that did not include a contextual stimulus (e.g., PO). Temperature measurements using a transponder wand were recorded within their housing room and after transfer to the testing location. Mice were given 1-2 h to acclimate to the testing room and location. At the completion of acclimation, baseline and consecutive measurements for 1 h were recorded for each mouse. This procedure was completed four times.
Overall, no sex differences were observed. Muscle temperatures significantly increased after the mice were moved to the testing room, then decreased by the baseline measurement after 60 min spent in the testing context. The combined-sex analysis of trial 4 showed no significant difference between "before move" and "baseline" temperature measurements (two-tailed, paired t-test, p > 0.10), showing the effectiveness of 1 h acclimation to the testing context. Furthermore, statistical comparison of the temperatures at baseline and 60 min showed a significant decrease in temperature (two-tailed, paired t-test, p < 0.01), providing evidence of the mice habituating to the investigator's movement during measurement. However, females (but not males) showed incremental responses where temperature measured from 5 min to 15 min was lower with successive habituation trials (Figure 3). When observing the acute effects of moving or rises in temperature after baseline, mice tend to respond less to transport into the testing room over successive habituation trials (Supplementary File 1, trial analysis).
Habituated adult WT mice described above were tested with Oxt, a pharmacological agent. Mice were given intraperitoneal injections (i.p., 2 mg/kg) of Oxt or vehicle (sterile saline) in a random order, and muscle temperatures were measured prior to movement into the testing room and after 5, 10, 15, 30, 45, 60, 90, 120, 150, and 180 min of injection. Each mouse received both treatments. A repeated-measures analysis of variance (ANOVA) revealed significant main effects of Oxt and time, where Oxt decreased muscle temperature relative to the vehicle. Oxt decreased muscle temperature relative to baseline as rapidly as 5 min after injection, with a maximal decrease seen 30 min after injection (Figure 4). Muscle temperatures were normalized by 60 min after Oxt injection (two-tailed, paired t-test, p > 0.10).
Adult male Sprague-Dawley rats (N = 4, age ~6 months) bilaterally implanted with transponders in the gastrocnemius were habituated and then tested in a home cage setting with a PO (ferret odor) stimulus. Baseline measurements were recorded, and each rat was presented with PO in the form of a towel. The odor was then removed after 10 min of exposure; consecutive measurements were taken before and after the removal of the stimulus. These preliminary data (Figure 5) suggest that PO has a continued impact on skeletal muscle thermogenesis after the removal of the stimulus.
Previously published data assessed predator threat activation of skeletal muscle thermogenesis in adult male Sprague-Dawley rats (age ~6 months)8. Rats with implanted bilateral gastrocnemius transponders were presented with predator (ferret) odor. Measurements were taken in a home cage setting (N = 8, Figure 6A). These data revealed a robust increase in temperature compared to control odors. To parse out aversive or stressful thermogenic responses to ferret odor, male rats (N = 7, Figure 6B) were presented with an aversive odor (butyric acid), a novel odor (2-methylbenzoxazole), or a fox odor, or restrained for 1 min prior to testing (moderate stress). Measurements were taken in a home cage setting over a 2 h period. Analysis of these data showed ferret odor to produce and maintain a strong change in thermogenesis compared to all other conditions. Together, these data provide evidence of the control odor's minimal and transient influence on skeletal muscle thermogenesis.
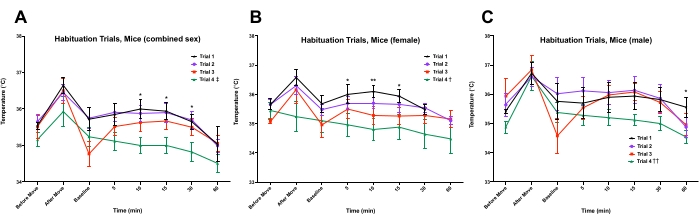
Figure 3: Analysis of muscle temperature during habituation for home cage temperature testing. Mice unilaterally implanted with transponders in the right gastrocnemius were habituated to the testing procedure. Mice were measured in the animal housing room, "Before Move", in the testing room, "After Move", after acclimation for 1-2 h, "Baseline", then consecutively over 1 h. All statistical comparisons shown were made between trial 1 and trial 4, * p < 0.05, ** p < 0.01 (t-test, N = 10); † p < 0.05, †† p < 0.01, ‡ p < 0.001 main effect trial (ANOVA, N [trials] = 4). Error bars shown display the standard error of the mean (SEM). Please click here to view a larger version of this figure.
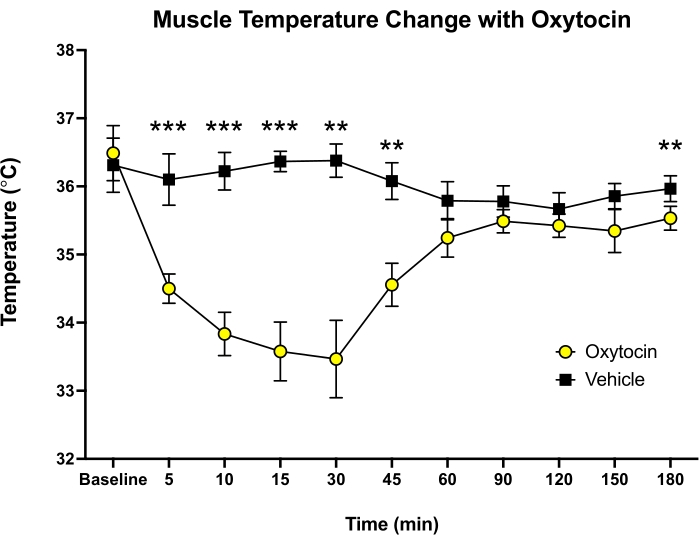
Figure 4: Muscle temperature during pharmacological stimulation of oxytocin in mice. Habituated mice, unilaterally implanted with transponders, were given 2 mg/kg (i.p.) of either oxytocin or vehicle (sterile saline). Significant decreases in muscle temperature were observed at 5 min after injection of oxytocin and normalized by 60 min, ** p < 0.01, *** p < 0.001 (two-tailed paired t-test, N = 9). Error bars shown display the standard error of the mean. Please click here to view a larger version of this figure.
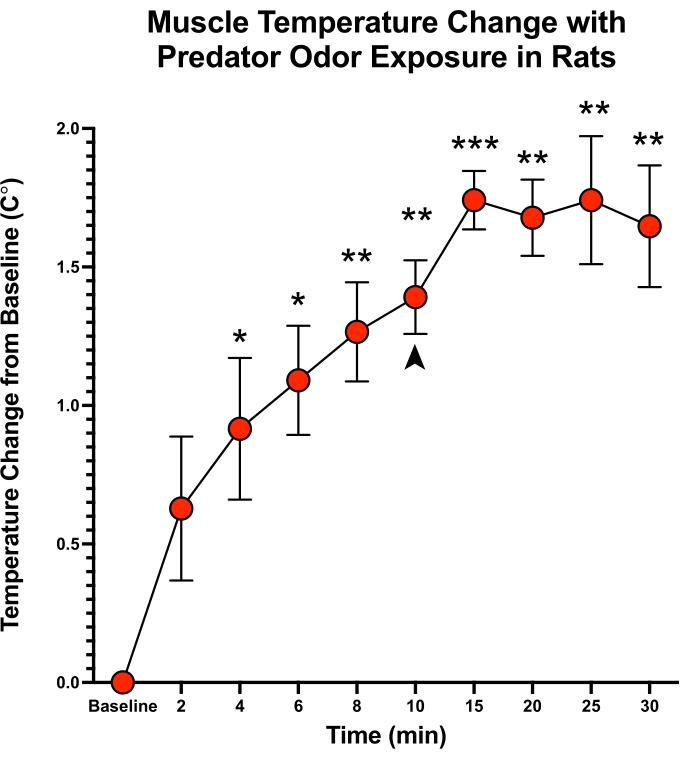
Figure 5: Predator-odor thermogenesis in rat home cage temperature testing. Temperature measurements in rats with transponders implanted bilaterally in the gastrocnemius after exposure to predator (ferret) odor for 10 min. After exposure for 10 min, towels containing the stimulus were removed, as indicated by the arrow. Rats maintained increased temperature 20 min after stimulus removal. Significantly greater than baseline temperature, * p < 0.05, ** p < 0.01, *** p < 0.001 (t-test, N = 4). Error bars shown display the standard error of the mean. Please click here to view a larger version of this figure.
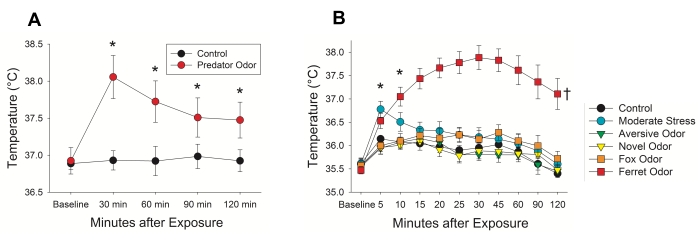
Figure 6: Ferret odor induces a rapid rise in muscle temperature compared to control. (A) Gastrocnemius temperature was significantly elevated after predator (ferret) odor compared with control exposure in male rats (two-tailed paired t-test, N = 8). (B) Novel, aversive, or fox odors did not significantly change muscle temperature compared to control. Temperature change induced by moderate stress quickly declined after 5 min. Ferret odor maintained a robust response, in comparison to other conditions, for the entirety of the test (ANOVA, N = 7). † p < 0.05, ferret odor > all other conditions; * p < 0.025, point comparison between ferret odor and moderate stress vs. control odor. This figure was modified with permission from Gorrell et al.8. Please click here to view a larger version of this figure.
Supplementary File 1: R markdown for Figure 3 habituation analysis. Markdown file for habituation analysis with R code shows example coding methods and ways in which sex can be probed within data. Please click here to download this File.
Discussion
This temperature testing protocol provides the field with an avenue to measure skeletal muscle thermogenesis directly. This is critical as research delves into identifying the mechanisms underlying muscle thermogenesis33. The method provides two cost-effective protocols for measuring skeletal muscle thermogenesis under contextual and pharmacological conditions. This protocol emphasizes the importance of both habituation and acclimation within these procedures. Habituation is used to repeatedly introduce the test subject to the testing procedure without the introduction of any pharmacological or contextual stimuli; it is a critical component of both home cage and treadmill temperature testing. This gives time for the animals to familiarize themselves with the environment while decreasing the salience of the experimental context. Omitting this step can lead to biased associations with the experimental stimulus, as well as elevated thermogenic responses to control stimuli8. Animals must learn the procedure to reduce stress responses to the general movement and manipulation required to test animals under these protocols. The example data collected provides evidence of the necessity of repeated habituation (Figure 3). In a similar effort, acclimation on the day of testing is necessary for each trial. Acclimation is a daily assimilation tool, giving the animals time to relax from the stressors of translocation to the testing room. Skipping acclimation can give inaccurate baseline temperature measurements, interreacting with any later assessment.
Here, muscle thermogenic measurements were used to demonstrate the hypothermic effect of intraperitoneal Oxt on mice. This outcome was surprising considering evidence supporting central Oxt's role in thermogenesis and, specifically, in social hyperthermia11,13. Others, however, have demonstrated the ability of both Oxt and vasopressin to suppress core temperature along with heart rate in rats, effects mediated by the Avpr1a receptor34. This apparent paradox has not been reconciled. It is possible that the ability of Oxt to increase or decrease the temperature in different contexts may stem from central versus peripheral action of Oxt or from the length of exposure13,35,36,37. Regardless, here, we demonstrate that mouse muscle temperature shows a sizable decrease in temperature rapidly after peripheral Oxt injection (Figure 4), consistent with the changes in rat core temperature reported by Hicks et al. (2014)33.
In accordance with the National Institute of Health's (NIH) expectation that investigators factor in sex as a biological variable, muscle thermogenesis is measured in males and females in both mice and rats. Thermogenesis data from males and females can be compared, though previous and current studies have failed to identify robust sex differences in contextual thermogenesis and variation across the estrous cycle in female rats8. One exception is the evident sex difference in muscle temperature at baseline and after transport to the testing area, particularly prior to habituation8. This may stem from differences in locomotion after transport, as female rats have a higher locomotor response to some stressful stimuli compared to males, separable from underlying anxiety measures38. This underscores the need for repeated habituation to the experimental context, in this case, to avoid misrepresenting a sex difference in thermogenesis that may be attributed to the experimental stimulus rather than underlying differences in the stress response.
The primary method of temperature testing within animal home cages has some limitations, one being the control of variable activity levels. This can be critical as increased activity leads to increased muscle temperature. To address this, a procedure for mice and rat treadmill walking has been outlined. Controlling the animal's movement minimizes the potential for an activity effect on the temperature, factoring out differences in contractile thermogenesis. While treadmill walking can be completed as a solo test, this method can be used in conjunction with home-cage temperature assessment. The combined analysis provides further evidence for assertions that skeletal muscle temperature changes stem from pharmacological or contextual stimuli rather than secondarily from changes in activity resulting from these stimuli8,14,15. Additionally, this method is limited in that it is mildly invasive, which does not meet the need of some research studies. However, this method only requires a single surgery, allowing researchers to avoid continual animal manipulation during testing while maintaining the specificity of the measurements. Furthermore, the currently available size of the IPTT-300 transponder does not allow the transponder to be placed directly within the gastrocnemius of a mouse. This can be completed within rat models due to their larger size. This method provides a mechanism of measurement adjacent to the muscle of interest; nevertheless, remodeled or smaller versions of transponders capable of measuring temperature would be a great asset to the field and future studies.
The broad use of the described method in our research program has given us the opportunity to manage variance in response to transponder implantation and testing procedures8,10,14,15. After implantation of the transponder, monitoring the temperatures of the animals immediately after surgery and during recovery is recommended. While this first gives insight into the animal's health (e.g., oddly low temperature as a sign of illness or impending fatality), it also provides evidence of the transponder still being active and secured in place. A rat or mouse may scratch at the incision location, potentially resulting in the transponder either partially or completely falling out. In compliance with institutional guidelines, this surgery is considered minor. Therefore, in cases of unilateral transponder placement, if a mouse loses their transponder or if the mouse's transponder no longer functions, the surgery can be repeated on an alternate limb. A marking (i.e., identification of new placement, or "R" for replacement) to indicate this repeated surgery noted during the programming of the transponder as a part of the animal identification name is recommended. Furthermore, since animals have free range of their cage, researchers may have difficulty finding the animal to take the reading. It is suggested that researchers utilize the habituation phase to practice measurements and assess their setup. Alterations may include increasing the number of experimenters and transponder scanners or decreasing the number of risers and, therefore, animals tested in each trial.
This protocol provides instruction for the direct temperature measurement of muscle without additional software analysis, resulting in a feasible and relatively low-cost avenue for studies where infrared cameras are typically used. Moreover, this procedure enables the collection of data that close the gap seen by some studies seeking to connect gene or protein changes to muscle thermogenesis37. All in all, increased interest in muscle thermogenesis and its mechanisms are facilitated by direct assessment of the heat generated in the target muscle. The described procedure directly addresses this methodological void within the field by providing a mechanism for studying the skeletal muscle of both mice and rats.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work is supported by R15 DK097644 and R15 DK108668. We thank Dr. Chaitanya K Gavini and Dr. Megan Rich for prior contributions and Dr. Stanley Dannemiller for ensuring our compliance with institutional animal use guidelines. A special thank you to Dr. Tim Bartness for providing the fundamental research necessary to build this method and its associated studies. Figures 1A, C, D and Figure 2A were created using Biorender.com.
Materials
| 1012M-2 Modular Enclosed Metabolic Treadmill for Mice, 2 Lanes w/ Shock | Columbus Instruments | ||
| 1012R-2 Modular Enclosed Metabolic Treadmill for Rats, 2 Lanes w/ Shock | Columbus Instruments | ||
| 1-1/4 in. Ratcheting PVC Cutter | BrassCraft | ||
| 1 mL Syringes | Fisher Scientific | BD 309659 | |
| Betadine Swabs | Fisher Scientific | 19-898-945 | |
| Booster Coil | BioMedic Data Systems | Transponder Accessory | |
| Electric Clippers | Andis | 40 Ultraedge Clipper Blade | |
| Flexible Mirror Sheets | Amazon | Self Adhesive Non Glass Mirror Tiles | |
| Forceps | Fisher Scientific | 89259-940 | |
| Heating Pad | |||
| Induction Chamber (isoflurane) | Kent Scientific | VetFlo-0730 | 3.0 L Low Cost Chambers for Traditional Vaporizers |
| Ketoprophen | Med-Vet Intl. | RXKETO-50 | |
| Magnetic Strips | Amazon | ||
| Magnets | Amazon | DIYMAG Magnetic Hooks 40lbs | |
| Needles | Med-Vet Intl. | 26400 | |
| Neomycin/Polymixin/Bacitracin with Hydrocortisone Ophthalmic Ointment, 3.5 g | Med-Vet Intl. | RXNPB-HC | |
| Oasis Absorbable Suture | Med-Vet Intl. | MV-H821-V | |
| Predator (Ferret) Odor Towels | Marshall BioResources | ||
| PVC pipe | |||
| Reflex Wound Clip Remover | CellPoint Scientific | ||
| Reflex Wound Clip, 7 mm (mouse) | CellPoint Scientific | ||
| Reflex Wound Clip, 9 mm (rat) | CellPoint Scientific | ||
| Srerile Autoclip, 7 mm (mouse) | CellPoint Scientific | Wound Clip Applier (mouse) | |
| Stainless Strainers Interval Seasonings Tea Infuser | Amazon | ||
| Sterile Autoclip, 9 mm (rat) | CellPoint Scientific | Wound Clip Applier (rat) | |
| Sterile Saline | Med-Vet Intl. | RX0.9NACL-10 | |
| Surgical Scissors | Fisher Scientific | 08-951-5 | |
| Surgical Sheets | |||
| Towels (Control/Habituation) | Amazon | 100% Cotton Towels, white | |
| Transponders | BioMedic Data Systems | Model: IPTT-300 | |
| Transponders Reader | BioMedic Data Systems | Model: DAS-8027-IUS/ DAS-7007R | |
| Versaclean | Fisher Scientific | 18-200-700 | liquid detergent |
| Webcol Alcohol Preps | Covidien | 22-246-073 | |
| Wedge pieces for PVC pipe |
References
- Periasamy, M., Herrera, J. L., Reis, F. C. G. Skeletal muscle thermogenesis and its role in whole body energy metabolism. Diabetes Metabolism Journal. 41 (5), 327-336 (2017).
- Rowland, L. A., Bal, N. C., Periasamy, M. The role of skeletal-muscle-based thermogenic mechanisms in vertebrate endothermy. Biological Reviews of the Cambridge Philosophical Society. 90 (4), 1279-1297 (2015).
- Maurya, S. K., et al. Sarcolipin is a key determinant of the basal metabolic rate, and its overexpression enhances energy expenditure and resistance against diet-induced obesity. Journal of Biological Chemistry. 290 (17), 10840-10849 (2015).
- Grigg, G., et al. Whole-body endothermy: Ancient, homologous and widespread among the ancestors of mammals, birds and crocodylians. Biological Reviews of the Cambridge Philosophical Society. 97 (2), 766-801 (2022).
- Franck, J. P. C., Slight-Simcoe, E., Wegner, N. C. Endothermy in the smalleye opah (Lampris incognitus): A potential role for the uncoupling protein sarcolipin. Comparative Biochemistry and Physiology – Part A: Molecular & Integrative Physiology. 233, 48-52 (2019).
- Nowack, J., et al. Muscle nonshivering thermogenesis in a feral mammal. Scientific Reports. 9, 6378 (2019).
- Oliver, S. R., Anderson, K. J., Hunstiger, M. M., Andrews, M. T. Turning down the heat: Down-regulation of sarcolipin in a hibernating mammal. Neuroscience Letters. 696, 13-19 (2019).
- Gorrell, E., et al. Skeletal muscle thermogenesis induction by exposure to predator odor. The Journal of Experimental Biology. 223, (2020).
- Gavini, C. K., et al. Leanness and heightened nonresting energy expenditure: Role of skeletal muscle activity thermogenesis. The American Journal of Physiology – Endocrinology and Metabolism. 306 (6), 635-647 (2014).
- Almundarij, T. I., Gavini, C. K., Novak, C. M. Suppressed sympathetic outflow to skeletal muscle, muscle thermogenesis, and activity energy expenditure with calorie restriction. Physiological Reports. 5 (4), 13171 (2017).
- Harshaw, C., Lanzkowsky, J., Tran, A. D., Bradley, A. R., Jaime, M. Oxytocin and ‘social hyperthermia’: Interaction with beta3-adrenergic receptor-mediated thermogenesis and significance for the expression of social behavior in male and female mice. Hormones and Behavior. 131, 104981 (2021).
- Caldwell, H. K. Oxytocin and vasopressin: Powerful regulators of social behavior. The Neuroscientist. 23 (5), 517-528 (2017).
- Harshaw, C., Leffel, J. K., Alberts, J. R. Oxytocin and the warm outer glow: Thermoregulatory deficits cause huddling abnormalities in oxytocin-deficient mouse pups. Hormones and Behavior. 98, 145-158 (2018).
- Gavini, C. K., Britton, S. L., Koch, L. G., Novak, C. M. Inherently lean rats have enhanced activity and skeletal muscle response to central melanocortin receptors. Obesity. 26 (5), 885-894 (2018).
- Gavini, C. K., Jones, W. C., Novak, C. M. Ventromedial hypothalamic melanocortin receptor activation: regulation of activity energy expenditure and skeletal muscle thermogenesis. The Journal of Physiology. 594 (18), 5285-5301 (2016).
- Zaretsky, D. V., Romanovsky, A. A., Zaretskaia, M. V., Molkov, Y. I. Tissue oxidative metabolism can increase the difference between local temperature and arterial blood temperature by up to 1.3(o)C: Implications for brain, brown adipose tissue, and muscle physiology. Temperature. 5 (1), 22-35 (2018).
- Yoo, Y., et al. Exercise activates compensatory thermoregulatory reaction in rats: A modeling study. Journal of Applied Physiology. 119 (12), 1400-1410 (2015).
- Langer, F., Fietz, J. Ways to measure body temperature in the field. Journal of Thermal Biology. 42, 46-51 (2014).
- Pence, S., et al. Central apolipoprotein A-IV stimulates thermogenesis in brown adipose tissue. International Journal of Molecular Sciences. 22 (3), 1221 (2021).
- Li, D., et al. Homeostatic disturbance of thermoregulatory functions in rats with chronic fatigue. Journal of Neuroscience Research. 165, 45-50 (2021).
- Carlier, J., et al. Pharmacodynamic effects, pharmacokinetics, and metabolism of the synthetic cannabinoid AM-2201 in male rats. Journal of Pharmacology and Experimental Therapeutics. 367 (3), 543-550 (2018).
- Pato, A. M., Romero, D. M., Sosa Holt, C. S., Nemirovsky, S. I., Wolansky, M. J. Use of subcutaneous transponders to monitor body temperature in laboratory rats. Journal of Pharmacological and Toxicological Methods. 114, 107145 (2022).
- Almeida, D. L., et al. Lean in one way, in obesity another: Effects of moderate exercise in brown adipose tissue of early overfed male Wistar rats. International Journal of Obesity. 46 (1), 137-143 (2022).
- Brito, M. N., Brito, N. A., Baro, D. J., Song, C. K., Bartness, T. J. Differential activation of the sympathetic innervation of adipose tissues by melanocortin receptor stimulation. Endocrinology. 148 (11), 5339-5347 (2007).
- Vaughan, C. H., Shrestha, Y. B., Bartness, T. J. Characterization of a novel melanocortin receptor-containing node in the SNS outflow circuitry to brown adipose tissue involved in thermogenesis. Brain Research. 1411, 17-27 (2011).
- Kort, W. J., Hekking-Weijma, J. M., TenKate, M. T., Sorm, V., VanStrik, R. A microchip implant system as a method to determine body temperature of terminally ill rats and mice. Laboratory Animals. 32 (3), 260-269 (1998).
- Mei, J., et al. Body temperature measurement in mice during acute illness: Implantable temperature transponder versus surface infrared thermometry. Scientific Reports. 8, 3526 (2018).
- Warn, P. A., et al. Infrared body temperature measurement of mice as an early predictor of death in experimental fungal infections. Laboratory Animals. 37 (2), 126-131 (2003).
- Hargreaves, K., Dubner, R., Brown, F., Flores, C., Joris, J. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain. 32 (1), 77-88 (1988).
- Fiebig, K., Jourdan, T., Kock, M. H., Merle, R., Thone-Reineke, C. Evaluation of infrared thermography for temperature measurement in adult male NMRI nude mice. Journal of the American Association for Laboratory Animal Science. 57 (6), 715-724 (2018).
- Franco, N. H., Geros, A., Oliveira, L., Olsson, I. A. S., Aguiar, P. ThermoLabAnimal – A high-throughput analysis software for non-invasive thermal assessment of laboratory mice. Physiology & Behavior. 207, 113-121 (2019).
- Koganti, S. R., et al. Disruption of KATP channel expression in skeletal muscle by targeted oligonucleotide delivery promotes activity-linked thermogenesis. Molecular Therapy. 23 (4), 707-716 (2015).
- Bal, N. C., Periasamy, M. Uncoupling of sarcoendoplasmic reticulum calcium ATPase pump activity by sarcolipin as the basis for muscle non-shivering thermogenesis. Philosophical Transactions of the Royal Society B. 375 (1793), 20190135 (2020).
- Hicks, C., et al. Body temperature and cardiac changes induced by peripherally administered oxytocin, vasopressin and the non-peptide oxytocin receptor agonist WAY 267,464: a biotelemetry study in rats. British Journal of Pharmacology. 171 (11), 2868-2887 (2014).
- Kasahara, Y., et al. Oxytocin receptor in the hypothalamus is sufficient to rescue normal thermoregulatory function in male oxytocin receptor knockout mice. Endocrinology. 154 (11), 4305-4315 (2013).
- Kasahara, Y., et al. Role of the oxytocin receptor expressed in the rostral medullary raphe in thermoregulation during cold conditions. Frontiers in Endocrinology. 6, 180 (2015).
- Yuan, J., Zhang, R., Wu, R., Gu, Y., Lu, Y. The effects of oxytocin to rectify metabolic dysfunction in obese mice are associated with increased thermogenesis. Molecular and Cellular Endocrinology. 514, 110903 (2020).
- Scholl, J. L., Afzal, A., Fox, L. C., Watt, M. J., Forster, G. L. Sex differences in anxiety-like behaviors in rats. Physiology & Behavior. 211, 112670 (2019).

