Automating Citrus Budwood Processing for Downstream Pathogen Detection Through Instrument Engineering
Summary
We engineered, fabricated, and validated an instrument that rapidly processes phloem-rich bark citrus budwood tissues. Compared to current methods, the budwood tissue extractor (BTE) has increased sample throughput and decreased the required labor and equipment costs.
Abstract
Graft-transmissible, phloem-limited pathogens of citrus such as viruses, viroids, and bacteria are responsible for devastating epidemics and serious economic losses worldwide. For example, the citrus tristeza virus killed over 100 million citrus trees globally, while "Candidatus Liberibacter asiaticus" has cost Florida $9 billion. The use of pathogen-tested citrus budwood for tree propagation is key for the management of such pathogens. The Citrus Clonal Protection Program (CCPP) at the University of California, Riverside, uses polymerase chain reaction (PCR) assays to test thousands of samples from citrus budwood source trees every year to protect California's citrus and to provide clean propagation units to the National Clean Plant Network. A severe bottleneck in the high-throughput molecular detection of citrus viruses and viroids is the plant tissue processing step.
Proper tissue preparation is critical for the extraction of quality nucleic acids and downstream use in PCR assays. Plant tissue chopping, weighing, freeze-drying, grinding, and centrifugation at low temperatures to avoid nucleic acid degradation is time-intensive and labor-intensive and requires expensive and specialized laboratory equipment. This paper presents the validation of a specialized instrument engineered to rapidly process phloem-rich bark tissues from citrus budwood, named the budwood tissue extractor (BTE). The BTE increases sample throughput by 100% compared to current methods. In addition, it decreases labor and the cost of equipment. In this work, the BTE samples had a DNA yield (80.25 ng/µL) that was comparable with the CCPP's hand-chopping protocol (77.84 ng/µL). This instrument and the rapid plant tissue processing protocol can benefit several citrus diagnostic laboratories and programs in California and become a model system for tissue processing for other woody perennial crops worldwide.
Introduction
Graft-transmissible phloem-limited pathogens of citrus, such as viroids, viruses, and bacteria, have caused devastating epidemics and serious economic losses in every citrus-producing area of the world. Citrus viroids are limiting production factors because of the exocortis and cachexia diseases they cause in economically important citrus types, such as trifoliate, trifoliate hybrids, mandarins, clementines, and tangerines1,2,3. In California, these viroid-sensitive citrus types are the basis of the growing and profitable market of "easy-peelers", following the shifting trend in consumers' preference for fruits that are easy to peel, segmented, and seedless4,5,6. Thus, citrus viroids are regulated under the California Department of Food and Agriculture (CDFA) "Citrus Nursery Stock Pest Cleanliness Program-Senate Bill 140", and the laboratories of CDFA's Plant Pest Diagnostics Branch perform thousands of citrus viroid tests annually7,8,9,10. Citrus tristeza virus (CTV) has been responsible for the death of over 100 million citrus trees since the beginning of the global epidemic in the 1930s3,9,10,11. In California, stem pitting and trifoliate breaking resistance isolates of the virus pose a serious threat to the $3.6 billion California citrus industry12,13,14. Consequently, CDFA classifies CTV as a regulated class-A plant pest, and the laboratory of the Central California Tristeza Eradication Agency (CCTEA) performs extensive field surveys and thousands of virus tests every year15,16. The bacterium "Candidatus Liberibacter asiaticus" (CLas) and the huanglongbing (HLB) disease are estimated to have caused close to $9 billion of economic damage to Florida as a result of a 40% reduction of citrus acreage, a 57% decrease in citrus operations, and a loss of almost 8,000 jobs17,18. In California, a hypothetical 20% reduction in citrus acreage due to HLB was predicted to result in more than 8,200 job losses and a reduction of over half a billion dollars in the state's gross domestic product. Therefore, the Citrus Pest and Disease Prevention Program spends over $40 million annually on surveys to test, detect, and eradicate CLas from California14,17,19,20.
A key element of the management of citrus viroids, viruses, and bacteria is the use of pathogen-tested propagative materials (i.e., budwood) for tree production. Pathogen-tested citrus budwood is produced and maintained within comprehensive quarantine programs that employ advanced pathogen elimination and detection techniques10,21. The Citrus Clonal Protection Program (CCPP) at the University of California, Riverside, tests thousands of budwood samples every year from citrus varieties newly imported into the state and the USA, as well as citrus budwood source trees, to protect California's citrus and support the functions of the National Clean Plant Network for Citrus10,17,22. To handle the large volume of citrus testing, high-throughput, reliable, and cost-effective pathogen detection assays are a fundamental component for the success of programs such as the CCPP7,10,22.
While molecular-based pathogen detection assays such as polymerase chain reaction (PCR) have allowed for significant increases in throughput in plant diagnostic laboratories, in our experience, one of the most critical bottlenecks in the implementation of high-throughput protocols is the plant tissue sample processing step. This is particularly true for citrus because the currently available protocols for the processing of phloem-rich tissues such as leaf petioles and budwood bark are labor-intensive, time-consuming, and require expensive and specialized laboratory equipment. These protocols require hand-chopping, weighing, freeze-drying, grinding, and centrifugation at low temperatures to avoid nucleic acid degradation8,23,24. For example, at the CCPP diagnostic laboratory, sample processing includes (i) hand-chopping (6-9 samples/h/operator), (ii) freeze-drying (16-24 h), (iii) pulverization (30-60 s), and (iv) centrifugation (1-2 h). The process also requires specialized supplies (e.g., heavy-duty safe-lock tubes, stainless steel grinding balls, adapters, blades, gloves) and multiple pieces of costly lab equipment (e.g., ultra-low freezer, freeze-dryer, tissue pulverizer, liquid nitrogen cryostation, refrigerated centrifuge).
As in any industry, equipment engineering and the automation of processes are key to lowering costs, increasing throughput, and providing high-quality, uniform product and services. The citrus industry needs low-cost tissue-processing instruments that require minimum skill to operate and, as such, are easy to transfer to diagnostic laboratories and field operations to allow high sample-processing capacity for rapid downstream pathogen detection. Technology Evolving Solutions (TES) and the CCPP developed (i.e., design and fabricate) and validated (i.e., tested with citrus samples and compared to standard laboratory procedures) a low-cost (i.e., eliminated the need for specialized laboratory equipment) instrument for the rapid processing of phloem-rich citrus tissues (i.e., budwood), named the budwood tissue extractor (BTE). As seen in Figure 1, the BTE includes a base component for power and controls, plus a removable chamber for the processing of citrus budwood. The BTE chamber is composed of a grinding wheel specifically designed to strip the phloem-rich bark tissues from the citrus budwood. The shredded bark tissue is ejected rapidly through a slide port into a syringe containing extraction buffer, filtered, and made ready for nucleic acid extraction and purification without any additional handling or preparation (Figure 1). The BTE system also includes a paperless sample tracking application and an integrated weighing application, which record the sample processing information in an online database in real time.
The BTE system has increased the CCPP's lab diagnostic capacity by over 100% and has consistently produced citrus tissue extracts suitable for the purification of high-quality nucleic acids and the downstream detection of graft-transmissible pathogens of citrus using PCR assays. More specifically, BTE has reduced the time for tissue processing from over 24 h to ~3 min per sample, replaced laboratory instruments costing over $60,000 (Figure 2, steps 2-4), and allowed for the processing of larger sample sizes.
This paper presents the BTE high-throughput citrus bark tissue processing, nucleic acid extraction, and pathogen detection validation data with citrus budwood samples from source trees, including all the appropriate positive and negative controls from the CCPP Rubidoux Quarantine Facility and Lindcove Foundation Facility, respectively. We also present the throughput and processing time changes compared to the current laboratory procedure (Figure 2). In addition, this work provides a detailed, step-by-step protocol for citrus pathogen testing laboratories and demonstrates how the BTE can support the functions of pathogen-clean nursery stock, survey, and eradication programs.

Figure 1: Budwood tissue extractor. The BTE includes a base component for power and controls, plus a removable chamber for the processing of citrus budwood. The BTE chamber is composed of a grinding wheel specifically designed to strip the phloem-rich bark tissues from citrus budwood. The shredded bark tissue is ejected rapidly through a slide port into a syringe, filtered, and made ready for nucleic acid extraction and purification without any additional handling or preparation. Abbreviation: BTE = budwood tissue extractor. Please click here to view a larger version of this figure.

Figure 2: Step-by-step comparison between the conventional hand-chopping lab procedure and BTE processing. BTE processing involves high-throughput citrus bark tissue processing, nucleic acid extraction, and pathogen detection. The time for each step is indicated in parentheses. Please click here to view a larger version of this figure.
Protocol
1. Collecting the citrus budwood samples to ship
- Send Technology Evolving Solutions a spreadsheet of tree information for them to load into their web server (eventually, the user will create new trees).
- Use the phone app TES tracker to select a tree, and hold a near-field communication (NFC) collar tag to the phone to load the tree information to the tag.
- Insert three to four citrus budwood samples into the BTE-compatible plastic bag carrier, and close with the lid.
- Ensure that the length of the budwood does not exceed 8 in and is not less than 5 in.
- If the length is greater than 8 in, use a sterile disposable razor blade or pruning shears decontaminated with 10% bleach solution (1% sodium hypochlorite) to make it shorter.
- If the length is less than 5 in, collect a different budwood sample to put into the bag.
- Ensure that any axillary growth or large thorns are removed by hand or using 10% bleach-sanitized cutting tools (1% sodium hypochlorite).
NOTE: This step is important so that the budwood is easier to maneuver in the opening of the chamber. - Ensure that there are no budwood samples that have curved features. If they do, remove them using 10% bleach-sanitized cutting tools (1% sodium hypochlorite), and place in the BTE bags only the straight portions of the sample.
NOTE: Curved budwood is very difficult to maneuver into the instrument, resulting in uneven bark striping.
- Ensure that the length of the budwood does not exceed 8 in and is not less than 5 in.
- Use the phone app TES tracker to scan the NFC collar tag of the tree and link it with the NFC clip tag on the sample bag.
- Pay attention to the thickness of the budwood as that determines how the operator will strip the phloem-rich budwood bark inside the BTE chamber.
- If the budwood has a thickness below 0.20 in, be careful while stripping the budwood because the high-velocity spinning threads in the chamber will pulverize the whole budwood to its core, beyond the bark layer and into the non-phloem-rich tissues of wood and pith.
- Select thicker budwood because it is easier to strip the bark tissues while avoiding the non-phloem-rich budwood tissues.
- Place the samples into an insulated shipping container with a few ice packs.
2. Setup in the fume hood
NOTE: It is preferred to operate the BTE inside a fume hood. This will reduce the risk of plant tissue cross-contamination and lab contamination.
- Disinfect using a 10% (1% sodium hypochlorite) spray bottle.
- Spray the surface of the hood, and let the bleach sit for around 1 min before wiping it with a paper towel.
- Spray a paper towel with the bleach solution. Wipe the TE base, weight scale components (scale, air shield, and tower), and a marker pen.
- Unwrap a syringe set for the number of samples to be processed, and place them inside a covered cardboard box.
- Have a pack of swabs available outside of the hood in case they are needed.
- Prepare the weight scale.
- Remove the weight tower from the scale, and hold the power button to turn it on. Place the tower on the center of the scale after the scale displays 0.
- Slide the weight scale air shield over the front of the scale.
- Prepare the tissue extractor base by flipping the switch on the back. Ensure the switch on the left side of the box is pressed down on the top. Wait for a blinking green LED, which indicates that the chamber is ready.
3. Set up the cleaning stations (Supplementary Figure S1).
- Place 1 L of water in the ultrasonic cleaner.
- Wrap two trash bags over the top of the ultrasonic cleaner.
- Pour ~5 L of 10% bleach (1% sodium hypochlorite solution) into the ultrasonic cleaner.
- Fill the water tub with enough water to submerge a chamber.
- Turn on the air compressor and open the valve.
- Set up a backdrop to catch the liquid while the chamber is drying.
4. Processing the material for the BTE for citrus budwood bark stripping
- Load the chamber onto the BTE Base.
- Prepare the BTE Chamber.
- Attach an empty sample bag to the back of the chamber. Use two O-rings to secure the empty bag to the back nozzle of the BTE chamber.
- Inspect the blade for signs of wear or damage, such as cuts forming on the blade where it continues into the plastic or cuts forming on the tip. Ensure the arrow on the blade lines up with the single lock symbol.
- Ensure the air release on the bottom of the chamber is turned toward the O symbol. Place the clear lid over the chamber opening, and slide the clear BTE slide onto the chamber using the track on the right of the chamber. Push the lock on the bottom of the chamber as far as it can go into the slide. Ensure the plunger cap is mounted on the top of the slide.
- Place the chamber on the BTE base with the BTE base's nozzle protruding into the back of the chamber. Wait for the blue LED to blink, indicating that the placement is successful.
- Prepare the BTE Chamber.
- Load the sample onto the BTE base by moving the white sticker on the NFC clip tag to a Z on the right side of the box. Move the tag in a slow circular motion on Z until the yellow light starts blinking. Attach the sample to the BTE base, and ensure that the sample bag has no holes in it; if there is a hole, patch it with tape. Place the O-ring over the sample bag to secure it to the front of the BTE nozzle.
- Load the unused syringe set.
- Tare the syringe set. Place the unused syringe set onto the weigh scale tower. Remove the syringe set when a red light begins flashing or the scale displays 0.
- Remove the plunger from the syringe with the filter. Ensure the fluid is in the bottom (non-filter) syringe. Place the plunger aside on a paper towel or on the tower where the black plunger is not touching any surfaces.
- Attach the syringe to the slide exit port by pressing the exit port into the syringe and turning 90°.
- Process the budwood sample.
CAUTION: BTE has high-velocity moving parts. All operation needs to occur inside a fume hood. All users should use protective eyeglasses, earmuffs (earplugs), and all other personal protective equipment (PPE), such as gloves and lab coats.- Press the top black button to start the machine. Press a second time to stop the motor at any time during processing.
NOTE: The box has speed and temperature sensing to ensure it functions properly. - Grab one budwood stick through the bag, and put it through the top of the BTE nozzle.
- Grab the budwood stick on the other side of the chamber with the other hand, and slowly inch down into the blade. Listen for a gentle buzzing sound indicating that the budwood is being stripped of its bark. Slowly move the budwood back and forth while rotating it.
- If a loud, aggressive chopping sound is heard, quickly move the branch to the top and/or press the top button to stop the motor. To stop the motor, use the right-side switch to finish processing (see step 4.4.3.2).
- If no material is coming out of the exit when cutting, then the chamber has a clog. Turn off the motor, remove the slide plunger cap, and use the plastic of the swab to push the clog out the exit of the machine. Use the right-side switch to finish processing: Up = Forward cutting; Middle = Motor off; Down = Reverse Cutting.
- Repeat step 4.4.2 and step 4.4.3 for the remaining branches.
NOTE: A general indicator that enough budwood bark has been stripped is when 25% of the syringe has been filled with plant tissue. Graft-transmissible pathogens of citrus could be unevenly distributed in the tree. Therefore, three to four budwood samples are collected from around the citrus tree canopy. It is important for every budwood sample in the BTE carrier bag to contribute to the final ground tissue sample to ensure that a full tree representative sample will be tested for pathogens. - Press the top black button to stop the processing. Wait for the light to begin flashing yellow again.
- Press the top black button to start the machine. Press a second time to stop the motor at any time during processing.
- Verify the sample weight.
- Rotate the syringe 90°, and pull it down to detach. Place the plunger back on the syringe, and place the syringe on the tower.
- Wait for the scale to auto-detect the sample, and determine if it is within the proper weight range (0.25 ± 0.05 g). If the sample weight is too low (<0.20 g), repeat step 4.4; the red LED will begin blinking. If the sample weight is too high (>0.30 g), remove some of the sample material; the yellow LED will begin blinking more slowly. If the sample is within the range, the green LED will begin blinking.
- Homogenizing the budwood sample
- Remove the plunger from the syringe with the sample, push the fluid into the syringe with the sample, and re-add the plunger. The plunger pushes the buffer and plant sap through the mesh filter (withholding any large bark tissue pieces) and via the rubber tubing into the empty syringe.
- Mix the sample by pushing the buffer and plant sap back and forth from one syringe to the other. Repeat ~3x-4x and until the sample becomes a homogeneous green liquid mixture.
- Once well homogenized, push the plant sap sample into the syringe without the mesh filter, and detach the rubber tubing and the syringe with the filter from the syringe with the sample.
- Expel the plant sap sample from the syringe into a 2 mL sterile microcentrifuge tube, and store at −20 °C until further use. Use a permanent marker to label the sample with the bag number.
NOTE: The plant sample sap from step 4.6.5 can now be processed with any of the available nucleic acid, RNA or DNA, extraction methods based on phenol-chloroform, silica column, or magnetic beads9,25,26,27 for nucleic acid extraction and purification (see step 7) and the downstream detection of graft-transmissible pathogens of citrus (see step 8).
5. Sanitizing the BTE removable chamber
CAUTION: If the buffer from the syringe set gets onto the chamber or slide, rinse, and follow all the safety rules of the laboratory before cleaning. The syringe set contains guanidine thiocyanate. If the buffer from the syringe set comes into contact with bleach, it will create cyanide gas.
- Perform the following steps after the 10th sample is processed in the chamber. Note that the green LED will continue blinking instead of moving to blue blinking.
- Disassemble the BTE chamber to clean all the possible contaminants; remove the clear plastic cover, clear the chamber slide, and clear the clog port on the chamber slide.
- Turn the air release valve on the bottom of the chamber to the open position (open padlock mark). Place the chamber components into the set-up ultrasonic cleaner (see step 3.1). Place the lid under the chamber to prevent it from floating. Run the ultrasonic cleaner for 15 min.
NOTE: The ultrasonic cleaning bath (5 L) can hold up to two chambers. - Rinse the chamber components in the water bath (step 3.4) for at least 30 s. Move the chamber components to the drying station.
CAUTION: Drying will cause fast-moving parts within the chamber, loud noises (~85 decibels [dB]), and possible splashing. All users should use protective eyeglasses, earmuffs (earplugs), lab coats, and all other PPE as required by the safety rules of the operating laboratory. - Dry the BTE chamber.
- Position the air gun in line with the groove of the upper opening of the chamber (where the slide would typically be). Press the trigger of the gun to dispense air for ~30 s.
NOTE: Ensure the opening is away from the user toward the backdrop. This should spin the blade set to remove the liquid trapped underneath it. - Position the air gun toward the top nozzles of the chamber. Press the trigger of the air gun, and slowly trace three full circles of each nozzle entrance.
- Position the air gun in the center of the BTE. Starting from the innermost point, hold the trigger down, and move until the air gun points toward where the slide would be.
- Quickly run air over the entire chamber, front and back, to get the surface water off the outside.
- Position the air gun in line with the groove of the upper opening of the chamber (where the slide would typically be). Press the trigger of the gun to dispense air for ~30 s.
- Dry the BTE slide.
- Position the air gun into the plunger slot of the slide, and press the trigger to eject water out of the slide exit.
- Run an air gun along the interior surface of the slide to dry it.
- Run an air gun along the slide groove to push out water.
- Quickly run air over the entire exterior to get surface water off it.
- Dry the components.
- Hold the slide plunger cap and O-rings in hand, and press the trigger.
- Run an air gun over the interior surface of the lid.
- Place the components in the chamber and slide to continue drying or reassemble them.
NOTE: For a high tissue-processing environment with multiple functional BTEs, a batch sanitizing system can be provided that is capable of decontaminating 10 chambers simultaneously.
6. Disposing and sanitizing
- Dispose of the syringes and rubber tubing in the designated guanidine waste container.
- Dispose of any plant material in the biohazardous waste container.
- Remove the BTE chamber from the BTE base.
- Disassemble the BTE chamber and proceed to their decontamination, as described in step 5.
7. Assessment of the tissue processing and the quality of RNA purified from the BTE citrus budwood extracts
NOTE: In this protocol, we used budwood samples from 255 citrus trees to compare the time required for citrus budwood tissue processing and the quality of RNA purified from bark tissue extracts prepared by BTE (Figure 2, right side, step 1, step 5, and step 6) versus that prepared following the regulatorily approved citrus budwood tissue-processing method utilizing hand peeling and chopping, freeze-drying, pulverization, and centrifugation of the bark tissue, as described by Dang et al.23 (Figure 2, left side, steps 1-6).
- Current laboratory procedure: Prepare budwood samples for tissue processing as per the regulatorily approved method23.
- Carry out hand peeling and chopping of the bark tissue, freeze-drying, pulverization, centrifugation, and transfer of the plant sap to the RNA extraction tube, as described by Dang et al.23 (Figure 2, left side, steps 1-6).
- After hand peeling the three to four budwood samples from each tested tree, place all the budwood inside a BTE-compatible plastic bag carrier, and store at 4 °C until the BTE processing (step 7.2).
- BTE procedure: Initiate the BTE citrus budwood tissue processing (section 4, steps 4.1-4.6; Figure 2, right side, step 1, step 5, and step 6).
- Use the budwood samples stored inside the BTE carrier bags from step 7.1.2.
- Extract and purify the RNA from the plant sap obtained from the current laboratory procedure (step 7.1.1) and the BTE procedure (step 7.2.2) using the regulatorily approved semi-automated magnetic bead-based method8,23,28.
- Assess the RNA quality by measuring its concentration, purity, and integrity8,23,24,29,33,31.
- To calculate the concentration, use spectrophotometry and optical density (OD) at a wavelength of 260 nm.
- To assess the purity, use a spectrophotometric OD ratio of 260/280.
- To check the integrity, use reverse transcription (RT) quantitative polymerase chain reaction (qPCR) reaction targeting the mRNA of the NADH dehydrogenase citrus gene24,32.
8. Assessment of the cross-contamination and detection of citrus viruses and viroids using RNA purified from BTE citrus budwood extracts
NOTE: In this protocol, we used budwood samples from 72 non-infected citrus trees and one tree mix-infected with viruses and viroids to assess the potential of cross-contamination between samples when processed by BTE (Figure 2, right side, step 1, step 5, and step 6) and the suitability of RNA purified from bark tissue extracts prepared by BTE to be used as a template for the RT-qPCR detection of citrus viruses and viroids.
- First BTE sample processing: Conduct a baseline experiment with 72 non-infected samples.
- Prepare three (A-C) BTE chambers (step 4.1.1).
- Prepare all the non-infected citrus budwood samples (1-72) in BTE carrier bags, and prepare an equal number (72) in the two-syringe BTE sample collection system, with one syringe system for each sample (step 2.4).
- Separate the sample carrier bags and sample collection syringes into six batches (I-VI) of 12 samples each.
- Initiate the BTE citrus budwood tissue processing (step 4) following the sequence below (steps 8.1.4.1-8.1.4.6) (See Table 1, First BTE Sample Processing).
- For Batch I/Samples 1-12, process 12 samples using chamber A. Sanitize chamber A after sample 12 (step 5).
- For Batch II/Samples 13-24, process 12 samples using chamber B. Sanitize chamber B after sample 24 (step 5).
- For Batch III/Samples 25-36, process 12 samples using chamber C. Sanitize chamber C after sample 36 (step 5).
- For Batch IV/Samples 37-48, process 12 samples using the sanitized chamber A (step 8.1.4.1).
- For Batch V/Samples 49–60, process 12 samples using the sanitized chamber B (step 8.1.4.2).
- For Batch VI/Samples 60-72, process 12 samples using the sanitized chamber C (step 8.1.4.3).
- Store the BTE carrier bags with all the samples at 4 °C until use at step 8.2.
- Extract and purify the RNA from the 72 plant sap samples generated in steps 8.1.4.1-8.1.4.6 using the regulatorily approved semi-automated magnetic bead-based method8,23,28.
- Perform RT-qPCR for the detection of citrus viruses and viroids, as previously described8,33.
- For the second BTE sample processing, perform a cross-contamination experiment with 70 non-infected samples and two mixed infected samples.
- Prepare three (A-C) BTE chambers (step 4.1.1).
- Prepare the mix-infected citrus budwood sample (73) in two BTE carrier bags (step 1.3) for a total number of two infected samples.
- Gather the BTE carrier bags with the non-infected citrus budwood samples from step 8.1.5, except Sample 3-Batch I and Sample 51-Batch V (see sample replacement in step 8.2.4), for a total number of 70 non-infected samples.
- Include the first BTE carrier bag with the mix-infected sample 73 in place of Sample 3 in Batch I and the second one in place of Sample 51 in Batch V for a total number of 72 samples.
- Prepare an equal number (72) in the two-syringe BTE sample collection system, with one double-syringe system for each sample (step 2.4).
- Separate the 72 sample carrier bags and sample collection syringes into six batches (I-VI) of 12 samples each.
- Initiate the BTE citrus budwood tissue processing (step 4), following the same sequence as in steps 8.1.4.1-8.1.4.6.
NOTE: The only difference is the replacement of Sample 3 in Batch I and Sample 51 in Batch V with the infected sample 73 (step 8.2.4) (Table 1, Second BTE Sample Processing). - Extract and purify the RNA from the 72 plant sap samples generated in step 8.2.7 as in step 8.1.6.
- Perform RT-qPCR for the detection of citrus viruses and viroids, as in step 8.1.7.
Representative Results
RNA extraction, purification, and quality using BTE-processed budwood citrus tissue and assessment of time for tissue processing
We used budwood samples from 255 representative citrus trees for this test to compare the RNA quality from the BTE versus the standard procedure. Samples were processed by the budwood tissue extractor (BTE) (protocol steps 4.1-4.6 and Figure 2, right side, step 1, step 5, and step 6) or prepared following the regulatorily approved citrus budwood tissue processing method, which utilizes hand peeling and chopping, freeze-drying, pulverization, and centrifugation of the bark tissue, as described by Dang et al.23(Figure 2, left side, steps 1-6).
The side-by-side comparison of the BTE with the conventional hand-chopping and laboratory equipment protocol for citrus tissue processing demonstrated that the quality (i.e., concentration, purity, and integrity) of the extracted nucleic acids (Figure 3A–C) and suitability for downstream use for the PCR detection of citrus pathogens (data not shown) were comparable. At the same time, the time spent processing samples was significantly reduced using the TE/BTE system. The BTE more than doubled the sample throughput of the CCPP laboratory, reducing the labor and the laboratory equipment costs by eliminating the need for tens of thousands of dollars of instruments, such as beads beaters, centrifuges, and cryostations.
The nucleic acids extracted with BTE had an average concentration of 76.96 ng/µL ± 26.23 ng/µL (n = 181) and were of high purity, with low protein contamination (A260/A280 2.27 ± 0.17, n = 181) (Figure 3B,C). These values were comparable to the nucleic acids produced by CCPP's standard manual protocol (concentration: 82.25 ng/µL ± 33.95 ng/µL, n = 181 and A260/A280 2.22 ± 0.10, n = 181) (Figure 3B,C). The nucleic acid integrity (RT-qPCR for the citrus gene nad5) was very similar for BTE (Cq 20.97 ± 2.26, n = 181) and the standard manual CCPP protocol (Cq 19.25 ± 1.53, n = 181) (Figure 3A). The results also demonstrated that the BTE instrument could process a higher sample volume in the same time frame compared to the conventional method. The conventional lab procedure required ~7-10 min for hand-chopping per sample and a total of 12 min for tissue processing (freeze-drying, grinding, and centrifuge), while the BTE could process citrus tissue for nucleic acid extraction in ~3 min per sample.
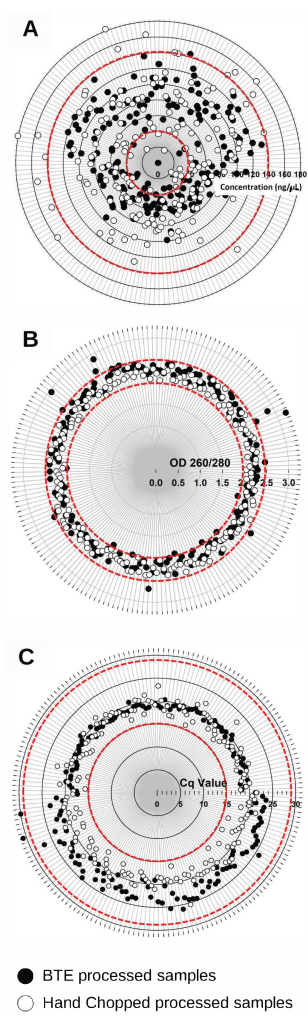
Figure 3: Quality of the nucleic acid extracts of the 181 representative citrus budwood samples, as defined by the concentration, purity, and integrity. The samples were processed by BTE and the conventional hand-chopping and laboratory equipment protocol. (A) The nucleic acid concentration was determined using absorbance at 260 nm; (B) the purity was determined as the ratio of the absorbances at 260 nm and 280 nm (A260/280). (C) The nucleic acid integrity was analyzed by RT-qPCR targeting the mRNA of the NADH dehydrogenase (Nad5) citrus gene. The ranges of optimal values are indicated between red dashed circles. Please click here to view a larger version of this figure.
Detection of graft-transmissible pathogens of citrus, assessment of cross-contamination, and detection of citrus viruses and viroids using RNA purified from BTE citrus budwood extracts
The validity of the tissue processing method was evaluated by processing 72 healthy citrus budwood samples side by side with all healthy samples and with the introduction of two samples from a tree mix infected with viruses and viroids (Table 1 and Figure 4A,B). Nucleic acids extracted from both batches were subjected to conventional lab-based q-PCR as previously described8,33. None of the 72 healthy samples from the first BTE sample processing gave amplification curves for the citrus pathogens tested (i.e., false positives) (Figure 4A). The results suggest that the BTE can process citrus tissue equally well compared to the standard laboratory protocol with hand-chopping methods. In the second BTE sample processing, we assessed the potential for cross-contamination between BTE heads and within samples processed with the same BTE head (steps 1-6) and the suitability of the nucleic acids for downstream applications (i.e., for use as a template for the RT-qPCR detection of citrus viruses and viroids). In the second BTE sample processing with two introduced mix-infected samples, the nucleic acids produced by the BTE protocol were successfully used to detect different citrus viruses and viroids (e.g., triplex virus, citrus leaf blotch virus [CLBV], citrus psorosis virus [CPsV], citrus tristeza virus [CTV]), apscaviroids (citrus bent leaf viroid [CBLVd], citrus dwarfing viroid [CDVd], citrus viroid V [CVd-V], citrus viroid VI [CVd-VI], and citrus viroid VII [CVd-VII]), non-apscaviroids hop stunt viroid (HSVd; hostuviroid), citrus bark cracking viroid (CBCVd; cocadviroid), and citrus exocortis viroid (CEVd; pospiviroid) in batch 1 and batch 5. Within batch 1 and batch 5, samples that followed the infected one were positive for the above plant diseases but had increasing Cq values. However, no cross-contamination between heads was detected nor any false positive or negative results (Figure 4B).
| Batch | Sample | BTE | First BTE | Second BTE |
| Chamber | Sample Processing | Sample Processing | ||
| I | 12-Jan | A | 1-12 Non-infected | 1-2 & 4-12 Non-infected |
| Sample #3 is replaced with mix infected | ||||
| II | 13-24 | B | 13-24 | 13-24 |
| Non-infected | Non-infected | |||
| III | 25-36 | C | 25-36 | 25-36 |
| Non-infected | Non-infected | |||
| IV | 37-48 | A-Sanitized | 37-48 | 37-48 |
| Non-infected | Non-infected | |||
| V | 49-60 | B-Sanitized | 49-60 | 49-50 & 52-60 Non-Infected |
| Non-infected | ||||
| Sample #51 is replaced with mix infected | ||||
| VI | 61-72 | C-Sanitized | 61-72 Non-infected | 61-72 Non-infected |
Table 1: Process followed for the validation of the BTE tissue processing method using 72 citrus budwood samples. Each processing was repeated twice. There were 6 batches with 12 samples each. In the first sample processing, all 72 citrus budwood samples were healthy. In the second BTE sample processing, sample 3 and sample 51 were replaced with two samples from a tree mix-infected with viruses and viroids in batch 1 and batch 5.
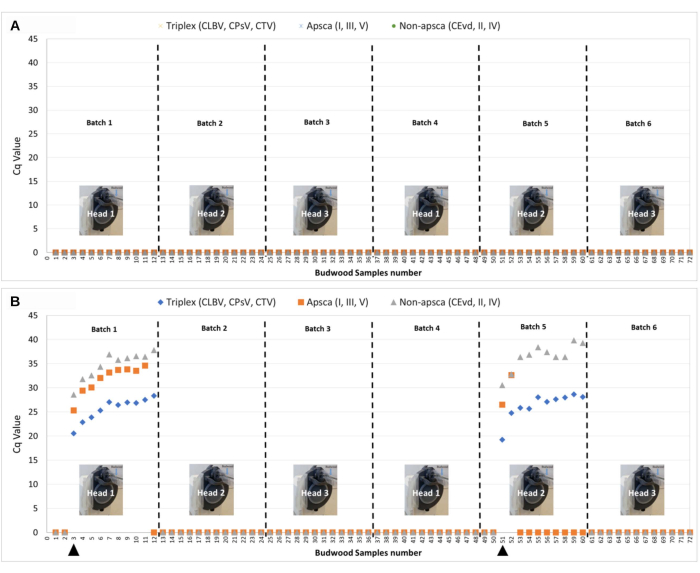
Figure 4: Validation of the BTE tissue processing method using 72 citrus budwood samples. Each processing was repeated twice. There were 6 batches with 12 samples each. (A) All 72 citrus budwood samples were healthy. (B) The same 72 citrus budwood samples with the introduction of two samples from a tree mix-infected with viruses and viroids in batch 1 (sample 3) and batch 5 (Sample 51). The NTC and water controls all had undetermined Cq values (i.e., DNA target not present in the sample). The positive controls for the triplex (CLBV, CPsV, CTV) had Cq values of 23.9, 25.2, and 22.4 respectively. The positive controls for apscaviroids (CBLVd, CDVd, and CBCVd) had Cq values of 23.39, 21.27, and 25.17 respectively. The positive controls for non-apscaviroids (CEVd, HSVd, IV) had Cq values of 26.9, 27.0, and 26.5 respectively. Abbreviations: BTE = budwood tissue extractor; NTC = no-template control. Please click here to view a larger version of this figure.
Supplementary Figure S1: Cleaning station setup. After the 10th sample is processed in the chamber, protocol steps 3.1-3.6 are to be followed to prepare the cleaning station to sanitize the chamber. As in protocol step 3.1, 1 L of water is placed in the ultrasonic cleaner. Two trash bags are wrapped over the top of the ultrasonic cleaner (protocol step 3.2), and ~5 L of 10% bleach (1% sodium hypochlorite solution) is poured into the ultrasonic cleaner (protocol step 3.3). The water tub is filled with enough water to submerge a chamber (protocol step 3.4), the air compressor turned off, and the valve is opened (protocol step 3.5). A backdrop is set up to catch the liquid while the chamber is drying (protocol step 3.6). Please click here to download this File.
Discussion
With the advent of HLB citrus disease, to reduce losses, the citrus industry, regulatory agencies, and diagnostic laboratories have been urged to rely on high-throughput nucleic acid extraction methods combined with low-throughput manual sample processing and pathogen detection assays such as qPCR34 for the testing of individual trees, in combination with disease management practices35. California’s HLB positivity rate has gone from 0.01% in 2012 to 1.2% in 2020. Even though qPCR is a powerful and reliable pathogen detection tool, the currently available technologies do not allow an adequate volume of plant tissue to be sampled and processed, resulting in clear undersampling and undertesting for CLas in citrus trees in California. Undersampling and undertesting occur in relation to both the number of trees tested and the quantity of plant material per tree being processed (i.e., number of leaves), and due to the sporadic distribution of infected leaves within the tree canopy, there is a high probability of missing early or mild infections. The current methods for sampling and CLas testing cannot scale with increased demand due to costs (e.g., labor and equipment). Currently, the sample processing method used by most citrus diagnostic laboratories in California to acquire nucleic acids suitable for citrus pathogen testing requires over 17 h for manual sample handling and specialized supplies and equipment costing over $100,000.
Here, we present the validation of a specialized instrument engineered to rapidly process citrus budwood bark tissues, named the budwood tissue extractor (BTE), and a detailed, step-by-step protocol of sample handling for citrus diagnostic laboratories. The research focused on developing an innovative tissue processing method to replace the current labor-intensive hand-chopping of budwood samples and build the highest-throughput and most cost-effective citrus budwood processing instrument possible. The results show that BTE increases the sample throughput and decreases the cost of equipment and supplies used at the CCPP diagnostic laboratory. The presented technology and method also include the use of NFC tags, a phone app, and an online database for real-time sample information tracking. After budwood samples are collected, the phone app links the NFC sample bag clip with the NFC tree tag. The samples are then shipped to the laboratory for processing with the BTE machine. Information for each sample is recorded with a quick swipe across the side of the BTE body. Through the sample NFC tag, the processing time for each sample and the sample’s weight are also recorded and uploaded to the online database in real time. This method provides a very time-efficient system, improves the quality control (i.e., by avoiding sample ID errors), and increases the laboratory efficiency.
The BTE high-throughput processing and validation were carried out with “real-life/field” citrus samples, demonstrating that other diagnostic laboratories can readily adopt the technology and methodology. Adopting this technology will allow for decreasing the operational costs and increasing the diagnostic capacity and laboratory efficiency, thus increasing the chances of identifying diseased trees at an earlier stage after infection occurs and lowering the chances of disease spread. The side-by-side comparison of BTE versus conventional tissue-processing methods demonstrates that the quality of extracted nucleic acid (i.e., concentration, purity, and integrity) and the results of the downstream pathogen detection are comparable (Figure 3A–C). However, the time spent to process the samples is significantly reduced using the BTE compared to manual processing protocols (i.e., 3.3 min vs. 6.8 min). The chambers and BTE base do require a regular cleaning schedule. Although the cleaning is time-consuming and constitutes a limitation of the method, the technology allows multiple samples to be rapidly processed in the BTE before the chamber needs to be cleaned. Conventional methods require less cleaning as each sample has its own processing container, which, in many cases, is disposable, but they need more containers and storage space (e.g., 4 °C refrigerators and −20 °C or −80 °C freezers).
The BTE process was conceived and built to increase the probability of identifying a problem area via bulk sample processing and testing (i.e., finding a diseased tree within a large citrus germplasm foundation block, nursery, or orchard) by running only a small number of bulk tests that can identify hot spots. Therefore, it only deviates when a positive result is found. When this happens (Figure 4B, batch 1 and batch 5), subsequent sample processing and the testing of individual samples from the same batch containing the positive material (i.e., only a subset of samples) are required to determine which samples are positive. Thus, it is important to note that this procedure is primarily suitable for cases with low infection rates, where it allows the testing of a high volume of material from many trees, which, in return, increases the probability of disease detection without having to test a large number of individual samples. Conventional methods will be the optimal option in cases with high infection rates, as they allow each sample to be tested individually. This is especially important in the case of HLB surveys in California20 (still at a low HLB positivity rate), where the utilized PCR-based methods for CLas detection are primarily meant to confirm the presence or absence of CLas in asymptomatic trees (i.e., no typical blotchy mottle, canopy yellowing, or tree decline) that have been exposed to CLas-positive Asian citrus psyllids (ACP). Given the fact that we do not know where in the tree an infective ACP has fed, the chances of identifying an infected but asymptomatic tree in the field or in any other large area or operation by testing a small number of samples are very low (e.g., in California, currently 20 leaves are collected, and 12 leaves are tested per tree20). Obviously, the larger the tree canopy, the lower the chances of CLas detection. Even though the costs of the PCR itself (i.e., reagents, basic laboratory equipment, and thermocycler), which follows the sample processing, and the nucleic acid extraction and purification continue unchanged in the BTE process, the uncertainty regarding where to collect the samples from asymptomatic trees to determine whether they are infected or not still persists and, in our opinion, is the Achilles’ heel of the HLB survey in California. The presented instrument, technology, method, and improvements will allow the processing of larger sample sizes in a shorter time and at a lower cost. Therefore, this method will increase the probability of identifying infected trees in a timely manner and improve the eradication efforts for HLB or other invasive pathogens of citrus in California (e.g., citrus yellow vein clearing virus reported in California in August 2022).
Combined with automated tissue processing methods, bulk sampling and testing would allow for the reduction of plant tissue processing costs. This cost reduction would allow many diagnostic laboratories to more effectively screen orchards in many states and to better track the movement of HLB and other diseases. Ultimately, this means growers would have a more effective tool to slow the spread of diseases through more efficient large-scale testing. While the BTE instrument can improve citrus laboratories’ current diagnostic capacity and benefit the whole citrus industry, at the same time, the technology can also be transferred to other tree crops. The preliminary data of nucleic acid concentration and purity, as estimated by the OD, have indicated that BTE can effectively process tissues from almond (59.87 ng/µL ± 7.43 ng/µL and A260/A280 1.17 ± 0.05, n = 9), grapevine (135.74 ng/µL ± 50.74 ng/µL and A260/A280 1.17 ± 0.06, n = 9), and peach (66.50 ng/µL ± 6.07 ng/µL and A260/A280 1.29 ± 0.05, n = 9) (samples kindly provided by Foundation Plant Services at UC Davis).
The budwood tissue processing platform presented here has inspired the development of additional plant tissue processing equipment. For example, a leaf tissue extractor (LTE) is currently under development. Preliminary testing results using citrus leaves have demonstrated that the LTE instrument could process about seven times more leaves with only a 35% increase in processing time over the current standard 12 leaf method used in California20. We have estimated that an LTE-supported bulk sampling and testing protocol could reduce the overall cost per citrus leaf test by a fourth. Research to prove the value of the practical application of this technology is currently ongoing under a CDFA Specialty Crop Block Grant in our laboratories.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors acknowledge the Cahuilla people as the Traditional Custodians of the Land on which the experimental work was completed. We are grateful to Professor Norman Ellstrand at the University of California, Riverside, for providing lab space to carry out research activities for this project under the UCR California Agriculture and Food Enterprise (CAFÉ) Initiative. This research was supported by the CDFA – Specialty Crop Block Grant Program (grant no. 18-0001-055-SC). Additional support was also provided by the CRB project 6100; USDA National Institute of Food and Agriculture, Hatch project 1020106; and the National Clean Plant Network-USDA Animal and Plant Health Inspection Service (AP17PPQS&T00C118, AP18PPQS&T00C107, AP19PPQS&T00C148, & AP20PPQS&T00C049) awarded to Georgios Vidalakis.
Materials
| 0.08" Hex Trimmer line | PowerCare | FPRO07065 | Needed to replace blades. |
| 1 Hp, 8 gal air compressor | California Air Tools | 8010 | Quickly dry chambers after rinsed |
| 1.5 mL microcentrifuge tube | Globe Scientific | 111558B | Store sample in after swishing with syinges |
| 10 mL Syringe Set | Technology Evolving Solutions | TE006-F1-10A-G1000-E1 | Syringe material is cut into. 1 L bottle with guanidine thiocyanate buffer. WARNING – contains guanidine thiocyanate, hazardous waste service required – do not mix with bleach |
| 12" Ruler | Westcott | 16012 | To measure trimmer line before cutting |
| 12% Sodium Hypochlorite | Hasa | 1041 | Disinfects chambers after processing |
| -20 C Freezer | Insignia | NS-CZ70WH0 | Store sample after processing |
| 4" x 12" plastic bags | Plymor | FP20-4×12-10 | Bags to hold branches during shipping. O-rings attach bag to BTE chamber to seal |
| 6" Cotton Swab | Puritan | 806-PCL | Swab to remove clogs |
| 7 Gallon Storage Tote | HDX | 206152 | Holds sodium hypochlorite solution to disinfect chambers and water to rinse chambers |
| Air blow gun | JASTIND | JTABG103A | Directs air into the chambers at high pressure |
| Black Sharpie | Sharpie | S-19421 | Mark 1.5 mL tubes so you can identify sample later |
| Bottle Top Dispensor | Brand | Z627569 | Adjustable bottle top dispensor to dispense guandine into syringe |
| BTE Chamber | Technology Evolving Solutions | TE002BB-A05-E1 | Used to process budwood. Includes O-rings, BTE Slide, slide plunger, drain valve, lid, blade set, and blade set removal tool |
| Dish Soap | Dawn | 57445CT | Surfectant to improve sodium hypochlorite penetration into chamber |
| Fume hood with hepa filter | Air Science | P5-36XT-A | Fume hood with hepa filter (ASTS-030) to limit possible contamination and protect against chemical spills |
| Insulated foam shipping container | PolarTech | 261/J50C | Insulated shipping container to ship samples on ice after they are collected |
| Lab coat | Red Kap | KP14WH LN 46 | Lab coat to limit possible contamination and protect against chemical spills |
| Laptop | Microsoft | Surface | Wifi capable laptop to run TES GUI. Needed for initial setup and provides more indepth information about the tissue processing base |
| NFC Capable Phone | Samsung | Galaxy S9 | Phone to download and use TES phone app |
| NFC clip tag | Technology Evolving Solutions | TE005-Clip-E1 | Sample tag that can be linked with trees. Made to function with TES phone app |
| NFC Collar Tag | Technology Evolving Solutions | TE005-Collar-E1 | Tag that is attached to a tree. Made to function with TES phone app |
| Nitrile Gloves | Usa Scientific | 3915-4400 | Gloves to limit possible contamination and protect against chemical spills |
| Noise-Reducing Earmuff | 3M | 90565-4DC-PS | Protect ears while operating air compressor and tissue processing base |
| Polyurethane Recoil Air Hose | FYPower | 510019 | Attaches air gun to compressor |
| Saftey glasses | Solidwork | SW8329-US | Protect eyes for chemical and physical hazards |
| Spray bottle | JohnBee | B08QM81BJV | Spray bleach to deconatinate surfaces |
| Tissue Extractor Base | Technology Evolving Solutions | TE001-A-E1 | System to process plant tissue. Needs BTE or LTE chambers to function. Includes power cable, blade adapter, and 8/32" allen wrench |
| Tissue Processing Base Weight Scale | Technology Evolving Solutions | TE003-A05-200g-01-E1 | 200 g, 0.01 resolution weight scale that connects to tissue processing base to enforce weight ranges and/or link weights with sample. Includes scale, power cable, connection cable, 5ml syringe holder, tower air shield |
| Vermiculite | EasyGoProducts | B07WQDZGRP | Needed to transport hazardous waste (guanidine thiocyanate) using a hazardous waste disposal service |
| Wire Cutter | Boenfu | BOWC-06002-US | Wire cutters to cut trimmer line |
References
- Vernière, C., et al. Interactions between citrus viroids affect symptom expression and field performance of clementine trees grafted on trifoliate orange. Phytopathology. 96 (4), 356-368 (2006).
- Vernière, C., et al. Citrus viroids: Symptom expression and effect on vegetative growth and yield of clementine trees grafted on trifoliate orange. Plant Disease. 88 (11), 1189-1197 (2004).
- Zhou, C., Talon, M., Caruso, M., Gmitter, F. G., et al. Chapter 19 – Citrus viruses and viroids. The Genus Citrus. , 391-410 (2020).
- Trends and issues facing the U.S. citrus industry. Choices Magazine Online Available from: https://www.choicesmagazine.org/choices-magazine/theme-articles/trends-and-challenges-in-fruit-and-tree-nut-sectors/trends-and-issues-facing-the-us-citrus-industry (2021)
- Fruit and Tree Nuts Outlook. United States Department of Agriculture-Economic Research Service Available from: https://www.ers.usda.gov/webdocs/outlooks/98171/fts-370.pdf?v=5697 (2020)
- Forsyth, J., Fruits Damiani, J. C. i. t. r. u. s. Citrus Fruits. Types on the market. Encyclopedia of Food Sciences and Nutrition. , 1329-1335 (2003).
- Bostock, R. M., Thomas, C. S., Hoenisch, R. W., Golino, D. A., Vidalakis, G. Plant health: How diagnostic networks and interagency partnerships protect plant systems from pests and pathogens. California Agriculture. 68 (4), 117-124 (2014).
- Osman, F., Dang, T., Bodaghi, S., Vidalakis, G. One-step multiplex RT-qPCR detects three citrus viroids from different genera in a wide range of hosts. Journal of Virological Methods. 245, 40-52 (2017).
- Wang, J., et al. Past and future of a century old Citrus tristeza virus collection: A California citrus germplasm tale. Frontiers in Microbiology. 4, 366 (2013).
- Gergerich, R. C., et al. Safeguarding fruit crops in the age of agricultural globalization. Plant Disease. 99 (2), 176-187 (2015).
- Moreno, P., Ambrós, S., Albiach-Martí, M. R., Guerri, J., Peña, L. Citrus tristeza virus: A pathogen that changed the course of the citrus industry. Molecular Plant Pathology. 9 (2), 251-268 (2008).
- Yokomi, R. K., et al. Identification and characterization of Citrus tristeza virus isolates breaking resistance in trifoliate orange in California. Phytopathology. 107 (7), 901-908 (2017).
- Selvaraj, V., Maheshwari, Y., Hajeri, S., Yokomi, R. A rapid detection tool for VT isolates of Citrus tristeza virus by immunocapture-reverse transcriptase loop-mediated isothermal amplification assay. PLoS One. 14 (9), 0222170 (2019).
- Babcock, B. A. Economic impact of California’s citrus industry in 2020. Journal of Citrus Pathology. 9, (2022).
- Gottwald, T. R., Polek, M., Riley, K. History, present incidence, and spatial distribution of Citrus tristeza virus in the California central valley. International Organization of Citrus Virologists Conference Proceedings (1957-2010). 15, (2002).
- Yokomi, R., et al. Molecular and biological characterization of a novel mild strain of citrus tristeza virus in California. Archives of Virology. 163 (7), 1795-1804 (2018).
- Fuchs, M., et al. Economic studies reinforce efforts to safeguard specialty crops in the United States. Plant Disease. 105 (1), 14-26 (2021).
- The real cost of HLB in Florida. Citrus Industry Magazine Available from: https://citrusindustry.net/2019/07/30/the-real-cost-of-hib-in-florida/ (2019)
- McRoberts, N., et al. Using models to provide rapid programme support for California’s efforts to suppress Huanglongbing disease of citrus. Philosophical Transactions of the Royal Society B: Biological Sciences. 374 (1776), 20180281 (2019).
- Albrecht, C., et al. Action plan for Asian citrus psyllid and huanglongbing (citrus greening) in California. Journal of Citrus Pathology. 7 (1), (2020).
- Navarro, L., et al. The Citrus Variety Improvement Program in Spain in the period 1975-2001. International Organization of Citrus Virologists Conference Proceedings (1957-2010). 15 (15), (2002).
- Vidalakis, G., Gumpf, D. J., Polek, M. L., Bash, J. A., Ferguson, L., Grafton-Cardwell, E. E. The California Citrus Clonal Protection Program. Citrus Production Manual. , 117-130 (2014).
- Dang, T., Rao, A. L. N., Lavagi-Craddock, I., Vidalakis, G., et al. High-throughput RNA extraction from citrus tissues for the detection of viroids. In Viroids: Methods and Protocols. 2316, (2022).
- Osman, F., Vidalakis, G., Rao, A. L. N., Lavagi-Craddock, I., Vidalakis, G. Real-time detection of viroids using singleplex and multiplex quantitative polymerase chain reaction. Viroids: Methods and Protocols. 2316, (2022).
- Li, R., et al. A reliable and inexpensive method of nucleic acid extraction for the PCR-based detection of diverse plant pathogens. Journal of Virological Methods. 154 (1-2), 48-55 (2008).
- Saponari, M., Manjunath, K., Yokomi, R. K. Quantitative detection of Citrus tristeza virus in citrus and aphids by real-time reverse transcription-PCR (TaqMan). Journal of Virological Methods. 147 (1), 43-53 (2008).
- Damaj, M. B., et al. Reproducible RNA preparation from sugarcane and citrus for functional genomic applications. International Journal of Plant Genomics. 2009, 765367 (2009).
- Dang, T., et al. First report of citrus leaf blotch virus infecting Bearss lime tree in California. Plant Disease. 104 (11), 3088 (2020).
- Manchester, K. L. Use of UV methods for measurement of protein and nucleic acid concentrations. BioTechniques. 20 (6), 968-970 (1996).
- Teare, J. M., et al. Measurement of nucleic acid concentrations using the DyNA QuantTM and the GeneQuantTM. BioTechniques. 22 (6), 1170-1174 (1997).
- Imbeaud, S. Towards standardization of RNA quality assessment using user-independent classifiers of microcapillary electrophoresis traces. Nucleic Acids Research. 33 (6), 56-56 (2005).
- Menzel, W., Jelkmann, W., Maiss, E. Detection of four apple viruses by multiplex RT-PCR assays with coamplification of plant mRNA as internal control. Journal of Virological Methods. 99 (1-2), 81-92 (2002).
- Vidalakis, G., Rao, A. L. N., Lavagi-Craddock, I., Vidalakis, G., et al. SYBR Green RT-qPCR for the universal detection of citrus viroids. Viroids: Methods and Protocols. , 211-217 (2022).
- Arredondo Valdés, R., et al. A review of techniques for detecting Huanglongbing (greening) in citrus. Canadian Journal of Microbiology. 62 (10), 803-811 (2016).
- Li, S., Wu, F., Duan, Y., Singerman, A., Guan, Z. Citrus greening: Management strategies and their economic impact. HortScience. 55 (5), 604-612 (2020).
- . CDFA California Citrus Pest and Disease Prevention Program Operations Subcomittee Meeting. Meeting Minutes Available from: https://www.cdfa.ca.gov/citrus/docs/minutes/2019/OpsSubcoMinutes-11062019.pdf (2019)

