Automated Measurement of Microcirculatory Blood Flow Velocity in Pulmonary Metastases of Rats
Summary
A method is presented to measure microcirculatory blood flow velocity in pulmonary cancer metastases of the pleural surface in rats in an automated fashion, using closed-chest pulmonary intravital microscopy. This model has potential to be used as a widespread tool to perform physiologic research on pulmonary metastases in rodents.
Abstract
Because the lung is a major target organ of metastatic disease, animal models to study the physiology of pulmonary metastases are of great importance. However, very few methods exist to date to investigate lung metastases in a dynamic fashion at the microcirculatory level, due to the difficulty to access the lung with a microscope. Here, an intravital microscopy method is presented to functionally image and quantify the microcirculation of superficial pulmonary metastases in rats, using a closed-chest pulmonary window and automated analysis of blood flow velocity and direction. The utility of this method is demonstrated to measure increases in blood flow velocity in response to pharmacological intervention, and to image the well-known tortuous vasculature of solid tumors. This is the first demonstration of intravital microscopy on pulmonary metastases in a closed-chest model. Because of its minimized invasiveness, as well as due to its relative ease and practicality, this technology has the potential to experience widespread use in laboratories that specialize on pulmonary tumor research.
Introduction
The lung is one of the most important target organs of metastatic disease, and because this condition is difficult to treat successfully with chemo- and radiation therapy, a cure is still rare1,2. Specific pathophysiological and microcirculatory features of solid primary and metastatic tumors, such as microregional hypoxia, diffusion limitation and inefficient tumor vasculature, greatly contribute to their resistance to anticancer treatment3,4. Due to the microscopic scale and dynamic nature of parameters such as microvascular blood flow, intravital microscopy of the tumor in the living animal has become a very important research tool in the field5. While intravital microscopy models have been applied to tumors in different organ sites, including the metastatic lung within an open rib cage, no protocol has been developed yet for the research of pulmonary metastases in a physiologically preserving, closed-chest environment6,7. Such an endeavor is particularly hampered by the necessity to surgically access the rib cage without affecting the overall function of the lung7-9. Recently, a method was introduced to image pulmonary microcirculatory blood flow in a close-chest setting in live rats, using fluorescence intravital microscopy10. This protocol enables the systematic quantification of blood flow velocity from injected, fluorescently labeled red blood cells, using computerized analysis, while keeping the animal physiologically stable and preserving the integrity of the lung11. In this present study, it is shown how this technology can be modified to image and quantify microcirculatory blood flow in tail vein-inoculated pulmonary metastases on the pleural surface in the immunocompromised rat. This model is also the first one to study metastatic lung tumors in a closed-chest intravital microscopy setting.
Protocol
NOTE: All animal related procedures described in this protocol have been previously approved by the Duke University Institutional Animal Care and Use Committee (DUIACUC).
1. Cancer Cell Culture and Injection
- Cultivate fluorescently labeled metastatic cancer cells (e.g. human MDAMB231-GFP breast cancer cells, gift from Dr. Patricia Steeg, NCI, and YFP-labeled mouse sarcoma cells, gift from Dr. David Kirsch, Duke University Medical Center, Department of Radiation Oncology) in appropriate culture medium (e.g. Dulbecco’s Modified Eagle Medium (DMEM) with 10% fetal bovine serum and 1% penicillin/streptavidin) at 37 °C until approximately 90% confluent.
- Trypsinize cells, wash them 2 times with PBS, count, and then inject them into the tail veins of isoflurane-anesthetized 10 weeks old female nude rats at 5 million cells per animal, using a syringe with a 27 G needle. Surgical level anesthesia is verified by the lack of reaction to toe pinch.
2. Monitoring of Metastases Using MicroCT
- Examine rats once a week using a micro-CT/micro-Irradiator, to detect the presence of metastases above approximately 2 mm in diameter in diameter. The micro-CT is commissioned as previously described12.
- Subject rats to 3% isoflurane anesthesia prior to imaging. Confirm deep anesthesia by toe pinch.
- After onset of anesthesia, quickly transfer rats to the imaging cradle in the imaging chamber and connect via a nose-cone to an isoflurane-air mixture at 2.5-3%. Adjust the position of the rat in the cradle in a way that its thorax is positioned in the photon beam of the MicroCT scanner, using the external position controls and laser delimiter on the imaging cradle. Ensure the door to the imaging chamber is closed, to shield the investigator from the gamma rays.
- Control the position of the animal again using the color video camera. Perform a low-resolution CT imaging test run, and use the resulting image to adjust the field of view to the xyz dimensions of the thoracic cavity.
- Image the rat thorax using a 2 mm Al filter at 40 kVp, 2.5 mA, and 0.008 voxel at 7 FPS and return the animal to its cage. Imaging of one animal should take not more than 15 or 20 min. Do not return an animal that has undergone surgery to the company of other animals until fully recovered. Do not leave an animal unattended until it has regained sufficient consciousness to maintain sternal recumbency.
- Confirm metastases by the appearance of relatively radio-opaque objects that cannot be explained by intrathoracic blood vessels (Figure 1A)
3. Window Chamber Surgery
- Anesthesia, vital signs and tail vein catheterization
- Select animals with presence of metastatic disease. Inject animal with an intraperitoneal dose of 50 mg/kg pentobarbital. Confirm surgical-level anesthesia by toe pinch before proceeding.
Note: Anesthesia protocols should be matched to the respective experimental setup. Pentobarbital was chosen here as a long-acting anesthetic, in order to induce deep anesthesia for lengthy procedures, while offering the option of facile re-dosing. However, loss of animals to overdosing is a common problem with pentobarbital anesthesia. Another option that preserves autonomic reflexes to a larger degree than pentobarbital is ketamine in combination with sedatives such as xylazine or medetomidine, which however permits only for a single re-dosing cycle. - Shave animals on the side of the body that has the metastatic disease, and in the neck area, using a clipper. Wipe off all remaining loose hair from the skin. After loose hair is removed, apply veterinary ointment to the eyes, to prevent them from drying out.
NOTE: Athymic nude rats may have residual hair that requires removal before proceeding with surgical procedures. It is very important to remove all hair thoroughly, as it may interfere with surgical procedures and imaging. - Fix the animal in a supine position on a metal plate that is placed on a 37 °C water circulated heating pad. The front and hind extremities are fixed on the plate with tape.
NOTE: It is useful to control and record vital signs, such as heart rates and arterial blood oxygenation by using a pulse oximeter, throughout surgical and experimental procedures.
- Select animals with presence of metastatic disease. Inject animal with an intraperitoneal dose of 50 mg/kg pentobarbital. Confirm surgical-level anesthesia by toe pinch before proceeding.
- Tracheal intubation
- In order to place a catheter for ventilation of the animal, first make a transversal cervical skin incision, followed by the median separation of the longitudinal musculature ventral to the trachea.
- Use repeated opening-to-closing action with a sharp forceps to create a passage for the suture through the dorsal side of the trachea.
- Make a small incision into the trachea on the ventral side, not more than semi-circular, approximately between the second and third tracheal ring. Leave a sufficiently long portion of the trachea exposed on the dorsal surface, to enable fixation of the tracheal catheter.
- Insert a 2.5 to 3.0 mm “Y” tracheal cannula into the trachea, and tighten with a 4-0 monofilament suture. Ensure the cannula is connected to a pressure cycled ventilator with a bottle connected to the expiration duct that is filled with 6 cm of water, to maintain positive pulmonary pressure. Inflowing gas should be 100% oxygen, unless experimentally desired otherwise.
- Insert a catheter with a 25-27 G needle, and filled with heparinized saline into one of the tail veins of the rat, and fix in place with tape.
NOTE: Ensure patency of the tail vein catheter throughout the procedure by repeatedly injecting a small amount of heparinized saline into the tail vein. Also, oro-tracheal intubation, i.e. the guidance of a tracheal tube via the mouth of the anesthetized animal and past the larynx into the trachea, is a possible alternative to the tracheotomy procedure that is described here. However,this method requires special training and experience, to avoid damage to the trachea, and also to preclude the accidental cannulation of the esophagus.
- Application of pulmonary window
- Remove the skin from the side of the chest where the metastatic disease is located, by creating an incision, and then detaching the skin using blunt dissection.
Note: The skin can be excised and removed subsequent to detachment - Proceed by dissecting the two layers of overlaying musculature (pectoralis, serratus, and latissimus dorsi), but leaving the intercostal muscles intact. Create a perforation in the chest cavity of approximately 1.5 cm in diameter, by removing portions of typically two adjacent ribs. Ideally, locate the perforation in of the region of the sixth and seventh ribs.
- Osteotomy:
- To minimize bleeding and damage to the lung surface, tightly hold the rib to be cut with a toothed surgical forceps during cutting. Using surgical scissors, cut the medial side of the rib first, at an angle of approximately 45°, leaving the pointed side of the remaining rib bone pointing outside.
- Subsequently, cut the lateral side of the rib bone in a similar manner, again leaving the pointed side of the rib bone pointing outwards, to prevent damage to the lung surface.
- Repeat the procedure for the adjacent rib, then cut the intercostal muscles and remove the excised piece. During this procedure, maintain lung pressure in a way that mechanical interaction between the lung surface and the rib cage is minimized. Do this by appropriately regulating inspiration pressure on the ventilator.
- Insertion of the window:
- Insert a custom made lung window, consisting of a coverglass that is attached to a Plexiglas socket (Figure 1B). Attach the window to the socket by gluing, or by applying a very small amount of vacuum grease. Insert the window in a way that surface metastases are located close to the center of the window. If necessary, adjust the inserted hole to bring the micrometastasis to the center of the window by enlarging the hole slightly to the respective side.
NOTE: Metastatic disease on the pleural surface can be identified as clearly recognizable white dots or areas within the otherwise pink-to-salmon colored healthy parts of the lung surface that appear predominantly along the fissures. While micrometastases can occur on other areas of the pleural exterior, the investigated cell lines almost always display surfacing micrometastases in the perforated area, once metastatic disease can be radiologically detected. - After inserting the socket into the perforation, and creating direct contact with the visceral pleura of the lung, suture the edges of the window frame to the surrounding intercostal muscle, using 4.0 monofilament suture (Figure 1C). Use a slight increase of inspiration pressure on the ventilator to help residual air to escape and to create a seal.
Note: The respiration rate of rats can vary greatly, depending on the status of anesthesia, status of excitement or anxiety, oxygen concentration of inspired air (FiO2), etc. It is recommendable to adjust the respiration rate between 70 and 90 bpm. The inspiration pressure should be adjusted with caution and not be set to more than approx. 8 cm H2O (0.6 mmHg), to avoid damage to the lung surface.
- Insert a custom made lung window, consisting of a coverglass that is attached to a Plexiglas socket (Figure 1B). Attach the window to the socket by gluing, or by applying a very small amount of vacuum grease. Insert the window in a way that surface metastases are located close to the center of the window. If necessary, adjust the inserted hole to bring the micrometastasis to the center of the window by enlarging the hole slightly to the respective side.
- Position the animal in a custom designed restrainer that is designed to eliminate Z-directional movement (Figure 1D) on a steel plate that is positioned on a thermostatic (electrical) heating blanket, under a fluorescence microscope. Control the animal’s body temperature using a rectal thermistor. Adjust the screws of the animal restrainer, and the inspiration pressure on the ventilator to achieve optimal control of lateral movement.
Note: Natural breathing in mammals involves all three directions of lung movement and chest extension: bilateral, dorso-ventral, and cranio-caudal. In order to preserve the natural breathing movement as much as possible, it is important to minimize Z-directional compression to the necessary extent. Because the Z-dimensional restraint has the potential to introduce artifacts that may affect blood flow and other parameters, it is advisable to keep conditions constant during series of repeated measurements in the same animal.
- Remove the skin from the side of the chest where the metastatic disease is located, by creating an incision, and then detaching the skin using blunt dissection.
4. Imaging and Measurement of Microcirculatory Blood Flow
- Collect red blood cells via cardiac puncture and label them with DiI (1,1=-dioctadecyl-3,3,3=,3=-tetramethyl-indocarbocyanine perchlorate), as described previously 10.
- Inject 300 µl of labeled red blood cells into the tail vein of the rat before the window chamber surgery is done, to avoid first pass adhesion effects to the glass window. Eliminate any air bubbles in the syringe or catheter, as introduction of air into the vein will inhibit any further injections.
- Image blood flow with a microscope CCD camera at -40 °C chip cooling temperature, and approximately 100X overall resolution (i.e. with a 10X microscope objective, and 10X pre-camera ocular). Use standard Rhodamine/ TRITC filter sets (excitation 450-490 nm, emission >515 nm). Record the actual frame rate and pixel resolution of the resulting image sequences. Record at least 200 (ideally around 300) images per stack, to ensure successful analysis of flow velocity.
- Replenish fluid loss in the animal by injecting approximately 1 ml of saline i.p. every hr.
NOTE: Experimental settings that involve an intervention, e.g. a drug that modifies the blood flow velocity in a pulmonary cancer metastasis, requires repeated measurements of blood flow velocity in pulmonary metastatic cancer. For these extended experiments, it is important to replenish the animal with sufficient fluid. - Euthanize the animals by infusion of 3 ml of 3N KCl into the tail vein
- Evaluate the image stacks using a published, publically available Matlab-based computer algorithm that will create flow velocity grayscale and color encoded maps for all blood flow traces 10,11. Subsequently evaluate the resulting grayscale images by using commercial or publicly available image analysis software, such as Image J, after thresholding off values that indicate no movement of blood cells, i.e. no active vasculature.
Representative Results
The vasculature in solid tumors is known to differ significantly from normal blood supply, showing greater degrees of tortuosity, and higher intervascular distances13. Accordingly, blood flow tracks in experimental pulmonary breast cancer and sarcoma metastases have irregular shapes and large intervascular gaps (Figure 2A, lower two panels) when compared to the normal pulmonary microcirculation (Figure 2A, upper panel). In a previous study, the ability of the pulmonary window method was demonstrated to perform automated measurements of changes in the blood flow velocity in normal lungs10. In order to investigate whether the pulmonary window method can also measure an increase in blood flow velocity in pulmonary metastases, the drug combination of the sympathomimetic drug ephedrine and the endothelin blocker ambrisentan that has recently been found to increase the microvascular circulation in healthy lungs, was applied here (data under review elsewhere). In this study, the ability of the method is shown to detect an increase in blood flow velocity in pulmonary metastases of mouse sarcoma, caused by dosing with the combination of ephedrine (20 mg/kg) and the endothelin receptor antagonist ambrisentan (0.5 mg/kg, both drugs intraperitoneally, Figure 2B). Every data point represents the pooled individual averages of three measurements with five minute intervals, averaged over the data acquired in five animals. The first injection significantly (p <0.01) increased blood flow velocity in the tumor area from 0.61 ± 0.12 mm/sec to 0.74 ± 0.19, whereas the second injection maintained the elevated flow velocity at 0.74 ± 0.19 mm/sec (). The second injection did not elicit any further increase in blood flow velocity. N = 5, all statistics: repeated measures ANOVA.

Figure 1: Procedure to prepare a lung metastases-bearing rat for live microscopy imaging. (A) MicroCT image of a rat bearing lung metastases (arrow). (B) Pulmonary window frame. (C) Rat with chest wall perforation and pulmonary window attached. (D) Restraining device to limit Z-directional chest movement. (E) Rat with window chamber and restraining device under the fluorescence microscope. Note: panel C and E show hair bearing Sprague Dawley rats, not athymic nude rats.
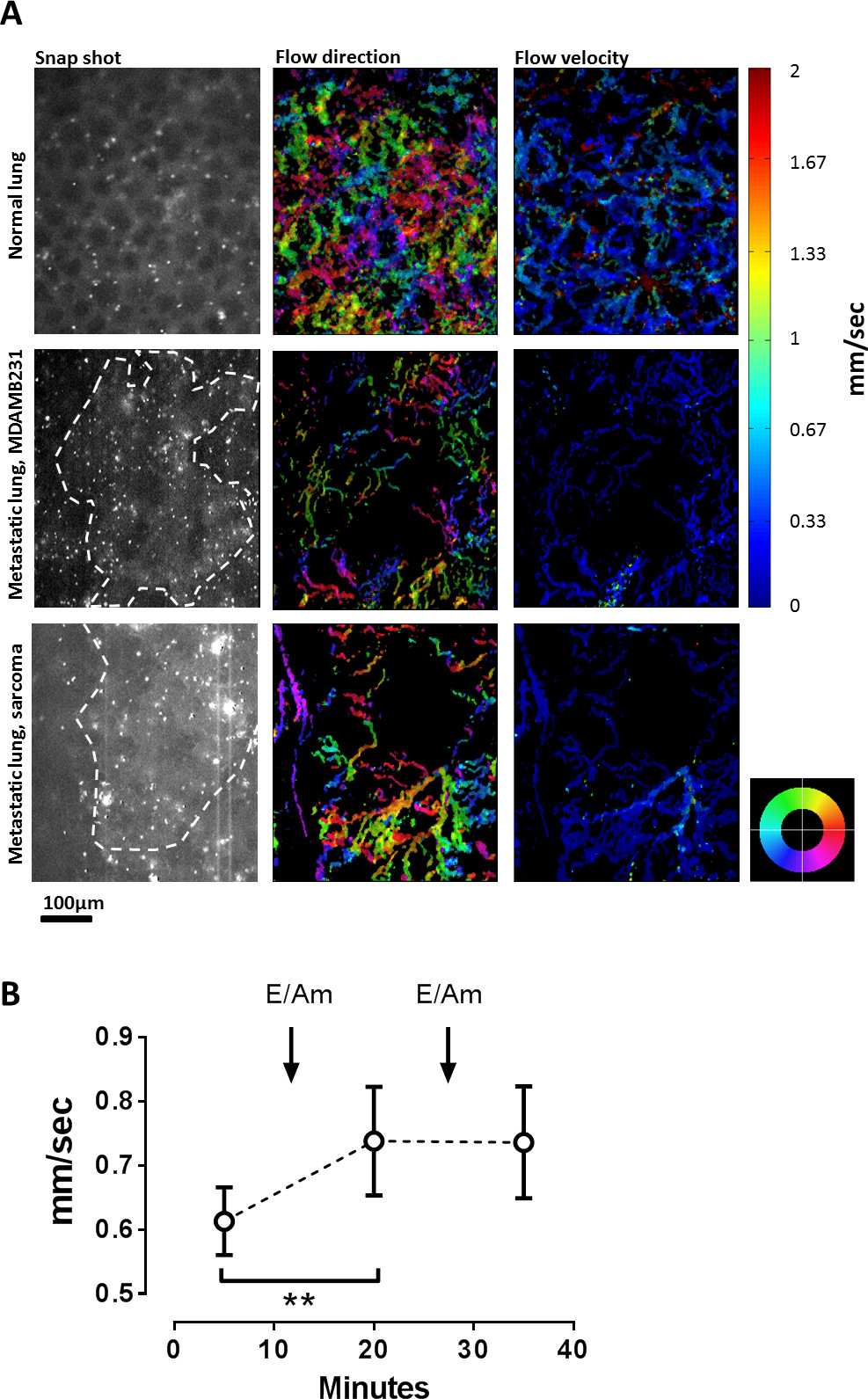
Figure 2: Quantification of increases in blood flow of pulmonary metastases. (A) blood flow direction and velocity in a normal lung, and a cancer metastasis from MDAMB-231 and mouse sarcoma. The column on the left side contains snap shots from image series of fluorescence micrographs. Alveolae can be identified as part of the background fluorescence pattern in normal lungs (upper panel). Metastases could be located by the fluorescence caused by endogenous fluorescing proteins of the injected cancer cells (dashed line). Columns to the right: color-encoded maps of blood flow direction (color indicator wheel marks the direction towards which cells flow) and flow velocity, measured in mm/second. (B) Changes in microcirculatory blood flow after repeated injection of combined ephedrine and ambrisentan (20 and 0.1 mg/kg per injection). A significant increase in overall flow velocity (repeated measures ANOVA, p <0.01) was observed after the first injection, but not after the second (N = 5).
Discussion
A model is presented that is feasible to image changes in microcirculatory blood flow and other dynamic processes in pulmonary metastases of rats, using intravital microscopy and computational blood flow analysis. While other methods exist to perform microscopy on exposed lungs in open ribcages of rodents, this model is also the first to image pulmonary metastases through a chest wall perforation in a closed-chest setting. Using this method, the feasibility is shown to measure pharmacologically induced changes in microcirculatory blood flow of pulmonary metastases.
Two basic methods exist to image the lungs of living rodents that are perfused by spontaneous blood flow, with direct contact to the lung surface: open chest models eliminate the problem of constant motion of the lung due to respiratory and cardiac activity by applying a suction window to the surface of the lung, while allowing the rest of the cardiopulmonary complex to expand and contract within an open chest cavity 7,14. While this method provides excellent experimental access, the open chest conditions and suction window introduce artifacts when compared to conditions of an intact lung8. Alternatively, closed-chest procedures that leave the rib cage largely intact bear the promise of preserving the original conditions in the intact chest cavity, such as the mutual influence of cardiac and pulmonary motion on each other’s blood flow dynamic. These models typically involve a frame with a window that is sutured to the chest wall15, or a transparent membrane that is brought in direct contact with the lung surface to prevent drying of the tissue9. The difficulty of negotiating the cardiopulmonary motion to accommodate and enable microscopy imaging is a major technical challenge of closed-chest procedures, and probably responsible for the low overall spread of this technique. In our case, the combination of a solid window-holding frame and a Z-directional restraining device has proven efficacious to sufficiently eliminate lateral movement of the lung to allow automated blood flow measurements of the pulmonary circulation in a closed-chest window10.
A common method to measure blood flow velocity from intravital microscopy image stacks is the use of spatial matching of blood flow patterns in consecutive images of a given vascular segment14. In order to reduce the time effort associated with the analysis of single blood vessels, a blood flow imaging algorithm has been introduced that produces blood flow velocity maps of the entire optical field11. Besides the time advantage, this method also allows simultaneous spatial analysis of blood flow in the whole optical field, irrespective of vascular morphology or branching points. This is particularly important when studying the tumor microenvironment, where multiple blood vessels contribute to the supply status of a given tumor area13,16,17. Indeed, the known tortuosity of tumor vasculature and the existence of large intervascular gaps can be clearly seen in both metastatic cancer types that have been investigated, (Figure 2A). The pulmonary window method has also been tested for its ability to report changes in blood flow velocity in the metastatic lesion, by measuring the effect of a pharmacological treatment that has recently been found to increase blood flow velocity in the pulmonary circulation: the hypertensive drug ephedrine increases cardiac output, whereas the endothelin receptor blocker ambrisentan reduces the pre-capillary arteriolar tone, which results, when the drugs are given in combination, in increased blood flow velocity in pulmonary capillaries. While these data are currently under review elsewhere, the ability of the combination of a hypertensive drug and an endothelin receptor antagonist to increase peripheral muscular perfusion has been published independently 18. While under normal conditions, a dose-dependent increase of blood flow velocity can be observed after both injections, the fact that the second injection does not lead to a further increase of flow velocity in sarcoma metastases might result from the fact that maximal tumor vessels vasodilation has already been reached after the first combined injection of ephedrine and ambrisentan.
The following limitations apply to the technique presented here: This protocol is applicable to rats (and theoretically, similar sized mammals) from approximately 180 to 300 g or more. Smaller mammals such as mice will require special modification to accommodate the more fragile anatomy and physiology of the rib cage. The maximum spatial resolution that can be achieved with the presented technique is theoretically only limited by the numerical aperture of the objective and the thickness of the cover slide that is used (~0.08-0.1 µm in standard brands), to 100X objectives, with the use of oil immersion. In practice however, the remaining movement of the lung might limit the use of high resolution objectives beyond 20X. If all autonomous motion of the lung is eliminated, the temporal resolution of the technique is only limited by the frame rate of the camera, and the signal strength of the fluorescent label, which determines the exposure time, to approximately 100 frames per sec. The following additional limitations apply to the presented technique: on one hand, the current microscopic and computational setting allows only the analysis of the surface of a given metastasis. The use of depth-penetrating microscopy, such as advanced confocal microscopy imaging strategies, may in the future enable measurement of blood flow velocity in 3 dimensions. Also, the presented technique cannot, in its current form, be used to visualize lesions further inside the lung parenchyma. Third, the insertion of a glass window in direct contact with active blood vessels has some potential to perturb the microvascular flow by itself, via spatial compression of blood vessels, or by impacting locoregional temperature. Fourth, the use of an external ventilator and positive pressure in the lung also has potential to alter pulmonary microcirculatory blood flow. Moreover, only limited areas of the rib cage are practically accessible for the described, relatively uncomplicated surgical procedures. Other areas of the lung surface, such as medial-ventral or dorsal access would require more elaborate surgical techniques, along with profound perturbations of the biomechanics of the rib cage. Because of the lack of alternatives to microscopy imaging to study microcirculatory blood flow, major developments can be expected in the near future to overcome these hurdles and study malignancies at high temporal-spatial resolution in all parts of the rodent lung.
In summary, a method is presented to measure patterns of, and changes in, microvascular blood flow velocity in surface metastases of the rat lung. Combining an acute closed-chest surgical model with an automated method of measuring blood flow velocity in the entire microscopic optical field, this technique offers relative preservation of the physiological environment of the lung, is capable of detecting changes in overall microcirculatory blood flow velocity and direction, and is relatively easy to use. It can be expected that this technique will be of great use to all groups that study the microcirculation of pulmonary metastases and other dynamic processes in this disease setting in rodent models.
Declarações
The authors have nothing to disclose.
Acknowledgements
The scientific advice of Drs. Timothy McMahon and Siqing Shan is appreciated. The presenters thank Drs. David Kirsch and Patricia Steeg for the generous gift of the fluorescently labeled Mouse Sarcoma and metastatic MDAMB-231 cells, respectively. This work was funded in part by the U.S. Defense Advanced Research Projects Agency (DARPA) Prime Award Number N66001-10-C-2134, and in part by the Department of Radiation Oncology, Duke University Medical Center.
Materials
| Name of Material/ Equipment | Company | Catalog Number | Comments/Description |
| Athymic nude rats | Charles River | Strain code 316 | Female 10 week-old athymic nude rats |
| micro-CT/micro-Irradiator | Precision X-ray Inc. | Xrad 225Cx | Use MicroCT to detect metastases |
| DiI (1,1=-dioctadecyl-3,3,3=,3=-tetramethyl-indocarbocyanine perchlorate) | Sigma Aldrich | 468495-100MG | Mix 100 ul packed red blood cells with 100 ul of 0.5 mg/ml DiI in 200 proof ethanol, 2 ml of 5% dextrose solution in water, and fill up to a 10-ml final volume with saline |
| Rodent ventilator | Kent Scientific | TOPO Small Animal Ventilator | Device is important to maintain positive lung pressure after application of pneumothorax |
| Zeiss Axioskop fluorescence microscope upright | Zeiss | Axioskop | Microscope for intravital imaging |
| Andor CCD camera | Andor | iXonEM 885 | CCD camera for live imaging of blood flow |
| Pulse oximeter | StarrLife | MouseOx | Pulse oximeter |
| Fluorescence microscope | Zeiss | Axioskop | Fluorescence microscope |
Referências
- Billingsley, K. G., et al. Pulmonary metastases from soft tissue sarcoma: analysis of patterns of diseases and postmetastasis survival. Ann Surg. 229, 602-610 (1999).
- Rashid, O. M., Takabe, K. The evolution of the role of surgery in the management of breast cancer lung metastasis. J Thorac Dis. 4, 420-424 (2012).
- Mayer, A., Vaupel, P. Hypoxia, lactate accumulation, and acidosis: siblings or accomplices driving tumor progression and resistance to therapy. Advances in experimental medicine and biology. 789, 203-209 (2013).
- Okunieff, P., O’Dell, W., Zhang, M., Zhang, L., Maguire, D. Tumor oxygen measurements and personalized medicine. Advances in experimental medicine and biology. 765, 195-201 (2013).
- Palmer, G. M., et al. In vivo optical molecular imaging and analysis in mice using dorsal window chamber models applied to hypoxia, vasculature and fluorescent reporters. Nature protocols. 6, 1355-1366 (2011).
- Palmer, G. M., et al. Optical imaging of tumor hypoxia dynamics. Journal of biomedical optics. 15, (2010).
- Funakoshi, N., et al. A new model of lung metastasis for intravital studies. Microvasc Res. 59, 361-367 (2000).
- Kuebler, W. M. Real-time imaging assessment of pulmonary vascular responses. Proc Am Thorac Soc. 8, 458-465 (2011).
- Tabuchi, A., et al. Precapillary oxygenation contributes relevantly to gas exchange in the intact lung. Am J Respir Crit Care Med. 188, 474-481 (2013).
- Hanna, G., et al. Automated measurement of blood flow velocity and direction and hemoglobin oxygen saturation in the rat lung using intravital microscopy. American journal of physiology. Lung cellular and molecular physiology. 304, 86-91 (2013).
- Fontanella, A. N., et al. Quantitative mapping of hemodynamics in the lung, brain, and dorsal window chamber-grown tumors using a novel, automated algorithm. Microcirculation. , (2013).
- Newton, J., et al. Commissioning a small-field biological irradiator using point, 2D, and 3D dosimetry techniques. Medical physics. 38, 6754-6762 (2011).
- Vaupel, P. Tumor microenvironmental physiology and its implications for radiation oncology. Seminars in radiation oncology. 14, 198-206 (2004).
- Tabuchi, A., Mertens, M., Kuppe, H., Pries, A. R., Kuebler, W. M. Intravital microscopy of the murine pulmonary microcirculation. J Appl Physiol. 104, 338-346 (2008).
- Fingar, V. H., Taber, S. W., Wieman, T. J. A new model for the study of pulmonary microcirculation: determination of pulmonary edema in rats. J Surg Res. 57, 385-393 (1994).
- Manzoor, A. A., Schroeder, T., Dewhirst, M. W. One-stop-shop tumor imaging: buy hypoxia, get lactate free. The Journal of clinical investigation. 118, 1616-1619 (2008).
- Evans, S. M., et al. Imaging and analytical methods as applied to the evaluation of vasculature and hypoxia in human brain tumors. Radiation research. 170, 677-690 (2008).
- Radiloff, D. R., et al. The combination of theophylline and endothelin receptor antagonism improves exercise performance of rats under simulated high altitude. Journal of applied physiology. 113, 1243-1252 (2012).

