Ex Vivo OCT-Based Multimodal Imaging of Human Donor Eyes for Research into Age-Related Macular Degeneration
Summary
Laboratory assays can leverage prognostic value from the longitudinal optical coherence tomography (OCT)-based multimodal imaging of age-related macular degeneration (AMD). Human donor eyes with and without AMD are imaged using OCT, color, near-infrared reflectance scanning laser ophthalmoscopy, and autofluorescence at two excitation wavelengths prior to tissue sectioning.
Abstract
A progression sequence for age-related macular degeneration (AMD) learned from optical coherence tomography (OCT)-based multimodal (MMI) clinical imaging could add prognostic value to laboratory findings. In this work, ex vivo OCT and MMI were applied to human donor eyes prior to retinal tissue sectioning. The eyes were recovered from non-diabetic white donors aged ≥80 years old, with a death-to-preservation time (DtoP) of ≤6 h. The globes were recovered on-site, scored with an 18 mm trephine to facilitate cornea removal, and immersed in buffered 4% paraformaldehyde. Color fundus images were acquired after anterior segment removal with a dissecting scope and an SLR camera using trans-, epi-, and flash illumination at three magnifications. The globes were placed in a buffer within a custom-designed chamber with a 60 diopter lens. They were imaged with spectral domain OCT (30° macula cube, 30 µm spacing, averaging = 25), near-infrared reflectance, 488 nm autofluorescence, and 787 nm autofluorescence. The AMD eyes showed a change in the retinal pigment epithelium (RPE), with drusen or subretinal drusenoid deposits (SDDs), with or without neovascularization, and without evidence of other causes. Between June 2016 and September 2017, 94 right eyes and 90 left eyes were recovered (DtoP: 3.9 ± 1.0 h). Of the 184 eyes, 40.2% had AMD, including early intermediate (22.8%), atrophic (7.6%), and neovascular (9.8%) AMD, and 39.7% had unremarkable maculas. Drusen, SDDs, hyper-reflective foci, atrophy, and fibrovascular scars were identified using OCT. Artifacts included tissue opacification, detachments (bacillary, retinal, RPE, choroidal), foveal cystic change, an undulating RPE, and mechanical damage. To guide the cryo-sectioning, OCT volumes were used to find the fovea and optic nerve head landmarks and specific pathologies. The ex vivo volumes were registered with the in vivo volumes by selecting the reference function for eye tracking. The ex vivo visibility of the pathology seen in vivo depends on the preservation quality. Within 16 months, 75 rapid DtoP donor eyes at all stages of AMD were recovered and staged using clinical MMI methods.
Introduction
Fifteen years of managing neovascular age-related macular degeneration (AMD) with anti-VEGF therapy under the guidance of optical coherence tomography (OCT) has offered new insights into the progression sequence and microarchitecture of this prevalent cause of vision loss. A key recognition is that AMD is a three-dimensional disease involving the neurosensory retina, retinal pigment epithelium (RPE), and choroid. As a result of the OCT imaging of trial patients and the fellow eyes of treated clinic patients, the features of pathology beyond those seen by color fundus photography, a clinical standard for decades, are now recognized. These include intraretinal neovascularization (type 3 macular neovascularization1, formerly angiomatous proliferation), subretinal drusenoid deposits (SDDs, also called reticular pseudodrusen)2, multiple pathways of RPE fate3,4, and intensely gliotic Müller cells in atrophy5,6.
Model systems lacking maculas (cells and animals) recreate some slices of this complex disease7,8,9. Further success in ameliorating the burden of AMD could come from the discovery and exploration of primary pathology in human eyes, understanding the unique cellular composition of the macula, followed by translation to model systems. This report portrays a three decade collaboration between an academic research laboratory and an eye bank. The goals of the tissue characterization methods described herein are two-fold: 1) to inform evolving diagnostic technology by demonstrating the basis of fundus appearance and imaging signal sources with microscopy, and 2) to classify AMD specimens for targeted (immunohistochemistry) and untargeted molecular discovery techniques (imaging mass spectrometry, IMS, and spatial transcriptomics) that preserve the cone-only fovea and rod-rich para- and perifovea. Such studies could accelerate the translation to clinical OCT, for which a progression sequence and longitudinal follow-up are possible through eye-tracking. This technology, which is designed to monitor treatment effects, registers scans from one clinic visit to the next using retinal vessels. Linking eye-tracked OCT to laboratory results obtained with destructive techniques could provide a new level of prognostic value to molecular findings.
In 1993, the research laboratory captured color photographs of postmortem fundus on film10. This effort was inspired by the superb photomicroscopy and histology of the human peripheral retina by Foos and colleagues11,12,13 and the extensive AMD clinicopathologic correlations by Sarks et al.14,15. Starting in 2009, ex vivo multimodal imaging (MMI) anchored on spectral domain OCT was adopted. This transition was inspired by the similar efforts of others16,17 and especially by the realization that so much of the ultrastructure described by the Sarks was available in three dimensions, over time, in the clinic18,19. The goal was to acquire eyes with attached maculas in a reasonable time frame for well-powered studies of cellular-level phenotypes in the retina, RPE, and choroid. The intent was to move beyond "per eye" statistics to "per lesion type," a standard influenced by the "vulnerable plaque" concepts from cardiovascular disease20,21.
The protocol in this report reflects experience with nearly 400 pairs of donor eyes accessioned in several streams. In 2011-2014, the Project MACULA website of AMD histopathology was created, which includes layer thicknesses and annotations from 142 archived specimens. These eyes were preserved from 1996-2012 in a glutaraldehyde-paraformaldehyde fixative for high-resolution epoxy-resin histology and electron microscopy. All the fundi had been photographed in color when received and were reimaged by OCT just prior to histology. An eye holder originally designed for optic nerve studies22 was used to accommodate an 8 mm diameter full-thickness tissue punch centered on the fovea. OCT B-scans through the foveal center and a site 2 mm superior, corresponding to histology at the same levels, were uploaded to the website, plus a color fundus photograph. The choice of the OCT planes was dictated by the prominence of AMD pathology under the fovea23 and the prominence of SDDs in rod-rich areas superior to the fovea24,25.
Starting in 2013, eyes imaged with OCT-anchored MMI during life were available for direct clinicopathologic correlations. Most (7 of 10 donors) involved patients at a retina referral practice (author: K.B.F.), which offered an advanced directive registry for patients interested in donating their eyes after death for research purposes. The eyes were recovered and preserved by the local eye bank, transferred to the laboratory, and prepared in the same way as the Project MACULA eyes. Pre-mortem clinical OCT volumes were seamlessly read in the laboratory, thus aligning the pathology features seen during life with the features seen under the microscope26.
Starting in 2014, prospective eye collection began by screening for AMD in donor eyes without a clinical history but preserved during a defined time limit (6 h). For this purpose, the eye holder was modified to accommodate a whole globe. This reduced the chance of detachment around the cut edges of the previously used 8 mm punch. The eyes were preserved in 4% buffered paraformaldehyde for immunohistochemistry and transferred to 1% the next day for long-term storage. In 2016-2017 (pre-pandemic), 184 eyes from 90 donors were recovered. The statistics and images in this report are generated from this series. During the pandemic era (2020 lockdowns and aftermath), prospective collections for transcriptomics and IMS collaborations continued at a reduced pace, essentially using the 2014 methods.
Other methods for donor eye assessment are available. The Minnesota Grading System (MGS)27,28 is based on the AREDS clinical system for color fundus photography29. The limitations of this method include the combining of atrophic and neovascular AMD into one stage of "late AMD". Further, the MGS entails the removal of the neurosensory retina before the photo-documentation of the RPE choroid. This step dislodges SDDs to varying degrees30,31 and removes the spatial correspondence of the outer retina and its support system. Thus, efforts to link metabolic demand and signaling from the retina to pathology in the RPE-choroid may be impeded. The Utah System implemented MMI using ex vivo color photography and OCT to categorize eyes destined for dissection into regions for RNA and protein extractions32. Although preferable to whole eyecup extractions, the 3 mm diameter area at the highest risk for AMD progression33,34 represents only 25% of a 6 mm diameter fovea-centered punch. Thus, techniques that can localize findings in reference to the fovea, such as serial sectioning for immunohistochemistry, are advantageous.
Protocol
The institutional review board at the University of Alabama at Birmingham approved the laboratory studies, which adhered to Good Laboratory Practices and Biosafety Level 2/2+. All US eye banks conform to the 2006 Uniform Anatomical Gifts Act and US Food and Drug Administration. Most US eye banks, including Advancing Sight Network, conform to the medical standards of the Eye Bank Association of America.
The Table of Materials lists the supplies and equipment. Supplementary Material 1 provides an overview of the dissection, color fundus photography, and OCT-based MMI. Supplementary Material 2 provides details of the OCT-based MMI.
1. Criteria for tissue collection
- To maximize the yield of AMD eyes in a screen of undocumented donors, set the following criteria for acceptable donors: age ≥80 years, white, non-diabetic, and ≤6 h death-to-preservation (DtoP).
NOTE: DtoP is defined as the time between death and when the eye is placed in a provided preservative, either in the hospital or in the laboratory.
2. Preservation medium and other preparation (laboratory)
- Make 4% phosphate-buffered paraformaldehyde from 20% stock (purchased). Prepare 1 L by adding 200 mL of 20% paraformaldehyde (dilution factor of 5) to 800 mL of 0.1 M Sorenson's phosphate buffer. Test and adjust to ensure the pH is 7.2, if necessary. Store at 4 ˚C.
- Dispense 30 mL of 4% phosphate-buffered paraformaldehyde into 40 mL jars.
- Stock labeled 40 mL containers of preservative at the eye bank so that the tissue can be recovered at any time and on any day.
- For the storage of preserved eyes, make 1% paraformaldehyde from the 4% solution. Prepare 1 L by adding 250 mL of 4% paraformaldehyde (dilution factor of 4) to 750 mL of 0.1 M Sorenson's phosphate buffer. Test and adjust the pH if necessary. Store at 4 ˚C.
- Prepare 1 L of a 0.1 M solution from the 0.2 M solution of Sorenson's phosphate buffer, pH 7.2, by combining 500 mL of distilled water and 500 mL of Sorenson's buffer.
- Adjust the pH drop by drop using 1 N hydrochloric acid or 1 N sodium hydroxide. Store at 4 ˚C.
- Create a holder to stabilize the eyes during dissection. Fill a Petri dish with dental wax heated until liquid. When it is slightly cool, make a hemispheric impression in it with a large ball bearing, and then freeze the dish to facilitate the removal of the ball bearing.
3. Eye bank methods
- To ensure the rapid recovery of research tissues, recover all the tissues rapidly, including those intended for transplantation.
- Receive death referrals, as required by law, within 1 h of death, and track each donor with a referral sequence number that follows the tissue.
- To find cases with potential clinical documentation, ask AMD- and eye disease-specific questions in the research Donor Risk Assessment Interview.
- To minimize the travel time, recover whole globes (as distinct from only corneas for transplantation) within a compact area (i.e., city and adjacent suburban county).
- Bring two jars of preservative (buffered 4% paraformaldehyde) to the decedent's hospital room for tissue recovery (as opposed to waiting for the body to be moved to a morgue).
- Before and during recovery, communicate with the investigators to confirm the delivery and timing.
- Recover the globes on-site in the hospital, open them by consistent handling methods, and immerse the opened eye in preservative (Figure 1).
- Hold the excised donor eye in place using a sheath of gauze stabilized by a hemostat.
- To facilitate the removal of the cornea with a 2 mm wide rim of sclera, use an 18 mm diameter trephine to score the globe.
- To free the cornea with an accompanying rim of sclera, cut along the scored circle using spring-loaded scissors with curved tips while stabilizing the globe with the hemostat-clipped gauze.
- Lift the cornea off the sclera, exposing the iris and ciliary body.
- To facilitate the penetration of the preservative into the vitreous chamber, make a 2 mm long slit in the iris perpendicular to the pupillary margin. Place the eyes into specimen jars with 30 mL of preservative at 4 °C, and transfer to the laboratory on wet ice.
- Transmit de-identified donor information electronically from the eye bank to the research laboratory database.
NOTE: The database maintains the referral number, the eye bank tissue number, and the laboratory ID number for tracking, plus other relevant information.
4. Tissue preparation for ex vivo color fundus photography
- Use two stereo microscopes, one for dissection and one for color fundus photography.
NOTE: For transillumination to visualize pigmentary changes, use a base for dark-field microscopy. - Remove the anterior segment remnants (iris and lens). To stabilize the eye during dissection, place it in the Petri dish filled with wax (Supplementary Material 1, slides 7-8). To prevent the ciliary body and attached retina from collapsing into the vitreous cavity, minimize the perturbation of the ring of thick vitreous attached to the ciliary body (vitreous base).
- Mark the superior pole for orientation. Place the globe anterior side down in the dish. Find the insertion tendons of the superior rectus and superior oblique muscles. Using a wooden applicator, sparingly apply a marking ink in a 10 mm line in an anterior to posterior direction (i.e., perpendicular to the tendinous insertion of the superior rectus muscle).
- Before photography, fill the fundus with cold Sorensen's phosphate buffer.
- Insert a 1 mm ruby bead into the fundus as an internal scale bar to appear in each image27.
- Acquire color images with a single-lens reflex camera mounted to a stereo microscope fitted with a ring flash. Use trans-, epi-, and flash illumination at each of three standard magnifications to capture images intended to highlight specific areas (Supplementary Material 1, slide 11): 1) the fundus to the equator, 2) the posterior pole (vascular arcades, optic nerve head, fovea), and 3) the fovea within the macula lutea (yellow spot).
5. Preparation for ex vivo color fundus photography
- Turn the camera and monitor on. Plug in the remote shutter, and release the actuator in the high-definition multimedia interface (HDMI) camera/television (TV) monitor and display cable.
- Set the camera settings to manual ISO function and the mirror lock-up position (locked in place to reduce vibration).
NOTE: Refer to the manufacturer's user manual for the settings on the camera used. Learn from the camera display the over/underexposure settings relative to the histogram and exposure readouts. - Arrange two light sources, each with two flexible light guides positioned perpendicular to each other, for illumination in the four cardinal directions on the microscope stage (Supplementary Material 1, page 10).
- Turn on the epi-illumination light sources to full power.
NOTE: It is helpful but not necessary to have a black felt shroud around the stage to limit light/flash scatter for the photographer. - At the dissecting microscope, use forceps to insert the posterior pole into a 30 mL quartz crucible filled with buffer. Allow the tissue to sink to the bottom. Insert a bracing, such as a tissue sponge, between the eye and the wall of the crucible to prevent movement. Insert the 1 mm ruby bead into the posterior pole.
NOTE: The bead may fall into the optic nerve cup. - Carefully place the globe in the crucible onto the stereo microscope stage, and observe the interior ocular fundus through the microscope eyepieces. Using the lowest magnification, orient the eye by identifying the tissue mark at the 12 o'clock position, the optic nerve head (ONH), and the fovea 5° below the ONH. Rotate the eye so that the fovea falls below a line through the ONH by 5°.
NOTE: If it is a right eye, the ONH is to the right of the fovea, as seen through the oculars of the microscope. If it is a left eye, the ONH is to the left of the fovea. - Turn on the remote monitor viewing from the camera. Ensure that the microscope beam-splitter slider is set to observe through the photo port and not through the port for the oculars. Depending on the light and mirror path, be prepared to rotate the tissue 180˚ on the stage for proper orientation.
6. Image acquisition using ex vivo color fundus photography
- With the epi-illumination turned on, set the magnification so that the entire 18 mm trephine incision occupies the entire field of view. Increase the magnification so that it is possible to focus on the bottom of the foveal pit. Reduce the magnification to the previous setting.
- Adjust the ISO settings in the range of 1,600-3,000 so that exposure times fall in the center range of the meter on the camera.
- Press the remote shutter trigger. Listen for the mirror to lock in the up position. Press the trigger again for exposure.
- Observe the image on the monitor, with the preset metadata showing red, green, blue (RGB), and a color histogram for the correct exposure. If the exposure is acceptable, proceed; if not, then delete, re-evaluate the parameters, and re-image.
- Turn off the gooseneck lamps for epi-illumination to highlight drusen, and turn on the flash, with a 1/4 s exposure, a camera shutter speed of 1/250 s, and an ISO of 100-320. Set the flash speed to the default, or modify it during the initial setup of the camera. Acquire an image, and check the histogram for proper exposure.
- Turn off the flash, and turn on the trans-illumination light source. Reset the ISO to above 5,000, and ensure the exposure time does not go below 1/30 s due to potential vibration in the photographic system. Acquire an image, and check the histogram for proper exposure.
- Increase the magnification to view both the ONH and fovea in the field of view. Turn on the epi-illumination lamps. Set the ISO range to 1,600-3,000. Ensure that the exposure times fall in the center range of the camera.
- Turn off the epi-illumination lamps, and turn on the flash, with a 1/4 s exposure, the preset camera shutter speed set to 1/250 s, and the ISO set to 400-800. Acquire an image, and check the histogram for proper exposure.
- Turn off the flash, and turn on the trans-illumination using the dark-field base. Reset the ISO to above 5,000. Ensure that the exposure time is not slower than 1/30 s due to potential vibration in the photographic system. Acquire an image, and check for a proper histogram.
- Turn off the transillumination, and turn on the epi-illumination lamps again.
- Increase the magnification to that used in step 6.7. Refocus if needed.
- Increase the ISO within the range of 3,000-6,000, and adjust the exposure time to fall within the proper exposure range of the camera. Acquire an image.
NOTE: The ruby bead may no longer appear in the field of view. If so, capture separate images of the bead at this magnification for reference. - Turn off the epi-illumination lamps. Turn on the flash with a 1/4 s exposure and with the camera shutter speed to 1/250 s. Set the flash speed to the default, or change it during the initial setup of the camera, and set the ISO to 500-1,000. Acquire the image. Check the histogram for proper exposure.
- Turn off the flash, and turn on the trans-illumination lamps. Reset the ISO to above 8,000 to allow a faster exposure time with a more sensitive image sensor. Ensure that the exposure time does not go below 1/30 s due to potential vibration in the photographic system. Acquire an image.
- Export the images from the camera to a computer. Review the images before removing the specimen from the microscope stage in case some need to be captured again.
7. Image acquisition overview for ex vivo OCT and scanning laser ophthalmoscopy (SLO)
- For spectral domain OCT, place the globes in phosphate buffer in a custom eye holder with a chamber with a 60 diopter lens35. Mount the eye holder to a bracket on a clinical OCT imaging device, and attach it to where a patient would rest their forehead. The OCT device automatically inserts a scale bar into each image.
- At the same time, using the same device and with the eye still in the holder, image the globes with a scanning laser ophthalmoscope (SLO) using near-infrared reflectance (NIR, used as a locator image by the indwelling software), 488 nm excitation fundus autofluorescence (FAF), and red-free (RF) reflectance.
NOTE: The 787 nm excitation FAF in this device is only occasionally suitable because a beam-splitter reduces the light transmittance to the SLO. For this reason, a second device for 787 nm FAF images (see next point) is used. - Separately, but with the eye still in the eye holder, image the globes with 787 nm FAF for detecting RPE disturbance36 using a separate device that displays this modality well.
8. Image acquisition protocol for ex vivo OCT/ SLO (see slides in Supplementary Material 2)
- Indicate the superior rectus muscle with tissue marking dye. Turn on the laser (blue arrow, Supplementary Material 2, page 1).
- Referring to page 2 of Supplementary Material 2, position the OCT head by moving the entire unit in two axes with respect to the base (green arrow) and then raising the height (y) by rotating the joystick clockwise/counterclockwise (cw/ccw, blue arrows). Focus the image by rotating the knob (orange arrow), with the black lever in position R (*). Lock down the unit by securing the thumb screw (purple arrow).
- Insert the eye into the holder, and stabilize from the posterior aspect with spacers (Supplementary Material 1, page 13). Exert as little pressure as possible to avoid denting the sclera. Orient with the tissue mark for the superior rectus muscle facing up.
NOTE: The approximate distance from the front of the eye holder to the OCT device is 25 mm. - Open the proprietary visualization and analysis software for the OCT device. A patient list will appear in the left column. The eye donors indexed by an internal code number are the "patients." Refer to the user manual from the manufacturer.
- Select the New Patient Icon. Complete the patient data information as needed. Select OK. Use a logically ordered numbering system for individual eyes, such as YYYYNNNL,R_agesex. For example, this could be 2017016R_97F.
- After continuing the data entry on the following window, press OK. Select the operator and study from the drop-down menu.
NOTE: The entered information will appear in the exportable meta-data. - After viewing a blank screen, touch the yellow button to start the image acquisition.
- Press IR + OCT (near infrared reflectance + OCT). Allow the laser to acquire a live SLO image of the fundus and OCT B-scan.
- Finalize the correct anatomical position based on the ONH and fovea, and use a wooden applicator to adjust the eye position in the holder. On the control panel, rotate the black round button to adjust the intensity.
- Press the same button to average 9-100 frames (red arrows; 9 is sufficient, 100 looks creamy). If the unit is oriented correctly, the fundus should be in focus, and the OCT B-scan should appear in the top third of the display (Supplementary Material 2, page 9, double red arrow).
- On the fundus image, use the cursor to move the blue line to center the fovea (Supplementary Material 2, page 9, white arrow). Press Acquire.
NOTE: Other default buttons are Retina, EDI (off), and Line Scan.
- Press RF + OCT for the next acquisition. Recheck the position to ensure that the image has not moved or degraded. Press the black button for averaging. Press Acquire.
- Switch the internal cube for autofluorescence imaging to the 488 nm and 787 nm excitation wavelengths (Supplementary Material 2, page 10).
NOTE: The cube position after the switch is shown. - Select Autofluorescence mode. Recheck the alignment. Press Acquire.
- For suspected cases of RPE disruption and atrophy, select ICGA (indocyanine green angiography) for 787 nm autofluorescence. Recheck the eye position, and then press Acquire.
NOTE: A fundus image often does not appear in this modality because the internal cube attenuates the beam. - Switch the internal cube back to R for IR and red free imaging for volume acquisition.
NOTE: The cube position after the switch is shown on page 13 of Supplementary Material 2. - To acquire the OCT volumes, select IR and the volume setting. Match all the settings on page 14 of Supplementary Material 2 by toggling the appropriate buttons on the control module (30° macula cube, 30 µm spacing, averaging depending on the experimental requirements).
- Notice that the near-infrared reflectance fundus view is covered in blue B-scan lines. Recheck the OCT position in the upper third of the right window. Press Acquire on the Control Module, and wait 5 min for the volume scan to complete. When the imaging acquisition is completed, select EXIT. The images will be saved (red arrow).
NOTE: The blue lines delimit the distances shown in the previous view in micrometers. The scans start numbering from the bottom (inferior) and proceed upward. Note the red line progression. - Allow the computer to process the acquired images, which will appear on the screen (Supplementary Material 2, page 16).
- When imaging the fellow eyes of one donor, do not change the position of the mounting bracket between the eyes. If the left eye is imaged first, the results will show in the OD (right eye) column. Right-click to select all the images, and then select exchange OS/OD. The images will shift to the OS column.
- Press Select > Window > Database. The screen defaults to panel 6 with the addition of the donor eye imaged in the right column (Supplementary Material 2, page 18). Right-click on the patient in the dropdown menu, choose Batch, and export the E2E file.
- Browse to the pre-determined folder created on the desktop for file transfers. Select OK. The folder contains the E2E files to be copied onto an external hard drive and archived.
- Bring the eye in its holder to the scanning laser ophthalmoscope, which is principally used for 787 nm autofluorescence.
NOTE: refer to the manufacturer's user guide for the acquisition and archiving of the images. - Turn on the computer and the laser.
- Select the New Patient icon. Complete the patient data as needed.
- To keep the eye datasheet the same, press OK. As the C-curve stays the same, press 계속.
- Select Study mode, and enter the password if required. Keep the C-curve at 7.7 mm. Press Ok.
- Select 계속 to verify the C-curve. Select the yellow indicator to start the camera.
- Select the R position. Align and orient the globe. Select the IR mode to focus and orient as above.
- Orient the camera head by moving the entire unit in two axes with respect to the base (Supplementary Material 2, page 28, green arrow) and then raising the height (y) of the unit (blue arrows). Focus the image by rotating the knob (orange arrow). The black lever is in position R (*). After this head is in position, lock down the unit by turning the thumb screw (purple arrow).
- Note that the screen appears as shown on page 29 of Supplementary Material 2.
- Move the selector knob from R 세스 A. Select ICGA (to achieve 787 nm autofluorescence) in blue, 100% intensity, a 30° field of view, and single-phase imaging.
NOTE: Like the dye indocyanine green, melanin is excited by 787 nm wavelength light. - Note that the screen appears as shown on page 31 of Supplementary Material 2. Press the black disc for averaging, and then select Acquire.
- Choose Window > Database. Select Import E2E files from the SLO device; these files are stored on an external hard drive and uploaded to the desktop.
- Select Open. Check the marks are the default, and select OK.
- Note that the patient now has two tabs: one showing the images acquired from the SLO (blue arrow), and the other tab showing the images acquired from the OCT device (red arrow) (Supplementary Material 2, page 34).
- Right-click on the 786 nm (ICGA) image, and export the picture to a file labeled SLO 786 on the desktop.
- To save the 786 nm autofluorescence SLO image, select the patient, and right-click to export images as RAW files (for .vol files) to a folder on the desktop.
- To export images from the OCT device for transfer to the archive computer, copy/paste from RAWEXPORT to a folder labeled RAW.
9. Imaging review
- Assemble the images (color, .vol file, SLO images) in folders for each donor with subfolders for each eye, and index the folders consecutively by laboratory ID.
- Record the tissue impressions (quality, pathology) in a database for a standardized report.
- Review the exported OCT volumes with an in-house ImageJ plugin.
- Find the fovea center, which can be recognized by the center of the foveal dip, the inward rise of the outer retinal bands, or the thinnest point. In most cases, these will coincide, but not always, as this depends on the foveal preservation, individual variability, and the presence of pathology.
- Find a standard scan in the superior perifovea (2 mm/67 B-scans away in the superior direction; i.e., increasing scan numbers).
- Save a .tif stack of the entire volume for quick reference in the future.
- Scroll through the OCT volume in its entirety, starting from scan 1 (inferior), while recording the scan numbers in which features are recognized.
- Check the peripapillary area in addition to the macula.
NOTE: Peripapillary chorioretinal atrophy in older eyes includes a distinctive basal laminar deposit and neovascularization. This is a common area of pigmentary change in myopic eyes and glaucoma, as well as in aging and AMD37.
- Inspect the color photographs, and link any findings to those seen by OCT if possible.
NOTE: In general, it is easier to see most findings by OCT first. Color photographs do provide a large view of the area outside the area on the SLO, choroidal pigmentation including nevi, the presence of heme and hard exudates, and black pigment in neovascular AMD. Dark pigment in the fovea may represent loose melanosomes from the anterior segment and should be washed away gently with buffer using a pipette. - Inspect the SLO images, and link any findings to those seen by OCT if possible.
- Save standard B-scans at the fovea and perifovea, extra B-scans with notable pathology or other features, and SLO images in a pathology report.
- Categorize the eyes as follows: Unremarkable, Questionable, Early-Intermediate AMD, Atrophic AMD, Neovascular AMD, Other, Unknown, Not Gradable, Not Recorded. "Questionable" is used if it is not clear if changes are severe enough for another categorization. "Not Gradable" is reserved for eyes lacking useful OCT scans, such as those with severely detached retinas. "Not Recorded" is reserved for eyes that are processed immediately upon receipt without photography.
- Use these criteria for AMD: severe RPE change with either drusen or subretinal drusenoid deposits, with or without signs of neovascularization, such as fluid or fibrosis, and without evidence of other causes (updated from Curcio et al.10 to accommodate SDDs).
NOTE: The final diagnosis is confirmed by histologic analysis.
Representative Results
Table 1 shows that during 2016-2017, 184 eyes from 94 white non-diabetic donors >80 years of age were recovered. The mean death-to-preservation time was 3.9 h (range: 2.0-6.4 h). Of the 184 eyes reviewed, 75 (40.2%) had certain AMD. The following categories were identified: Unremarkable (39.7%), Questionable (11.4%), Early-Intermediate AMD (22.8%), Atrophic (7.6%), Neovascular (9.8%), Other (8.7%), and Unknown/Not Recorded/Not Gradable (<1%). Figure 2, Figure 3, Figure 4, and Figure 5 show multiscale, multimodal ex vivo imaging of exceptionally well-preserved eyes from this series. The eyes were reviewed in collaboration with an ophthalmologist specializing in retinal disease (J.A.K.). While some eyes showed individual features better than others, these cases were chosen for all-around high quality.
As described38, ex vivo color photography differs from the corresponding in vivo photography. Retinal edema and/or detachment can reduce the visibility of the posterior pigmented tissues. Observations in fresh eyes indicate that these changes occur postmortem and do not worsen markedly with prompt fixation. In addition, choroidal vessels empty after death. Due to an undulating background of pale vessels and dark interstitial tissue, the assessment of pigmentary variations in the plane of the RPE should be assisted by modalities other than color. In ex vivo OCT, more information is available than in color photography. Ex vivo OCT also differs significantly from in vivo OCT. The major differences include the overall increased reflectivity of tissue, especially in the inner retina, the consistent reflectivity of some bands (nerve fiber layer, inner and outer plexiform layer, RPE), the lower visibility of the choroid details, especially under the edematous retina, and the visibility of tissue layer detachments (see below). The outer retinal hyperreflective bands, involving photoreceptors and the RPE (ellipsoid zone, EZ; interdigitation zone, IZ), are inconsistently visible ex vivo and are not used as landmarks in this context. The clinical consensus for spectral domain OCT uses the term RPE-Bruch's membrane (BrM) band. However, the term RPE-basal lamina (BL)-BrM band is preferred because it accommodates the appearance of basal laminar deposits in AMD24.
Figure 2 shows an unremarkable macula and hyporeflective large choroidal vessels, with a reflective RPE-BL-BrM band between the two. A large vessel in the inner retina casts a shadow on the posterior layers. The IPL and OPL are moderately reflective. In the NIR SLO, both the retinal and choroidal vasculatures are visible. The red-free reflectance SLO works best for features of the inner retina and vitreoretinal interfaces like the arcuate fibers and the papillomacular bundle of the NFL. In normal eyes, the 488 nm autofluorescence SLO shows an area of overall reduced signal in the central macula due to thickened parafovea and, in some cases, absorption by the yellow xanthophyll pigment, as well as hyperautofluorescence lining the large retinal vessels, which is suggestive of connective tissue sheaths. The autofluorescence at 787 nm shows a small region of increased signal in the central macula from the RPE, a signal in the choroidal stroma, and hypoautofluorescent stripes corresponding to the choroidal vessels.
Figure 3 shows a macula with early-intermediate AMD. The visible features include a soft drusen (dome-shaped RPE elevation near the fovea), SDDs (intermittent reflectivity with a dentate appearance internal to the RPE-BL-BrM band), hyper-reflective foci (HRF, reflective material with the same reflectivity as the in-layer RPE, located in the retina), and vitelliform change (an inward expansion of the RPE organelles, both intracellular and extracellular, in conjunction with basal laminar deposits39). The color photography shows strong pigmentation corresponding to the vitelliform lesion, surrounded by reduced pigmentation. Neither the drusen nor the SDDs are clearly visible by color. The NIR reflectance shows the reflectivity in the fovea. The autofluorescence at 488 nm excitation shows a mottled signal in the fovea. SDDs appear occasionally on SLO modalities; this is more likely if the SDDs are abundant, and SDDs are most easily seen as a regularly spaced punctate pattern (see Spaide and Curcio19, Figure 6). The pattern superior and temporal to the fovea in Figure 3I is not SDD, because it is irregular and localized to a superficial focal plane. All the en face modalities show fine folds radiating from the fovea. In less well-preserved eyes, similar folds may be large enough to be visible at the lowest viewing magnifications.
Figure 4 shows a macula with atrophic AMD. The color fundus photography shows circular atrophic areas, central hyperpigmentation, and small hyperpigmented dots in the parafovea that correspond in the OCT to HRF at the level of Henle fiber layer-outer nuclear layer (HFL-ONL). Additionally, in OCT, a low flat RPE elevation may represent a basal laminar deposit, non-exudative type 1 neovascularization, or both. Atrophy in the foveal B-scan is recognizable by the subsidence of the HFL-ONL, an area of hypertransmission (light shining through to choroid), a vitelliform change with increased shadowing in the foveal center, and HRF that cast shadows. In this eye, the NIR reflectance shows hyper-reflective spots in the fovea. The autofluorescence at 787 nm excitation effectively shows a signal corresponding to foveal hyperpigmentation and the absence of a signal in the circular atrophic areas. The red-free and 488 nm autofluorescence show the inner retinal features.
Figure 5 shows a macula with macular atrophy secondary to neovascular AMD. The color fundus photography shows black pigmentation within the atrophic area. The OCT shows atrophy by the sagging (subsidence) of HFL-ONL and increased hypertransmission. The foveal B-scan shows a mound of subfoveal hyperreflective material and intraretinal cysts. The near-infrared reflectance shows reflectivity in the atrophic area due to the loss of RPE and choroidal vessels. A small, intensely reflective area in the fovea is not visible on color. The red-free reflectance shows retinal vessels and, within an annular zone, choroidal vessels. The autofluorescence (488 nm) clearly shows a roughly circular atrophic border and islands of incipient atrophy. A central area devoid of signal is surrounded by an annulus of moderate signal and visible choroidal vessels.
Figure 6 shows common artifacts in ex vivo OCT imaging. Edema can be prominent in the inner retina, creating bulges and folds through the fovea (Figure 6A,I). Mechanical damage can occur with traction on the vitreous or by direct contact of the retina with the dissecting tools, which results in the dislocation of material and, sometimes, loss of that material (Figure 6F,G). Detachments can occur along multiple tissue planes and may represent relative tensile forces between the layers that also occur in vivo. Any detachment can widen further upon subsequent processing. The most common detachment is retinal (i.e., between the photoreceptor outer segments and the apical processes of the RPE) (Figure 6B–D,F–I). The apical processes may either detach from the outer segments or remain with the RPE cell bodies, as determined by histology. Retinal detachments may be large and billowing (Figure 6B,D,I) or narrow and barely discernible (Figure 6C,F,G). Bacillary layer detachment (BALAD40) was first seen in the laboratory and then later found in clinical OCT. BALAD is attributed to a split through the myoid portion of the photoreceptor inner segments, which leaves the ellipsoid portion of the inner segment and the outer segment attached to the RPE (Figure 6A,D,I). BALAD should not be mistaken for SDD in ex vivo OCT. A third detachment plane is the inner limiting membrane (ILM), and there is often residual reflective fluid between the ILM and the remaining retinal layers (Figure 6A,B,I). The least common detachment is between the choroid and sclera (Figure 6C). The fovea often exhibits cystoid spaces that should not be considered pathological without supporting evidence, such as signs of neovascularization (Figure 6H). In single B-scans, undulations of the RPE can give the impression of drusen. The 3D view of an OCT volume clarifies that these travel along the choroidal vessels (Figure 6E,J). Undulations may be due to differential volume changes between the vessels and intervening stroma after death and during fixation.
To calibrate the expectations for quality and to explore the limitations of ex vivo OCT, Figure 7 compares the in vivo imaging, ex vivo imaging, and histology of three clinically documented eyes with AMD. These three eyes were preserved differently from those in Figure 2, Figure 3, Figure 4, and Figure 5. To confirm the structural OCT reflectivity sources, which are subcellular41, the eyes in Figure 7 were preserved in 2.5% glutaraldehyde and 1% glutaraldehyde to allow high-resolution epoxy resin histology and correlative electron microscopy. Glutaraldehyde adds opacity to these specimens relative to those in Figure 2, Figure 3, Figure 4, and Figure 5. The effect of a shorter versus longer DtoP is apparent (Figure 7A–F, 2.1 h vs. Figure 7G–I, 8.9 h). In the eye with a longer DtoP, postmortem edema changed the retinal contour, and the ILM was detached. The pathology of interest (outer retinal tubulation) was subtle in the ex vivo imaging. It was found because the eye-tracked in vivo OCT pinpointed the relevant B-scan, and the choroidal vessels could be matched. In the two eyes with a shorter DtoP, some major pathologies (type 3 macular neovascularization) were immediately apparent (Figure 7A–C). Others were found with assistance from eye tracking (Figure 7D–F).
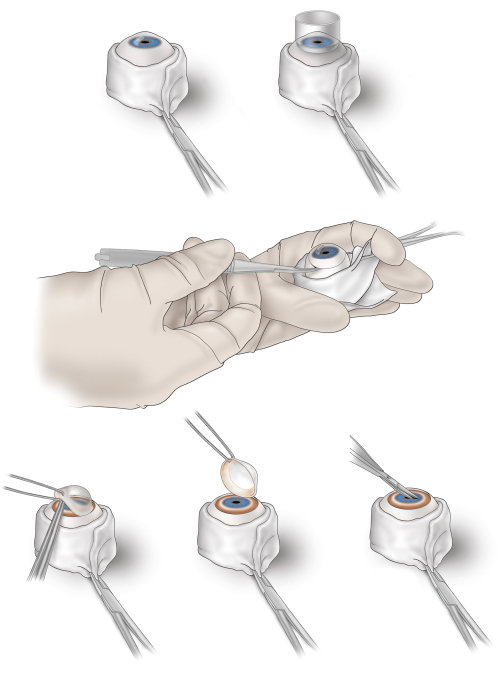
Figure 1: Corneal excision from a human donor eye for immersion fixation of the retina. Top left, the excised donor eye is held in place by a sheath of gauze stabilized by a hemostat; top right, an 18 mm trephine is used to make a circular score including the cornea and a 2 mm wide rim of sclera; middle, the scored circle is finished by a cut with spring-loaded curved tipped scissors, while the globe is stabilized; bottom left, the cornea and scleral rim are lifted, exposing the iris (blue) and ciliary body (tan-brown); bottom middle, the cornea with the rim is lifted off completely; bottom right, the iris is snipped perpendicular to the pupillary margin to facilitate the penetration of the preservative into the vitreous chamber. Please click here to view a larger version of this figure.
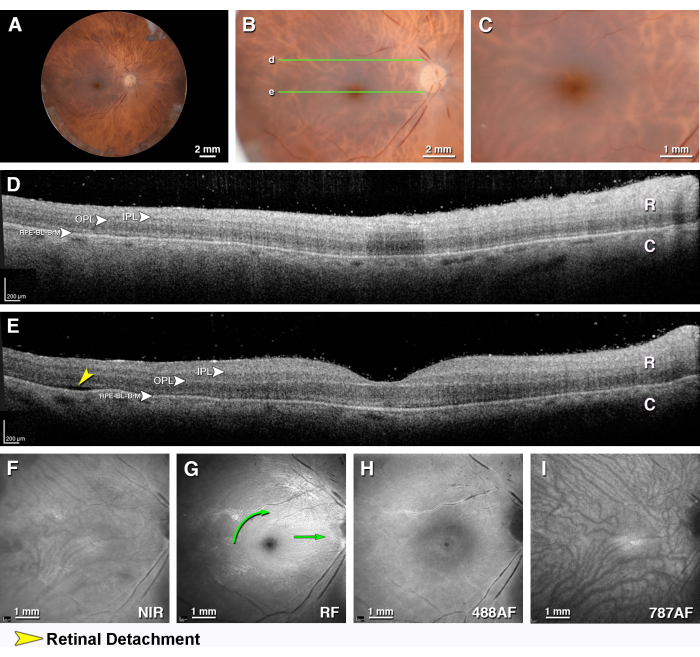
Figure 2: Multimodal ex vivo imaging of an unremarkable macula. A macula from an 82 year old female donor with a death-to-preservation interval of 1.97 h. (A) The fundus of the right eye viewed with the anterior segment removed (epi-illumination). (B) Close-up of the macula (epi-illumination). (C) Close-up view of the fovea (epi-illumination). The green lines indicate the locations of the OCT B-scans in panels D and E. (D,E) OCT B-scans through the (D) superior perifovea (E) and fovea. The retina (R), the choroid (C) with hyporeflective large vessels, and the intervening hyper-reflective RPE-BL-BrM band are visible. In this exceptionally well-preserved eye, the moderately reflective IPL and OPL are also visible. (D) A large vessel in the inner retina casts a shadow on the posterior layers. (E) The separation between the retina and RPE in panel E (yellow arrowhead) is artifactual. (F) The near-infrared reflectance shows the detail of both the retinal and choroidal vasculatures. (G) The red-free reflectance shows the arcuate fibers (left green curved arrow) and the papillomacular bundle (green arrow) of the nerve fiber layer. (H) The 488 nm wavelength autofluorescence shows an area of overall reduced signal in the central macula due to a thickened edematous parafovea, as well as rings and a point of low signal in the foveal center and hyperautofluorescence lining the large retinal vessels, which is suggestive of connective tissue sheaths. (I) The autofluorescence at 787 nm shows a small region of increased signal in the central macula from the RPE, a signal in the choroidal stroma, and hypoautofluorescent stripes corresponding to choroidal vessels. Please click here to view a larger version of this figure.
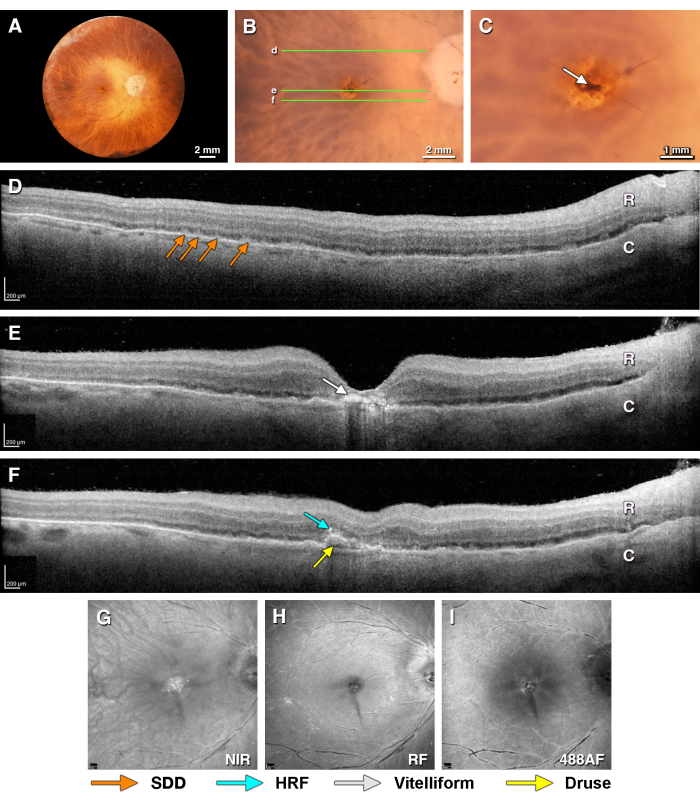
Figure 3. Multimodal ex vivo imaging of a donor eye with early intermediate AMD. Donor eye from a 97 year old female donor with a death-to-preservation interval of 3.1 h. (A) The fundus of the right eye viewed with the anterior segment removed (epi-illumination). (B) Close-up of the macula (epi-illumination). The green lines indicate the locations of the OCT B-scans in panels D, E, and F. (C) Close-up view of the fovea showing hyperpigmentation (arrow, flash illumination). (D) An SDD in the superior perifovea (orange arrows) is seen on OCT. I Vitelliform change in the RPE under the fovea (white arrows). (F) Inferior to the fovea is a soft drusen with a hyporeflective line at the base (yellow arrow) and a hyper-reflective focus above (light blue arrow). (G–I) The scanning laser ophthalmoscope images show very fine stellate folds in the fovea (also seen in C). (G) The near-infrared reflectance shows reflectivity material in the fovea corresponding in part to vitelliform material. (H) The red-free reflectance shows the retinal surface. (I) The 488 nm wavelength autofluorescence shows an area of overall reduced signal in the central macula due to a slightly thickened parafovea. SDDs are not clearly visible. Please click here to view a larger version of this figure.
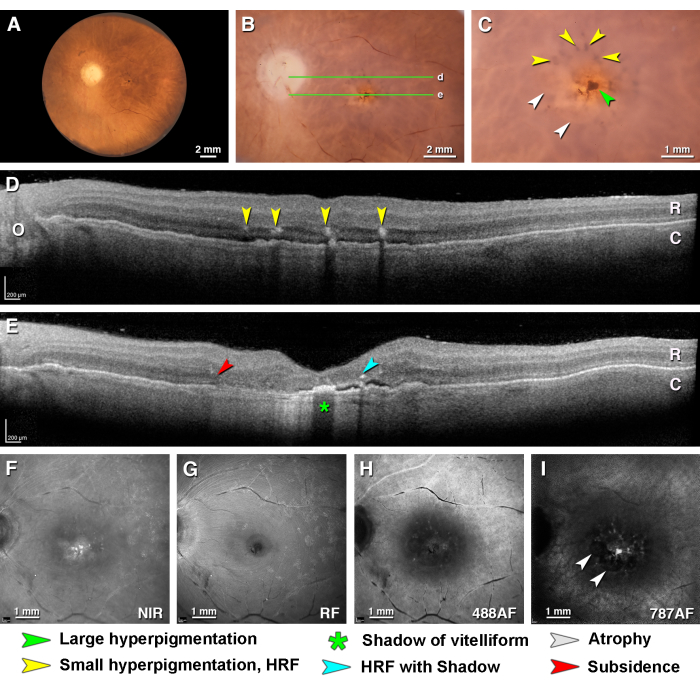
Figure 4: Multimodal ex vivo imaging of a complete RPE and retinal atrophy in age-related macular degeneration. Donor eye from a 97 year old female with a death-to-preservation interval of 2.33 h. (A) Fundus of left eye viewed with trans-illumination. (B) Close-up of the macula (epi-illumination). The green lines indicate the locations of the OCT B-scans in panels D and E. (C) Close-up of the fovea (epi-illumination) showing central hyperpigmentation (green arrowhead) and small hyperpigmented dots (yellow arrowheads). The central dot is intense brown because the overlying retina is very thin. The dots appear desaturated because the overlying retina is thick. Circular atrophic areas are indicated (white arrowheads). (D,E) OCT B-scans through the (D) perifovea (E) and fovea. The retina (R) and thin choroid (C) are visible. (D) Hyper-reflective foci (yellow arrowheads) at the level of the HFL-ONL correspond to the hyperpigmented dots in C. The low flat RPE elevation under them may represent a basal laminar deposit, non-exudative type 1 neovascularization, or both. (E) The B-scan through the fovea shows atrophy recognizable by the subsidence of the HFL-ONL (red arrowhead), an area of hypertransmission, a vitelliform change with increased shadowing in the foveal center (green asterisk), and hyper-reflective foci (teal arrowhead) with shadowing. The hyporeflective space between the retina and the RPE band may represent subretinal fluid. (F) The near-infrared reflectance shows hyper-reflective spots in the fovea. (G) The red-free image shows the arcuate fibers of the NFL and reflective blooms on the retinal surface. (H) The 488 nm autofluorescence focused on the retina shows a signal associated with the retinal vessels, no signal associated with the RPE, and faint autofluorescent spots in the central macular area. (I) The 787 nm autofluorescence shows a signal corresponding to pigmentation in C. Circular atrophic areas are apparent. Please click here to view a larger version of this figure.
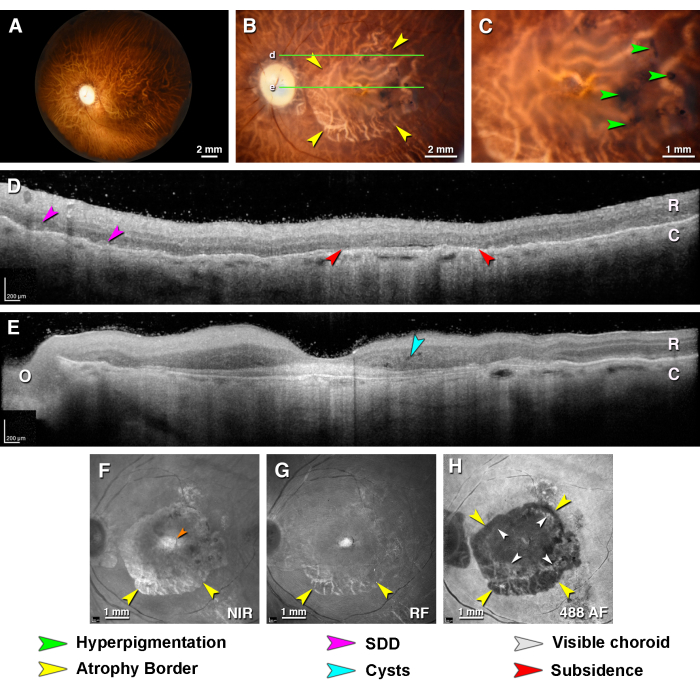
Figure 5: Multimodal ex vivo imaging of type 1 neovascularization and macular atrophy in age-related macular degeneration. A donor eye from an 86 year old female donor with a death-to-preservation interval of 3.5 h. (A) The fundus of the left eye viewed with the anterior segment removed (trans-illumination). (B) Close-up of the macula detailing atrophic borders (yellow arrowheads). The green lines indicate the locations of the OCT B-scans in panels D and E. (C) Close-up of the fovea shows black pigmentation (green arrowheads) within the atrophic area. (D,E) OCT B-Scans through the (D) perifovea (E) and fovea. The retina I and thin choroid (C) are visible. In this exceptionally well-preserved eye, the moderately reflective IPL and OPL are also visible. (D) The perifoveal B-scan grazes the superior edge of the atrophic area. Atrophy is evidenced by the sagging of the HFL-ONL and increased hypertransmission (red arrowheads). Possible subretinal drusenoid deposits are indicated (fuchsia arrowheads). (E) The foveal B-scan shows a mound of subfoveal hyper-reflective material and intraretinal cysts (asterisk). O = optic nerve head. (F) The near-infrared reflectance shows a reflective area of atrophy and choroidal vessels (teal arrowheads), including a small intense area in the central macula (orange arrowhead) not visible by color imaging. (G) The red-free reflectance shows retinal vessels and, within an annular zone, choroidal vessels. (H) The 488 nm autofluorescence depicts a roughly circular atrophic border (yellow arrowheads) and islands of incipient atrophy. A central area devoid of autofluorescence is surrounded by an annulus of moderate autofluorescence and visible choroidal vessels (white arrowheads). The 787 nm autofluorescence was not possible in this eye. Please click here to view a larger version of this figure.
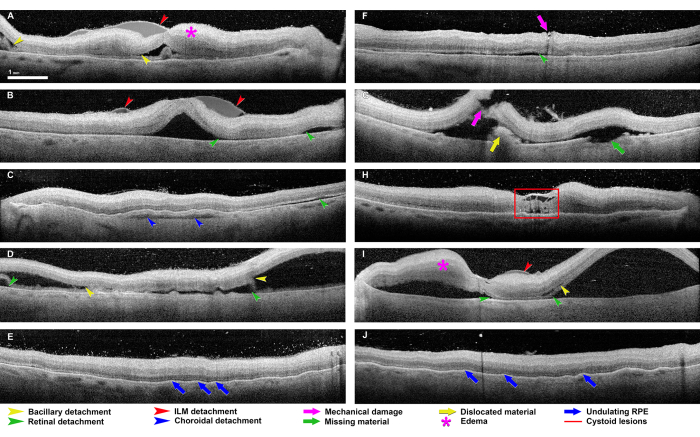
Figure 6: Common artifacts seen in the ex vivo OCT imaging of donor eyes. These eyes come from the 2016-2017 series of eyes. Most retinas are hyper-reflective relative to the retinas imaged in vivo, and the inner retinal layers are more reflective than the outer retinal layers. Bacillary detachment (yellow arrowheads in panels A, D, G, and I) is defined as a split at the level of the photoreceptor inner segment myoid, which creates a distinctive intraretinal cavity40. Retinal detachment (green arrowheads in panels B, C, D, F, G, and I) describes a separation of the entire neurosensory retina from the underlying RPE42. Detachments of the internal limiting membrane (ILM) separate the ILM and nerve fiber layer (red arrowheads in panels A, B, and I). A choroidal detachment (blue arrowhead in panel C) is a separation within the choroid or between the choroid and sclera. Mechanical damage (purple arrow in panels F and G) can appear at any level and with any dimensions, as applies to missing material (green arrow in panel G) and dislocated material (yellow arrow in panel G). The retinal edema (purple stars in panels A and I) appears as a thickening of the retinal tissue with poorly defined boundaries between the separate retinal layers. An undulating RPE (blue arrows in panels E and J) appears as an uneven, wavy RPE layer. Cystoid lesions (red square in panel H) correlate to hyporeflective chamber-like alterations within the retinal tissue, commonly in the fovea. Please click here to view a larger version of this figure.
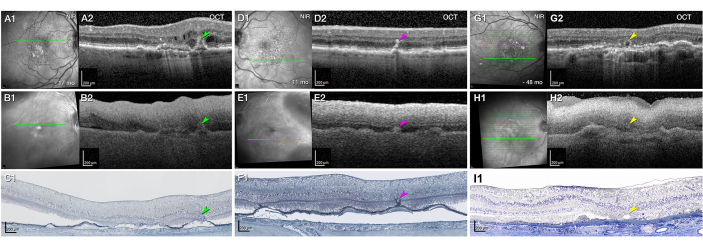
Figure 7: Ex vivo visibility of the pathology seen in vivo dependent on the preservation quality. (A–I) Panels A, B, and C, panels D, E, and F, and panels G, H, and I represent three clinically documented eyes from two donors, each seen by eye-tracking in vivo (A1–A2,D1–D2,G1–G2) and ex vivo (B1–B2,E1–E2,H1–H2) imaging, followed by histology (C,F,I). The green lines on the near-infrared reflectance (NIR, A1,B1,D1,E1,G1,H1) represent the levels of optical coherence tomography (OCT) B-scans and panoramic histology. Panels A, B, and C and panels D, E, and F, present the right and left eye, respectively, of one female donor in her 90s. Panels G, H, and I show the right eye of a second female donor, also in her 90s. (A–C) Well-preserved ocular tissue (DtoP: 2.1 h) enables good visibility of the major pathology. (A) A hyper-reflective intraretinal macular neovascularization (type 3 MNV, green arrowhead) is surrounded by intraretinal fluid 17 months before death (A2). The RPE/Bruch's membrane complex is split by hyporeflective material and appears as a "double layer" sign (A2). To the left is another double layer sign with barcode hypertransmission into the choroid (orange arrowhead). On ex vivo OCT (B2), the type 3 MNV and barcode hypertransmission are clearly visible and delineated. The retina is artifactually detached, as shown by the white arrowhead. The panoramic histology shows a vertically oriented type 3 MNV lesion (green arrowhead, C1). See previous research43,44 for details of the original case. (D–F) Well-preserved ocular tissue (DtoP: 2.1 h) can result in transparency that is reduced yet still sufficient for detecting major pathology. The OCT (D2) shows a stack of intraretinal hyper-reflective foci (HRF, fuchsia arrowhead) in an eye followed for exudative type 3 MNV. No intraretinal cysts are visible at 11 months before death. On ex vivo OCT (E2), the stack of HRF is compressed vertically (fuchsia arrowhead) but clearly delineated. On panoramic histology (F1), a retinal pigment epithelium tower (fuchsia arrowhead) rises upward from a soft druse. (G–I) Less well-preserved ocular tissue (DtoP: 8.9 h) results in reduced visibility of major pathology. The OCT indicates an outer retinal tubulation (ORT, yellow arrowhead) at 48 months before death (G2). On the ex vivo OCT, the ORT appears as a subtle disturbance, and this would have been difficult to discern without prior knowledge (H2). The inner limiting membrane is artifactually detached (white arrowhead). Edema has distorted the retinal contour. The histological analysis shows the ORT lumen delimited by the external limiting membrane and the photoreceptors protruding into it (I1). See previous research26,45 for details of the original case. Please click here to view a larger version of this figure.
| Numbers | Percentages | |||||
| Diagnostic category | Right Eyes | Left Eyes | Total | Right Eyes | Left Eyes | Total |
| Unremarkable | 39 | 34 | 73 | 41.50% | 37.80% | 39.70% |
| Questionable AMD | 10 | 11 | 21 | 10.60% | 12.20% | 11.40% |
| Early AMD | 20 | 22 | 42 | 21.30% | 24.40% | 22.80% |
| Atrophic AMD | 6 | 8 | 14 | 6.40% | 8.90% | 7.60% |
| Neovascular AMD | 11 | 7 | 18 | 11.70% | 7.80% | 9.80% |
| 기타 | 7 | 8 | 15 | 7.40% | 8.90% | 8.20% |
| Unknown | 0 | 0 | 0 | 0.00% | 0.00% | 0.00% |
| Not recorded | 1 | 0 | 1 | 1.10% | 0.00% | 0.50% |
| Total | 94 | 90 | 184 | 100.00% | 100.00% | 100.00% |
| Certain AMD | 37 | 37 | 75 | 39.40% | 41.10% | 40.20% |
| Possible AMD | 47 | 48 | 95 | 50.00% | 53.30% | 51.60% |
| Eyes were accessioned during the period 6/17/16 – 9/14/17. Criteria: ≥ 80 years, white, non-diabetic, ≤6 h death-to-preservation. Target: 184 eyes (180 eyes of 90 donors, preserved; 4 eyes of 4 donors, preserved) Death-to-preservation time (mean, maximum, minimum): 3.9 h, 6.4 h, 2.0 h | ||||||
Table 1: Donor eye recovery from 2016-2017.
Supplementary Material 1: Overview of the dissection, color fundus photography, and OCT-based multimodal imaging. Please click here to download this File.
Supplementary Material 2: Details of the OCT-based multimodal imaging to illustrate the steps in sections 5-8. Please click here to download this File.
Discussion
Using a population-based screening approach during a 16 month period in the pre-COVID era, it was possible to procure 75 donor eyes with AMD. All were recovered with a short DtoP and staged using OCT-anchored MMI. The age criterion (>80 years) is outside the typical age range for tissue recoveries intended for transplantable corneas. Despite the advanced age, our criteria resulted in eyes at all stages of AMD. Many RPE phenotypes are common to all AMD stages, and some are exclusive to neovascular AMD3,46. The direct comparison of ex vivo and in vivo imaging (Figure 7) confirmed that short DtoP is a critical factor, along with expert handling (Figure 1), in producing ex vivo images of the outer retina sufficient for top-level diagnostic classification, and some of these samples were suitable for direct correlations between imaging and histology4,35,47. Not all pathology is visible ex vivo. However, far more is visible when using OCT compared to color photography-based methods10, especially those involving separating the retina from the RPE/choroid27. Further, eye tracking from clinical OCT directs attention to focal and sometimes small features (Figure 7).
This preservation system involves typical eye banking procedures and tools for cornea removal, followed by the immersion of an opened eye in a preservative provided in advance. In this way, the eye bank personnel can recover research tissues continuously (24/7). The latter characteristic is critical, as tissues destined for immediate complex dissections27,32 require around-the-clock staffing by either the investigators, the eye bank, or both. Other stabilizers that would enable tissue recovery at any hour followed by specialized dissection and extractions during working hours, such as the PAXgene Tissue System and Hibernate-A, may be helpful in the future if these are compatible with OCT imaging.
This approach yields retinas that remain largely attached to the RPE and are, thus, capable of generating data translatable to OCT imaging. Precise localization is essential because the macula is small (<3% of the retinal area). Further, deposit-driven AMD progression aligns with the topography of cones and rods48, and the earliest onset and most persistent visual defects occur in a specific place (i.e., the rod-containing parafovea next to the all-cone fovea)49. For comparison to OCT, immunohistochemistry with colorimetric dyes is compatible with comprehensive microscopy (e.g., bright-field) to account for all the tissue elements, both labeled and unlabeled4. Untargeted molecular assays based on sampling arrays can leverage the horizontal alignment of vertically compartmentalized photoreceptors and the RPE to effectively increase the resolution. Imaging mass spectrometry with ionizing laser pulses of 8 µm in diameter and 10-15 µm apart can localize dozens of lipids to subcellular compartments of the outer retinal cells50. Spatially resolved transcriptomics uses a pre-arranged set of barcoded reverse transcription primers on glass slides, to which tissue sections are applied51. This technology is currently limited to 55 µm diameter capture and 100 µm spacing; improvements in the resolution that would make this technology suitable for AMD research are anticipated.
This protocol has limitations. The recovery criterion omits diagnoses of diabetes (30% among Medicare recipients in Alabama)52 due to the importance of the choroid in both diseases. This exclusion reduces the overall donor pool and lengthens the time needed to collect enough eyes for a study. For historical reasons, the 2016-2017 criteria omitted black donors, who now represent 14% of state-wide eye donors, and black donors are now included in current prospective projects. The advance directive registry for persons wishing to be eye donors is extensive but does not yet include convenient registration at local retina specialty practices that care for AMD patients; this project is currently in development. During a population-based screen such as this one, eyes with conditions overlapping in phenotype with AMD will appear and must be recognized in consultation with the evolving clinical literature and an ophthalmologist specializing in retinal diseases. For example, this series of eyes included nevi with drusen53 and a line of RPE disturbance over a pachyvessel (a thick vessel in the inner choroid)54. Finally, early and intermediate AMD were combined due to the lack of an OCT-based grading system for AMD, which is under development by the Classification of Atrophy Meeting group55. Nevertheless, the optimization of tissue recovery and OCT-based characterization allows AMD research focusing on leveraging the time element of eye-tracked OCT-anchored MMI to be planned, powered, scheduled, and budgeted for in funding applications.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank Heidelberg Engineering for the instrumentation and the design of the original eye holder, Richard F. Spaide MD for the introduction to OCT-based multimodal imaging, Christopher Girkin MD for facilitating access to clinical imaging devices, and David Fisher for Figure 1. The recovery of the human donor eyes for research was supported by National Institutes of Health (NIH) grants R01EY06019 (C.A.C.), P30 EY003039 (Pittler), R01EY015520 (Smith), R01EY027948 (C.A.C., T.A.) R01EY030192 (Li), R01EY031209 (Stambolian), and U54EY032442 (Spraggins), IZKF Würzburg (N-304, T.A.), the EyeSight Foundation of Alabama, the International Retinal Research Foundation (C.A.C.), the Arnold and Mabel Beckman Initiative for Macular Research (C.A.C.), and Research to Prevent Blindness AMD Catalyst (Schey).
Materials
| Beakers, 250 mL | Fisher | # 02-540K | |
| Bottles, 1 L, Pyrex | Fisher | # 10-462-719 | storage for preservative |
| Bunsen burner or heat source | Eisco | # 17-12-818 | To melt wax |
| Camera, digital | Nikon D7200 | D7200 | |
| Computer and storage | Apple | iMac Pro; 14 TB external hard drive | Image storage |
| Container, insulated | Fisher | # 02-591-45 | For wet ice |
| Containers, 2 per donor, 40 mL | Fisher | Sameco Bio-Tite 40 mL # 13-711-86 | For preservative |
| Crucible, quartz 30 mL | Fisher | # 08-072D | Hold globe for photography |
| Cylinder, graduate, 250 mL | Fisher | # 08-549G | |
| Disinfectant cleaning supplies | https://www.cardinalhealth.com/en/product-solutions/medical/infection-control/antiseptics.html | ||
| Eye holder with lens and mounting bracket | contact J. Messinger | jeffreymessinger@uabmc.edu | custom modification of Heidelberg Engineering original design |
| Face Protection Masks | Fisher | # 19-910-667 | |
| Forceps, Harmon Fix | Roboz | # RS-8247 | |
| Forceps, Micro Adson | Roboz | # RS-5232 | |
| Forceps, Tissue | Roboz | # RS-5172 | |
| Glass petri dish, Kimax | Fisher | # 23064 | |
| Gloves Diamond Grip | Fisher | # MF-300 | |
| Gowns GenPro | Fisher | # 19-166-116 | |
| Image editing software | Adobe | Photoshop 2021, Creative Suite | |
| KimWipes | Fisher | # 06-666 | |
| Lamps, 3 goosenecks | Schott Imaging | # A20800 | |
| Microscope, stereo | Nikon | SMZ 1000 | for dissection |
| Microscope, stereo | Olympus | SZX9 | color fundus photography |
| Paraformaldehyde, 20% | EMS | # 15713-S | for preservative; dilute for storage |
| pH meter | Fisher | # 01-913-806 | |
| Phosphate buffer, Sorenson’s, 0.2 M pH 7.2 | EMS | # 11600-10 | |
| Ring flash | B & H Photo Video | Sigma EM-140 DG | |
| Ruby bead, 1 mm diameter | Meller Optics | # MRB10MD | |
| Safety Glasses 3M | Fisher | # 19-070-940 | |
| Scanning laser ophthalmoscope | Heidelberg Engineering | HRA2 | |
| Scissors, curved spring | Roboz | # RS-5681 | |
| Sharps container | Fisher | # 1482763 | |
| Shutter cord, remote | Nikon | MC-DC2 | |
| Spectral Domain OCT device | Heidelberg Engineering | Spectralis HRA&OCT | https://www.heidelbergengineering.com/media/e-learning/Totara-US/files/pdf-tutorials/2238-003_Spectralis-Training-Guide.pdf |
| Stainless steel ball bearing, 25.4 mm diameter | McMaster-Carr | # 9529K31 | |
| Tissue marking dye, black | Cancer Diagnostics Inc | # 0727-1 | |
| Tissue slicer blades | Thomas Scientific | # 6767C18 | |
| Trephine, 18-mm diameter | Stratis Healthcare | # 6718L | |
| TV monitor (HDMI) and cord for digital camera | B&H Photo Video | BH # COHD18G6PROB | for live viewing and remote camera display features |
| Wax, pink dental | EMS | # 72670 | |
| Wooden applicators | Puritan | # 807-12 |
References
- Spaide, R. F., et al. Consensus nomenclature for reporting neovascular age-related macular degeneration data: Consensus on neovascular age-related macular degeneration nomenclature study group. Ophthalmology. 127 (5), 616-636 (2020).
- Spaide, R. F., Ooto, S., Curcio, C. A. Subretinal drusenoid deposits a.k.a. pseudodrusen. Survey of Ophthalmology. 63 (6), 782-815 (2018).
- Curcio, C. A., Zanzottera, E. C., Ach, T., Balaratnasingam, C., Freund, K. B. Activated retinal pigment epithelium, an optical coherence tomography biomarker for progression in age-related macular degeneration. Investigative Ophthalmology & Visual Science. 58 (6), 211-226 (2017).
- Cao, D., et al. Hyperreflective foci, OCT progression indicators in age-related macular degeneration, include transdifferentiated retinal pigment epithelium. Investigative Ophthalmology & Visual Science. 62 (10), 34 (2021).
- Zanzottera, E. C., et al. Visualizing retinal pigment epithelium phenotypes in the transition to geographic atrophy in age-related macular degeneration. Retina. 36, S12-S25 (2016).
- Edwards, M. M., et al. Subretinal glial membranes in eyes with geographic atrophy. Investigative Ophthalmology & Visual Science. 58 (3), 1352-1367 (2017).
- Zhang, Z., Shen, M. M., Fu, Y. Combination of AIBP, apoA-I, and aflibercept overcomes anti-VEGF resistance in neovascular AMD by inhibiting arteriolar choroidal neovascularization. Investigative Ophthalmology & Visual Science. 63 (12), 2 (2022).
- Jiang, M., et al. Microtubule motors transport phagosomes in the RPE, and lack of KLC1 leads to AMD-like pathogenesis. Journal of Cell Biology. 210 (4), 595-611 (2015).
- Collin, G. B., et al. Disruption of murine Adamtsl4 results in zonular fiber detachment from the lens and in retinal pigment epithelium dedifferentiation. Human Molecular Genetics. 24 (24), 6958-6974 (2015).
- Curcio, C. A., Medeiros, N. E., Millican, C. L. The Alabama Age-related Macular Degeneration Grading System for donor eyes. Investigative Ophthalmology and Visual Science. 39 (7), 1085-1096 (1998).
- Bastek, J. V., Siegel, E. B., Straatsma, B. R., Foos, R. Y. Chorioretinal juncture. Pigmentary patterns of the peripheral fundus. Ophthalmology. 89 (12), 1455-1463 (1982).
- Lewis, H., Straatsma, B. R., Foos, R. Y., Lightfoot, D. O. Reticular degeneration of the pigment epithelium. Ophthalmology. 92 (11), 1485-1495 (1985).
- Lewis, H., Straatsma, B. R., Foos, R. Y. Chorioretinal juncture. Multiple extramacular drusen. Ophthalmology. 93 (8), 1098-1112 (1986).
- Sarks, J. P., Sarks, S. H., Killingsworth, M. C. Evolution of geographic atrophy of the retinal pigment epithelium. Eye. 2 (5), 552-577 (1988).
- Sarks, J. P., Sarks, S. H., Killingsworth, M. C. Evolution of soft drusen in age-related macular degeneration. Eye. 8 (3), 269-283 (1994).
- Ghazi, N. G., Dibernardo, C., Ying, H. S., Mori, K., Gehlbach, P. L. Optical coherence tomography of enucleated human eye specimens with histological correlation: Origin of the outer "red line". American Journal of Ophthalmology. 141 (4), 719-726 (2006).
- Brown, N. H., et al. Developing SDOCT to assess donor human eyes prior to tissue sectioning for research. Graefe’s Archive for Clinical and Experimental Ophthalmology. 247 (8), 1069-1080 (2009).
- Helb, H. M., et al. Clinical evaluation of simultaneous confocal scanning laser ophthalmoscopy imaging combined with high-resolution, spectral-domain optical coherence tomography. Acta Ophthalmologica. 88 (8), 842-849 (2010).
- Spaide, R. F., Curcio, C. A. Drusen characterization with multimodal imaging. Retina. 30 (9), 1441-1454 (2010).
- Naghavi, M., et al. From vulnerable plaque to vulnerable patient: a call for new definitions and risk assessment strategies: Part 1. Circulation. 108 (14), 1664-1672 (2003).
- Garcia-Garcia, H. M., Gonzalo, N., Regar, E., Serruys, P. W. Virtual histology and optical coherence tomography: from research to a broad clinical application. Heart. 95 (16), 1362-1374 (2009).
- Strouthidis, N. G., et al. Comparison of clinical and spectral domain optical coherence tomography optic disc margin anatomy. Investigative Ophthalmology & Visual Science. 50 (10), 4709-4718 (2009).
- Sarks, S. H. Ageing and degeneration in the macular region: A clinico-pathological study. British Journal of Ophthalmology. 60 (5), 324-341 (1976).
- Sura, A. A., et al. Measuring the contributions of basal laminar deposit and Bruch’s membrane in age-related macular degeneration. Investigative Ophthalmology & Visual Science. 61 (13), 19 (2020).
- Chen, L., Messinger, J. D., Kar, D., Duncan, J. L., Curcio, C. A. Biometrics, impact, and significance of basal linear deposit and subretinal drusenoid deposit in age-related macular degeneration. Investigative Ophthalmology & Visual Science. 62 (1), 33 (2021).
- Litts, K. M., et al. Clinicopathological correlation of outer retinal tubulation in age-related macular degeneration. JAMA Ophthalmology. 133 (5), 609-612 (2015).
- Olsen, T. W., Feng, X. The Minnesota grading system of eye bank eyes for age-related macular degeneration. Investigative Ophthalmology & Visual Science. 45 (12), 4484-4490 (2004).
- Mano, F., Sprehe, N., Olsen, T. W. Association of drusen phenotype in age-related macular degeneration from human eye-bank eyes to disease stage and cause of death. Ophthalmology Retina. 5 (8), 743-749 (2021).
- Age-related eye disease study research group. The Age-Related Eye Disease Study system for classifying age-related macular degeneration from stereoscopic color fundus photographs: The Age-Related Eye Disease Study Report Number 6. American Journal of Ophthalmology. 132 (5), 668-681 (2001).
- Arnold, J. J., Sarks, S. H., Killingsworth, M. C., Sarks, J. P. Reticular pseudodrusen. A risk factor in age-related maculopathy. Retina. 15 (3), 183-191 (1995).
- Olsen, T. W., Bottini, A. R., Mendoza, P., Grossniklausk, H. E. The age-related macular degeneration complex: linking epidemiology and histopathology using the Minnesota grading system (the inaugural Frederick C. Blodi Lecture). Transactions of the American Ophthalmological Society. 113, (2015).
- Owen, L. A., et al. The Utah protocol for postmortem eye phenotyping and molecular biochemical analysis. Investigative Ophthalmology & Visual Science. 60 (4), 1204-1212 (2019).
- Wang, J. J., et al. Ten-year incidence and progression of age-related maculopathy: The Blue Mountains Eye Study. Ophthalmology. 114 (1), 92-98 (2007).
- Joachim, N., Mitchell, P., Burlutsky, G., Kifley, A., Wang, J. J. The incidence and progression of age-related macular degeneration over 15 years: The Blue Mountains Eye Study. Ophthalmology. 122 (12), 2482-2489 (2015).
- Pang, C., Messinger, J. D., Zanzottera, E. C., Freund, K. B., Curcio, C. A. The onion sign in neovascular age-related macular degeneration represents cholesterol crystals. Ophthalmology. 122 (11), 2316-2326 (2015).
- Keilhauer, C. N., Delori, F. C. Near-infrared autofluorescence imaging of the fundus: Visualization of ocular melanin. Investigative Ophthalmology & Visual Science. 47 (8), 3556-3564 (2006).
- Curcio, C. A., Saunders, P. L., Younger, P. W., Malek, G. Peripapillary chorioretinal atrophy: Bruch’s membrane changes and photoreceptor loss. Ophthalmology. 107 (2), 334-343 (2000).
- Curcio, C. A. Imaging maculopathy in the post-mortem human retina. Vision Research. 45 (28), 3496-3503 (2005).
- Brinkmann, M., et al. Histology and clinical lifecycle of acquired vitelliform lesion, a pathway to advanced age-related macular degeneration. American Journal of Ophthalmology. 240, 99-114 (2022).
- Ramtohul, P., et al. Bacillary layer detachment: Multimodal imaging and histologic evidence of a novel optical coherence tomography terminology. Literature review and proposed theory. Retina. 41 (11), 2193-2207 (2021).
- Wilson, J. D., Foster, T. H. Mie theory interpretations of light scattering from intact cells. Optics Letters. 30 (18), 2442-2444 (2005).
- Ghazi, N. G., Green, W. R. Pathology and pathogenesis of retinal detachment. Eye. 16 (4), 411-421 (2002).
- Berlin, A., et al. Correlation of optical coherence tomography angiography of type 3 macular neovascularization with corresponding histology. JAMA Ophthalmology. 140 (6), 628-633 (2022).
- Berlin, A., et al. Histology of type 3 macular neovascularization and microvascular anomalies in anti-VEGF treated age-related macular degeneration. Ophthalmology Science. 3 (3), 100280 (2023).
- Schaal, K. B., et al. Outer retinal tubulation in advanced age-related macular degeneration: optical coherence tomographic findings correspond to histology. Retina. 35 (7), 1339-1350 (2015).
- Chen, L., et al. Histology and clinical imaging lifecycle of black pigment in fibrosis secondary to neovascular age-related macular degeneration. Experimental Eye Research. 214, 108882 (2022).
- Balaratnasingam, C., et al. Histologic and optical coherence tomographic correlations in drusenoid pigment epithelium detachment in age-related macular degeneration. Ophthalmology. 124 (1), 644-656 (2017).
- Curcio, C. A., et al. Subretinal drusenoid deposits in non-neovascular age-related macular degeneration: Morphology, prevalence, topography, and biogenesis model. Retina. 33 (2), 265-276 (2013).
- Owsley, C., et al. Biologically guided optimization of test target location for rod-mediated dark adaptation in age-related macular degeneration: ALSTAR2 baseline. Ophthalmology Science. 3 (2), 100274 (2023).
- Anderson, D. M. G., et al. The molecular landscape of the human retina and supporting tissues by high resolution imaging mass spectrometry. Journal of the American Society for Mass Spectrometry. 31 (12), 2426-2436 (2020).
- Lee, J., Yoo, M., Choi, J. Recent advances in spatially resolved transcriptomics: challenges and opportunities. BMB Reports. 55 (3), 113-124 (2022).
- Diabetes. Alabama Public Health Available from: https://www.alabamapublichealth.gov/healthrankings/diabetes.html (2022)
- Francis, J. H., et al. Swept-source optical coherence tomography features of choroidal nevi. American Journal of Ophthalmology. 159 (1), 169-176 (2015).
- Inoue, M., Dansingani, K. K., Freund, K. B. Progression of age-related macular degeneration overlying a large choroidal vessel. Retina Cases Brief Reports. 10 (1), 22-25 (2016).
- Jaffe, G. J., et al. Imaging features associated with progression to geographic atrophy in age-related macular degeneration: CAM Report 5. Ophthalmology Retina. 5 (9), 855-867 (2021).

