Isolation, Culture, and Characterization of Primary Dermal Fibroblasts from Human Keloid Tissue
Summary
This study describes an optimized protocol for establishing primary fibroblasts from keloid tissues that can effectively and steadily provide pure and viable fibroblasts.
Abstract
Fibroblasts, the major cell type in keloid tissue, play an essential role in the formation and development of keloids. The isolation and culture of primary fibroblasts derived from keloid tissue are the basis for further studies of the biological function and molecular mechanisms of keloids, as well as new therapeutic strategies for treating them. The traditional method of obtaining primary fibroblasts has limitations, such as poor cellular state, mixing with other types of cells, and susceptibility to contamination. This paper describes an optimized and easily reproducible protocol that could reduce the occurrence of possible issues when obtaining fibroblasts. In this protocol, fibroblasts can be observed 5 days after isolation and reach nearly 80% confluency after 10 days of culture. Then, the fibroblasts are passaged and verified using PDGFRα and vimentin antibodies for immunofluorescence assays and CD90 antibodies for flow cytometry. In conclusion, fibroblasts from keloid tissue can be easily acquired through this protocol, which can provide an abundant and stable source of cells in the laboratory for keloid research.
Introduction
Keloid, a fibroproliferative disease, manifests as the continuous growth of plaques that often invade the surrounding normal skin without self-limitation and cause various degrees of itching, pain, and cosmetic and psychological burdens for patients1. Fibroblasts, the primary cells involved in keloids, play an essential role in the formation and development of this disease through excessive proliferation, redundant extracellular matrix production, and disorganized collagens2,3. However, the underlying pathogenesis remains unclear, and an effective therapeutic method for keloid is still lacking; therefore, there is an urgent need for further research4,5.
As there is no ideal animal model for keloid research in vivo6,7,building an in vitro model by acquiring primary fibroblasts from keloid tissues can offer feasibility and reliability for keloid research2,6. Primary cells are those derived directly from living tissue, and it is generally recognized that these cells can more closely resemble the physiological state and genetic background of multiple individuals compared with cell lines8,9. Culturing primary cells provides a powerful means to study the growth and metabolism of cells, as well as other cell phenotypes.
At present, there are two methods for acquiring primary fibroblasts: enzyme digestion and explant culture. However, several obstacles have been identified to obtaining primary fibroblasts, such as the risk of contamination by various bacteria or fungi, mixing with other types of cells that are not easily removed, the long period of the culture cycle, the subsequent changes to the cell characteristics compared to the original cells, and so on9. Therefore, developing a feasible and effective process for obtaining primary fibroblasts is the foundation for further studies and applications.This study describes an optimized protocol for extracting primary fibroblasts from keloid tissues that can effectively and steadily provide pure and viable fibroblasts.
Protocol
This study was approved by the institutional review board of the Dermatology Hospital, Southern Medical University (2020081). Informed patient consent was obtained before tissues were collectedfrom the individuals.
1. Preparation
NOTE: The following procedures should be performed in a sterile environment under a biological safety cabinet.
- Prepare complete culture medium by adding 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin-amphotericin B solution (PSA) to high-glucose Dulbecco's modified Eagle medium (DMEM).
- Prepare phosphate-buffered saline solution (PBS) with PSA by adding 1% PSA into 1x PBS. Prepare PBS with FBS by adding 1% FBS to 1x PBS.
- Prepare several sterilized scissors, forceps, and scalpels by autoclaving.
2. Obtaining removed tissues
- Obtain tissues from keloid patients through surgery. Collect the fresh keloid tissues in a sterile packing bag or sterile centrifuge tube, and transfer them to a biological safety cabinet in the laboratory as soon as possible.
NOTE: In this protocol, the size of the acquired keloid tissue was approximately ~20 x 20 x 10 mm3, and the size may vary depending on the surgery.
3. Isolation
- Take out the keloid tissue using sterile tweezers, and place it in a 50 mL sterile centrifuge tube containing 10-25 mL of PBS with 1% PSA for 10 min. Then, prepare a 6-well plate, and add 4 mL of PBS supplemented with 1% PSA to each well. Take out the tissue using sterilized tweezers, and wash it twice with PBS supplemented with 1% PSA. Using sterile forceps, transfer the tissue sequentially from one well to the next.
- Remove the adipose andepidermis layers using surgical scissors or a surgical scalpel, and leave the dermis layer untouched. Trim and dissect the dermis layer into 3-5 mm2 pieces with scissors, transfer these pieces using sterile forceps to the next well, and wash them in PBS with 1% PSA solution again.
4. Culture
- Place the dermis tissue pieces in Petri dishes using sterilized forceps; ensure that the number of pieces is between 10 and 30 and the distance between each piece is >5 mm. Put the Petri dishes upside down in a 5% CO2 incubator at 37 °C for 30-60 min until the pieces of tissues dry a little and stick to the Petri dish. Then, add DMEM supplemented with 10% FBS and 1% PSA, and carefully place the Petri dishes into a 5% CO2 incubator at 37 °C.
NOTE: Fibroblasts are a morphologically and functionally heterogeneous cell population. Considering this complexity, the isolation and culture steps should be performed with care; select the keloid dermal tissue pieces evenly, and mix these pieces well before placing them in the Petri dish. - After 3 days, replace half of the supernatant with a complete culture medium. Change the culture medium every 2-3 days. Observe the fibroblasts under a microscope at 40x magnification every day.
NOTE: All the steps should be performed gently. Do not move the Petri dish for 2 days after isolating, as the tissue pieces take time to adhere to the Petri dishes. - When the fibroblasts growing around the tissue pieces reach approximately 90% confluency, remove the tissue pieces and the culture medium. Wash the fibroblasts with sterile 1x PBS, and add 2 mL of sterile 1x trypsin-EDTA solution to the plates.Incubate the cells for approximately ~3-5 min at 37 °C in a humidified 5% CO2 incubator.Gently tap the culture dish, and observe it under a microscope. When the majority of the cells detach from the plate, add 2 mL of complete medium to end the digestion process.
- Transfer the cell suspension to a 15 mL sterile centrifuge tube, and centrifuge the tube at 300 × g for 3 min at room temperature. Discard the supernatant carefully, and resuspend the cell pellet in complete medium.Seed the fibroblasts into a 9 cm cell culture dish, and incubate at 37 °C in a humidified 5% CO2 incubator.
5. Maintenance and preservation
- Approximately 3-4 days later, when the fibroblasts have grown to 80% confluency, repeat steps 4.2-4.4, and passage the fibroblasts at a ratio of 1:3. Use the passaged fibroblasts for further experiments.
NOTE: The fibroblast culture should be stopped after 10 passages, because the cells may start to show changed characteristics compared to the original cells. - Cryopreserve the passaged cells of P1-P3 in liquid nitrogen for further usage.
- Repeat steps 4.2-4.3. Transfer the cell suspension to a 15 mL sterile centrifuge tube, and centrifuge the tube for 3 min at 300 × g. Discard the supernatant carefully, resuspend the cell pellet in 1 mL of cell freezing medium containing 90% FBS and 10% DMSO, and transfer the suspension into cell cryotubes.
- Move the cells into a frozen box, and then put the frozen box in a −80 °C freezer. After 1 day, transfer the cells to liquid nitrogen for long-term preservation.
6. Identification of fibroblasts by immunofluorescence staining
- Place round coverslips in a 24-well plate, and culture the passaged fibroblasts at a concentration of 1 × 104 cells/well. When the fibroblasts reach 60% confluency, remove the culture medium. Add 1 mL of 4% paraformaldehyde to fix the fibroblasts for 20 min at room temperature, and wash 3x with PBS for 1 min each time.
NOTE: Count the number of cells by using a hemocytometer slide under a microscope or an automatic cell counter. - Remove the PBS, incubate with 0.5% Triton X-100 for 20 min for the permeabilization of cell membranes, and then wash 3x with PBS for 1 min each time. Add PBS with 0.5% bovine serum albumin, and soak for 30 min. Then, remove it, and add the PDGFR-α/vimentin antibody diluted in antibody diluent at 1:1,000. Incubate overnight at 4 °C.
- The next day, remove the primary antibody, wash the cells 3x with PBS for 3 min, and add the secondary antibody Alexa Fluor-555 goat anti-rabbit IgG diluted with antibody diluent at 1:200 to soak for 1 h.
- Remove the secondary antibody, and wash the cells 3x with PBS. Take out the round coverslips using forceps, put them on glass slides, and add 50 µL of 5 µg/mL 4',6-diamidino-2-phenylindole (DAPI) solution to stain the cellular nuclei. Keep the samples in a wet, dark box, and observe them under a laser confocal fluorescence microscope.
NOTE: Add a negative control group without a primary antibody but with a secondary antibody to exclude nonspecific staining.
7. Identification of fibroblasts by flow cytometry
- Collect the cell pellet into a 1.5 mL sterile centrifuge tube, and resuspend with 50 µL of PBS containing 1% FBS. Incubate with anti-CD90 for 30 min in the dark, and add an anti-IgG isotype control to the control group. Then, add 200 µL of PBS containing 1% FBS, and centrifuge the tube for 10 min at 300 × g at 4 °C.
- Remove the supernatant, and add 200 µL of PBS containing 1% FBS. Resuspend and filter the suspension through a cell strainer (70 mesh). Add a 5 µg/mL stock solution of DAPI at 1:100 to the filtrate, and then acquire the results by flow cytometry. Set the forward scatter (FSC) as the abscissa and the side scatter (SSC) as the ordinate, and circle the main cell population. Select the cell population, and set the phycoerythrin as the abscissa and the count as the ordinate for analysis.
Representative Results
The timeline of the protocol is summarized in Figure 1A. Some representative images of the isolation process are shown in Figure 2; the epidermis and adipose layers were carefully removed, and the dermis layer was separated into small fragments of 3-4 mm2, which were inoculated into the Petri dishes.
As shown in Figure 3A, several fibroblast outgrowths of the tissue pieces were observed under the microscope 5 days after processing. As shown in Figure 3B, the fibroblasts displayed high proliferation rates and reached a high level of confluency after 10 days. These fibroblasts had elongated, spindle-like cell bodies and were aligned in bundles when they reached a high level of confluency. By following this protocol, the fibroblasts were passaged and expanded to the desired quantity within 2-3 weeks.
To verify the identity and purity of the fibroblasts, an immunofluorescence staining assay was employed for detecting the fibroblast-specific markers PDGFRα10 and vimentin. As expected, Figure 4A and Figure 4C show that all the fibroblasts were positively stained by the PDGFRα/vimentin antibody, with red immunofluorescence and blue immunofluorescence in the cellular nucleus. As demonstrated in Figure 4C, the flow cytometry assay also showed CD90 positivity in almost all of the fibroblasts.
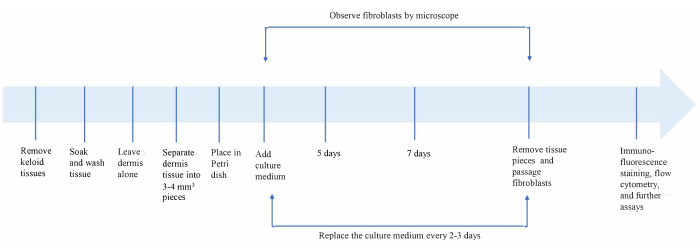
Figure 1: Overview of the keloid fibroblast isolation and culture procedure. Please click here to view a larger version of this figure.

Figure 2: Representative images of the isolation and culture of fibroblasts from keloid tissues. (A) Wash the tissue. (B) Remove the epidermis and adipose layers. (C) Dissect the dermis into 3-4 mm3 pieces, and wash with PBS again. (D) Place the tissue fragments in a Petri dish. (E) Put the Petri dishes upside down into the 5% CO2 incubator at 37 °C for 30-60 min to let the pieces of tissue dry a little and stick to the Petri dish. (F) Add complete culture medium into the Petri dish, and culture in the CO2-containing cell incubator. Please click here to view a larger version of this figure.
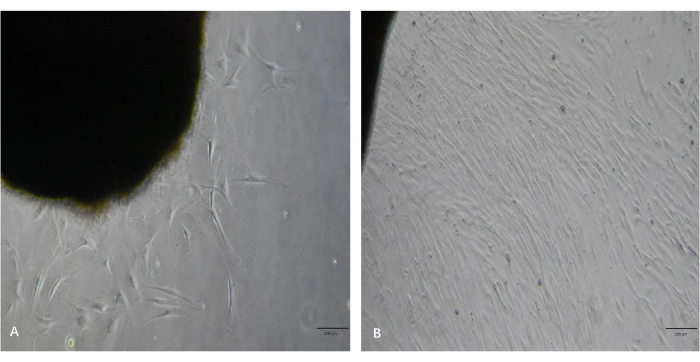
Figure 3: Representative images of fibroblast outgrowths from the keloid tissue pieces. (A) Fibroblast outgrowths from the tissue pieces in ~5 days. (B) The fibroblasts reached a high level of confluency in ~10 days. Scale bars = 200 µm. Please click here to view a larger version of this figure.
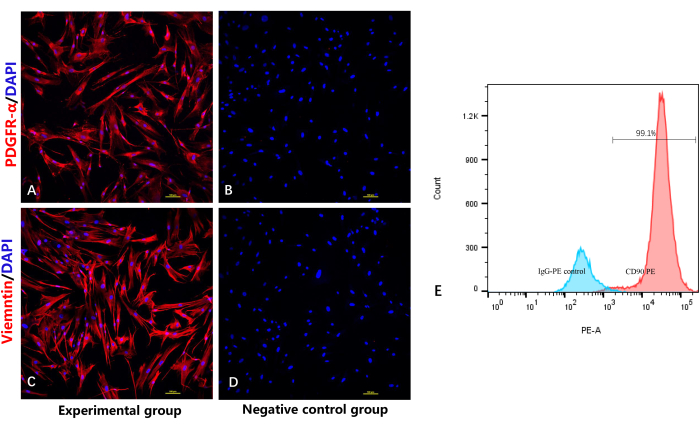
Figure 4: Identification of highly purified fibroblasts by immunofluorescence staining and flow cytometry. (A) Immunofluorescencestaining of fibroblasts by anti-PDGFRα. (B) Negative control group without the primary antibody for PDGFRα. (C) Immunofluorescencestaining of the fibroblasts by anti-vimentin. (D) Negative control group without the primary antibody for vimentin. (E) Flow cytometry analysis of the fibroblasts. Please click here to view a larger version of this figure.
Discussion
Obtaining primary fibroblasts from keloid tissues is a critical basis for further research. Up until now, there have been two methods for acquiring primary fibroblasts: enzyme digestion and explant culture11,12,13,14. However, both traditional methods have limitations, such as susceptibility to contamination, mixing with other types of cells, a long culture period, and a low rate of success15,16. This study has described an optimized method and provided clear instructions to solve these existing challenges and increase the chance of success for the isolation and culture of keloid fibroblasts.
The possibility of microbial contamination is one of the major problems in obtaining primary fibroblasts. All the equipment and solutions must be sterilized, and standard aseptic techniques must be implemented during all the steps in the protocol. The protocol contains reminders to remain aseptic to reduce the risk of contamination. The first critical step is the isolation process; the aim of soaking the tissues in PBS supplemented with 1% PSA and washing several times is to remove existing microbes and residual bloodstains. To prevent the possible transfer of contamination, an extra pair of sterilized scissors and forceps should be prepared; these instruments could be replaced by unused ones in the next operation. Furthermore, PSA is added to the culture medium to prevent the outgrowth of microbes. Periodic testing for the mycoplasma during the isolation and subsequent culture of the keloid fibroblasts should be incorporated into the protocol. Through these processes, the possibility of microbial contamination can be significantly reduced.
The other challenge is mixing with other types of cells, such as keratinocytes. This cell type also has an intense proliferative ability, making it difficult to remove. The purity of fibroblasts is highly dependent on the isolation process; the epidermis and adipose layers should be entirely removed to avoid mixing with other cells. PDGFR-α is a fibroblast-specific marker that is expressed in the membrane and cytoplasm of fibroblasts10,17,18. CD90 and vimentin are also specific mesenchymal markers that are specifically expressed in the membranes of fibroblasts19,20,21. The immunofluorescence staining assay in this work showed that all the cells positively expressed PDGFR-α and vimentin. The flow cytometry assay also directly demonstrated CD90 positivity in almost all the fibroblasts and greater CD90 positivity compared with the isotype control, thus confirming that we obtained relatively high-purity fibroblasts using this protocol.
The crucial recommended steps to promote the outgrowth and enhance the proliferative efficiency of fibroblasts are as follows. First, the tissue pieces should be cut into appropriate sizes of 3-5 mm2, as smaller tissue pieces are more likely to float during the movement of the Petri dish, and the cells will not easily outgrow larger pieces.Second, the first medium change should be after the third day, as the tissue pieces take time to adhere to the Petri dishes. Moving the Petri dishes too early tends to disturb the tissues and decrease the possibility of fibroblast outgrowth. Third, all the steps in this protocol should be conducted gently; improper handling will cause unnecessary movement of the tissue pieces and impair the cell outgrowth.
Fibroblasts are a morphologically and functionally heterogeneous cell population that can be divided into several subpopulations. One limitation of this study is that we can obtaingeneral fibroblasts from the entire keloid tissue but not the different subpopulations of fibroblasts. We are trying to find better methods to solve this problem in the future.
As demonstrated in our previous studies by RNA-seq, there are many differences between keloid fibroblasts and normal fibroblasts. Keloid fibroblasts produce more collagen I and collagen III. In addition, keloid fibroblasts express specific markers such as POSTN, among others. After passaging, the fibroblasts still maintain these characteristics, which proves that primary fibroblasts could also exhibit some features found in keloidpatients.
In conclusion, this study provides an optimized method and gives clear instructions to solve the existing difficulties in keloid fibroblast extraction, such as microbial contamination, the mixing with other cell types, and the long culture period, and to increase the chance of success. The current work provides a simple and reproducible method for isolating and culturing keloid primary fibroblasts, which will provide a rich resource for future studies and research or could be frozen in liquid nitrogen for banking.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (grant numbers 81903189 and 82073418) and the Science and Technology Foundation of Guangzhou (grant number 202102020025).
Materials
| 1.5 mL sterile centrifuge tube | JETBIOFIL | CFT002015 | |
| 15 mL sterile centrifuge tube | JETBIOFIL | 8076 | |
| 4% polyformaldehyde | Beyotime Biotechnology | P0099 | Cell fixation |
| 50 mL sterile centrifuge tube | JETBIOFIL | 8081 | Put keloid tissue |
| Alexa Fluor-555 goat anti-rabbit IgG | Abcam | Alexa Fluor 555 | second antibody for immunofluorescence staining assay |
| Anti human CD90 | BioLegend | B301002 | Identify the purity of fibroblasts |
| Antibody diluent | Beyotime Biotechnology | P0262 | |
| Biological safety cabinet | Thermo Scientific | 1300 series A2 | Isolation and culture cells |
| Bovine serum albumin | aladdin | B265993 | Blocking for immunofluorescence staining assay |
| Carbon dioxide incubator | ESCO | CCL-170B-8 | Using for culturing cells |
| Cell cryotubes | Corning | 43513 | Store the cells in low temperature |
| centrifugal machine | Thermo Fisher | ST 16R | Discard supernatant |
| DAPI | Beyotime Biotechnology | C1006 | Stain the cellular nucleus |
| DMSO | MP Biomedicals | 196055 | Using for preserving cells |
| Dulbecco's modified eagle medium | Gibco | C11995500BT | Culture medium solution |
| Fetal bovine serum | BI | 04-001-1A | |
| Flow cytometer | BD | BD FACSCelesta | Observing the identity of cells |
| frozen box | Thermo Scientific | 5100-0050 | |
| Inverted microscope | Nikon | ECLIPSE Ts2 | |
| Laser confocal microscope | Nikon | AIR-HD25 | Observing the immunofluorescence staining assay |
| PDGFR-α antibody | CST | 3174T | First antibody for immunofluorescence staining assay |
| Penicillin-streptomycin-Am solution | Solarbio | P1410 | Add in culture medium solution to avoid contamination |
| petri dish | JETBIOFIL | 7556 | Culture fibroblasts |
| Phosphate buffered saline solution | Gibco | C10010500BT | Culture medium solution |
| Rabbit (DAIE) mAB IgG XR (R) Isotuge Control (PE) | Cell Signaling Technology | 5742S | As a control for flow cytometry |
| Round coverslip | Biosharp | 801007 | Cell culture |
| Triton X 100 | Solarbio | T8200 | Punch holes in the cell membrane |
| Trypsin-EDTA | Gibco | 25200072 | Used for passaging cells |
| Vimentin antibody | Abcam | ab8978 | First antibody for immunofluorescence staining assay |
References
- Zhu, Y. Q., et al. Genome-wide analysis of Chinese keloid patients identifies novel causative genes. Annals Of Translational Medicine. 10 (16), 883 (2022).
- Feng, F., et al. Biomechanical regulatory factors and therapeutic targets in keloid fibrosis. Frontiers in Pharmacology. 13, 906212 (2022).
- Cohen, A. J., Nikbakht, N., Uitto, J. Keloid disorder: Genetic basis, gene expression profiles, and immunological modulation of the fibrotic processes in the skin. Cold Spring Harbor Perspectives in Biology. , (2022).
- Wang, W., et al. Current advances in the selection of adjuvant radiotherapy regimens for keloid. Frontiers in Medicine. 9, 1043840 (2022).
- Ghadiri, S. J., Kloczko, E., Flohr, C. Topical treatments in the management of keloids and hypertrophic scars: A critically appraised topic. British Journal of Dermatology. 187 (6), 855-856 (2022).
- Neves, L. M. G., Wilgus, T. A., Bayat, A. In vitro, ex vivo, and in vivo approaches for investigation of skin scarring: Human and animal models. Advances in Wound Care. 12 (2), 97-116 (2023).
- Supp, D. M. Animal models for studies of keloid scarring. Advances in Wound Care. 8 (2), 77-89 (2019).
- Künzel, S. R., et al. Ultrasonic-augmented primary adult fibroblast isolation. Journal of Visualized Experiments. (149), e59858 (2019).
- He, Y., et al. An improved explants culture method: Sustainable isolation of keloid fibroblasts with primary characteristics. Journal of Cosmetic Dermatology. 21 (12), 7131-7139 (2022).
- Philippeos, C., et al. Spatial and single-cell transcriptional profiling identifies functionally distinct human dermal fibroblast subpopulations. Journal of Investigative Dermatology. 138 (4), 811-825 (2018).
- Li, L., et al. Hydrogen sulfide suppresses skin fibroblast proliferation via oxidative stress alleviation and necroptosis inhibition. Medicine and Cellular Longevity. 2022, 7434733 (2022).
- Zhou, B. Y., et al. Nintedanib inhibits keloid fibroblast functions by blocking the phosphorylation of multiple kinases and enhancing receptor internalization. Acta Pharmacologica Sinica. 41 (9), 1234-1245 (2020).
- Wang, X. M., Liu, X. M., Wang, Y., Chen, Z. Y. Activating transcription factor 3 (ATF3) regulates cell growth, apoptosis, invasion and collagen synthesis in keloid fibroblast through transforming growth factor beta (TGF-beta)/SMAD signaling pathway. Bioengineered. 12 (1), 117-126 (2021).
- Sato, C., et al. Conditioned medium obtained from amnion-derived mesenchymal stem cell culture prevents activation of keloid fibroblasts. Plastic and Reconstructive Surgery. 141 (2), 390-398 (2018).
- Li, J., et al. Long-term explant culture: An improved method for consistently harvesting homogeneous populations of keloid fibroblasts. Bioengineered. 13 (1), 1565-1574 (2022).
- Wang, Q., et al. Altered glucose metabolism and cell function in keloid fibroblasts under hypoxia. Redox Biology. 38, 101815 (2021).
- Fan, C., et al. Single-cell transcriptome integration analysis reveals the correlation between mesenchymal stromal cells and fibroblasts. Frontiers in Genetics. 13, 798331 (2022).
- Yao, L., et al. Temporal control of PDGFRα regulates the fibroblast-to-myofibroblast transition in wound healing. Cell Reports. 40 (7), 111192 (2022).
- Domdey, M., et al. Consecutive dosing of UVB irradiation induces loss of ABCB5 expression and activation of EMT and fibrosis proteins in limbal epithelial cells similar to pterygium epithelium. Stem Cell Research. 40, 102936 (2022).
- Lin, Z., et al. Renal tubular epithelial cell necroptosis promotes tubulointerstitial fibrosis in patients with chronic kidney disease. FASEB Journal. 36 (12), e22625 (2022).
- Korosec, A., et al. Lineage identity and location within the dermis determine the function of papillary and reticular fibroblasts in human skin. Journal of Investigative Dermatology. 139 (2), 342-351 (2019).

