Optogenetic Activation of Afferent Pathways in Brain Slices and Modulation of Responses by Volatile Anesthetics
Instructor Prep
concepts
Student Protocol
All procedures involving animals described in this protocol were approved by the University of Wisconsin-Madison School of Medicine and Public Health Animal Care and Use Committee.
1. Breeding mice to express fluorescent reporter protein in interneuron subpopulations
- Pair homozyogous, Cre-dependent tdTomato male mouse with either homozygous SOM-Cre female or homozygous PV-Cre female mouse.
NOTE: Other specific neuronal populations may be targeted by using the appropriate Cre lines. - Allow heterozygous offspring to mature to at least 3 weeks of age before proceeding. For experiments described here, genotyping is not necessary, as homozygous parents produce offspring that are all heterozygous for both cell type-specific Cre recombinase and Cre-dependent reporter alleles.
2. Performing unilateral stereotaxic injection of viral construct
- Adjust settings of the micropipette puller for injection pipettes as indicated in the instrument user manual (see Table 1 for recommended settings). Pull the glass micropipette.
- Break the tip of the sharp end of the pipette such that the tip diameter is approximately 30 µm with minimal taper over several millimeters.
- Using previously documented procedures, decide on the appropriate titer and volume of virus to be injected. In the experiments described here, 1.0 μL (titer: 3.1-5.7 TU/mL) injected unilaterally produced good results.
- Backfill the full volume of the pipette with mineral oil. Load the pipette onto the microsyringe and flow a small amount of mineral oil through the tip to ensure the tip is not clogged.
- Frontfill at least 1.0 μL of viral construct. The recombinant adeno-associated viral vector used in these experiments was AAV2-hSyn-hChR2(H134R)-EYFP.
- Arrange sterile drape in a surgical area. Sterilize tools for stereotaxic procedure and place it on the drape.
- Anesthetize SOM-tdTomato or PV-tdTomato heterozygous animal using isoflurane (3% for induction, 1.5-2% for maintenance) and oxygen mixture. Periodically confirm surgical level of anesthesia with toe pinch throughout surgery. Ensure animal does not move beyond the surgical plan of anesthesia by monitoring respirations every 10-15 min.
- Shave the top of the animal’s head. Apply 70% isopropyl alcohol and iodine-based solution liberally to surgical area and ophthalmic ointment to eye sockets to prevent drying of the membrane. Administer bupivacaine/lidocaine (1:1 ratio, 1.0 mg/kg) subcutaneously to surgical site for local anesthetic.
- Fit the animal into stereotaxic frame.
- Use scalpel to make incision along sagittal axis of skin overlying the dorsal surface of the skull. Retract skin using forceps. Hydrate skull with 0.9% saline as necessary.
- On the surface of the skull, lightly mark the intersection of anterior and lateral coordinates with a cross in pencil. Drill a hole at the appropriate coordinates in the transverse plane (in mm relative to Bregma, for cingulate cortex (Cg) injection: anterior 0.2, lateral 0.3; for posterior thalamus (Po) injection: posterior 2.25, lateral 3.4).
NOTE: Markings should extend beyond the boundaries of the burr hole to provide guidance for accurate placement of pipette. - Turn on and balance the air table.
- Reposition electrode manipulator of the stereotaxic frame at 0° for injections into Cg, or 45° in the coronal plane for injections into Po.
- Attach the syringe pump to the electrode manipulator. Attach the microsyringe pump controller to the syringe pump.
- Navigate pipette tip near (but not touching) the surface of the brain, at the intersection of markings created in Step 2.11. Advance the pipette at approximately 1 mm/s along its longitudinal axis into the brain either 0.9 mm (injection in Cg) or 3.1 mm (injection in Po). Wait for 10 min before proceeding.
- Inject 1.0 µL of viral construct over a period of 10 min (100 nL/min). If welling of virus from pipette insertion site is observed, slow the injection rate to 50 nL/min.
- After injection, wait for 10 min before slowly retracting the injection pipette.
- Suture to close the scalp incision and administer 2-5 mg/kg meloxicam subcutaneously.
- Discontinue isoflurane and monitor animal during emergence from anesthesia. Allow to recover according to procedures described by Institution’s Animal Care and Use Committee, including further administration of analgesics.
3. Preparation of acute brain slices
- Allow at least 3 weeks for the expression of viral construct before harvesting tissue.
- Prepare 1 L of artificial cerebral spinal fluid for slicing procedure (slicing artificial cerebral spinal fluid, sACSF). See Table 2 for ingredients.
- Throughout the slicing procedure, supply sACSF with dissolved 95% O2/5% CO2 mixture, delivered via gas dispersion tube.
- Prepare ice cold bath for vibrating blade microtome. Mount ice-cold specimen stage onto microtome and fix sapphire blade in place for tissue sectioning.
- Anesthetize mouse with 3% isoflurane and oxygen until loss of righting reflex.
- Decapitate the mouse using guillotine and immediately submerge head in 4°C sACSF. To preserve the health of the tissue, complete the following steps as swiftly as possible.
- Open skull cavity by making a small incision at the base of the skull and gently removing each skull plate. Gently remove underlying dura mater.
- While the brain is still in the skull cavity, use the razor blade to remove the cerebellum. Make a second vertical cut along the sagittal plane in the left hemisphere, just lateral to the midline.
- Prepare tissue block for sectioning.
- Gently lift the brain from the skull cavity. Place the brain on the filter paper with the flat, sagittal plane down. Guide the filter paper over the blocking template and align the brain to the underlying template outline (Supplementary Figure 1).
- Make two parallel cuts in the coronal plane as indicated by the lines on the template. Add a small drop of sACSF to keep filter paper wet, if necessary.
- Place the tissue block in 4°C sACSF briefly while step 3.8.4 is conducted.
- Apply a small amount of super glue to the ice-cold specimen stage.
- Lift the tissue block from cold sACSF. Use the corner of an absorbent towel to wick away excess sACSF. Glue the posterior coronal plane of the tissue block to the specimen stage, with the dorsal surface of the brain facing the sapphire blade.
- Collect 500 µm thick coronal brain slices. Place slices of interest on nylon mesh (Supplementary Figure 2) in 34 °C sACSF and allow the container to reach room temperature.
NOTE: For experiments described here, electrophysiological recordings were collected from a coronal section centered approximately 2.25 mm posterior to bregma to study a non-primary sensory area, medial secondary visual cortex (V2MM).
4. Preparation of experimental artificial cerebral spinal fluid (eACSF) bags containing dissolved volatile anesthetic isoflurane
- Prepare 300 mL of a stock mixture of 3.0% isoflurane.
- In a sealed polytetrafluoroethylene gas bag, add ~100 mL of 95% O2/5% CO2 gas mixture to 20-30 mL of liquid isoflurane and a small amount of 0.9% saline. Wait at least for 30 min to allow equilibration of isoflurane between liquid and gas phases.
- Determine the amount of saturated isoflurane gas, Vsat, to add to the stock bag using the following equation:

where P%stock is the target composition of the stock gas (3% in this case), Vstock is the final volume of the stock gas bag, Pisoflurane is the partial pressure of isoflurane at room temperature (~240 mmHg), and Ptotal is the atmospheric pressure (~760 mmHg). - Add the calculated amount of saturated gas to an empty gas bag and fill the bag with a volume of 95% O2/5% CO2 gas mixture to bring the total volume of stock bag to 300 mL.
- Prepare 2 L of artificial cerebral spinal fluid for perfusion of the slice during the experiment (experimental ACSF, eACSF). See Table 2 for ingredients. Dissolve 95% O2/5% CO2 gas mixture into solution.
- Prepare two separate bags of Control and Isoflurane solutions.
- To an empty polytetrafluoroethylene gas bag, add 600 mL eACSF and 600 mL of 95% O2/5% CO2 gas mixture. Label this bag as Control.
- To another empty polytetrafluoroethylene gas bag, add 300 mL of eACSF. Label this bag as Isoflurane.
- Choose a physiologically relevant equilibrated gas phase concentration of isoflurane. Experiments were conducted using gas concentrations equivalent to 1.3% isoflurane. Mice lose righting reflex, and presumably consciousness, at 0.9% inhaled isoflurane.
- Use the following equation to calculate the equivalent gas phase concentration at room temperature, P%(Troom)31:
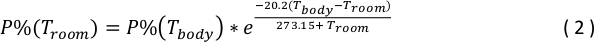
where P%(Tbody) is the physiologically relevant gas phase concentration chosen in Step 4.3.3, Troom is 25 °C, and Tbody is 37 °C. - Use the following equation to determine volume of gas from stock gas bag, Vstock, to add to the Isoflurane solution.
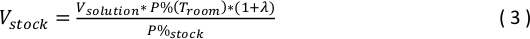
where Vsolution is the volume of eACSF in ISOFLURANE bag (300 mL), P%(Troom) is entered from equation (2), λ is the saline/gas Ostwald partition coefficient of isoflurane (λ = 1.232), and P%stock is the gas phase concentration of the stock gas bag (P%stock = 3.0%). - To the Isoflurane solution bag, add the volume of gas from the stock gas bag, Vstock, calculated in Step 4.3.5.
- To the Isoflurane solution bag, add a volume of 95% O2/5% CO2 gas mixture to bring the total volume of gas in the Isoflurane solution bag to 300 mL.
- Shake both Control and Isoflurane bags on shaker for at least 1 h to allow isoflurane phase equilibration.
- After all data has been collected, the correct concentration may be verified by using an anesthetic gas monitor to measure equilibrated gas concentration of isoflurane above the remaining solution in bag.
- Report experimental concentrations of volatile gases in aqueous units, as millimolar concentrations are more robust to changes in temperature. Use the following equation to convert room temperature gas phase concentration, P%(Troom), to equivalent aqueous concentration (Caqueous, in mM)31:
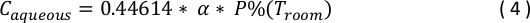
where α is the saline/gas Bunsen partition coefficient for isoflurane at 25°C32.
5. Preparation of hardware and software for multi-channel recordings
- Set up 16-channel data acquisition system according to manufacturer instructions.
NOTE: Several commercially available amplifiers and data acquisition systems can be used to collect multi-channel recordings. In the experiments described here, analog signals are delivered via an electrode reference panel to two amplifiers, where they are amplified (2000x) and filtered (0.1-10kHz). Analog inputs to the data acquisition system are digitized at 40kHz. - Fasten the appropriate 16-channel headstage adaptor to a microscope micromanipulator. Orient the adaptor such that the female connector ports are facing downward.
- Adjust the angle of operation of this micromanipulator such that it is oriented downward toward the recording chamber, at an angle approximately 70° relative to horizontal.
- Connect the headstage input to a 16 x 1 probe for in vitro electrophysiology via the headstage adaptor anchored to the micromanipulator.
- Connect the headstage output connector to the data acquisition system.
- Install appropriate software for data acquisition. Configure 15 input channels to correspond to input signals from the first 15 multi-channel probe contacts. Configure the remaining channel to receive input from the intracellular electrode.
NOTE: Take care to consider electrode and adaptor maps when collecting and analyzing data, to ensure the appropriate signal corresponds to the electrode contact from which it was collected.
6. Configuration of light stimulation protocols
- Set up light delivery system and install the accompanying software.
- Open the software. Choose hardware wiring configuration in which a Trigger Source (Digital/TTL Out) provides Trigger In signal to the light delivery system, and the light delivery system provides Trigger Out signal to a 470 nm LED.
- Mount high-power objective lens. Using digital camera, calibrate high-power objective for use with light delivery system.
- Create new profile sequence of light stimulation profiles.
- Create a pattern of choice. In the experiments described here, a circle of diameter 150 µm is used to allow layer-specific activation of axon terminals.
- To construct a profile sequence, copy and paste this profile for each of any number of trials.
- Create a waveform list that contains waveforms of any light intensity, pulse duration, or pulse number.
- Randomly assign waveforms to each profile. Each profile with its assigned waveform corresponds to one trigger pulse from a Digital TTL input, or one trial.
- Save the profile sequence.
- In the data acquisition software, create a new protocol.
- Set the number of trials to equal the number of profiles in the profile sequence just created.
- Choose signal inputs to match those configured in Step 5.6. Configure a protocol that provides a single digital TTL output, recording from these 16 input channels for an appropriate amount of time before and after the digital trigger.
7. Placing multi-channel probe in ex vivo brain tissue slice
- Perfuse bubbled eACSF (not in sealed bags) at 3-6 mL/min.
- Transfer the brain slice containing area of interest onto mesh grid in microscope perfusion chamber. Anchor with platinum harp (see Supplementary Figure 3).
- Rotate mesh grid such that the line of electrode contacts on the distal end of the multi-channel probe is approximately perpendicular to the pial surface.
- Under broadfield illumination and under fine control of the micromanipulator, lower the multi-channel probe toward the surface of the slice.
- Rotate the filter cube turret to engage the appropriate filter cube for visualization of the fluorescent reporter protein expressed in axon terminals of cortical afferents. If necessary, rotate the slice to more precisely align the probe with the pial surface.
- Position the probe just above the plane of the slice, ~200 µm short of the final target position along the x-axis, leaving at least one channel outside the boundary of the area of tissue being recorded
- Slowly insert the probe into the slice by moving the manipulator along its longitudinal axis. To minimize damage to the tissue, only advance the probe to the extent that the sharp tips are just visible below the tissue surface. This will minimize damage to the tissue while still ensuring the electrode contacts are in contact with the tissue.
8. Patch clamping targeted neurons and obtaining whole-cell configuration
- Switch eACSF source to bagged Control solution.
- Identify fluorescently labeled cell for targeted patch clamp recording.
- Restrict the aperture iris diaphragm to the smallest diameter. Engage a low-power objective lens and bring the tissue into focus.
- Center the light over an area of tissue adjacent to (but not overlapping) the multi-channel probe.
- Engage the high-power (40x or 60x) water immersion objective, using caution to avoid contact between the multi-channel probe and objective lens.
- Rotate the filter cube turret to engage the appropriate filter set to allow imaging of cells expressing Cre-dependent fluorescent marker.
- Identify a fluorescently labeled cell as a target for patch clamp recording. Raise the objective lens to create ample space to lower a patch pipette.
- Load a patch pipette (see Table 1) with internal solution (Table 2) and mount pipette into electrode holder. Using 1 mL syringe, apply positive pressure corresponding to ~0.1mL air.
- Lower patch pipette into the solution. Bring the pipette tip into focus under visual guidance.
- Obtain whole-cell recording from the targeted cell using the steps previously demonstrated33.
- If planning to assess changes to intrinsic properties of the cell (e.g., input resistance, action potential firing rate in response to current steps), conduct these recordings. Otherwise, move to axon stimulation protocol in below.
9. Layer-specific optogenetic activation of axon terminals
- Manipulate field of view in the x-y plane to align light stimulation profile with desired location on slice.
- Load light stimulus protocol and prepare the light delivery system to receive a digital TTL pulse.
- Optogenetically activate axon terminals while simultaneously recording extracellular field potentials and intracellular membrane fluctuations.
- Switch eACSF source to Isoflurane solution and wash drug in for 15 min. If necessary, collect spontaneous recordings during the wash-in.
- Repeat step 9.2-9.3.
- Switch eACSF source to Control solution and wash drug out for 20 min. If necessary, collect spontaneous recordings during wash-out.
- Repeat step 9.2-9.3.
Optogenetic Activation of Afferent Pathways in Brain Slices and Modulation of Responses by Volatile Anesthetics
Learning Objectives
A timeline of steps described in the protocol is shown in Figure 1. Cortical inputs arriving from higher order cortical areas or from non-primary thalamic nuclei have partially overlapping terminal fields in layer 1 of non-primary visual cortex24. To isolate independent thalamocortical or corticocortical afferent pathways, a viral vector containing ChR2 and an eYFP fluorescent reporter into either Po or Cg was injected. Cells within the injection radius take up the viral vector and, after 2-4 weeks, express the non-specific cation channel ChR2 and the reporter in both the soma and projecting axons (Figure 2A). Coronal slices were collected. With the appropriate filter cube engaged, axons expressing the viral construct were imaged (Figure 2B). The use of ChR2 to activate axon terminals allows for activation of afferents without the prerequisite for an attached soma.
The animals used in the experiments described here were SOM-tdTomato or PV-tdTomato hybrid animals, which express the fluorescent reporter protein tdTomato in either somatostatin- (SOM+) or parvalbumin-positive (PV+) interneurons, respectively. SOM+ or PV+ interneurons in layer 2/3 were targeted for patch clamping under visual guidance with the appropriate filter cube engaged (Layer 1C). These interneurons have dendrites in layer 1 and are targets of corticocortical inputs (Figure 3A).
Addition of 125 mL of 3.0% isoflurane gas and 175 mL of 95% O2/5% CO2 to a sealed bag resulted in a pre-equilibrium concentration of gas of 1.3%. Gas dissolved into eACSF according to its partition coefficient; the predicted gas phase equilibrium concentration of isoflurane at room temperature was 0.6% (Figure 2D). This was confirmed via gas monitor.
The tissue slice was transferred to the recording chamber and the 16×1 multi-channel recording probe was placed orthogonally to the cortical laminae (Figure 2E). A 150 μm circle of 470 nm light centered over cortical layer 1 was delivered via the objective light path, while extracellular field potentials were collected using the 16 x 1 multi-channel probe and targeted whole-cell patch clamp recordings were conducted in interneurons. A schematic of the recording set-up is shown in Figure 2F.
Post-synaptic potentials (PSPs) were observed in interneurons in response to a train of four 2 ms pulses of light (10 Hz; Figure 3A). Local field potentials were also recorded (Figure 3B). Current source density (CSD; Figure 3C) and multi-unit activity (MUA; Figure 3D) were extracted from local field potentials. Ten trials at several different light intensities were used to conduct post hoc analyses. The amplitude of current sinks extracted from the CSD increased as a function of light intensity (Figure 4A). A three-parameter nonlinear logistic equation was fit to the data for comparisons across pathways. PSP amplitude also increased with current sink amplitude (Figure 4B).
Synaptic responses to thalamocortical and corticocortical inputs were measured during control, isoflurane (0.28 mM), and recovery conditions. Post-synaptic responses of somatostatin- (Figure 5A) to corticocortical stimuli were suppressed during isoflurane, as were evoked current sinks (Figure 5B).
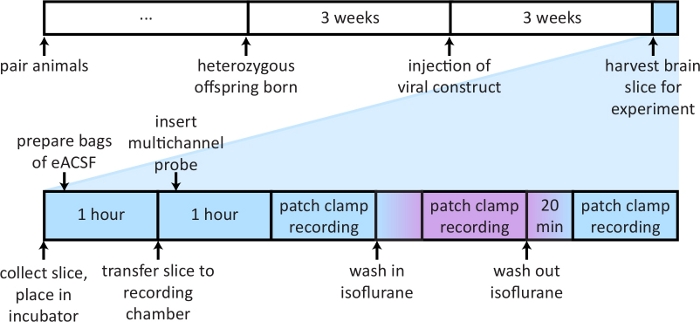
Figure 1: A schematic outlining timeline of important steps in protocol.
Top: Describes timeline of steps necessary for breeding of transgenic animals and expression of viral vector. Bottom: Depicts steps and timeline for preparing materials and conducting experiment on the day of slice preparation. Please click here to view a larger version of this figure.
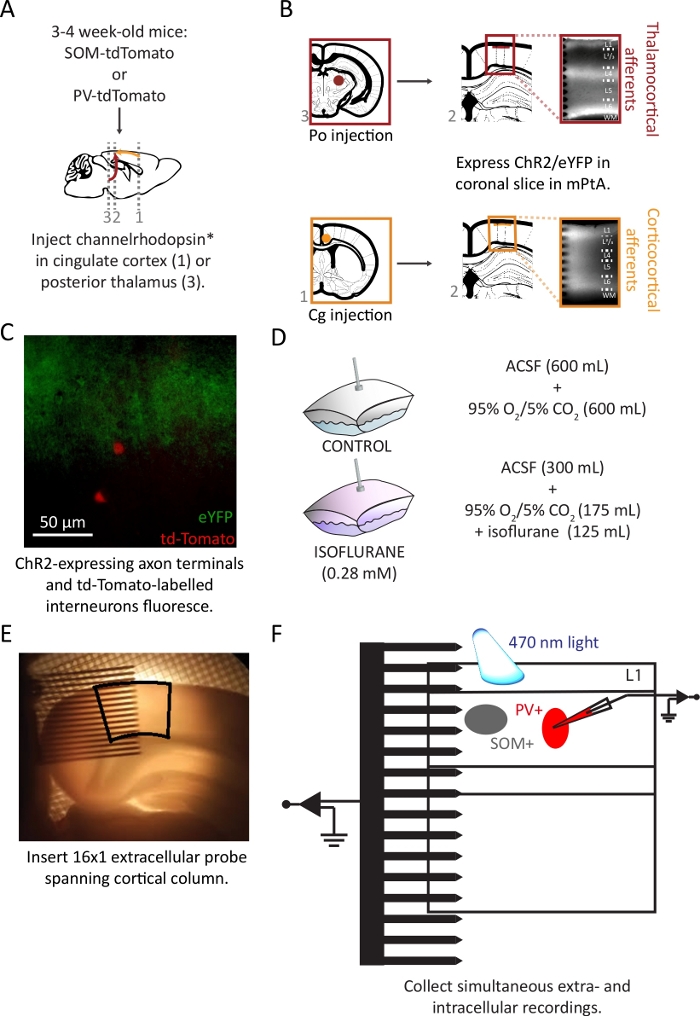
Figure 2: Injection of viral vector and preparation ex vivo coronal brain slices.
(A) Schematic representation of injection of viral vector into SOM-tdTomato or PV-tdTomato hybrid mice. (B) Coronal slices of the medial parietal association area (mPtA) were harvested, and thalamocortical (top) or corticocortical (bottom) afferent fibers were identified by their eYFP reporter in layer 1. This figure is modified with permission from24. (C) Overlay of eYFP-labeled axon terminals in layer 1 (green) and tdTomato-labelled SOM+ interneurons (red) in superficial layer 2/3. (D) Sealed bags were prepared with a 50:50 solution-to-gas mixture. (E) Placement of a 16 x 1 probe into mPtA (black outline). (F) Schematic of the recording set-up in the cortical slice. Please click here to view a larger version of this figure.
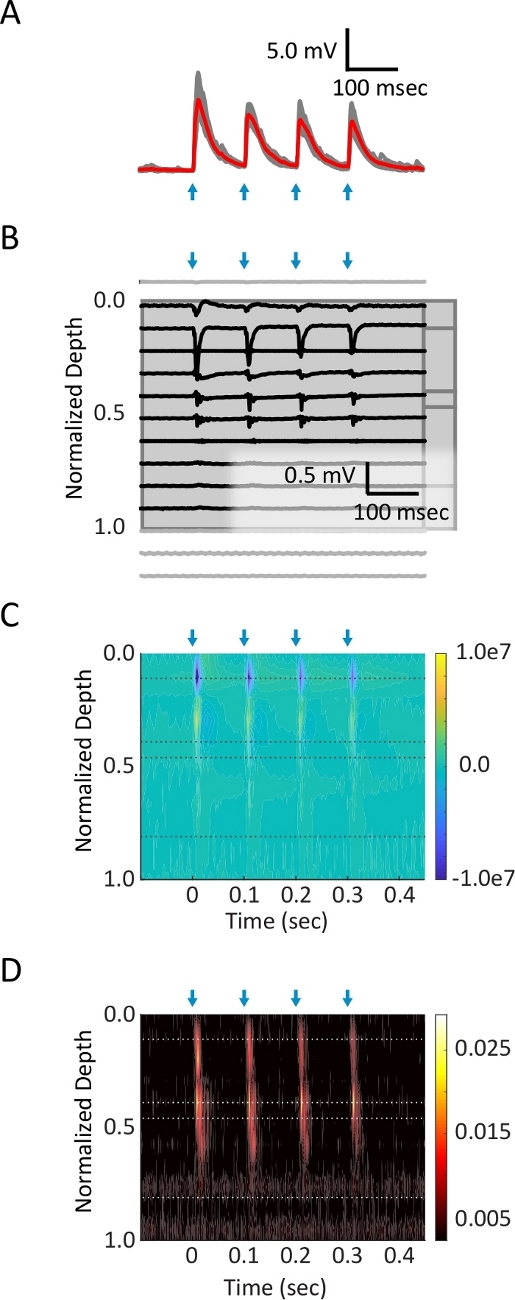
Figure 3: Simultaneous intracellular and multi-channel extracellular recordings in cortical slice.
(A) Whole-cell current clamp patch recording from the soma of a layer 2/3 PV+ interneuron. Four pulses (2 ms each, blue arrows) of blue light (2.2 mW) at 10 Hz were delivered to corticocortical axon terminals in L1. Average (red trace) of ten trials (grey traces) are shown. (B) Raw data from 16 channels of extracellular 16 x 1 probe. Channels placed in cortical tissue are shown in black, and those lying outside of cortex in grey. (C) A current source density diagram, extracted from the local field potential signal, shows synaptic current sinks (blue) in layer 1. (D) Multi-unit activity, generated by applying a high-pass filter to the local field potential signal, isolates spiking activity evoked in lower layers. Please click here to view a larger version of this figure.
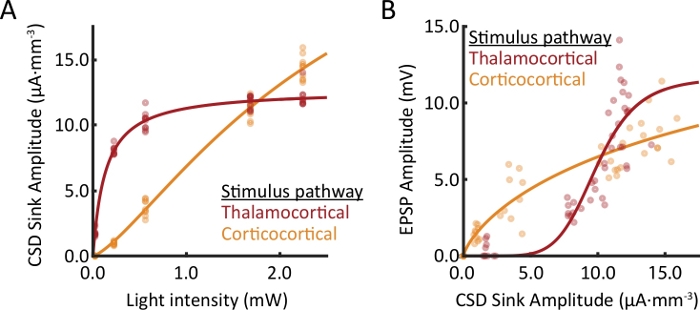
Figure 4: Comparison of responses from recordings in two different slices.
Multiple light intensities were used to evoke synaptic responses in cortical layer 1. For each trial, the peak amplitude of the evoked response was extracted from the layer 1 extracellular current sink and EPSPs in layer 2/3 PV+ interneurons. (A) Extracellular response profiles of thalamocortical and corticocortical afferents are compared as a function of light intensity. (B) The relationship between current sink amplitude and EPSP amplitude is pathway dependent. Within each stimulus pathway, data from (A) and (B) were collected simultaneously. Please click here to view a larger version of this figure.
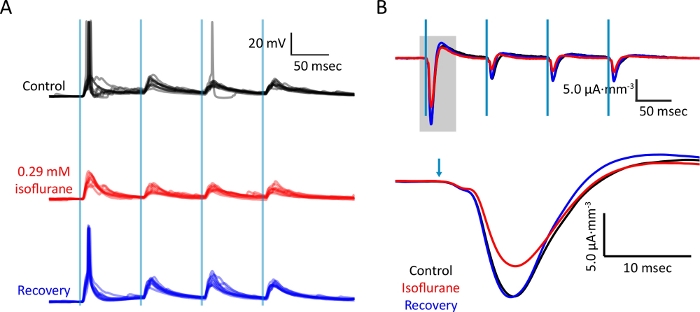
Figure 5: Bath application of isoflurane dissolved in eACSF during simultaneous recordings.
(A) Intracellular whole-cell current clamp recording from layer 2/3 SOM+ interneuron upon activation of corticocortical afferents during control, isoflurane, and wash conditions. Vertical blue lines indicate light stimuli (2 ms; 1.65 mW). (B) Current source density trace extracted from electrode in layer 1. Data were collected simultaneously with those collected in (A). Recovery of responses upon wash demonstrates depression of synaptic responses by isoflurane. Please click here to view a larger version of this figure.
| Micropipette for virus injection | ||||
| Glass | ID: 0.05 mm, OD: 0.11 mm | |||
| Loops | 1 | |||
| Heat | Pull | Vel | Time | Pressure |
| Ramp + 10 | 20 | 40 | 200 | 300 |
| Micropipette for whole-cell patch clamp recordings | ||||
| Glass | ID: 1.1 mm, OD: 1.7 mm | |||
| Loops | 4 | |||
| Heat | Pull | Vel | Time | Pressure |
| Ramp | 0 | 25 | 250 | 500 |
Table 1: Recommended glass and parameters for pulling micropipettes for viral injections and whole-cell patch clamp recordings. Glass used for viral injections and whole-cell patch clamp recordings is described, as well as the parameters for pulling micropipettes using the micropipette puller. Consult instruction manuals for micropipette puller for further recommendations or fine-tuning of settings.
| Slicing ACSF, sACSF (in mM) | Experiment ACSF, eACSF (in mM) | |
| NaCl | 111 | 111 |
| NaHCO3 | 35 | 35 |
| HEPES | 20 | 20 |
| KCl | 1.8 | 1.8 |
| CaCl2 | 1.05 | 2.1 |
| MgSO4 | 2.8 | 1.4 |
| KH2PO4 | 1.2 | 1.2 |
| glucose | 10 | 10 |
| Internal Solution | ||
| K-gluconate | 140 | |
| NaCl | 10 | |
| HEPES | 10 | |
| EGTA | 0.1 | |
| MgATP | 4 | |
| NaGTP | 0.3 | |
| pH = 7.2 |
Table 2: Composition of artificial cerebral spinal fluid and intracellular solution. Reagents and concentrations for sACSF, eACSF, and intracellular pipette solution for patch clamp recordings are listed.
Supplementary Figure 1: Template for preparing block of tissue to collect brain slices. The template is adjusted to the appropriate size, printed, and glued to a microscope slide. A cover slip is glued over the template to prolong its use. The tissue block is placed on a piece of filter paper with the sagittal plane down, aligned to the pink background, and a vertical cut is made in the coronal plane along the black line. Please click here to download this figure.
Supplementary Figure 2: Incubation chamber for harvested brain slices. The chamber is filled with sACSF and bubbled with 95% O2/5% CO2 gas mixture via a bent needle attached to tubing. Incubation platform is made of nylon stretched over a plastic circular fitting. Please click here to download this figure.
Supplementary Figure 3: Platinum structures for slice in recording chamber. Brain slice is transferred to recording chamber via pipette and placed on top of nylon mesh, which is stretched over a horseshoe-shaped piece of flattened platinum wire and super glued in place. Platinum harp is placed over brain slice to anchor it in place during recording. Please click here to download this figure.
Supplementary Table 1: Ostwald (λ) and Bunsen (α) coefficients for other volatile anesthetics. Adapt this protocol for study of other volatile gas anesthetics, such as halothane, sevoflurane, or desflurane. Substitute the equations described in the protocol with the appropriate coefficients as listed in this table. Please click here to download this table.
List of Materials
| 2.5x broadfield objective lens | Olympus | MPLFLN2.5X | |
| 40x water immersion objective lens | Olympus | LUMPLFLN40XW | |
| 95% O2/5% CO2 mixture | Airgas | Z02OX95R2003045 | |
| A16 probe | NeuroNexus | A16x1-2mm-100-177-A16 | 16-channel probe |
| AAV2-hSyn-hChR2(H134R)-EYFP | Karl Deisseroth Lab, UNC Vector Core | ||
| Anesthetic gas monitor (POET II) | Criticare | 602-3A | |
| ATP, Magnesium Salt | Sigma Aldrich | A9187 | intracellular solution |
| B6.Cg-Gt(ROSA)26Sortm14(CAG-tdTomato)Hze/J | The Jackson Laboratory | 007914 | Cre-dependent tdTomato mouse |
| B6;129P2-Pvalbtm1(cre)Arbr/J | The Jackson Laboratory | 008069 | PV-Cre mouse |
| Belly Dancer Shaker | Thomas Scientific | 1210H86-TS | for equilibration of sealed gas bags |
| Betadine solution | Generic brand | ||
| Bleach | Generic brand | for silver chloriding patch clamp electrode | |
| Bupivicaine | |||
| Calcium Chloride (CaCl2) | Dot Scientific | DSC20010 | ACSF |
| Capillary glass (patch clamp recordings) | King Precision Glass, Inc. | KG-33 | Borosilicate, ID: 1.1mm, OD: 1.7mm, Length: 90.0mm |
| Capillary glass (viral injections) | Drummond Scientific Company | 3-000-203-G/X | 3.5" |
| Control of junior micromanipulator | Luigs and Neumann | SM8 | for control of junior micromanipulator |
| Control of manipulators and shifting table | Luigs and Neumann | SM7 | for control of multichannel electrode and shifting table |
| Digidata 1440A + Clampex 10 | Molecular Devices | 1440A | Digitizer and software |
| E-3603 tubing | Fisher Scientific | 14171208 | for delivery of 95% O2/5% CO2 gas mixture to incubation chamber + application of pressure during patch clamping |
| EGTA | Dot Scientific | DSE57060 | intracellular solution |
| ERP-27 EEG Reference/Patch Panel | Neuralynx | Retired | |
| Filling needle | World Precision Instruments | 50821912 | for filling patch clamp pipettes |
| Filter cube for imaging EYFP | Olympus | U-MRFPHQ | |
| Filter paper | Fisher Scientific | 09801E | lay over slice template during preparation of tissue block |
| Flaming/Brown micropipette puller | Sutter Instrument | P-1000 | 2.5×2.5 Box filament |
| Gas dispersion tube | Sigma Aldrich | CLS3953312C | |
| Glass syringe (100 mL) | Sigma Aldrich | Z314390 | for filling gas-sealed bags |
| Gluconic Acid, Potassium Salt (K-gluconate) | Dot Scientific | DSG37020 | intracellular solution |
| Glucose | Dot Scientific | DSG32040 | ACSF |
| GTP, Sodium Salt | Sigma Aldrich | G8877 | intracellular solution |
| Headstage-probe adaptor | NeuroNexus | A16-OM16 | adaptor to connect 16-channel probe to headstage input |
| Hemostatic Forceps | VWR International | 76192-096 | |
| HEPES | Dot Scientific | DSH75030 | ACSF,intracellular solution |
| HS-16 Headstage | Neuralynx | Retired | |
| Isoflurane | Patterson Veterinary | 07-893-1389 | |
| Isopropyl alcohol (70%) | VWR International | 101223-746 | |
| Junior micromanipulator | Luigs and Neumann | 210-100 000 0090-R | for manipulation of patch clamp electrode |
| LED Light Source Control Module | Mightex | BLS-PL02_US | optogenetic light source control |
| Lidocaine | |||
| Lynx-8 Amplifier | Neuralynx | Retired | |
| Lynx-8 Power Supply | Neuralynx | Retired | |
| Magnesium Sulfate (MgSO4) | Dot Scientific | DSM24300 | ACSF |
| mCherry, Texas Red filter cube | Chroma | 49008 | for imaging tdTomato fluorescent reporter |
| Meloxicam | |||
| Micropipette holder | Fisher Scientific | NC9044962 | |
| Microsyringe pump | World Precision Instruments | UMP3-4 | |
| Mineral oil | Generic brand | ||
| MultiClamp 700A | Molecular Devices/Axon Instruments | 700A | Amplifier |
| Nitrogen (for air table) | Airgas | NI200 | |
| Nylon mesh | Fisher Scientific | 501460083 | stretched over horseshoe of flattened platinum wire, slice rest on top of this during recordings |
| Nylon, cut from pantyhose | Generic brand | small piece to create slice platform in incubation chamber, single fibers to create platinum harp | |
| Ophthalmic ointment | Fisher Scientific | NC1697520 | |
| Pipette | Dot Scientific | 307 | For transferring tissue to rig |
| Platinum wire | VWR International | BT124000 | 2 cm, flattened, to make platinum harp |
| Polygon400 | Mightex | DSI-E-0470-0617-000 | optogenetic light delivery system, comes with PolyScan2 software |
| Potassium Chloride (KCl) | Dot Scientific | DSP41000 | ACSF |
| Potassium Phosphate (KH2PO4) | Dot Scientific | DSP41200 | ACSF |
| Razor blade | Fisher Scientific | 12-640 | |
| Sapphire blade (for vibratome) | VWR International | 100492-502 | |
| Scalpel blade | Santa Cruz Biotechnology, Inc. | sc-361445 | |
| Sealed gas bag | Fisher Scientific | 109236 | |
| Shifting table for microscope | Luigs and Neumann | 380FMU | |
| Sodium Bicarbonate (HCO3-) | Dot Scientific | DSS22060 | ACSF |
| Sodium Chloride (NaCl) | Dot Scientific | DSS23020 | ACSF, intracellular solution |
| Ssttm2.1(cre)Zjh/J (SOM-IRES-Cre) | The Jackson Laboratory | 013044 | SOM-Cre mouse |
| Stereotaxic instrument | Kopf | Model 902 | Dual Small Animal |
| Super glue | Staples | 886833 | to fix tissue block to specimen stage during slice preparation |
| Surgical drill | RAM Products Inc. | DIGITALMICROTORQUE | Microtorque II |
| Syringe (1 mL) with LuerLock tip | Fisher Scientific | 309628 | for application of pressure during patch clamping |
| Syringe (1 mL) with slip tip | WW Grainger, Inc. | 19G384 | for filling patch clamp pipettes |
| Syringe Filters | VWR International | 66064-414 | |
| Upright microscope | Olympus | BX51 | |
| Vibrating microtome | Leica Biosystems | VT1000S | |
| Wypall towels | Fisher Scientific | 19-042-427 |
Lab Prep
Anesthetics influence consciousness in part via their actions on thalamocortical circuits. However, the extent to which volatile anesthetics affect distinct cellular and network components of these circuits remains unclear. Ex vivo brain slices provide a means by which investigators may probe discrete components of complex networks and disentangle potential mechanisms underlying the effects of volatile anesthetics on evoked responses. To isolate potential cell type- and pathway-specific drug effects in brain slices, investigators must be able to independently activate afferent fiber pathways, identify non-overlapping populations of cells, and apply volatile anesthetics to the tissue in aqueous solution. In this protocol, methods to measure optogenetically-evoked responses to two independent afferent pathways to neocortex in ex vivo brain slices are described. Extracellular responses are recorded to assay network activity and targeted whole-cell patch clamp recordings are conducted in somatostatin- and parvalbumin-positive interneurons. Delivery of physiologically relevant concentrations of isoflurane via artificial cerebral spinal fluid to modulate cellular and network responses is described.
Anesthetics influence consciousness in part via their actions on thalamocortical circuits. However, the extent to which volatile anesthetics affect distinct cellular and network components of these circuits remains unclear. Ex vivo brain slices provide a means by which investigators may probe discrete components of complex networks and disentangle potential mechanisms underlying the effects of volatile anesthetics on evoked responses. To isolate potential cell type- and pathway-specific drug effects in brain slices, investigators must be able to independently activate afferent fiber pathways, identify non-overlapping populations of cells, and apply volatile anesthetics to the tissue in aqueous solution. In this protocol, methods to measure optogenetically-evoked responses to two independent afferent pathways to neocortex in ex vivo brain slices are described. Extracellular responses are recorded to assay network activity and targeted whole-cell patch clamp recordings are conducted in somatostatin- and parvalbumin-positive interneurons. Delivery of physiologically relevant concentrations of isoflurane via artificial cerebral spinal fluid to modulate cellular and network responses is described.
Procedure
Anesthetics influence consciousness in part via their actions on thalamocortical circuits. However, the extent to which volatile anesthetics affect distinct cellular and network components of these circuits remains unclear. Ex vivo brain slices provide a means by which investigators may probe discrete components of complex networks and disentangle potential mechanisms underlying the effects of volatile anesthetics on evoked responses. To isolate potential cell type- and pathway-specific drug effects in brain slices, investigators must be able to independently activate afferent fiber pathways, identify non-overlapping populations of cells, and apply volatile anesthetics to the tissue in aqueous solution. In this protocol, methods to measure optogenetically-evoked responses to two independent afferent pathways to neocortex in ex vivo brain slices are described. Extracellular responses are recorded to assay network activity and targeted whole-cell patch clamp recordings are conducted in somatostatin- and parvalbumin-positive interneurons. Delivery of physiologically relevant concentrations of isoflurane via artificial cerebral spinal fluid to modulate cellular and network responses is described.
