Reduced Procedure Time and Variability with Active Esophageal Cooling During Radiofrequency Ablation for Atrial Fibrillation
Summary
This study utilized advanced informatics techniques to compare procedure duration in patients undergoing radiofrequency atrial ablation treated with active esophageal cooling to those treated with traditional luminal esophageal temperature monitoring. Contextual inquiry, workflow analysis, and data mapping were utilized. The findings demonstrated reduced procedure time and variability with active cooling.
Abstract
Various methods are utilized during radiofrequency (RF) pulmonary vein isolation (PVI) for the treatment of atrial fibrillation (AF) to protect the esophagus from inadvertent thermal injury. Active esophageal cooling is increasingly being used over traditional luminal esophageal temperature (LET) monitoring, and each approach may influence procedure times and the variability around those times. The objective of this study is to measure the effects on procedure time and variability in procedure time of two different esophageal protection strategies utilizing advanced informatics techniques to facilitate data extraction. Trained clinical informaticists first performed a contextual inquiry in the catheterization laboratory to determine laboratory workflows and observe the documentation of procedural data within the electronic health record (EHR). These EHR data structures were then identified in the electronic health record reporting database, facilitating data extraction from the EHR. A manual chart review using a REDCap database created for the study was then performed to identify additional data elements, including the type of esophageal protection used. Procedure duration was then compared using summary statistics and standard measures of dispersion. A total of 164 patients underwent radiofrequency PVI over the study timeframe; 63 patients (38%) were treated with LET monitoring, and 101 patients (62%) were treated with active esophageal cooling. The mean procedure time was 176 min (SD of 52 min) in the LET monitoring group compared to 156 min (SD of 40 min) in the esophageal cooling group (P = 0.012). Thus, active esophageal cooling during PVI is associated with reduced procedure time and reduced variation in procedure time when compared to traditional LET monitoring.
Introduction
With a rise in the incidence of atrial fibrillation (AF) and an aging population, there is an increased demand for left atrial ablation to achieve pulmonary vein isolation (PVI) for the treatment of AF1. Optimizing procedure duration and minimizing variability are of increased interest among electrophysiologists and hospitals to meet population needs. During PVI procedures, one major risk is thermal injury to the esophagus due to the anatomical proximity of the left atrium to the esophagus2. Many methods exist to protect the esophagus from injury, including the current standard, luminal esophageal temperature (LET) monitoring, and other more recent developments including mechanical esophageal deviation and active esophageal cooling3.
Recent studies have found that LET monitoring may offer limited benefits over using no protection at all4,5,6. Additionally, LET monitoring necessitates frequent pauses of the procedure in response to luminal temperature alerts, which notify operators that the esophagus has reached dangerous temperatures. Recent data have shown that the distance between the temperature sensor and the radiofrequency (RF) catheter influences the sensitivity of LET monitoring, with greater than 20 mm of distance resulting in the absence of detection of significant temperature rises7. Moreover, large lag times (up to 20 s) in temperature rises and large gradients in temperature (up to 5 °C) across the esophageal wall exist, further challenging the ability of LET monitoring to detect temperature elevations quickly enough to avert tissue damage8. Depending on the electrophysiology lab, the use of LET monitoring also requires frequent fluoroscopy exposure to patients and staff to reposition the temperature probe. These additional burdens may prolong the procedure, as reported in a recent study of a community hospital system in which a reduction in procedure duration when using active esophageal cooling instead of LET monitoring was found9.The use of active esophageal cooling allows the placement of contiguous point-to-point ablation lesions in the left atrium without the need to pause radiofrequency ablation due to temperature alarms or heat stacking. As a result, procedural pauses are reduced, and the contiguity of lesions is enhanced. This effect allows a reduction in procedure time and fluoroscopy time, and an improvement in the long-term efficacy of the ablation in reducing the recurrence of arrhythmias9,10,11,12,13.
As practice in an academic setting can vary drastically from a community hospital lab due to the introduction of trainees performing procedures while undergoing their education, the impact of the esophageal protection method is less certain. Moreover, advances in human factors analysis to ensure the identification of clinical data structures documenting the critical steps of each ablation case can be leveraged to facilitate studies of this type. Multiple individuals representing various specialties are involved during an ablation, making contextual inquiry useful for understanding the clinical workflows and pairing key activities with electronic health record (EHR) data structures14,15. Consequently, this study aimed to leverage medical informatics with contextual inquiry to compare the procedural efficiency of PVI procedures conducted with active esophageal cooling to those performed with LET monitoring.
Protocol
This research was performed in compliance with the institutional guidelines of the University of Texas, Southwestern Medical Center, approval number STU-2021-1166. Data were collected retrospectively through chart review, and thus the need for patient consent was waived.
1. User workflow analysis
- During the user workflow analysis, use contextual inquiry to identify the key procedural steps and identify the personnel responsible for documenting these steps. Identify the EHR data structures that represent them, and map these data structures to tables in the EHR's reporting database.
NOTE: Contextual inquiry is a method combining real-time field observations with the interactive probing of workers during work activities14,15. - Identification of key procedural events and the personnel responsible for documentation
- Observe and interview faculty and trainee physicians, circulating and scrub staff, anesthesiology staff, and device representatives to develop a process map of the key procedural events required for tracking performance.
- Note the following key procedural events: patient arrival time, the time out procedure, vascular access attainment, sheath insertion and removal, esophageal cooling device or temperature probe insertion and removal, vascular closure, patient emergence, and patient departure time.
2. Observation of placement and use of an esophageal cooling device
NOTE: The placement and use of the active esophageal cooling device have been previously demonstrated and can be seen in Zagrodzky et al.10.
- In brief, first connect the esophageal cooling device to the external heat exchanger. Turn on the power and activate the water flow to provide adequate device stiffness and ensure the absence of any leaks. Apply a generous amount of lubrication to the distal 15 cm and place the device in a similar fashion to a standard orogastric tube.
- Determine proper esophageal cooling device placement using standard fluoroscopy demonstrating the device tip below the patient's diaphragm; if zero-fluoroscopy techniques are used, visualize the device on intracardiac echocardiography.
- If using fluoroscopy, use standard settings as chosen by the lab with an anterior-posterior view, and center the image at the patient's xiphoid.
- If using intracardiac echocardiography, rotate the catheter to obtain a posterior view to allow for visualization of the device in the esophagus, posterior to the left atrium.
3. Structured data extraction
- Identification of the data elements representing procedural events: after identification of the users responsible for procedural documentation (i.e., circulating or documenting nurse), which may be facility-specific, identify and record the documentation workflows and data elements representing the procedural activities described in step 1.2. Data elements in this step include correlating the sheath insertion to the EHR flowsheet elements representing this data point.
- Map and extract the data elements to database structures for bulk reporting: after identification of the data structures representing the key procedural steps, use EHR database mapping tools to translate these structures from the operational data structures to relational database tables in the reporting database. Extract the data into a tabular format for integration with the results of the manual chart review.
4. Identification of data necessitating manual extraction
- Identify any necessary data that cannot be easily extracted via database structures.
- For this protocol, perform manual extraction for the following data elements: energy used in ablation; esophageal protection method used, type of atrial fibrillation, episode of postoperative pain during admission, episode of postoperative pain following discharge (within 30 days).
5. Manual data extraction
- Create a REDCap database instrument to facilitate manual chart review16,17. The extracted data are shown in Supplementary File 1 (REDCap data storage extraction form).
- Create a new project within REDCap by clicking the New Project button. After naming the project, this will lead to a page titled: Project Setup. Navigate to the second section titled: Design your data collection instruments and click the Online Designer button.
- In the online designer, click Create a new instrument from scratch. In the instrument, add all the fields listed in step 4.2, in addition to a patient medical record number in order to correlate the manual data collected, to the data that was collected via EHR database structure extraction.
- Once the instrument is finalized, click the Move project to production button. From the left panel, click Add / Edit Records to view the finalized data instruments for inputting the data during the chart review.
- Identify patients that fit within the study inclusion criteria, in this case, all patients that received ablations for AF between January 2020 and January 2022.
- Perform a manual chart review of the included patients, adding the data collected into the project created in REDCap for future analysis.
Representative Results
Patient characteristics
In this analysis, a total of 164 patients were identified who underwent radiofrequency PVI between January 2020 and January 2022. Patients were included regardless of whether they received only PVI or received additional lesions such as roof lines, floor lines, mitral isthmus lines, etc. LET monitoring was performed with a single-sensor temperature probe and was performed by the same teams and in the same labs as the cases with active cooling. There were 63 patients who received LET monitoring for their PVI during the study period and 101 patients who received active esophageal cooling for esophageal protection. There were similar proportions of AF type in both groups (Table 1).
Procedure duration and procedure variability
Procedure duration was defined as the time from the first sheath placed to the time of the last sheath removal. The mean procedure time in patients that underwent LET monitoring was 176 min ± 52 min. In the actively cooled group, the mean procedure time was 156 min ± 40 min, representing a 20 min overall reduction in procedure duration (P = 0.012). The median procedure time was 172 min (interquartile range [IQR] = 144 to 198 ) in the LET monitored group, and 151 min (IQR = 129 to 178 ; P = 0.025) in active esophageal cooling group. Overall, there was a median reduction of 21 min (FIGURE 1). Other than differences in the operator, no other factors differed between the groups other than the type of esophageal protection utilized. As such, the difference in procedural duration is believed to be due entirely to the pauses required with LET monitoring, reacting to temperature elevations, as well as the need to reposition repeatedly while ablating around the pulmonary veins. Although a long-term efficacy analysis has not yet been performed at this clinical site, data from elsewhere have shown improved efficacy with cooling when compared to LET monitoring. This is believed to be due to the improved point-to-point lesion sequencing that can be completed without interruption from local overheating alarms.
In the context of the technique described here, these results highlight the utility of the technique of workflow analysis, human factors analysis, and contextual inquiry to facilitate uncovering and analyzing data that can provide important insights into clinical practice. Traditional analyses of this type often rely on the manual extraction of large quantities of data, adding time and cost burdens to clinical investigations while reducing reliability and consistency. Incorporating advanced informatics techniques as described here opens new avenues for investigation without requiring extensive time and funding.
| Esophageal Protection | |||
| Active Esophageal Cooling (n=101) | LET Monitoring (n=63) | ||
| Patient age (years), mean (SD) | 67.9 ± 11.3 | 64.5 ± 11.6 | |
| Gender | Male | 66 | 46 |
| Female | 35 | 17 | |
| AF Type | Paroxysmal Atrial Fibrillation | 55 | 36 |
| Persistent Atrial Fibrillation | 38 | 23 | |
| Long-standing Persistent Atrial Fibrillation | 8 | 4 | |
Table 1: Patient characteristics, including age, gender, and type of atrial fibrillation treated.
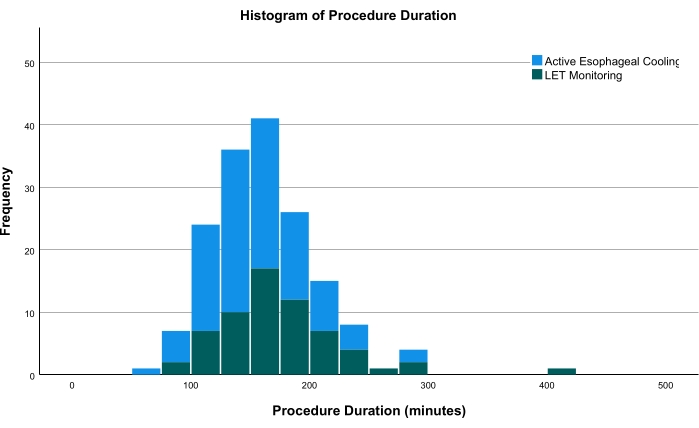
Figure 1: Histogram comparing the procedure times of both groups. The green bars show patients receiving LET monitoring; the blue bars show patients receiving active esophageal cooling. Abbreviation: LET = luminal esophageal temperature. Please click here to view a larger version of this figure.
Supplementary File 1: REDCap data storage extraction form. An example of the data extraction form used for this protocol, demonstrating the specific data elements recorded. Please click here to download this File.
Discussion
This investigation demonstrates the use of advanced informatics techniques, including contextual inquiry, workflow analysis, and the pairing of key activities with electronic health record (EHR) data structures, to analyze the impact on procedural times of two different esophageal protection methods utilized during cardiac ablation. This is the first study of the effects of esophageal cooling on procedure time and variability to be performed in an academic medical center, where trainees (fellows) receive clinical training in electrophysiologic procedures and perform many of the procedures as a part of this training while being overseen by experienced electrophysiologists. The main finding of this study is that the use of active esophageal cooling was associated with shorter procedure times and less variability around procedure times. Leveraging expertise from trained informaticists ensured accuracy in data identification and facilitated the data acquisition.
The reduction in procedure time and the variability around procedure time offers several benefits. Better predictability of procedure duration improves hospital scheduling, and reducing procedure times may allow additional cases to be scheduled, further improving hospital operations. More importantly, patient risk is reduced as procedure time is shortened. Increased operative duration, in general, increases the risk of complications such as surgical site infections, venous thromboembolism, bleeding, pneumonia, urinary tract infections, renal failure, and hematoma formation18. The likelihood of developing a complication increases with increasing operative time increments (i.e., 1% for every 1 min, 4% for every 10 min, 14% for every 30 min, and 21% for every 60 min increase in operative time)18. In the case of left atrial ablation, access time in the left atrium is the most significant procedural variable for the risk for post-operative cognitive dysfunction19.
A previous study in a community medical center also found procedural time savings associated with the use of active esophageal cooling during left atrial ablation for the treatment of atrial fibrillation9. The mechanism behind this effect relates to the elimination of frequent pauses from overheating that result in ablations and the temperature alarms that are utilized in LET monitoring. As active cooling eliminates overheating and therefore, the need for temperature alarms, it allows electrophysiologists to proceed without pauses20,21,22.
The critical steps in this protocol include properly identifying the individuals and their roles in the procedure to accurately record real-time field observations, probing to uncover any unconscious behaviors involved in experts' workflows, and identifying specific elements of interest related to outcomes to determine where these variables are recorded and located in the Epic chronicles database. With careful completion of these steps, similar analyses can be undertaken for countless outcomes of interest.
The limitations of this analysis include the non-randomized allocation of patients and the retrospective collection of data recorded as the standard of care in the EHR. Although non-randomization introduces the potential for unmeasured confounders to influence results, no secular changes to treatment protocols occurred during the time period investigated in this analysis. Likewise, the use of data recorded as the standard of care in the hospital EHR may reduce the potential for bias in the data.
In conclusion, using contextual inquiry, workflow analysis, and data mapping to analyze procedural timing, this study demonstrated reduced procedure time and variability with active cooling when compared to traditional LET monitoring.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors would like to acknowledge the staff of the UT Southwestern Department of Electrophysiology: Cheryl Thomas RN, Roma Alfonso RN, Eileen Dwyer RN, Anish Varghese RN, Josey George RCIS, Pam Harrison RCIS, and Carolyn Carlson RN. Data are available upon request from the authors.
Materials
| Blanketrol III hyper-hypothermia system | Gentherm Medical, Cincinnati, OH | Model 233 | Programmable heat exchanger for temperature regulation |
| ensoETM | Attune Medical, Chicago, IL | ECD02A | Active esophageal cooling device |
| EPIC Clarity | Epic System Corporation, Verona, WI | Electronic Health Record reporting database | |
| REDCap | Nashville, TN | Secure web application for building and managing online surveys and databases, including compliance with 21 CFR Part 11, FISMA, HIPAA, and GDPR |
References
- McCarthy, P. M., et al. Surgery and catheter ablation for atrial fibrillation: History, current practice, and future directions. Journal of Clinical Medicine. 11 (1), 210 (2021).
- Della Rocca, D. G., et al. Clinical presentation, diagnosis, and treatment of atrioesophageal fistula resulting from atrial fibrillation ablation. Journal of Cardiovascular Electrophysiology. 32 (9), 2441-2450 (2021).
- Leung, L. W. M., et al. Preventing esophageal complications from atrial fibrillation ablation: A review. Heart Rhythm O2. 2, 651-664 (2021).
- Schoene, K., et al. Oesophageal Probe Evaluation in Radiofrequency Ablation of Atrial Fibrillation (OPERA): Results from a prospective randomized trial. Europace. 22 (10), 1487-1494 (2020).
- Chen, S., et al. Catheter ablation of atrial fibrillation using ablation index-guided high power (50 W) for pulmonary vein isolation with or without esophageal temperature probe (the AI-HP ESO II). Heart Rhythm. 17 (11), 1833-1840 (2020).
- Meininghaus, D. G., et al. Temperature monitoring and temperature-driven irrigated radiofrequency energy titration do not prevent thermally-induced esophageal lesions in pulmonary vein isolation: A randomized study controlled by esophagoscopy before and after catheter ablation. Heart Rhythm. 18 (6), 926-934 (2021).
- Barbhaiya, C. R., et al. Esophageal temperature dynamics during high-power short-duration posterior wall ablation. Heart Rhythm. 17 (5), 721-727 (2020).
- Kar, R., Post, A., John, M., Rook, A., Razavi, M. An initial ex vivo evaluation of temperature profile and thermal injury formation on the epiesophageal surface during radiofrequency ablation. Journal of Cardiovascular Electrophysiology. 32 (3), 704-712 (2021).
- Joseph, C., et al. Procedural time reduction associated with active esophageal cooling during pulmonary vein isolation. Journal of Interventional Cardiac Electrophysiology. , (2022).
- Zagrodzky, J., et al. Cooling or warming the esophagus to reduce esophageal injury during left atrial ablation in the treatment of atrial fibrillation. Journal of Visualized Experiments. (157), e60733 (2020).
- Joseph, C., et al. Arrhythmia recurrence reduction with an active esophageal cooling device during radiofrequency ablation. EP Europace. 24, (2022).
- Joseph, C., et al. Reduction of procedure time with active esophageal cooling during left atrial ablation in zero-fluoroscopy cases. Journal of the American College of Cardiology. 79, 161 (2022).
- Joseph, C., et al. One-year outcomes after active cooling during left atrial radiofrequency ablation. Journal of the American College of Cardiology. 79, 114 (2022).
- Holtzblatt, K., Wendell, J. B., Wood, S. . Rapid Contextual Design: A How-to Guide to Key Techniques for User-Centered Design. , (2005).
- Karen, H., Sandra, J., Schuler, D., Namioka, A. Contextual inquiry: A participatory technique for system design. Participatory Design. , 177-210 (2017).
- Harris, P. A., et al. The REDCap consortium: Building an international community of software platform partners. Journal of Biomedical Informatics. 95, 103208 (2019).
- Harris, P. A., et al. Research electronic data capture (REDCap)-A metadata-driven methodology and workflow process for providing translational research informatics support. Journal of Biomedical Informatics. 42 (2), 377-381 (2009).
- Cheng, H., et al. Prolonged operative duration is associated with complications: A systematic review and meta-analysis. Journal of Surgical Research. 229, 134-144 (2018).
- Medi, C., et al. Subtle post-procedural cognitive dysfunction after atrial fibrillation ablation. Journal of the American College of Cardiology. 62 (6), 531-539 (2013).
- Mercado, M., Leung, L., Gallagher, M., Shah, S., Kulstad, E. Modeling esophageal protection from radiofrequency ablation via a cooling device: An analysis of the effects of ablation power and heart wall dimensions. Biomedical Engineering Online. 19 (1), 77 (2020).
- Zagrodzky, J., Bailey, S., Shah, S., Kulstad, E. Impact of active esophageal cooling on fluoroscopy usage during left atrial ablation. The Journal of Innovations in Cardiac Rhythm Management. 12 (11), 4749-4755 (2021).
- Leung, L., et al. Oesophageal thermal protection during AF ablation: Effect on left atrial myocardial ablation lesion formation and patient outcomes. EP Europace. 23, (2021).

