Author Spotlight: Evaluation of Entomopathogenic Fungi in Wild Monochamus alternatus Populations for Biocontrol Applications in Forest Wood Borers
Summary
Here we present a protocol for obtaining entomopathogenic fungi from a forest wood borer and a substitutive way to evaluate their entomopathogenic activities using a Coleopteran model insect. This method is efficient and convenient for exploring entomopathogenic fungal resources from wood-boring insect pests in natural forests.
Abstract
Forest wood borers (FWB) cause severe tree damage and economic losses worldwide. The release of entomopathogenic fungi (EPF) during the FWB emergence period is considered an acceptable alternative to chemical control. However, EPF resources have been significantly less explored for FWBs, in contrast to agricultural insect pests. This paper presents a protocol for exploring EPF resources from FWBs using wild Monochamus alternatus populations as an example. In this protocol, the assignment of traps baited with M. alternatus attractants to different populations guaranteed the collection of adequate samples with natural infection symptoms, during the emergence periods of the beetle. Following finely dissecting integuments and placing them onto a selective medium, fungal species were isolated from each part of beetle bodies and identified based on both molecular and morphological traits.
Several fungal species were certified as parasitic EPFs via re-infection of healthy M. alternatus with spore suspensions. Their behavioral phenotypes on M. alternatus were observed using scanning electron microscopy and further compared with those on the Coleopteran model insect Tribolium castaneum. For EPFs that present consistent parasitism phenotypes on both beetle species, evaluation of their activities on T. castaneum provided valuable information on lethality for future study on M. alternatus. This protocol helped the discovery of EPF newly reported on M. alternatus populations in China, which could be applied as an efficient approach to explore more EPF resources from other FWBs.
Introduction
The devastation caused by insect pests has led to great ecological and economic losses in both forest and agricultural ecosystems. Most agricultural pests expose themselves to natural enemies or artificial control agents while damaging host plants. Instead, forest wood borers (FWB) nearly complete their whole developmental cycles inside host tree trunks1, which raises large challenges to explore efficient biocontrol organisms from FWB in the wild field. What is even worse is that FWBs carry a great number of phytopathogens2 or have an intimate relationship with these pathogens as their potential vectors3,4, dramatically amplifying the negative effects of FWB on forest health. Excessive use of chemical insecticides can alleviate FWB severity, but the emergence of insecticidal resistance5,6 limits their environmental application. In certain cases, insect parasitoids, predatory arthropods as well as entomopathogenic microbes were released as biocontrol agents to the distribution areas of FWB7 and were proven to be efficient and economically acceptable alternatives to chemical control8,9,10.
Entomopathogenic fungi (EPF) are regarded to have the advantage in controlling FWB over most other microbial groups. Their spores can be carried by insect hosts and stably fixed on body surfaces via penetration into the cuticle or integument8,11. EPF also present excellent adaptability to environmental stresses and some species colonize well in the tissue of trees as endophytes12,13, facilitating their growth, survival, and transmission. However, compared to that in agricultural industries, the species diversity of EPF used in natural forest ecosystems is remarkably restricted14,15,16. Beauveria bassiana (strain PPRI 5339) was evidenced as the most promising strain to promote an IPM program to Eucalyptus weevils in South Africa17 and the combination of two promising isolates of B. bassiana provided an opportunity for the practical microbial control of red palm weevil, Rhynchophorus ferrugineus, at different life stages in palm tree fields18. In addition to Beauveria and the well-known Metarhizium, other EPF genera of the order Hypocreales, especially species of Lecanicillium (many of which are now classified into the genus Akanthomyces19,20), showed strong pathogenicity and high potential in management of forest pests, such as the Cypress aphid in Chile21.
The pine sawyer beetle Monochamus alternatus is a notorious pine forest pest in China and neighboring countries, which burrows into branches and trunks of pine trees to impede the transportation of nutrients and water22,23,24. Moreover, M. alternatus also promotes the invasion of the plant-parasitic pine wood nematode (Bursaphelenchus xylophilus, PWN) as its main vector beetle. Another congeneric species of the beetle, M. galloprovincialis, has spread PWN in several countries in Europe in recent years25. Previous research reported several genera of natural EPFs from Monochamus spp., such as Beauveria, Metarhizium, and Lecanicillium (Verticillium, an even former name of Lecanicillium), in Spain, Japan, and the Anhui/Zhejiang Provinces of China26,27,28,29. Nevertheless, these collections of EPFs seem to be commonly restricted in a certain location, compared to the wide occurrence of Monochamus beetles in natural fields. As the M. alternatus beetle has a wide geographical distribution in China, it could be regarded as a representative wood borer to explore more potential EPFs across different populations.
In the present protocol, we introduce a specific procedure exploring EPFs from several geographical populations of M. alternatus in southern China. This protocol uses a model Coleopteran beetle as a substitute to perform entomopathogenicity assays, under the condition that the tested fungal species has a consistent behavioral phenotype on both beetle species. This protocol can also provide insights into EPF exploration for other forest wood borers, in which the diversity of their entomopathogenic fungal species is underestimated or less investigated.
Protocol
1. Isolation of fungi from M. alternatus (Figure 1)
- Collect the beetle sample
- Collect the pines sawyer beetles M. alternatus using commercial traps (see Table of Materials) baited with attractants before the predicted emergence periods of the beetle populations in naturally infected pine forests.
NOTE: In this study, beetles were collected from five geographical regions of southern China (Huzhou/Zhejiang, Liangshan/Sichuan, Xiamen/Fujian, Shaoguan/Guangdong, Yulin/Guangxi). The trap setting from top to bottom is as follows: a round top (50 cm in diameter), a cross-shaped panel (35 cm in width and 66 cm in length), a funnel (35.5 cm in top round diameter and 5.5 cm in bottom round diameter), a collection cup (10.5 cm in diameter and 26.5 cm in length). The host volatiles (e.g., ethanol and α-pinene) and the aggregation pheromone (2-undecyloxy-1-ethanol) are used as the bait30. - Label the specimen before transferring it to the laboratory. Put each beetle in individual sterilized tubes with fresh twigs. Replace twigs with fresh ones every 2 days.
- Rear the alive beetles at 25 ± 1 °C in a non-humidified incubator (16-8 L/D cycles) and observe them daily.
- Record samples showing decreased feeding and mobility. Transfer dead M. alternatus that become hardened and stiff to wet chambers to observe the growth of fungal mycelia and conidia.
NOTE: Once infected by entomopathogenic fungi, the beetles displayed a decrease in feeding and mobility at the initial stage of infection31,32. After death, their bodies became hardened and stiff, with mycelia and conidia appearing several days after storage in a wet chamber33. - Store beetle cadavers with fungal infections at 4 °C for future use. To follow this protocol, process the samples immediately within a few hours to avoid contamination with more saprotrophic fungi.
- Collect the pines sawyer beetles M. alternatus using commercial traps (see Table of Materials) baited with attractants before the predicted emergence periods of the beetle populations in naturally infected pine forests.
- Preparation of growth medium
- Add 39 g of potato dextrose agar (PDA) powder into 1 L of pure water and autoclave it for 30 min at 121 °C.
- Cool to an operable temperature (~50 °C), add 0.05 g of streptomycin, 0.05 g of penicillin G, and 0.05 g of tetracycline, and shake to distribute the medium.
- Plate the medium into Petri dishes under a clean bench and use after solidification.
NOTE: The PDA medium with antibiotics is used for isolation from beetles, which prevents the growth of bacteria without affecting fungi growth during isolation. However, the PDA medium without antibiotics is used for daily cultivation and conservation. - Put 35 g of potato dextrose broth (PDB) powder into 1 L of ddH2O and autoclave it for 30 min at 121 °C. Cool it for use.
- Dissection of the beetle sample with fungal infection symptoms
- Prepare sterilized scissors, scalpels, and insect pins; then, work under a clean bench with an alcohol burner.
NOTE: The use of dissection tools can be adjusted according to personal preferences. Insect pins are sharper than standard dissecting needles, and different specifications can meet the need for dissection. - Dissect the beetle body integuments with tools in sterile and single-use Petri dishes. Take care to avoid possible damage to the midgut and hindgut tissues that may exude internal contents.
- Divide the beetle bodies into the main positions as follows: antennae, head, thorax, abdomen, wings (each pair), and legs.
- Prepare sterilized scissors, scalpels, and insect pins; then, work under a clean bench with an alcohol burner.
- Fungi purification
- Cut the integuments of each position into small pieces with scissors. Gently press the outer surface onto the surface of PDA plates with antibiotics.
NOTE: Make sure that fungal mycelia on the integuments have been inoculated to the plate. - Seal with parafilm carefully, and culture in an incubator at 25 ± 1 °C until the tissues are thoroughly covered by mycelium.
- Transfer single colonies according to phenotypic properties (color, shape) onto a new PDA with antibiotics for culture at 25 ± 1 °C.
- Repeat step 1.4.3 twice or three times on PDA with antibiotics until pure colonies are isolated separately.
NOTE: If multiple fungal strains grow in the original plates, pick out the mycelium precisely to facilitate the sorting of more fungal species.
- Cut the integuments of each position into small pieces with scissors. Gently press the outer surface onto the surface of PDA plates with antibiotics.
- Storage of the fungal isolates
- Transfer agar blocks (~5 mm in diameter) from colonies to new PDA plates.
- Culture in an incubator at 25 ± 1 °C for 1-2 weeks.
- Place in a 4 °C refrigerator for daily storage for further use.
- Inoculate fungal strains into 50 mL of sterile PDB in a 250 mL flask, shaking at 180 rpm/min at 25 °C for 5-7 days.
- Harvest 1 mL of the freshly grown fungal mycelium and suspend it in 1 mL of sterilized 20% glycerol in 2 mL sterile freezer tubes (the final concentration of glycerol is 10%).
- Seal the tubes with parafilm and precool them at -20 °C and -40 °C. Then, maintain the tubes at -80 °C as stocks.
2. Molecular and morphological identification of fungal isolates
- Extraction of fungal genomic DNA
- Culture fungi in PDB as described in step 1.5.4.
- Harvest the mycelium and filter it to separate it from the PDB. Homogenize in liquid nitrogen using a precooled mortar and pestle.
- Extract the genomic DNA using the Fungi Genomic DNA Isolation Kit according to the manufacturer's instructions (see Table of Materials).
- PCR amplification and DNA sequencing
- Prepare the PCR reaction mixture as follows: 25 µL of Taq DNA polymerase, 1 µL of template, 2 µL of forward and reverse primers, and ddH2O to a total volume of 50 µL.
- Amplify the rDNA-ITS region of the DNA sample using primer pairs ITS1 and ITS434 (see Table 1) with the following procedure: initial denaturing at 95 °C for 4 min, followed by 35 cycles of denaturing at 94 °C for 60 s, annealing at 58 °C for 60 s, elongation at 72 °C for 2 min, and a final elongation at 72 °C for 10 min.
- Electrophoresis of amplified products using 1.5% agarose gel at 100 V, 100 mA.
- Sequence the PCR.
- Align the sequence using Clustal X2.0 and MEGA 6.0.
NOTE: During sequence alignment, sites with ambiguous alignment are excluded, and gaps are treated as missing data. - Compare the obtained sequence with those in the ITS sequence database in GenBank using BLAST on the National Center for Biotechnology Information (NCBI) website. Identify the fungal strain species information preliminarily.
- Morphological Identification
- Use a camera to capture the mature fungal pure colony morphology on both the front and reverse sides of the PDA plates.
- Pluck conidia from the pure culture fungal colonies with an inoculation needle and transfer them to a glass slide with a drop of sterile water.
- Hold the cover slip and slowly put it down at an angle of ~45°, so that the cover slip can cover the specimen without bubbles.
- Cut a 5 mm2 agar block with a scalpel from colonies at the edge of the fungal colony, and transfer it to a clean glass slide with a drop of sterile water. Carefully tease apart fungi with a fine needle to see specific structures.
- Repeat step 2.3.3.
- Observe the asexual morph of the fungi under an optical microscope (OM), including the shape, transparency, and posture of the hyphae, conidiophores, phialides, and conidia. Record and measure the shape and size of the asexual characteristics to distinguish the different fungal isolates from each other.
NOTE: To maximize the ability to observe fungal structures, use stains and mounting medium35.
3. Induction of the fungal infection symptoms on M. alternatus to observe their behavioral phenotypes
- Preparation of conidial suspension
- Prepare 0.01% Tween-80 with pure water and autoclave it for 25 min at 121 °C. Use after cooling.
- Add an appropriate amount of sterile small glass beads and 0.01% Tween-80 solution to a 2 mL centrifuge tube.
- Scrape fungal colonies into the tube and shake it sufficiently with a vortex shaker until the colony is broken up. Filter through two layers of gauze to remove the mycelium.
NOTE: Shaking is done to ensure the suspension of conidiospore in 0.01% Tween-80 solution. - Count the number of spores under the OM with a hemocytometer, adjust to 1 × 108 conidial/mL with sterile 0.01% Tween-80, and keep at 4 °C for use.
- Preparation of beetles
- Autoclave the pine twigs (about the same size) and dry in the oven (~ 60 °C).
- Keep field-collected M. alternatus adults with pine twigs in an incubator at 25 ± 1 °C.
- Starve beetles for 24 h and sterilize the beetles' surface with bleach, ethanol, and distilled water [10:10:80 (v:v)] before use.
- Induction of the fungal infection
- Work under a clean bench, immerse the pine twigs into conidial suspension for 10 s, and dry them in the air.
- Dip beetles in conidial suspension for 10 s, then transfer to 50 mL sterile tubes (one beetle and one twig per tube). Replace the twigs with new ones every 2 days.
- For negative control, change the conidial suspension into only sterile 0.01% Tween-80 solution. Make five replicates for each fungal strain and control group.
- Assess the activity of beetles based on the amount of frass produced on pine twigs. When feeding ceases, consider them dead.
- Transfer the dead beetles into new sterile 50 mL tubes and place a piece of sterile moist cotton. Incubate at 25 ± 1 °C.
NOTE: The wet cotton is used to maintain humidity, as the growth of fungi requires appropriate moisture, but not too much. - Observe and photograph the change of beetle surface at the same time every day.
- Re-isolation and confirm fungal species
- Until clear fungal infection phenotypes appear on the surface of beetles, pick mycelium from different tissues individually into new PDA plates.
- Culture at 25 ± 1 °C and observe to ensure the morphology is consistent with that of the inoculated fungi.
- Treatment of model beetle
- Repeat steps 3.2.1-3.3.6 on the model beetle Tribolium castaneum, using wheat bran as food, and sterilize wheat bran with UV light.
- Touch beetles lightly with sterile tweezers. Assume they are dead if there is no response.
4. Confirm the infection phenotypes of fungi on M. alternatus and model beetle
- Sample preparation for observation
- Dissect the infected M. alternatus cadavers carefully as in step 1.3.
- Pick T. castaneum bodies with obvious symptoms of fungal infection.
- Cut 5 mm disc plugs of four mature fungal colonies growing on PDA.
- Sample pretreatment
- Put plugs, infected beetle tissues, and bodies into prechilled 2.5% glutaraldehyde at 4 °C for 2 days.
- Wash samples thrice in 0.1% phosphate buffer (pH 7.2-7.4) for 5 min and dehydrate in 30%, 50%, 70%, 80%, 90%, 95%, 100% ethanol for 10 min.
- Dry the samples in a vacuum freeze dryer.
NOTE: The entire pretreatment process should be careful, including cutting, soaking, dehydration, washing, and drying, to prevent sample damage.
- Scanning electron microscopy (SEM) observation
- Coat the pretreated samples with platinum using a sputter coater, and observe under a scanning electron microscope36.
- Record the morphological characteristics of hyphae and conidia growing on PDA, M. alternatus tissues, and model beetle bodies.
- Compare if the results are consistent with those under OM in step 2.3.
5. Evaluation of entomopathogenic activity
- Preparation of conidial suspension
- The preparation of conidial suspension is the same as in steps 3.1.1-3.1.4.
- Pretreatment of model beetle
- Culture, feed, sterilize, and starve T. castaneum as in step 3.5.1.
- Sterilize wheat bran by UV, and place equal weight to sterile Petri dishes.
- Entomopathogenic activity test
- Treat wheat bran with the conidial suspension and dry them in the air under a clean bench at room temperature.
- Immerse T. castaneum beetles in the conidial suspension for 10 s and transfer them to a Petri dish (n = 20 in each Petri dish). Replace new wheat bran with the same conidial treatment every 4 days.
- For negative control, apply only sterile 0.01% Tween-80 solution.
NOTE: Set aside at least three replicates for each treatment group and the control. - Count the dead beetles daily.
NOTE: Touch beetles lightly with sterile tweezers. Assume they are dead if there is a lack of response.
- Multi-gene phylogenetic analysis
- Extract entomopathogenic fungal genomic DNA as described in step 2.1.
NOTE: Multi-gene identification is conducted for the parasitic EPFs that show significant infection phenotypes and strong entomopathogenic activities against the model beetle. - Amplify the following genes, including a region spanning the nuclear ribosomal SSU34 gene, a segment of the large subunit rRNA gene (LSU37,38), part of the elongation factor 1-alpha (tef-1α39) gene, the second largest subunit sequences of RNA polymerase ІІ (rpb240), and part of the β-tubulin41 gene, using the primer pairs NS1/NS4, LR7/LROR, EF-983F/EF-2218R, RPB2-5'F/RPB2-5'R, and TUB1/TUB22 (see Table 1), respectively.
- Prepare the PCR reaction mixture as in step 2.2.1.
- Carry out amplification as follows: initial denaturing at 95 °C for 4 min, followed by 35 cycles (tef-1α, rpb2, SSU), 38 cycles (LSU) or 39 cycles (β-tubulin) of denaturing at 94 °C for 60 s, annealing at 47 °C for 60 s (LSU), 55 °C for 60 s (tef-1α), 54 °C for 40 s (β-tubulin), and 50 °C for 30 s (rpb2, SSU), elongation at 72 °C for 1 min, and a final elongation at 72 °C for 10 min.
- Sequence and align following the same steps as shown in steps 2.2.4-2.2.5.
- Perform the phylogenetic analysis by MEGA 6.0 based on the Maximum Likelihood (ML) method42.
- Extract entomopathogenic fungal genomic DNA as described in step 2.1.
Representative Results
Isolation and identification of fungal isolates from M. alternatus
With the aid of attractant traps, a large number (approximately 500 beetles in total) of M. alternatus were collected from five geographical regions. Beetle cadavers with typical symptoms of infection by entomopathogenic fungi were picked; then, body integuments of every beetle were dissected into several positions as described in protocol step 1.3. As a result, more than 600 fungal isolates were isolated from different body positions. This allows richer fungal candidates for screening pathogens against pine sawyer beetles. Sequencing of the ITS region preliminarily revealed that these isolates can be categorized into 15 fungal genera and 39 species. In addition, there were significant differences in the fungal community composition among geographical populations of M. alternatus adults. The fungal species, Aspergillus austwickii, Akanthomyces attenuatus (syn. Lecanicillium attenuatum), Penicillium citrinum, Scopulariopsis alboflavescens were the dominant species in Sichuan, Zhejiang, Guangdong, Fujian populations, respectively. Under OM and SEM, the asexual reproductive morphology of these four fungi grown on PDA plates was identified based on the macroscopic characteristics, including the color, texture, and sizes of colonies, and microscopic characteristics, including shape, size of conidia, conidiophores, sporangia and hyphae (Figure 2). These observations can be photographed for measuring the shape, size and arrangement of conidia, hyphae, and conidiophores.
The parasitic fungal infection phenotypes on M. alternatus and T. castaneum
Through the induction method (step 3.3), four region-representative fungal species from different geographical populations were confirmed to function in parasitic or non-parasitic modes. As a result, there was no visible infection phenotypes displayed on M. alternatus by P. citrinum, neither on T. castaneum. Conversely, significant infection symptoms were appeared both on M. alternatus (Figure 3) and T. castaneum body surface after infection by A. austwickii, A. attenuatus (syn. L.attenuatum), and S. alboflavescens. Evidence from SEM also well confirmed the consistency of the morphological characteristics of the parasitic fungi on both beetle surfaces. Furthermore, the process of fungal infection on beetles was clearly observed, in which mycelium carrying conidiophores penetrated from the inside of the beetle to the body surface (Figure 4), indicating strong parasitic abilities of these fungi. The above results indicated that the parasitism of these fungi was consistent for both M. alternatus and T. castaneum. In addition, those pictures and data also suggested the spatial preference and localized niches of three fungi on host beetle bodies.
The entomopathogenic activity of parasitic fungal species
T. castaneum has been regarded as a general model organism of Coleopteran species widely, combined with the results above, it has been used as a substitute to assess entomopathogenic activity in this study. The behavioral phenotypes of the three parasitic fungi against M. alternatus and T. castaneum were similar, so the lethal effect on the model beetle would provide valuable information on the entomopathogenic activity against M. alternatus. In a 9-day pilot assay, respectively inoculated with conidial suspension of parasite fungi (A. austwickii, A. attenuatus (syn. L. attenuatum), S. alboflavescens), three groups performed significantly higher mortality on T. castaneum adults than those treated with Tween-80 (control group), and differences were also found between the survival of three groups (Figure 5). These results suggested parasitic fungal species had varying degrees of pathogenicity to model beetles, revealing that these fungi can also exhibit different levels of entomopathogenic activity to M. alternatus adults. Thus, we provide new candidates for the repository of pathogenic fungi.
Multi-gene phylogenetic analysis
The ITS regions of all fungal isolates were subjected to molecular analysis to gain some preliminary insight into the fungal classification. Then, the sequences of the SSU, LSU, tef-1α, rpb2, and β-tubulin region were performed specifically for parasitic entomopathogens. Based on the five independent loci, the accurate taxonomic status of the three parasitic fungi was classified. As shown in Figure 6, the multi-gene phylogenetic treeclearly indicated the genetic distances of the three fungal species from other species in their respective genera, which also matched the morphological characteristics of these fungi.
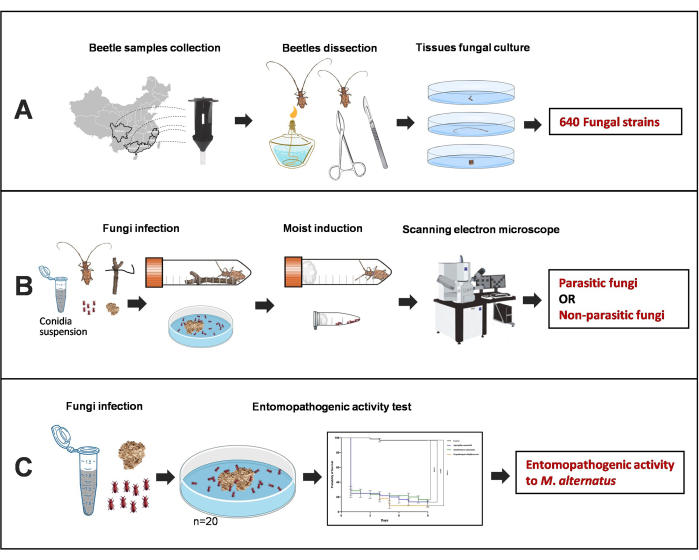
Figure 1: Illustration of screening out entomopathogenic fungi from multiple populations of M. alternatus adults. (A) Fungal isolation from different tissues of M. alternatus with naturally fungal infection. (B) Screening of entomopathogenic fungi. (C) Pathogenicity testing of parasitic entomopathogenic fungi against model beetles. Please click here to view a larger version of this figure.
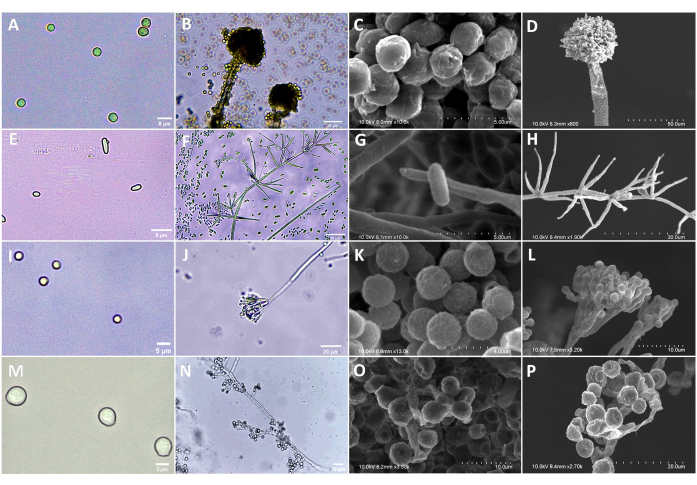
Figure 2: Morphology of A. austwickii, A. attenuatus (syn. L.attenuatum), P. citrinum, S. alboflavescens under OM and SEM. (A) Conidia of A. austwickii (OM). (B) Conidiophores of A. austwickii (OM). (C) Conidia of A. austwickii (SEM). (D) Conidiophores of A. austwickii (SEM). (E) Conidia of A. attenuatus (OM). (F) Conidiophores of A. attenuatus (OM). (G) Conidia of A. attenuatus (SEM). (H) Conidiophores of A. attenuatus (SEM). (I) Conidia of P. citrinum (OM). (J) Conidiophores of P. citrinum (OM). (K) Conidia of P. citrinum (SEM). (L) Conidiophores of P. citrinum (SEM). (M) Conidia of S. alboflavescens (OM). (N) Conidiophores of S. alboflavescens (OM). (O) Conidia of S. alboflavescens (SEM). (P) Conidiophores of S. alboflavescens (SEM). Scale bars are shown in the bottom right corner of each image. Please click here to view a larger version of this figure.

Figure 3: Phenotypes of M. alternatus cadavers infected by A. austwickii, A. attenuatus (syn. L. attenuatum), S. alboflavescens. Beetle cadaver surrounded by mycelium of (A) A. austwickii, (B) A. attenuatus, (C) S. alboflavescens. Please click here to view a larger version of this figure.
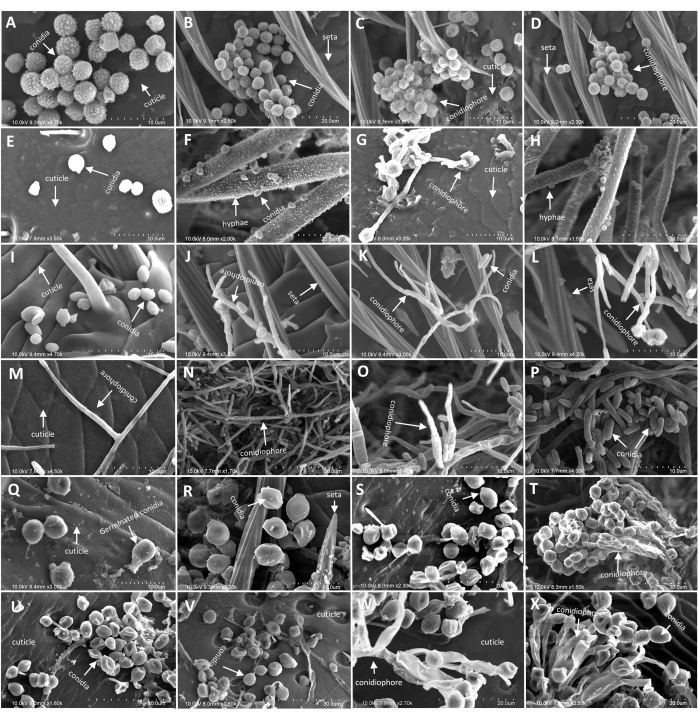
Figure 4: Morphology of M. alternatus and T. castaneum cadaver infected by A. austwickii, A. attenuatus (syn. L.attenuatum), S. alboflavescens under SEM. (A–D) Hyphae and conidiophores of A. austwickii on M. alternatus abdomen. (E–H) Hyphae and conidiophores of A. austwickii on T. castaneum cuticle surface. (I–L) Hyphae and conidiophores of A. attenuatus on M. alternatus antennae. (M–P) Hyphae and conidiophores of A. attenuatus on T. castaneum cuticle surface. (Q–T) Hyphae and conidiophores of S. alboflavescens on M. alternatus head. (U–X) Hyphae and conidiophores of S. alboflavescens on T. castaneum cuticle surface. Scale bars are shown in the bottom right corner of each image. Please click here to view a larger version of this figure.
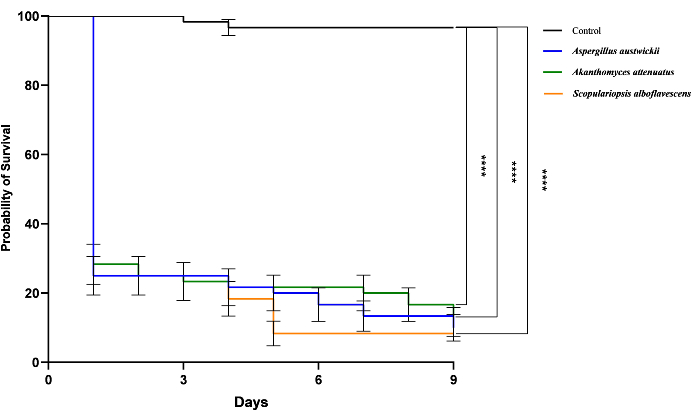
Figure 5: Pathogenicity activities of three parasitic fungi isolated from wild M. alternatus. The survival of T. castaneum beetles infected by three parasitic fungi is shown by Kaplan-Meier curves. Log-rank tests were performed and the significance level were denoted: ns, not significant; ****, P < 0.0001. Please click here to view a larger version of this figure.
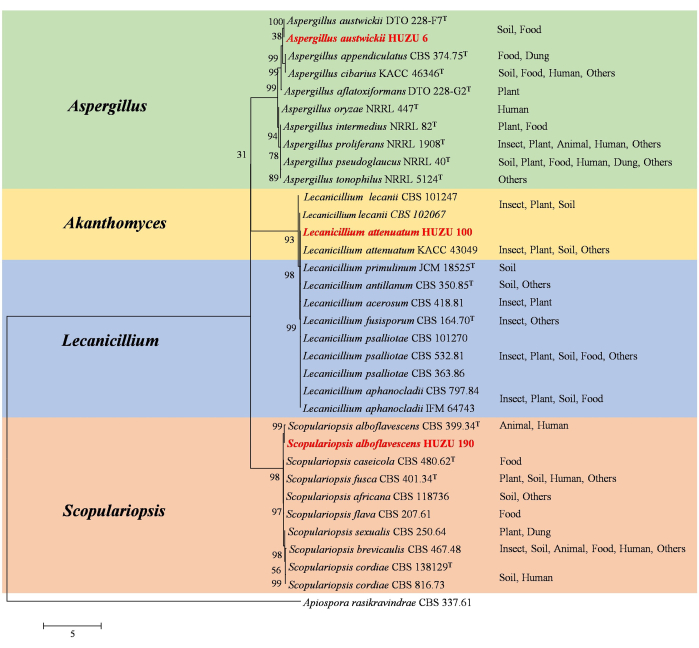
Figure 6: Phylogenetic tree construction of three entomopathogenic fungi inferred from a multi-gene dataset (ITS, LSU, SSU, EF-1α, rpb2, and β-tubulin). The fungal isolates in this study are in red. The nodes indicate supportive values greater than 50%. This figure was modified from Wu et al.43. The habitats where the species were found are indicated behind the species names as Insect, Plant, Soil, Animal, Food, Human, Dung, and others. The T indicates the isolates were from type materials. The GenBank accession numbers of fungal species sequences used for phylogenetic construction see Supplemental Table S1. Please click here to view a larger version of this figure.
| Primer name | Region | Sequence (5'-3') | Bibliography | |
| ITS1 | ITS | TCCGTAGGTGGACCTGCGG | 34 | |
| ITS4 | TCCTCCGCTTATTGATATGC | |||
| NS1 | SSU | GTAGTCATATGCTTGTCTC | 34 | |
| NS4 | CTTCCGTCAATTCCTTTAAG | |||
| LR0R | LSU | ACCCGCTGAACTTAAGC | 37, 38 | |
| LR7 | TACTACCACCAAGATCT | |||
| EF-983 | tef-1α | GCYCCYGGHCAYGGTGAYTTYAT | 39 | |
| EF-2218 | GACTTGACTTCRGTVGTGAC | |||
| RPB2-5’F | rpb2 | CCCATRGCTTGTYYRCCCAT | 40 | |
| RPB2-5’R | GAYGAYMGWGATCAYTTYGG | |||
| TUB1 | β-tubulin | AACATGCGTGAGATTGTAAGT | 41 | |
| TUB22 | TCTGGATGTTGTTGGGAATCC | |||
Table 1: Primer pairs for fungal identification.
Supplemental Table S1: GenBank accession numbers of fungal species sequences used for the phylogenetic construction. Please click here to download this File.
Discussion
Different geographical populations of FWB may develop varied interactions with the natural entomopathogenic fungi, due to long-term environmental adaptation of EPF species to local climate factors and the specific genotypic population of the host insect44,45. Expansion of the sampling sites to multiple insect occurrence regions helps increase the possibility of acquiring diverse strains or species of EPF from their natural hosts, as described by previous studies on agricultural insect pests46. In this protocol, with the assistance of traps baited with attractants, the pine sawyer beetle M. alternatus is collected at a cross-latitudinal level in five different sites of southern China, which is regarded as an ideal and rich resource reservoir for exploring novel EPF, considering the higher temperature and humidity. This labor-saving method traps beetles alive, which should be individually transported to the laboratory in separate sterile tubes. The natural infection symptoms on beetles require several days to occur. Representative EPF species A. austwickii, A. attenuatus (syn. L.attenuatum), and S. alboflavescens, obtained by the protocol, show significant regional specificity in different M. alternatus populations, which have not been reported on Monochamus beetles by other studies before. In addition, considering the potential distribution areas of the vector beetle M. alternatus in China, more EPF could be expected through deeper field investigation in the future. The result of our study demonstrates the great value of geographical EPF exploration for widely distributed FWB insect species.
This protocol also emphasizes the importance that different body positions of beetle cadavers should be dissected carefully into fine pieces followed by selective growth of mycelium on antibiotics-supplementing plates. As shown in our study with this mode of isolation, more than 600 fungal strains were acquired from different parts of M. alternatus bodies, which could guarantee an adequate quantity of fungal candidates for screening out potential EPF. Although the addition of antibiotics into medium plates seems to cause the loss of certain fungal species during isolation47, this traditional technology would still be the most suitable for various FWB systems to protect EPF growths from being contaminated by bacteria. The fineness of operation in dissection and isolation can compensate for possible species loss owing to the use of antibiotics. Additionally, the isolation of fungi from specific parts of insect bodies promotes the understanding of EPF colonization preferences on the hosts. For instance, A. attenuatus (syn. L.attenuatum) seems to infect M. alternatus antennae more frequently than other body positions, as observed in our previous study43, which would inspire further studies on its behavioral traits.
During strain identification, it is necessary to use a combination of morphological and molecular identification to distinguish closely related fungal species. For morphological identification, both macroscopic and microscopic characteristics should be clearly observed, including the different characteristics of fungal colonies, conidia, conidiophores, and hyphae of each different fungal strain. For molecular identification, the sequencing of the ITS region is not sufficient to reveal the species-level discrimination in the fungal community48. Therefore, it requires the application of different combinations of primer sets targeting more molecular markers (such as SSU, LSU, tef-1α, rpb2, and β-tubulin) to determine the phylogeny of entomopathogenic fungi more accurately.
Observation of fungal infection phenotype on beetles using an optical microscope has the shortcoming that light transmission and resolution affect discrimination of fungal characteristics. Conversely, the use of scanning electron microscopy (SEM) is much more reliable to obtain details on the infection process, which has been applied to record parasitic phenomena on several agricultural nematodes and insect pests49,50. Following artificial infection, the three parasitic entomopathogenic fungi, A. austwickii, A. attenuatus (syn. L.attenuatum), and S. alboflavescens, can be re-isolated from M. alternatus and T. castaneum body surfaces. Their morphological characteristics of conidia, infection pegs, and conidiophores can be determined from the two beetle species via SEM and shown the same with their respective colonies grown on PDA medium. Another insecticidal fungal species, P. citrinum, however, could not be re-isolated from both beetle species and was not observed by SEM from their body surfaces. Thus, the parasitism phenotypes induced by this protocol present excellent consistency between M. alternatus and the model beetle species.
The red flour beetle, T. castaneum is an important postharvest pest51 and also a general model organism of Coleopteran species used for genetics, immunology, and other research52,53,54,55 because it is highly fertile, well-adapted, and easy to manipulate experimentally. The consistency in parasitism phenotypes indicates that T. castaneum could be an appropriate substitute or tool to evaluate entomopathogenic activities of EPF on M. alternatus. This can release the demand for a great number of adult beetles of uniform genetic background, which is difficult for FWB collection in the wild field within a limited period. Following the entomopathogenic bioassay using this model insect, valuable information on EPF lethality can guide field studies on M. alternatus and even other Coleopteran FWB in the future.
To sum up, this present protocol facilitates the EPF resource exploration from forest wood borers, which considers a model beetle as an alternative for entomopathogenic evaluation. It greatly reduces the problems associated with having no access to an adequate quantity of beetles for testing purposes. Hence, this is a practical and efficient protocol for preliminary screening the entomopathogenic candidates on forest wood borers, which will facilitate the provision of natural resources for the future development of biological control strategies.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This research was supported by the National Key Research and Development Program of China (2021YFC2600100) and the Natural Science Foundation of Zhejiang Province (LY21C040001).
Materials
| 1.5 mL, 2 mL centrifuge tubes | Biosharp | BS-15-M | |
| 10 µL pipet tips | Sangon Biotech | F601216 | |
| 10 µL, 20 µL, 100 µL, 200 µL, 1,000 µL pipettes | Rainin | ||
| 1,000 µL pipet tips | Sangon Biotech | F630102 | |
| 2 mL cryogenic vials | Corning | 430659 | |
| 20x PBS buffer | Sangon Biotech | B548117-0500 | |
| 200 µL pipet tips | Sangon Biotech | F601227 | |
| 2,000 bp maker | TaKaRar | SD0531 | |
| 50 mL tubes | Nest | 602052 | |
| 50% glutaraldehyde solution | Sangon Biotech | G916054 | |
| 50x TAE buffer | Sangon Biotech | B548101 | |
| 6x loading buffer | TaKaRar | SD0503 | |
| Agarose | Sangon Biotech | A610013 | |
| Anhydrous ethanol | Jkchemical | LB10V37 | |
| Biochemistry Cultivation Cabinet | Shanghaiyiheng | LRH-250F | |
| Chloroform | Juhua | 61553 | |
| Commercial beetle traps | FEIMENGDI | BF-8 | www.yinyouji.com |
| Gel imager | Bio-Rad | GelDoc XR+ | |
| Glycerol | Sangon Biotech | A600232 | |
| High speed refrigerated centrifuge | Sigma | D-37520 | |
| High-Pressure Steam Sterilization Pot | Mettler Toledo | JA5003 | |
| Isopropyl alcohol | General-reagent | G75885B | |
| Nucleic acid dye | Sangon Biotech | A616696 | |
| Optical Microscope, OM | Leica | DM2000 | |
| Parafilm | Parafilm | PM996 | |
| PCR meter | Heal Force | Trident960 | |
| Penicillin G | Marklin | GB15743 | |
| Potato dextrose agar, PDA | Oxoid | CM0139 | |
| Potato dextrose broth, PDB | Solarbio | P9240 | |
| Primers | Sangon Biotech | / | |
| Primers Taq | TaKaRar | RR902A | |
| Rapid Fungi Genomic DNA Isolation Kit | Sangon Biotech | B518229 | |
| Scanning Electron Microscope, SEM | Hitachi | S-3400N | |
| Streptomycin | Marklin | S6153 | |
| Tetracycline | Marklin | T829835 | |
| Tween-80 | Marklin | T6336 | |
| Vacuum freeze dryer | Yamato | DC801 | |
| Vortex Shaker | HLD | WH-861 | |
| β-Mercaptoethanol | Marklin | M6230 |
References
- Hajek, A. E., Bauer, L. S. Microbial control of wood-boring insects attacking forest and shade trees. Field Manual of Techniques in Invertebrate Pathology. 10, 505-525 (2007).
- Linnakoski, R., Forbes, K. M. Pathogens-the hidden face of forest invasions by wood-boring insect pests. Frontiers in Plant Science. 10, 90 (2019).
- Hulcr, J., Dunn, R. R. The sudden emergence of pathogenicity in insect-fungus symbioses threatens naive forest ecosystems. Proceedings of the Royal Society B: Biological Sciences. 278 (1720), 2866-2873 (2011).
- Humble, L. M., Allen, E. A. Forest biosecurity: alien invasive species and vectored organisms. Canadian Journal of Plant Pathology. 10, 90 (2006).
- Ahmed, R., Freed, S. Biochemical resistance mechanisms against chlorpyrifos, imidacloprid and lambda-cyhalothrin in Rhynchophorus ferrugineus (Olivier) (Coleoptera: Curculionidae). Crop Protection. 143, 105568 (2021).
- Al-Ayedh, H., Hussain, A., Rizwan-Ul-Haq, M., Al-Jabr, A. M. Status of insecticide resistance in field-collected populations of Rhynchophorus ferrugineus (Olivier) (Coleoptera: Curculionidae). International Journal of Agriculture and Biology. 18 (01), 103-110 (2015).
- Garcia, F. R. M., Ovruski, S. M., Suarez, L., Cancino, J., Liburd, O. E. Biological control of Tephritid fruit flies in the Americas and Hawaii: a review of the use of parasitoids and predators. Insects. 11 (10), 662 (2020).
- Lacey, L. A., Shapiro-Ilan, D. I. Microbial control of insect pests in temperate orchard systems: potential for incorporation into IPM. Annual Review of Entomology. 53, 121-144 (2008).
- Wang, Z. Z., Liu, Y. Q., Shi, M., Huang, J. H., Chen, X. X. Parasitoid wasps as effective biological control agents. Journal of Integrative Agriculture. 18 (4), 705-715 (2019).
- Islam, W., et al. Insect-fungal-interactions: a detailed review on entomopathogenic fungi pathogenicity to combat insect pests. Microbial Pathogenesis. 159, 105122 (2021).
- Ali, S., Huang, Z., Ren, S. Production of cuticle degrading enzymes by Isaria fumosorosea and their evaluation as a biocontrol agent against diamondback moth. Journal of Pest Science. 83 (4), 361-370 (2010).
- Vidal, S., Jaber, L. R. Entomopathogenic fungi as endophytes: plant-endophyte-herbivore interactions and prospects for use in biological control. Current Science. 109 (1), 46-54 (2015).
- Rasool, S., et al. Seed inoculations with entomopathogenic fungi affect aphid populations coinciding with modulation of plant secondary metabolite profiles across plant families. New Phytololgist. 229 (3), 1715-1727 (2021).
- Dara, S. K., Montalva, C., Barta, M. Microbial control of invasive forest pests with entomopathogenic fungi: a review of the current situation. Insects. 10 (10), 341 (2019).
- Rajula, J., Rahman, A., Krutmuang, P. Entomopathogenic fungi in Southeast Asia and Africa and their possible adoption in biological control. Biological Control. 151, 104399 (2020).
- Sharma, L., et al. Advances in entomopathogen isolation: a case of bacteria and fungi. Microorganisms. 9 (1), 16 (2020).
- Echeverri-Molina, D., Santolamazza-Carbone, S. Toxicity of synthetic and biological insecticides against adults of the Eucalyptus snout-beetle Gonipterus scutellatus Gyllenhal (Coleoptera: Curculionidae). Journal of Pest Science. 83 (3), 297-305 (2010).
- Yang, T. H., et al. Entomopathogenic fungi-mediated biological control of the red palm weevil Rhynchophorus ferrugineus. Journal of Asia-Pacific Entomology. 26 (1), 102037 (2023).
- Kepler, R. M., et al. A phylogenetically-based nomenclature for Cordycipitaceae (Hypocreales). IMA Fungus. 8 (2), 335-353 (2017).
- Zhou, Y. M., Zhi, J. R., Qu, J. J., Zou, X. Estimated divergence times of Lecanicillium in the family Cordycipitaceae provide insights into the attribution of Lecanicillium. Frontiers in Microbiology. 13, 859886 (2022).
- Montalva, C., et al. Lecanicillium attenuatum isolates affecting the invasive cypress aphid (Cinara cupressi) in Chile. BioControl. 62 (5), 625-637 (2017).
- Futai, K. Pine wood nematode, Bursaphelenchus xylophilus. Annual Review of Phytopathology. 51 (1), 61-83 (2013).
- Kobayashi, F., Yamane, A., Ikeda, T. The Japanese pine sawyer beetle as the vector of pine wilt disease. Annual Review of Entomology. 29 (1), 115-135 (1984).
- Zhao, L., Sun, J. Pinewood nematode Bursaphelenchus xylophilus. (Steiner and Buhrer) Nickle. Biological invasions and its management in China. 13, 3-21 (2017).
- Akbulut, S., Stamps, W. T. Insect vectors of the pinewood nematode: a review of the biology and ecology of Monochamus species. Forest Pathology. 42 (2), 89-99 (2012).
- Shimazu, M. Effects of temperature on growth of Beauveria bassiana F-263, a strain highly virulent to the Japanese pine sawyer, Monochamus alternatus, especially tolerance to high temperatures. Applied Entomology and Zoology. 39 (3), 469-475 (2004).
- Han, B., Piao, C. G., Wang, L. F., Li, Y., Zheng, R. Z. Survey, identification and virulence test of pathogens of the pine sawyer beetle, Monochamus alternatus, at forest farm of maanshan, anhui province. Forest Research. 20 (20), 204-208 (2007).
- Ma, L., Zhang, L., Lin, H., Mao, S. Investigation of pathogens of Monochamus alternatus in east China and virulence. Chinese Journal of Biological Control. 25 (3), 220-224 (2009).
- Alvarez-Baz, G., Fernandez-Bravo, M., Pajares, J., Quesada-Moraga, E. Potential of native Beauveria pseudobassiana strain for biological control of pine wood nematode vector Monochamus galloprovincialis. Journal of Invertebrate Pathology. 132, 48-56 (2015).
- Dong, Y., Xie, P., Zheng, K., Gu, Y., Fan, J. Teflon coating and anti-escape ring improve trapping efficiency of the longhorn beetle, Monochamus alternatus. Applied Sciences. 13 (3), 1664 (2023).
- Gul, H. T., Saeed, S., Khan, F. Z. A. Entomopathogenic fungi as effective insect pest management tactic: a review. Applied Sciences and Business Economics. 1 (1), 10-18 (2014).
- Takuji Noma Strickler, K. Effects of Beauveria bassiana on Lygus hesperus (Hemiptera: Miridae) feeding and oviposition. Environmental Entomology. 29 (2), 394-402 (2000).
- Thakur, R., Sandhu, S. S. Distribution, occurrence and natural invertebrate hosts of indigenous entomopathogenic fungi of Central India. Indian Journal of Microbiology. 50 (1), 89-96 (2010).
- White, T. J., Bruns, T., Lee, S., Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protocols. 18 (1), 315-322 (1990).
- Zaman, G., Chayen, J. An aqueous mounting medium. Journal of Clinical Pathology. 34 (5), 567-568 (1981).
- Koon, M. A., et al. Preparation of prokaryotic and eukaryotic organisms using chemical drying for morphological analysis in scanning electron microscopy (SEM). Journal of Visualized Experiments. (143), e58761 (2019).
- Vilgalys, R., Hester, M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. Journal of Bacteriology. 172 (8), 4238-4246 (1990).
- Rehner, S. A., Samuels, G. J. Taxonomy and phylogeny of Gliocladium analysed from nuclear large subunit ribosomal DNA sequences. Mycological Reasearch. 98 (6), 625-634 (1994).
- Aphidech, S., Kusavadee, S. Isolation, identification, culture and production of adenosine and cordycepin from cicada larva infected with entomopathogenic fungi in Thailand. African Journal of Microbiology Research. 7 (2), 137-146 (2013).
- Wang, Y., et al. Polycephalomyces yunnanensis (Hypocreales), a new species of Polycephalomyces parasitizing Ophiocordyceps nutans and stink bugs (hemipteran adults). Phytotaxa. 208 (1), (2015).
- Qi, M., Xie, C. X., Chen, Q. W., Yu, Z. D. Pestalotiopsis trachicarpicola, a novel pathogen causes twig blight of Pinus bungeana (Pinaceae) in China. Antonie Van Leeuwenhoek. 114 (1), 1-9 (2021).
- Tamura, K., Stecher, G., Peterson, D., Filipski, A., Kumar, S. MEGA6: molecular evolutionary genetics analysis version 6.0. Molecular Biology and Evolution. 30 (12), 2725-2729 (2013).
- Wu, S., et al. Discovery of entomopathogenic fungi across geographical regions in southern China on pine sawyer beetle Monochamus alternatus and implication for multi-pathogen vectoring potential of this beetle. Frontiers in Plant Science. 13, 1061520 (2022).
- Brodeur, J. Host specificity in biological control: insights from opportunistic pathogens. Evolutionary Applications. 5 (5), 470-480 (2012).
- Croll, D., Mcdonald, B. A. The genetic basis of local adaptation for pathogenic fungi in agricultural ecosystems. Molecular Ecology Resources. 26 (7), 2027-2040 (2017).
- Perez-Gonzalez, V. H., et al. Specific diversity of the entomopathogenic fungi Beauveria and Metarhizium in Mexican agricultural soils. Journal of Invertebrate Pathology. 119, 54-61 (2014).
- Debono, M., Gordee, R. S. Antibiotics that inhibit fungal cell wall development. Reviews of Microbiol. 48, 471-497 (1994).
- Li, S., et al. Exploring the accuracy of amplicon-based internal transcribed spacer markers for a fungal community. Molecular Ecology Resources. 20 (1), 170-184 (2019).
- Wan, J., Dai, Z., Zhang, K., Li, G., Zhao, P. Pathogenicity and metabolites of endoparasitic nematophagous fungus Drechmeria coniospora YMF 1.01759 against nematodes. Microorganisms. 9 (8), 1735 (2021).
- Ammon, V., Wyllie, T. D., Brown, M. F. Investigation of the infection process of macrophomina phaseolina on the surface of soybean roots using scanning electron microscopy. Mycopathologia. 55 (2), 77-81 (1975).
- Throne, J. E., Hallman, G. J., Johnson, J. A., Follett, P. A. Post-harvest entomology research in the United States department of agriculture-agricultural research service. Pest Management Science. 59 (6-7), 619-628 (2003).
- Silver, K., et al. The Tribolium castaneum cell line TcA: a new tool kit for cell biology. Scientific Reports. 4 (1), 6840 (2014).
- Fatehi, S., et al. Characterization of iflavirus in the red flour beetle, Tribolium castaneum (Coleoptera; Tenebrionidae). Insects. 14 (3), 220 (2023).
- Li, C., et al. Identification and characterization of development-related microRNAs in the red flour beetle, Tribolium castaneum. International Journal of Molecular Sciences. 24 (7), 6685 (2023).
- Li, J., et al. The TGF-β receptor gene saxophone influences larval-pupal-adult development in Tribolium castaneum. Molecules. 27 (18), 6017 (2022).

.
