Disruption of the Mouse Blood-Brain Barrier by Small Extracellular Vesicles from Hypoxic Human Placentas
Summary
A protocol is presented to evaluate whether small EVs (sEVs) isolated from placental explants cultured under hypoxic conditions (modeling one aspect of preeclampsia) disrupt the blood-brain barrier in nonpregnant adult female mice.
Abstract
Cerebrovascular complications, including cerebral edema and ischemic and hemorrhagic stroke, constitute the leading cause of maternal mortality associated with preeclampsia. The underlying mechanisms of these cerebrovascular complications remain unclear. However, they are linked to placental dysfunction and blood-brain barrier (BBB) disruption. Nevertheless, the connection between these two distant organs is still being determined. Increasing evidence suggests that the placenta releases signaling molecules, including extracellular vesicles, into maternal circulation. Extracellular vesicles are categorized according to their size, with small extracellular vesicles (sEVs smaller than 200 nm in diameter) considered critical signaling particles in both physiological and pathological conditions. In preeclampsia, there is an increased number of circulating sEVs in maternal circulation, the signaling function of which is not well understood. Placental sEVs released in preeclampsia or from normal pregnancy placentas exposed to hypoxia induce brain endothelial dysfunction and disruption of the BBB. In this protocol, we assess whether sEVs isolated from placental explants cultured under hypoxic conditions (modeling one aspect of preeclampsia) disrupt the BBB in vivo.
Introduction
Approximately 70% of maternal deaths due to preeclampsia, a hypertensive pregnancy syndrome characterized by impaired placentation processes, maternal systemic endothelial dysfunction, and, in severe cases, multi-organ failure1,2, are associated with acute cerebrovascular complications3,4. Most maternal deaths occur in low and middle-income countries5. However, the underlying mechanisms are still unclear despite the clinical and epidemiological relevance of cerebrovascular complications associated with preeclampsia.
On the other hand, extracellular vesicles (EVs) (diameter ~30-400 nm) are essential mediators of intercellular communication among tissues and organs, including maternal-placental interaction6. In addition to proteins and lipids on the external surface, EVs carry cargo within (proteins, RNA, and lipids). EVs can be categorized into (1) exosomes (diameter ~50-150 nm, also named small EVs (sEVs)), (2) medium/large EVs, and (3) apoptotic bodies, which differ by size, biogenesis, content, and potential signaling function. The composition of EVs is determined by the cells from which they originate and the disease type7. Syncytiotrophoblast-derived EVs express placental alkaline phosphatase (PLAP)8,9, which detects placentae-derived circulating small EVs (PDsEVs) in pregnancy. Also, PLAP helps discern changes in the PDsEVs cargo and their effects in preeclampsia versus normotensive pregnancies10,11,12,13,14,15.
The placenta has been recognized as the necessary component in the pathophysiology of preeclampsia16 or cerebral complications associated with this disease17,18,19. However, how this distant organ might induce alterations in brain circulation is unknown. Since sEVs play pivotal roles in cell-to-cell communication due to their capacity to transfer bioactive components from donor to recipient cells6,20,21, a growing number of studies have associated placental sEVs with the generation of maternal endothelial dysfunction21,22,23,24, including brain endothelial cells25,26in women with preeclampsia. Thus, the compromise of brain endothelial function may lead to disruption of the blood-brain barrier (BBB), a critical component in cerebrovascular complications associated with preeclampsia3,27.
Nevertheless, preclinical findings using rat cerebral vessels exposed to serum of women with preeclampsia28 or human brain endothelial cells exposed to plasma of women with preeclampsia29 reported that circulating factor(s) induce disruption of the BBB. Despite several candidates with the potential to harm the BBB present in the maternal circulation during preeclampsia, such as elevated levels of proinflammatory cytokines (i.e., tumor necrosis factor)18,28 or vascular regulators (i.e., vascular endothelial growth factor (VEGF))29,30,31, or oxidative molecules such as oxidized-lipoproteins (oxo-LDL)32,33, among others34, none of them establishes a direct connection between the placenta and the BBB. Recently, sEVs isolated from hypoxic placentas have shown the capacity to disrupt the BBB in nonpregnant female mice25. Since placental sEVs may carry most of the listed circulating factors with the capacity to disrupt the BBB, sEVs are considered suitable candidates to connect the injured placenta, be the carrier of harmful circulating factors, and disrupt the BBB in preeclampsia.
This protocol allows us to investigate whether sEVs isolated from placental explants cultured under hypoxic conditions can disrupt the BBB in nonpregnant female mice as a proxy for understanding the pathophysiology of cerebral complications during preeclampsia.
Protocol
The research was carried out following the principles expressed in the Declaration of Helsinki and under the authorization of the respective Ethical Review Boards. All human participants gave their informed consent before sample collection, as reported previously25. Additionally, the Bioethics and Biosafety Committee of the Bío-Bío University approved this project (Fondecyt grant 1200250). The animal work was conducted in accordance with the cardinal principles of the three R's in the use of animals in experimentation35, and according to the recommendations of the guidelines for the Care and Use of Laboratory Animals published by the US National Institute of Health. Animals were kept in appropriate environments at the Vivarium of the University of Bío-Bío. Fresh placentas (n = 4) were obtained within 1 h after elective cesarean section from mothers (ages 28-31 years old) with normal pregnancies at term (38 to 41 weeks of gestation). Cesarean sections were performed at the Herminda Martin Clinical Hospital, Chillan, Chile, as previously reported25. To apply the in vivo model, 4-6-month-old female nonpregnant mice (strain C57BLACK/6) were used. They were divided into three experimental groups: (1) control (without treatment), (2) treated with sEVs from normoxia (sEVs-Nor), and (3) treated with sEVs from hypoxic cultured placentas (sEVs-Hyp), which were used to evaluate the disruption of the BBB in vivo25. All the injected solutions were sterile. Also, the preparation of sEVs was performed in aseptic conditions and under a class II biosafety hood to avoid contamination.
1. Placental culture explants
- To extract explants of normal pregnancy placentas, check the method published by Miller et al. 200536.
- Carefully remove the clots from the basal plate of the human placenta using sterilized forceps. Extract small explants to obtain a final tissue extraction of 10 g.
- Ensure that placental explants are removed from the four quadrants of the maternal portion of the placenta. Ensure that devitalized tissue and calcified areas are excluded.
- To manipulate placental tissue, do so on ice, under sterile conditions, and within a biosafety cabinet.
- Wash the explants (at least three times) with copious amounts (five volumes of the explants) of cold (4 °C) phosphate buffer solution (PBS 1x, pH 7.4) to remove as much blood as possible. Centrifuge (252 x g for 10 min) at room temperature between washes.
- Resuspend 10 g of washed explants in 20 mL of culture medium supplemented with 2% fetal bovine serum, 100 IU/mL penicillin, and 100 µg/mL of streptomycin (see Table of Materials).
- Put the suspension in a 100 mm culture dish. Leave the explants for 2 h in an incubator at 37 °C with 21% oxygen and 5% CO2.
- After that, rewash the explants with 1x PBS at 37 °C (three times). Centrifuge (252 x g for 10 min) at room temperature between washes.
- Further, resuspend the tissue in 20 mL of culture medium previously depleted of nanoparticles.
NOTE: For nanoparticle depletion, perform ultracentrifugation (120,000 x g for 18 h at room temperature) and microfiltration (0.22 µm filter). - Split the resuspended explants into two cultures dishes (100 mm). Each culture dish must include 10 mL of resuspended explants.
- After that, place one of the dishes containing placental explants in standard culture conditions (37 °C with 8% oxygen and 5% CO2), while placing the other one in a hypoxic chamber with 1% O2.
NOTE: (OPTIONAL) Analysis of tissue viability can be performed using the same tissue extractions and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay (MTT assay) as described elsewhere37,38. Use total protein concentration measured in the explants homogenates to normalize MTT values. - After 18 h in culture, harvest the conditioned media of the explants into a new 15 mL tube.
NOTE: At this step, conditioned media can be frozen (-80 °C) for an extended period (weeks).
2. Placental-derived sEVs isolation
- Isolate sEVs from the conditioned medium using the differential centrifugation and microfiltration protocol following previously published reports25,39.
- Perform sequential centrifugation for the harvested conditioned medium. Sequential steps of centrifugations are (1) 300 x g for 10 min, (2) 2000 x g for 10 min, (3) 10,000 x g for 30 min, and (4) 120,000 x g for 2 h. Perform the centrifugations room temperature.
- In all these centrifugations, using a 20-100 μL pipette, carefully collect the supernatant and discard the pellet.
- Once done, pass the last collected supernatant through a 0.22 µm filter (see Table of Materials). Subsequently, perform one additional centrifugation at 120,000 x g for 18 h at room temperature.
- After that, discard the supernatant while resuspending the pellet (which contains placental sEVs) in 500 µL of PBS (pH 7.4) and again pass through a 0.22 µm filter. Finally, perform one last centrifugation at 120,000 x g for 3 h at room temperature.
- Next, resuspend the pellet (which contains placental sEVs) in 500 µL of PBS (pH 7.4, previously depleted of sEVs) and pass through a 0.22 µm filter.
- Label the samples as stock of sEVs-Normoxia (sEVs-Nor) or sEVs-Hypoxia (sEVs-Hyp).
NOTE: Prepare 50-100 µL aliquots of the isolated sEVs for further experimentation. Small EVs can be stored at -80 °C for an extended period (months). - Characterize the placental sEVs by size, count, and sEVs protein marker, as reported previously25. The ideal mean particle diameter of sEVs is 50-150 nm.
NOTE: sEVs characterization includes positive detection of CD63, Tsg101, Alix, and HSP70 (i.e., to characterize sEVs population enriched in exosomes). Also, use PLAP as a marker of placental origin25. - Measure the total amount of protein in the sEVs solution using the BCA protein assay kit following the manufacturer's instructions (see Table of Materials).
NOTE: Figure 1 shows an overview of the placental explant culture and extracellular vesicle isolation.
3. Mice injection
- Anesthetize the animal in an anesthetic induction chamber with 5% isoflurane distribution (see Table of Materials). Verify the level of anesthesia via a toe pinch.
- After ~40 s, put the mouse into the anesthesia system using a facemask and maintain an anesthesia level of 2% isoflurane.
- Next, position the animal on a heating platform (see Table of Materials) at a room temperature of 28 °C. Keep the eyes lubricated with artificial tears.
- Before injecting, dilute placental sEVs (200 μg of total protein) with phosphate buffer (pH, 7.4) until reaching a final volume of 70 µL.
- Disinfect the injection area using 70% ethanol and cotton swabs. Ensure to use an insulin syringe with a 30 G needle to inject the sEVs solution.
NOTE: The syringe can be positioned on the heating platform for 5 min to acquire a physiological temperature. - Then, inject the solution into the external jugular vein. This step needs special training.
NOTE: The procedure must be performed cautiously, slowly retracting the needle and gently aspirating to evidence of blood reflux. - After injection, apply light pressure to the injected area for ~15 s with a dry cotton swab. After that, return the animals to their respective cages.
- Ensure to evaluate consciousness and behavior for 15 min. An indicator of consciousness achievement is the animal recovery of total motor activity.
NOTE: Ensure to keep a warm temperature in animal's cages.
4. Rapid murine coma and behavior scale (RMCBS)
- Use the RMCBS to evaluate the neurological and well-being parameters of the animals40.
NOTE: RMCBS allows a fast evaluation (~ 3 min) with minimal animal stress due to operator intervention. RMCBS has ten parameters (each score is 0-2). - Evaluate mouse well-being with the RMCBS scale at 0 h (before injecting sEVs) and 3 h, 6 h, 12 h, and 24 h after injection.
NOTE: Perform analysis of claudin-5 (at 6 h after sEVs injection) and Evan's blue extravasation (at 24 h after sEVs injection) to evaluate the disruption of the BBB (Figure 2).
5. Analysis of Evan's blue extravasation
- After placental sEVs injection (24 h), anesthetize the mice as described in step 3.1.
- Prepare Evan's Blue solution (2%, diluted in phosphate buffer solution, 1x) (see Table of Materials).
- Using retro-orbital access25, inject Evan's Blue solution at 2 mL/kg.
- Allow Evan's blue solution to circulate for 20 min under maintenance of anesthesia.
- Next, perform intracardiac perfusion following previously described protocol41 with saline solution (~3 mL, 0.9%, w/v) to remove Evan's blue dye from the circulation. This step is mandatory.
NOTE: For this procedure, increase anesthesia to 5% and verify a deep plane of anesthesia (i.e., drastic reduction in respiratory rate). - Expose the heart via medial thoracotomy41 (Figure 3).
- After that, fix the whole animal with an intracardial infusion of paraformaldehyde solution (4% in PBS, v/v).
NOTE: Check the rigidity of the tail for monitoring the appropriate fixation. - Further, once fixation is complete, carefully extract the brain, weigh it, and photograph it41. Take pictures with the whole brain close to a ruler42.
- Next, put the brain in a brain mouse slicer (see Table of Materials).
- Dissect the brain into nine sections, crane-caudal, including the cerebellum (100 μm each section).
- After dissection, visualize and capture images of the brain slices in a stereo-zoom microscope (see Table of Materials).
- Use the captured images to quantify Evan's blue extravasation using ImageJ software (see Table of Materials).
NOTE: To analyze the presence of Evan's blue dye, set Evan's blue values using a positive control of the brain section (from an animal control animal injected with dye but without intracardial perfusion). Evan's blue values reached from positive control are generally between 75 and 110 in the intensity histogram of the blue channel (Figure 4). - Ensure that brain images are analyzed blindly by the respective experimental group to avoid observer bias. Alternatively, quantify Evan's blue extravasation by brain homogenization followed by spectroscopy43.
NOTE: In separate experiments, using freshly isolated brains from sEVs injected mice (6 h), analyze the protein levels of claudin 5 (CLND-5) (see Table of Materials), a critical tight junction involved in the tightness of the BBB44 (Figure 5).
Representative Results
This protocol evaluates the capacity of sEVs derived from placentas cultured in hypoxia to disrupt the BBB in nonpregnant mice. This method allows one to understand better the potential connection between the placenta and the brain in normal and pathological conditions. In particular, this method may constitute a proxy to analyze placental sEVs participation in the onset of cerebral complications in preeclampsia.
Contrary to mice injected with sEVs-Nor, mice injected with sEVs-Hyp show a progressive decline in the neurological score until 24 h (Table 1), which suggests the sEVs-Hyp capacity to impair brain function.
Also, mice brains of the sEVs-Hyp injected group have higher fresh weight than those isolated from mice injected with sEVs-Nor or control mice (0.51 ± 0.008; 0.46 ± 0.008; 0.47 ± 0.01 g, respectively), which may constitute a gross indicator of brain edema45.
Compatible with this finding, this protocol allows one to identify Evan's blue extravasation as an indicator of disruption of the BBB. In that regard, brains from mice injected with sEVs-Hyp have higher Evan's blue extravasation than brains from the sEVs-Nor group (Figure 5A).
Although the underlying mechanism of disruption of the BBB induced by sEVs-Hyp was not analyzed with this protocol, results also indicate that sEVs-Hyp injected mice showed reduced protein amounts of CLND-5 in the areas in which the BBB was most affected (i.e., posterior areas) (Figure 5B). Therefore, it is feasible that sEVs-hyp impairs the expression of the function of this critical endothelial tight junction protein.
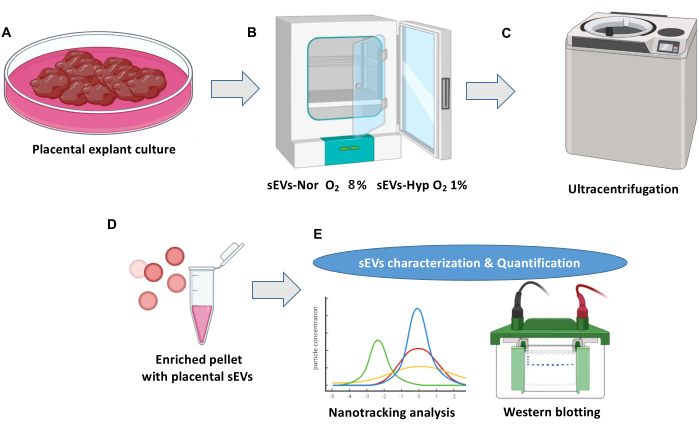
Figure 1: Placental explant culture and extracellular vesicle isolation protocol. (A) Normal placenta explant cultures. (B) Explants are distributed into two conditions for the biogenesis of placental small extracellular vesicles (sEVs). Normoxia (sEVs-Nor, 8% O2) or hypoxia (sEVs-Hyp, 1% O2) for 18 h. (C) Conditioned media is harvested, filtered, and centrifuged to eliminate cell debris. (D) sEVs are isolated by ultra-centrifugations. (E) sEVs are characterized using nano-tracking analysis and western blot. Please click here to view a larger version of this figure.
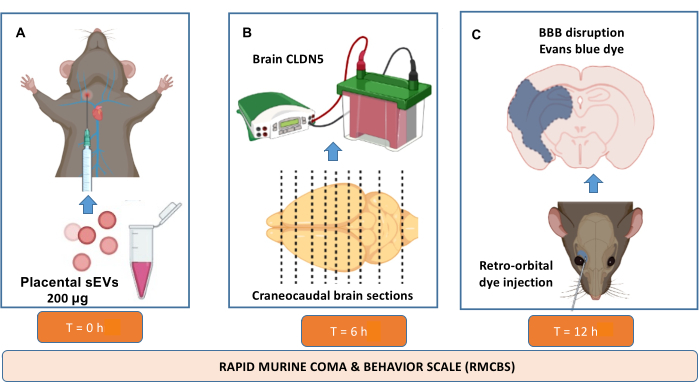
Figure 2: In vivo evaluation of blood-brain barrier disruption protocol. Nonpregnant C57BL6/J mice, 4-6 months old, are used. (A) Via external jugular vein, animals received sEVs (200 μg of total protein) isolated from Normoxic (sEVs-Nor, 8% O2) or hypoxic (sEVs-Hyp, 1% O2) placental cultures. RMCBS is monitored at 0-24 h after injection. (B) 6 h after sEVs injection, brains are extracted and sectioned into nine segments for protein extraction. Claudin 5 (CLDN5) is analyzed in homogenates of those nine sections. (C) Evan's blue extravasation analysis (24 h after sEVs injection) was analyzed after retro-orbital puncture injection in each of the nine segments. Please click here to view a larger version of this figure.
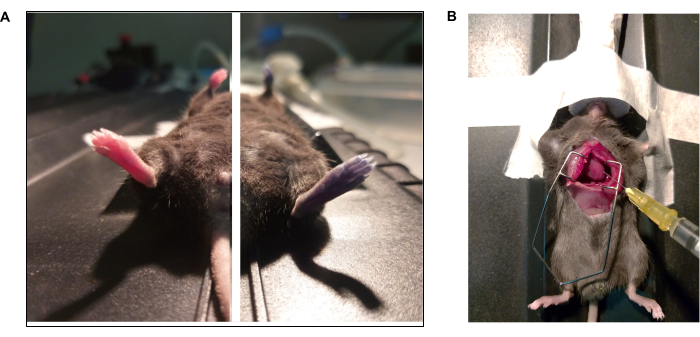
Figure 3: Photo documentaries of Evan' s blue dye and intracardial perfusion protocol. (A) Mouse received Evan's blue via retro-orbital injection. (Left) Animal before and (Right) after injection (15 s) of Evan's blue. (B) Thoracotomy to perform intracardial perfusion of phosphate buffer solution (1x PBS) and paraformaldehyde (4% PFA). The left ventricle is pointed with the tip of the needle. Please click here to view a larger version of this figure.
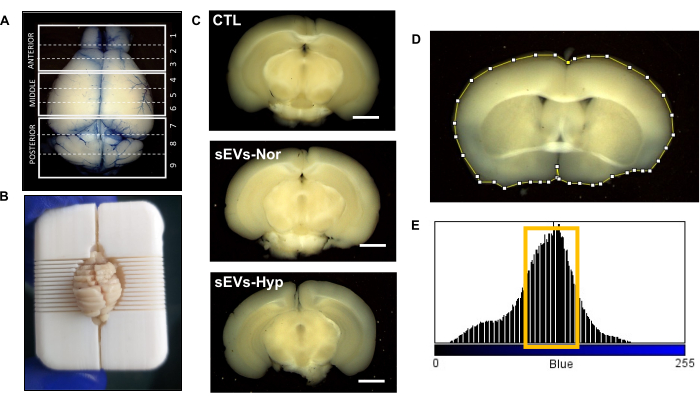
Figure 4: Analysis of Evan's blue extravasation after injection of sEVs. (A) Representative image of the whole brain showing Evan's blue extravasation. The dashed line represents nine sections obtained from the whole brain. (B) Brain dissected using a brain mice slicer. (C) Representative images of brain slices at 24 h after sEVs injection. Control (CTL), placenta in normoxic (sEVs-Nor) or hypoxic conditions (sEVs-Hyp). Scale bar = 0.4 cm. (D) Digital outlining of brain slice using ImageJ. (E) Histogram in the blue channel. Values between 75-110 are associated with Evan's blue extravasation. Please click here to view a larger version of this figure.
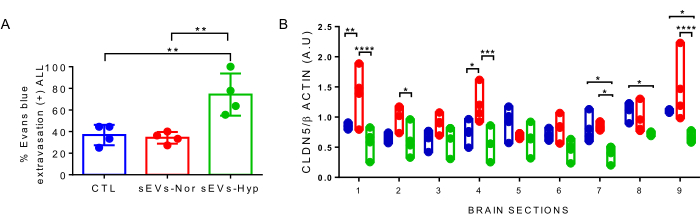
Figure 5: Evan's blue extravasation and claudin-5 levels in mice injected with sEVs isolated from placental explants. (A) Percentage of Evan's blue (EB) extravasation considering the whole brain sections. Control (CTL, blue), placenta in normoxic (sEVs-Nor, red) or hypoxic conditions (sEVs-Hyp, green). (B) Relative levels of claudin 5 (CLDN5) in the nine brain sections were obtained from the three experimental groups. β-actin is used as a loading control. Values are mean ± interquartile range. Each dot represents an individual experimental subject. *p < 0.05, **p < 0.005. ****p < 0.0001. ***p < 0.001; ANOVA test followed by Bonferroni post-test. Please click here to view a larger version of this figure.
| Time (h) | Control | sEVs-Normoxia | sEVs-Hypoxia | ANOVA |
| 0 | 18.75 ± 0.250 | 18.5 ± 0.288 | 18.75 ± 0.250 | ns |
| 3 | 18.5 ± 0.866 | 17 ± 0.707 | 13.25 ± 1.750*α | 0.006 |
| 6 | 19.25 ± 0.750 | 17 ± 0.577 | 11.75 ± 1.250*α | 0.002 |
| 12 | 18.5 ± 0.645 | 16.75 ± 1.109 | 13 ± 0.816*α | 0.001 |
| 24 | 19.5 ± 0.288 | 17.75 ± 0.250 | 10.25 ± 0.853*α | <0.0001 |
| *p < 0.01 versus control. αp < 0.01 versus sEVs-Normoxia | ||||
Table 1: Rapid murine coma and behavior scale (RMCBS) after 24 h post sEVs injection. The score is expressed as mean ± SEM. Animals' scores closest to 20 are standard, while the lower the score, the higher the dysfunction of the CNS.
Discussion
This study unveils fresh insights into potential harm resulting from sEVs isolated from placental explants cultured in hypoxic conditions on the disruption of the rodent blood-brain barrier. The pathological mechanism involves a reduction in CLND-5 in the posterior brain region25.
Prior investigations have revealed that plasma-sEVs from individuals with preeclampsia induce endothelial dysfunction in various organs using in vitro models46,47. This investigation particularly scrutinized the blood-brain barrier, offering a novel perspective on sEVs isolated from hypoxia-cultured placenta, which disrupts this vital barrier. These discoveries introduce a new realm of research wherein sEVs may serve as a communication channel between the placenta and the brain in both normal and pathological scenarios, like preeclampsia.
The presented protocol is straightforward, but several critical steps warrant mention. This protocol necessitates fresh placentas within 1 h post-delivery. We also advise against extending the culture period beyond 24 h to prevent tissue degradation. This protocol mitigates potential contamination from sEVs produced by mouse placentas. Digital analysis of Evan’s blue extravasation is time-intensive. Furthermore, Evan’s blue staining is less conspicuous after brain sectioning. Therefore, an initial setup for identifying the blue range using a positive control is recommended. A negative control, such as a brain from a mouse not subjected to Evan’s blue injection, can also be employed. Blind analysis of experimental groups is imperative to avert potential observer bias.
Several challenges may arise during this protocol. A significant limitation lies in biological variability originating from both human placentas and injected mice. To ensure the reproducibility of Evan’s blue extravasation experiments, establishing sEV doses isolated from human placentas is suggested. We opted for a dose of 200 µg of total protein; however, this quantity may fluctuate based on vesicle purity, the efficacy of jugular injection, Evan’s blue administration, its clearance from the circulatory system, and, not least, the biological impact of sEVs considering their contents.
The cellular mechanisms by which sEVs from the human placenta can disrupt the blood-brain barrier necessitate additional experimentation. Nevertheless, this method holds relevance, hinting at potential communication between the placenta and the brain, warranting further exploration. Therefore, future investigations are encouraged, concentrating on sEVs and their interactions with the blood-brain barrier, as well as the impact of their cargo on neuronal tissues. Whether blood-brain barrier impairment leads to enduring consequences for the maternal brain warrants further examination.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors would like to thank the researchers belonging to GRIVAS Health for their valuable input. Also, midwives and clinical staff from the Obstetrics and Gynecology Service belong to the Hospital de Chillan, Chile. Founded by Fondecyt Regular 1200250.
Materials
| Adult mice brain slecer matrice 3D printed | Open access file | Adult mice | Adult mice brain slicer. Printed in PLA filament. |
| Anti β-Actin primary antibody | Sigma-Aldrich | Clon AC-74 | Antibody for loading control (Western blot) |
| Anti-Claudin5 primary antibody | Santa cruz Biotechnology | sc-374221 | Primary antibody for tight junction protein CLDN5 of mice BBB (Western blot) |
| BCA protein kit | Thermo Scientific | 23225 | Kit for measuring protein concentration |
| Culture media #200 500 mL | Thermo Fisher Scientific | m200500 | Culture media for placental explants |
| D180 CO2 incubator | RWD Life science | D180 | Standard incubator to estabilize explants and culture sEVs-Nor |
| Evans blue dye > 75% 10 g | Sigma-Aldrich | E2129.10G | Dye to analize blood brain barrier disruption IN VIVO |
| Fetal bovine serum 500 mL | Thermo Fisher Scientific | 16000044 | Additive growth factor for culture media 200 |
| Himac Ultracentrifuge CP100NX | Himac eppendorf group | 5720410101 | Ultracentrifuge for condicioned media > 1,20,000 x g |
| ImageJ software | NIH | https://imagej.nih.gov/ij/download.html | |
| Isoflurane x 100 mL | USP Baxter | 212-094 | Volatile inhalated anaesthesia agent for mice |
| Kit CellTiter 96 Non-radioactive | Promega | 0000105232 | In vitro assay for placental explants viability |
| Mouse IgG Secondary antibody | Thermo Fisher Scientific | MO 63103 | Secondary antibody for CLDN5 (western blot) |
| NanoSight NS300 | Malvern Panalytical | 90278090 | Nanotracking analysis of particles from placental explants condicioned media |
| Paraformaldehide E 97% solution 500 mL | Thermo Fisher Scientific | A11313.22 | Fixative solution for brain tissue slices and intracardial perfusion (once diluted) |
| PBS 1 X pH 7.4 500 mL | Thermo Fisher Scientific | 10010023 | Wash solution for placenta explants |
| Peniciline-streptomicine 100x 20 mL | Thermo Fisher Scientific | 10378016 | Antiobiotics for placental explants culture media |
| ProOX C21 Cytocentric O2 and CO2 Subchamber Controller | BioSpherix | SCR_021131 | CO2 regulator to induce Hypoxia in sealed chamber for sEVs-Hyp |
| Sodium Thiopental 1 g | Chemie | 7061 | humanitarian euthanasia agent |
| Somnosuite low flow anesthesia system | Kent Scientifics | SS-01 | Isoflurane vaporizer for small rodents |
| Surgical Warming platform | Kent Scientifics | A41166 | Warming platform for mainteinance anesthesia in mice |
| Syringe Filters, Polytetrafluoroethylene (PTFE), Hydrophobic, 0.22 µm, Sterile, 25 mm | Southern labware | 10026 | Filtration of condicioned media harvested from placental explants |
| Tabletop High-Speed Micro Centrifuges HITACHI himac CT15E/CT15RE | Hitachi medical systems | 6020 | Serial centrifugations of condicioned media < 1,20, 000 x g |
| Trinocular stereomicroscope transmided and reflective light 10x-160x | Center Medical | 2597 | Stereomicroscope to register brain slices |
References
- Lisonkova, S., Joseph, K. S. Incidence of preeclampsia: risk factors and outcomes associated with early- versus late-onset disease. Am J Obstet Gynecol. 209 (544), 544.e1-544.e12 (2013).
- Sibai, B., Dekker, G., Kupferminc, M. Preeclampsia. Lancet. 365 (9461), 785-799 (2005).
- Hammer, E. S., Cipolla, M. J. Cerebrovascular dysfunction in preeclamptic pregnancies. Curr Hypertens Rep. 17 (8), 64 (2015).
- Okanloma, K. A., Moodley, J. Neurological complications associated with the preeclampsia/eclampsia syndrome. Int J Gynaecol Obstet. 71, 223-225 (2000).
- Frias, A. E., Belfort, M. A. Post magpie: how should we be managing severe preeclampsia. Curr Opin Gynecol Obstet. 15 (6), 489-495 (2003).
- Familari, M., Cronqvist, T., Masoumi, Z., Hansson, S. R. Placenta-derived extracellular vesicles: Their cargo and possible functions. Reprod Fertil Dev. 29 (3), 433-447 (2017).
- Montoro-Garcia, S., Shantsila, E., Marin, F., Blann, A., Lip, G. Y. Circulating microparticles: new insights into the biochemical basis of microparticle release and activity. Basic Res Cardiol. 106, 911-923 (2011).
- Germain, S. J., Sacks, G. P., Sooranna, S. R., Sargent, I. L., Redman, C. W. Systemic inflammatory priming in normal pregnancy and preeclampsia: the role of circulating syncytiotrophoblast microparticles. J Immunol. 178 (9), 5949-5956 (2007).
- Tannetta, D., Masliukaite, I., Vatish, M., Redman, C., Sargent, I. Update of syncytiotrophoblast derived extracellular vesicles in normal pregnancy and preeclampsia. J Reprod Immunol. 119, 98-106 (2017).
- Collett, G. P., Redman, C. W., Sargent, I. L., Vatish, M. Endoplasmic reticulum stress stimulates the release of extracellular vesicles carrying danger-associated molecular pattern (DAMP) molecules. Oncotarget. 9 (6), 6707-6717 (2018).
- Cooke, W. R., et al. Maternal circulating syncytiotrophoblast-derived extracellular vesicles contain biologically active 5′-tRNA halves. Biochem Biophys Res Commun. 518 (1), 107-113 (2019).
- Gill, M., et al. Placental syncytiotrophoblast-derived extracellular vesicles carry active nep (neprilysin) and are increased in preeclampsia. Hypertension. 73 (5), 1112-1119 (2019).
- Kandzija, N., et al. Placental extracellular vesicles express active dipeptidyl peptidase IV; levels are increased in gestational diabetes mellitus. J Extracell Vesicles. 8 (1), 1617000 (2019).
- Motta-Mejia, C., et al. Placental vesicles carry active endothelial nitric oxide synthase and their activity is reduced in preeclampsia. Hypertension. 70 (2), 372-381 (2017).
- Sammar, M., et al. Reduced placental protein 13 (PP13) in placental derived syncytiotrophoblast extracellular vesicles in preeclampsia – A novel tool to study the impaired cargo transmission of the placenta to the maternal organs. Placenta. 66, 17-25 (2018).
- Burton, G. J., Woods, A. W., Jauniaux, E., Kingdom, J. C. Rheological and physiological consequences of conversion of the maternal spiral arteries for uteroplacental blood flow during human pregnancy. Placenta. 30 (6), 473-482 (2009).
- Warrington, J. P., et al. Placental ischemia in pregnant rats impairs cerebral blood flow autoregulation and increases blood-brain barrier permeability. Physiological Reports. 2 (8), e12134-e12134 (2014).
- Warrington, J. P., Drummond, H. A., Granger, J. P., Ryan, M. J. Placental Ischemia-induced increases in brain water content and cerebrovascular permeability: Role of TNFα. Am J Physiol Regul Integr Comp Physiol. 309 (11), R1425-R1431 (2015).
- Johnson, A. C., et al. Magnesium sulfate treatment reverses seizure susceptibility and decreases neuroinflammation in a rat model of severe preeclampsia. PLoS ONE. 9 (11), e113670 (2014).
- Escudero, C. A., et al. Role of extracellular vesicles and microRNAs on dysfunctional angiogenesis during preeclamptic pregnancies. Front Physiol. 7, 1-17 (2016).
- Salomon, C., et al. Placental exosomes as early biomarker of preeclampsia: Potential role of exosomalmicrornas across gestation. J Clin Endocrinol Metab. 102 (9), 3182-3194 (2017).
- Knight, M., Redman, C. W., Linton, E. A., Sargent, I. L. Shedding of syncytiotrophoblast microvilli into the maternal circulation in pre-eclamptic pregnancies. Br J Obstet Gynaecol. 105 (6), 632-640 (1998).
- Gilani, S. I., Weissgerber, T. L., Garovic, V. D., Jayachandran, M. Preeclampsia and extracellular vesicles. Curr Hypertens Rep. 18 (9), 68 (2016).
- Dutta, S., et al. Hypoxia-induced small extracellular vesicle proteins regulate proinflammatory cytokines and systemic blood pressure in pregnant rats. Clin Sci (Lond). 134 (6), 593-607 (2020).
- Leon, J., et al. Disruption of the blood-brain barrier by extracellular vesicles from preeclampsia plasma and hypoxic placentae: attenuation by magnesium sulfate. Hypertension. 78 (5), 1423-1433 (2021).
- Han, C., et al. Placenta-derived extracellular vesicles induce preeclampsia in mouse models. Haematologica. 105 (6), 1686-1694 (2020).
- Amburgey, O. A., Chapman, A. C., May, V., Bernstein, I. M., Cipolla, M. J. Plasma from preeclamptic women increases blood-brain barrier permeability: role of vascular endothelial growth factor signaling. Hypertension. 56 (5), 1003-1008 (2010).
- Cipolla, M. J., et al. Pregnant serum induces neuroinflammation and seizure activity via TNFalpha. Exp Neurol. 234 (2), 398-404 (2012).
- Bergman, L., et al. Preeclampsia and increased permeability over the blood brain barrier – a role of vascular endothelial growth receptor 2. Am J Hypertens. 34 (1), 73-81 (2021).
- Torres-Vergara, P., et al. Dysregulation of vascular endothelial growth factor receptor 2 phosphorylation is associated with disruption of the blood-brain barrier and brain endothelial cell apoptosis induced by plasma from women with preeclampsia. Biochim Biophys Acta Mol Basis Dis. 1868 (9), 166451 (2022).
- Schreurs, M. P., Houston, E. M., May, V., Cipolla, M. J. The adaptation of the blood-brain barrier to vascular endothelial growth factor and placental growth factor during pregnancy. FASEB J. 26 (1), 355-362 (2012).
- Schreurs, M. P., Cipolla, M. J. Cerebrovascular dysfunction and blood-brain barrier permeability induced by oxidized LDL are prevented by apocynin and magnesium sulfate in female rats. J Cardiovasc Pharmacol. 63 (1), 33-39 (2014).
- Schreurs, M. P. H., et al. Increased oxidized low-density lipoprotein causes blood-brain barrier disruption in early-onset preeclampsia through LOX-1. FASEB J. 27 (3), 1254-1263 (2013).
- Escudero, C., et al. Brain vascular dysfunction in mothers and their children exposed to preeclampsia. Hypertension. 80 (2), 242-256 (2023).
- Russell, W. M. S., Burch, R. L. The principles of humane experimental technique. Universities Federation of Animal Welfare. , (1959).
- Miller, R. K., et al. Human placental explants in culture: approaches and assessments. Placenta. 26 (6), 439-448 (2005).
- Troncoso, F. A. J., Herlitz, K., Ruiz, F., Bertoglia, P., Escudero, C. Elevated pro-angiogenic phenotype in feto-placental tissue from gestational diabetes mellitus. Placenta. 36 (4), 2 (2015).
- Zhang, H. C., et al. Microvesicles derived from human umbilical cord mesenchymal stem cells stimulated by hypoxia promote angiogenesis both in vitro and in vivo. Stem Cells Dev. 21 (18), 3289-3297 (2012).
- Thery, C., Amigorena, S., Raposo, G., Clayton, A. Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protoc Cell Biol. Chapter 3 (Unit 3), 22 (2006).
- Carroll, R. W., et al. A rapid murine coma and behavior scale for quantitative assessment of murine cerebral malaria. PLoS One. 5 (10), e13124 (2010).
- Wu, J., et al. Transcardiac perfusion of the mouse for brain tissue dissection and fixation. Bio Protoc. 11 (5), e3988 (2021).
- Walchli, T., et al. Quantitative assessment of angiogenesis, perfused blood vessels and endothelial tip cells in the postnatal mouse brain. Nat Protoc. 10 (1), 53-74 (2015).
- Wang, H. L., Lai, T. W. Optimization of Evans blue quantitation in limited rat tissue samples. Sci Rep. 4, 6588 (2014).
- Morita, K., Sasaki, H., Furuse, M., Tsukita, S. Endothelial claudin: claudin-5/TMVCF constitutes tight junction strands in endothelial cells. J Cell Biol. 147 (1), 185-194 (1999).
- Lara, E., et al. Abnormal cerebral microvascular perfusion and reactivity in female offspring of reduced uterine perfusion pressure (RUPP) mice model. J Cereb Blood Flow Metab. 42 (12), 2318-2332 (2022).
- Chang, X., et al. Exosomes from women with preeclampsia induced vascular dysfunction by delivering sflt (soluble fms-like tyrosine kinase)-1 and seng (soluble endoglin) to endothelial cells. Hypertension. 72, 1381-1390 (2018).
- Smarason, A. K., Sargent, I. L., Starkey, P. M., Redman, C. W. The effect of placental syncytiotrophoblast microvillous membranes from normal and pre-eclamptic women on the growth of endothelial cells in vitro. BJOG. 100 (10), 943-949 (1993).

