An Experimental System to Study Mechanotransduction in Fetal Lung Cells
Summary
Mechanical forces play a key role in lung development and lung injury. Here, we describe a method to isolate rodent fetal lung type II epithelial cells and fibroblasts and to expose them to mechanical stimulation using an in vitro system.
Abstract
Mechanical forces generated in utero by repetitive breathing-like movements and by fluid distension are critical for normal lung development. A key component of lung development is the differentiation of alveolar type II epithelial cells, the major source of pulmonary surfactant. These cells also participate in fluid homeostasis in the alveolar lumen, host defense, and injury repair. In addition, distal lung parenchyma cells can be directly exposed to exaggerated stretch during mechanical ventilation after birth. However, the precise molecular and cellular mechanisms by which lung cells sense mechanical stimuli to influence lung development and to promote lung injury are not completely understood. Here, we provide a simple and high purity method to isolate type II cells and fibroblasts from rodent fetal lungs. Then, we describe an in vitro system, The Flexcell Strain Unit, to provide mechanical stimulation to fetal cells, simulating mechanical forces in fetal lung development or lung injury. This experimental system provides an excellent tool to investigate molecular and cellular mechanisms in fetal lung cells exposed to stretch. Using this approach, our laboratory has identified several receptors and signaling proteins that participate in mechanotransduction in fetal lung development and lung injury.
Protocol
1. Coating of plates with ECM proteins
- Under sterile conditions, mix 120 μg of laminin with 12 ml of cold sterile 1X PBS per plate.
- Take Bioflex untreated plates and add 2ml of the solution to each well (final concentration of laminin is 2 μg/cm2). Alternatively, other ECM proteins could be used, such as collagen-1 [10 μg/cm2], fibronectin [5 μg/cm2], vitronectin [0.5 μg/cm2] or elastin [10 μg/cm2]. The coating technique is similar to what it is described for laminin-coating. Make sure the wells are completely covered by the solution.
- Wrap the plates with plastic and put at 4°C on a flat surface overnight to allow the laminin to adsorb to the bottom of the wells. In these conditions, plates can be stored for at least one week while still retaining good ECM function.
- Next day, under sterile conditions, wash the wells 3 times with 1X PBS.
- Add 1ml of 1% BSA in 1X PBS to each well and incubate at 37°C for 1h to block non-specific binding sites on the membranes.
- Rinse the plates 3 times with 1X PBS to remove unadsorbed proteins.
- Keep the plates at 37°C in DMEM until cells are isolated from step 2.14.
2. Isolation of fetal rodent lung type II epithelial cells
The day before isolation have the screen cups with different size nylon meshes autoclaved and ready.
- Obtain fetal lungs from timed-pregnant mouse/rat on E17-19 of gestation. Transfer the tissue into a 50 ml conical tube containing DMEM medium and place it on ice.
- Make digestion buffer in a 50 ml conical tube: (10 ml). For this volume, lung tissue from approximately 20 fetuses from mouse/rat is used.
8.5 ml DMEM
0.5 ml 500mM pH 7.4 HEPES
5 mg Collagenase 1
5 mg Collagenase 1A
1 ml Chicken serum, heat inactivated - Mix well until all components are dissolved and filter through a 0.2 micron syringe filter inside the sterile hood. Place the conical tube containing the digestion buffer in a water bath at 37°C.
- Remove the DMEM medium from the conical tube from step 2.1 and transfer the tissue into a sterile Petri dish.
- Mince the lungs well with sterile scissors or razor blade to make tissue pieces less than 1 mm and transfer the tissue into the conical tube containing the pre-warmed digestion buffer from step 2.3.
- Digest the tissue by pipetting up and down:
100 times with 10 ml pipette
100 times with 5 ml pipette
100 times with Pasteur pipette - Centrifuge the homogenate at 1,300 rpm for 5 minutes at room temperature.
- Carefully remove the supernatant by aspiration and re-suspend the pellet containing the cells in 15 ml of DMEM plus 20% FBS.
- Take out the screen cups with 100-, 30- and 15-micron nylon meshes and place them on sterile 150 ml beakers.
- Add cell homogenate from step 2.8 and sequentially filter through 100-, 30-, and 15-micron mesh screen cups.
- Collect clumped non-filtered cells from the 30- and 15-micron meshes after several washes with DMEM plus 10% FBS to facilitate the filtration of non-epithelial cells.
- Discard the filtrate from the 15-micron mesh which contains mostly fibroblasts.
- Purify further type II cells by incubating cells from step 2.11 in 75-cm2 flasks for 30 min (approximatelly10 ml of cell suspension per flask).
- Collect the supernatant and centrifuge at 1,300 rpm for 5 minutes at room temperature to pellet the cells.
- Carefully remove the supernatant by aspiration and re-suspend the pellet containing the cells in serum-free DMEM. The volume of resuspension depends on the amount of tissue processed. Approximately, we plate the equivalent of cells obtained from one fetus in each well (2 ml) in order to achieve between 60-70% confluency the following day.
- Plate the cell suspension on Bioflex six-well plates precoated with laminin or fibronectin.
3. Isolation of fetal rodent lung fibroblasts
- Follow the same steps described above for type II cells isolation up to 2.11.
- Take the filtrate from the 15-micron mesh (without previous washing; approximate volume is 10-15 ml) and plate onto 75-cm2 at 37°C for 30-60 min to allow fibroblasts to adhere.
- Aspirate the supernantant and replace with serum-free DMEM and incubate overnight.
- Next day, harvest the cells with 0.25% (wt/vol) trypsin in 0.4 mM EDTA and plate them on Bioflex plates precoated with fibronectin.
4. Experimental system to provide mechanical stimulation to lung cells
- Seed the cells (around 50% confluent) on Bioflex culture plates in serum-free DMEM (2 ml/well) and let them attach and spread for at least 6h before experiments. In general our laboratory maintains cells in culture for around 24h before applying mechanical strain. If a specific number of cells are required for the experiments, after isolation, type II cells are maintained in serum-free DMEM 75-cm2 flasks and the next day, cells are trypsinized, counted and then seeded.
- Next day, media are replaced with fresh serum-free DMEM (2 ml/well) and bioflex plates are mounted in a Flexcell FX-5000 Strain Unit. Monolayers should be no greater than 80% confluent before the initiation of the experiments.
- Equibiaxial strain is applied to the membranes. The regimen of strain varies depending on simulation of mechanical forces in vivo (see discussion).
- Cells grown on non-stretched membranes cultured in parallel are used as controls.
- At the end of the experiments, monolayers can be processed to analyze changes on gene expression (for example surfactant protein C) by real time-PCR, protein abundance by Western blot, etc. Also, monolayers can be fixed for immunocytochemistry experiments. For this technique, after fixation, silastic membranes are cut out from the plates and mounted on glass slides using 10-20 μl of water as a mounting agent before permeabilization and incubation with antibodies. Supernatant can also be used to investigate the presence of growth factors, cytokines, etc.
5. Representative Results
Figure 1 and Figure 2 show representative phase-contrast photographs of E18 fetal mouse type II cells isolated using the technique described in this manuscript.
Figure 3 demonstrates that mechanical strain induces differentiation of fetal type II epithelial cells using surfactant protein-C as a marker.

Figure 1. Representative phase-contrast photograph taken right after isolation showing the clumped appearance of fetal type II cells.
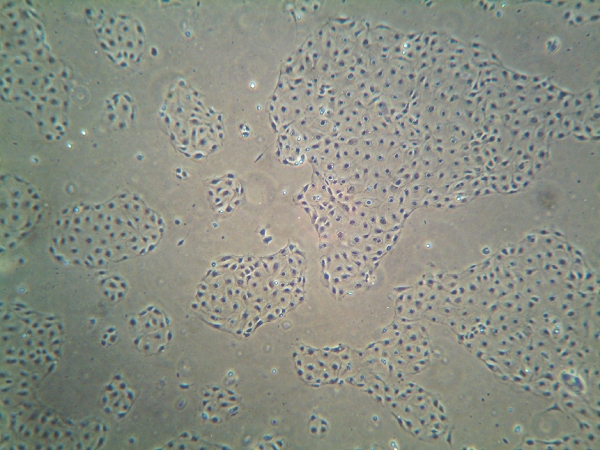
Figure 2. E18 fetal type II epithelial cells were isolated as described here and plated on bioflex plates coated with laminin. Photograph was taken the following day after non-stretched cells were fixed in paraformaldehyde. The purity of the cells was determined to be 90 ± 5% by microscopic analysis of epithelial cell morphology and immunostaining for SP-C.

Figure 3. Fetal type II epithelial cells were exposed to 5% cyclic strain at 40 cycles/min for 16h. A) Northern blot of surfactant protein C (SP-C) mRNA expression showing that strain induces type II cell differentiation using different ECM substrates. +/- signs represent exposure or not to strain, respectively. Data are presented as mean +/- SEM, n=3; * P<0.05. B) Fluorescence immunocytochemistry images demonstrating SP-C protein levels (green) in fetal type II cells exposed or not (control) to mechanical strain . Nuclei were counterstained with dapi (blue). Bar, 10 μm. C) Western blot results from three experiments showing that mechanical stretch increases SP-C protein. N=3; * P<0.05.
Discussion
In this manuscript, we describe a method to isolate fetal type II epithelial cells and fibroblasts and to expose them to mechanical stimulation using the Flexcell Strain Apparatus. We have used this technique to assess differentiation of epithelial cells 1,2 and to study receptor and signaling pathways activated by stretch 3-9. In addition, this method can also be used to investigate cell responses induced by mechanical injury 10,11. The procedure is based on digestion of the lung tissue with collagenase. It takes advantage of the tendency of type II cells to clump together after digestion. Important steps while performing this technique are mincing the tissue well to facilitate digestion of the lung and washing thoroughly the non-filtered cells on 30 and 15 micron meshes to facilitate the filtration of non-epithelial cells and therefore to achieve greater purity of type II cells.
Our laboratory uses membranes coated with collagen or laminin to simulate the ECM composition of the basement membranes. However, when cells are exposed to 20% elongation, cells might detach from the substrate during stretch and fibronectin should be considered as an alternative substrate.
Another important consideration is to avoid 100% confluence of cell monolayers prior to stretch, given that this may promote cell-to-cell contact rather than cell-to-matrix contact and therefore cells might not “sense” the percentage of elongation set up by the protocol.
The Flexcell Strain Unit uses a vacuum to deform a flexible bottom culture plate. Application of vacuum stretches each membrane over the central cylinder post, creating uniform radial and circumferential strain across the membrane surface and therefore providing equibiaxial distension, similar to in vivo conditions (see cartoon, schematic overview video, with permission, Dr. A.J. Banes Flexcell International Corporation, www.flexcellint.com). This system provides a defined, controlled static or cyclic deformation by the specified strain regimen. To mimic fetal breathing movements a cyclic stretch regimen between 2.5-5% stretch at 40-60 cycles/minute is applied. There is not agreement on the percentage of elongation that fetal lung senses during fetal breathing. Some authors believe that 5% distension is appropriate 12 whereas other investigators argue that 2.5% is more representative of the in vivo conditions 13,14. To simulate constant distension pressure, a 2.5% continuous stretch protocol is used. To mimic lung injury, 20% cyclic stretch at 40 cycles/min is used. Potential limitations of this technique include the increase of deformation of cells seeded in the area of the membrane peripheral to the loading post. Another limitation is the inability to provide elongation greater than 15-20% if a high cycle rate is used (above 40 cycles/min).
Declarações
The authors have nothing to disclose.
Acknowledgements
Supported by NIH grant HD052670.
Materials
| Name of the reagent | Company | Catalogue number | Comments |
| DMEM | Sigma | D5648 | |
| HEPES | Sigma | H3375 | |
| Collagenase 1 | Sigma | C0130 | |
| Collagenase 1A | Sigma | C9891 | |
| Chicken serum | Sigma | C5405 | |
| Screen cups | Sigma | CD1-1KT | |
| Syringe filters | Fisher Scientific | 09-754-25 | |
| 100 micron nylon mesh | Small Parts, INC | CMN-100-D | |
| 30 micron mesh | Small Parts, INC | CMN-30-D | |
| 15 micron mesh | Dynamic Aqua-Supply Ltd. | NTX15 | |
| Laminin | Sigma | L2020 | |
| Collagen-1 | Collagen Biomed | PC0701 | |
| Fibronectin | Sigma | F1141 | |
| Vitronectin | Sigma | V-0132 | |
| Elastin | Sigma | E-6402 | |
| Bioflex plate | Flexcell International | BF-3001U | Uncoated |
| Flexcell Strain Unit | Flexcell International | FX-5000 |
Referências
- Sanchez-Esteban, J., Tsai, S. W., Sang, J., Qin, J., Torday, J. S., Rubin, L. P. Effects of mechanical forces on lung-specific gene expression. Am. J. Med. Sci. 316, 200-204 (1998).
- Sanchez-Esteban, J., Cicchiello, L. A., Wang, Y., Tsai, S. W., Williams, L. K., Torday, J. S., Rubin, L. P. Mechanical stretch promotes alveolar epithelial type II cell differentiation. J. Appl. Physiol. 91, 589-595 (2001).
- Sanchez-Esteban, J., Wang, Y., Cicchiello, L. A., Rubin, L. P. Cyclic mechanical stretch inhibits cell proliferation and induces apoptosis in fetal rat lung fibroblasts. Am. J. Physiol. Lung. Cell. Mol. Physiol. 282, L448-L456 (2002).
- Sanchez-Esteban, J., Wang, Y., Cicchiello, L. A., Rubin, L. P. Cyclic mechanical stretch inhibits cell proliferation and induces apoptosis in fetal rat lung fibroblasts. Am. J. Physiol. Lung. Cell. Mol. Physiol. 282, L448-L456 (2002).
- Sanchez-Esteban, J., Wang, Y., Gruppuso, P. A., Rubin, L. P. Mechanical stretch induces fetal type II cell differentiation via an epidermal growth factor receptor-extracellular-regulated protein kinase signaling pathway. Am. J. Respir. Cell. Mol. Biol. 30, 76-83 (2004).
- Wang, Y., Maciejewski, B. S., Lee, N., Silbert, O., McKnight, N. L., Frangos, J. A. Strain-induced fetal type II epithelial cell differentiation is mediated via cAMP-PKA-dependent signaling pathway. Am. J. Physiol. Lung. Cell. Mol. Physiol. 291, L820-L827 (2006).
- Sanchez-Esteban, J., Wang, Y., Filardo, E. J., Rubin, L. P., Ingber, D. E. Integrins {beta}1, {alpha}6, and {alpha}3 contribute to mechanical strain-induced differentiation of fetal lung type II epithelial cells via distinct mechanisms. Am. J. Physiol. Lung. Cell. Mol. Physiol. 290, L343-L350 (2006).
- Wang, Y., Maciejewski, B. S., Soto-Reyes, D., Lee, H. S., Warburton, D., Sanchez-Esteban, J. Mechanical stretch promotes fetal type II epithelial cell differentiation via shedding of HB-EGF and TGF-alpha. J. Physiol. 587, 1739-1753 (2009).
- Wang, Y., Maciejewski, B. S., Drouillard, D., Santos, M., Hokenson, M. A., Hawwa, R. L. A role for caveolin-1 in mechanotransduction of fetal type II epithelial cells. Am. J. Physiol. Lung. Cell. Mol. Physiol. 298, L775-L783 (2010).
- Lee, H. S., Wang, Y., Maciejewski, B. S., Esho, K., Fulton, C., Sharma, S. Interleukin-10 protects cultured fetal rat type II epithelial cells from injury induced by mechanical stretch. Am. J. Physiol. Lung. Cell. Mol. Physiol. 294, L225-L232 (2008).
- Hawwa, R. L., Hokenson, M. A., Wang, Y., Huang, Z., Sharma, S., Sanchez-Esteban, J. IL-10 inhibits inflammatory cytokines released by fetal mouse lung fibroblasts exposed to mechanical stretch. Pediatric Pulmonology. , (2011).
- Kitterman, J. A. The effects of mechanical forces on fetal lung growth. Clin. Perinatol. 23, 727-740 (1996).
- Harding, R., Crystal, R. G., West, J. B., Banes, P. J. Fetal breathing movements. The Lung: Scientific Fountations. , 2093-2104 (1997).
- Harding, R., Liggins, G. C. Changes in thoracic dimensions induced by breathing movements in fetal sheep. Reprod. Fertil. Dev. 8, 117-124 (1996).

