Real-time X-ray Imaging of Lung Fluid Volumes in Neonatal Mouse Lung
Summary
We present a protocol to assess the rate of alveolar fluid clearance or pulmonary edema in neonatal mouse lung using X-ray imaging technology.
Abstract
At birth, the lung undergoes a profound phenotypic switch from secretion to absorption, which allows for adaptation to breathing independently. Promoting and sustaining this phenotype is critically important in normal alveolar growth and gas exchange throughout life. Several in vitro studies have characterized the role of key regulatory proteins, signaling molecules, and steroid hormones that can influence the rate of lung fluid clearance. However, in vivo examinations must be performed to evaluate whether these regulatory factors play important physiological roles in regulating perinatal lung liquid absorption. As such, the utilization of real time X-ray imaging to determine perinatal lung fluid clearance, or pulmonary edema, represents a technological advancement in the field. Herein, we explain and illustrate an approach to assess the rate of alveolar lung fluid clearance and alveolar flooding in C57BL/6 mice at post natal day 10 using X-ray imaging and analysis. Successful implementation of this protocol requires prior approval from institutional animal care and use committees (IACUC), an in vivo small animal X-ray imaging system, and compatible molecular imaging software.
Introduction
At birth, the newborn lung must transition from a fluid secreting to a fluid reabsorbing organ to establish adequate ventilation and oxygenation of the body. The mechanisms that facilitate (or hinders) effective clearance of lung fluid at the time of birth remain unclear. Modeling the rate of alveolar fluid clearance in C57BL/6 newborn mouse pups will lead to a better understanding of regulatory factors that can enhance or attenuate the rate of fluid absorption. It could also be applied to other neonatal models of acute lung injury or infection, and could lead to novel therapeutic strategies for newborn infants with respiratory distress.
Since newborn lungs are minuscule compared to adult lungs, conventional measures of alveolar fluid clearance that rely on lavage or gravimetrical measurements may not be suitable to accurately study lung fluid clearance in neonatal lung models. In this protocol, we demonstrate an assay that allows for the accurate determination of alveolar fluid clearance rates in postnatal day 10 C57BL/6 mouse pups using a small animal imager. One major benefit of using a fluoroscopic approach is that the animals are imaged in vivo. They are freely breathing and may recover from this minimally invasive assay for future observation and study. The overall goal of this method is to model pulmonary edema in the newborn lung, and evaluate the rate of alveolar fluid clearance in neonatal lung. This technique was developed, in part, as a reduction strategy to decrease the number of animals needed, yet maximize experimental output. This technique also allows for superior detection of lung fluid volumes using X-ray imaging and requires proficiency in basic animal restraint and handling1; small animal surgeries and tracheal instillation2, a small animal imager, and basic image analysis software. Investigators who wish to evaluate lung fluid volumes in vivo (freely breathing anesthetized animal models) may find this procedure suitable for their application. Lastly, this protocol could augment other existing models of neonatal lung injury used in the mechanistic study of bronchopulmonary dysplasia, including hyperoxia-induced lung injury, mechanical ventilation, and models of lung inflammation3.
Protocol
All experimental techniques must be conducted in accordance with Institutional Care and Use Committee guidelines.
1. X-ray Imaging Acquisition
- Software Overview (See Figure 1 for an Overview of Software Settings).
- Select the Capture button directly below the File tab.
- In the Settings drop-down menu, select Current Session.
- Select Standard Exposure in drop-down box located under Exposure Time.
- Set the Exposure Time to 2.00 min, and No. Exposures para 1. Set the X Binning and Y Binning to 2 pixels. See Figure 1.
- Select Final Accumulation from the Export Options drop-down menu.
- In the Illumination settings, select X-Ray from the Illumination Source drop-down menu. The default KVP is set at 35.
- Select Autoselect in the Apply Reference File drop-down menu to apply the corresponding X-ray reference file. (See Section 2).
- In the Set Camera To settings, set the f-stop to 2.51.
- Set the FOV (Field of View) to 125.64 for PN10 mice.
- Set the Focal Plane to 13, and in the drop-down menu beside the Focal Plane slider, select X-Ray.
- For X-rays, set the Excitation Filter and the Emission Filter to 0 in the drop-down menu.
- Save current session by clicking novo on the top next to Settings. Enter name for New Acquire Settings File, e.g., "Jove Single Exposure Event for making a Protocol." See Figure 2.
- Creating an X-ray Imaging Protocol
- Click on the Create/Edit Protocols button on the right side of the open window.
- As new Protocol pop-up window appears, click the novo button in the upper right-hand corner
- Enter the name that the protocol will save under, for example "Jove Demo Protocol", and click OK. (See Figure 3).
- On the bottom of the Protocol pop-up window in Protocol Steps, ensure that Step 1 is highlighted in red text, Capture New Images is selected, and Do Nothing is selected in the Before Image Capture drop down menu.
- In Capture Setting, select the recently generated single exposure event from Step 1.1.12.
- Select Wait (sec) from the After Image Capture drop-down menu.
- Click the Edit button, type 180 into the pop-up box, and click OK to add a 3 min wait time after each 2 min acquisition.
- Duplicate Step 1 by right clicking the Step 1 tab and selecting Duplicate Step. Create 23 duplicates for a 2 hr observation period. (See Figure 4).
- On the final step (Step 24), change the After Image Capture setting to Do Nothing from Wait (sec).
- Click the Save button and exit the Protocol Editor.
2. Illumination Reference Files
Note: Apply X-ray illumination reference files to an X-ray image in order to automatically correct for variations in detector uniformity of the X-ray images obtained throughout experiment. The procedures outlined below are specific to the Bruker In Vivo Animal Imaging Systems; other in vivo imaging systems may be used.
- Generate an illumination reference file by opening the molecular imaging software and clicking on the Capture button.
- Using the pop-up menu, establish the X-ray capture settings in Illumination Source (see suggested settings in section 1 above) and then select Illumination Reference in the exposure type. This can be found under the Standard Exposures drop down box. (See Figure 5).
- Remove all samples from the image station. Set the X and Y binning to 4 x 4 binning. Refer to Table 1: Illumination reference file exposure times to determine the accurate exposure time.
- Press Expose.
- Apply the reference file by selecting Auto Select from the drop down menu under Apply Reference File. (See Figure 6). The illumination reference file will now be automatically applied to all X-ray images captured with the same camera settings. Steps 2.1 – 2.4 will not need to be repeated if same camera settings are utilized in subsequent experiments.
Note: An illumination reference file can be applied after image acquisition, or if an error message occurs after auto-selection of reference files. Apply illumination reference files post-image capture using the following series of commands in the Navigation Panel of the molecular imaging software: Image > Image Math > Type: Task > Calculate: Illumination correction. Select the input image (X) that you would like to apply the illumination reference file (Y) to. Rename the corrected file (Z). (See Figure 7).
3. Animal Handling
- Acquiring Animals
- Purchase pregnant dams from commercial breeders or breed female mice in house at 12 weeks of age (or older) in accordance with institutional guidelines.
- House newborn mice with lactating mothers up to postnatal day (PN) 10.
- Animal Anesthesia (PN 10)
- Prepare a Ketamine/Xylazine cocktail to anesthetize PN 10 mice for prolonged anesthetic effects lasting up to 40 min. Add 500 µl of Ketamine (100 mg/ml) to 75 µl of Xylazine (100 mg/ml). Dilute 1:10 in a 0.9% saline solution to make a Ketamine (100 mg/kg)/Xylazine (10 mg/kg) anesthetic cocktail.
- Weigh the newborn mice.
- Using a 3/10 syringe with a 31 G 5/16 inch (8 mm) needle, administer 10 µl/g of body weight of anesthesia with an intraperitoneal injection.
- Keep animals dry and insulated to prevent excessive loss of body heat.
4. Tracheal Instillations
- Prepare an intratracheal saline solution comprised of 140 mM NaCl, 5 mM KCl, 1 mM CaCl2, and 10 mM HEPES; pH = 7.4. The osmolality of this solution should be 319 mosmol/kg H2O.
- Mount anesthetized animals ventral side up onto a sloped surgical board using surgical tape. Ensurethat the animals' heads are at the top of the incline.
- Perform a toe pinch to ensure that animals are anesthetized and ready for surgery. Disinfect all surgical areas, instruments, and thoracic region of the animal.
- Make a small (3 mm) incision in the anterior-medial ventral aspect of the neck (throat region) using a surgical scalpel, size 11. Push aside the platysma and anterior tracheal muscles using blunted forceps in order to visualize and access the trachea.
- Instill 3 µl/g of weight (approximately 10 – 30 µl final volume) of saline via the exposed trachea using a 31 G 5/16 inch (8 mm) needle. The minor incision is left open during animal imaging and usually heals itself well. Consult with local Division of Animal Resource to determine if the incision can likewise be left open. Otherwise, sutures may be required.
5. Animal Imaging
- Position the pups ventrally on a clear-bottom, removable, animal imaging tray. Center the animals so that the X-ray beam will be directly over the thoracic area.
- For odd numbered animal cohorts, place the first pup directly in the middle of the tray, and for even numbered cohorts place the first pup just left of center so that when the other animals are placed on the tray all of the animals are centered.
- Return the imaging tray to the X-ray imaging cabinet and close the cabinet door.
- Turn on the animal thermal control unit to maintain the anesthetized animals' body temperature. Use the high setting to achieve a chamber temperature of approximately 35 – 37 °C. Turn on animal anesthesia unit (vaporized isoflurane delivered via oxygen) to ensure animals are anesthetized and immobilized throughout duration of 2 hr imaging procedure.
- Executing X-ray imaging protocol
- Click on Capture and select the appropriate protocol, e.g., "Jove Demo Protocol," from the Protocol drop-down menu. (See Figure 8).
- Click the Execute Selected Protocol button on the molecular imaging software.
Note: A pop-up window will appear to track the status of the image acquisition, when the protocol is complete the window will disappear. The X-ray dose is low, < than 0.3 mRem or about 10 times less than a dental X-ray. As with other X-Ray procedures, there is no residual radiation. - When the 2 hr acquisition session is complete, remove the animals from the imaging tray and return them to their cage. Monitor animals for full recovery before returning to racks.
6. Data Analysis
Note: Molecular imaging software allows for quantification and the translation of X-ray pixel intensity into rate of lung fluid clearance. The steps below outline the procedures needed to normalize X-ray images and quantify intensities in defined regions of interest (ROI).
- Design an ROI template
Note: A region of interest template must be created specific to the captured X-ray images during the 2 hr study, and should be used in order to compare X-ray intensities amongst experimental groups. Since small volumes of saline challenges typically accumulate in the left upper lobe of the lung4-6, the ROI(s) should focus on this portion of the lung.- Open the first and last x-ray image in the 2 hr set. Select the window of the first x-ray image.
- In the Navigation toolbar, select Manual-ROIs > New ROI Set.
- Click ROI Ellipse and create a ROI that adequately covers the mouse's left lung.An ROI is not defined until the red, outlined ROI is dragged to a different position. A defined ROI will be outlined in blue with a number.
- If multiple pups are imaged, click on the red, outlined ROI and drag to the left lungs of the other mice to create more individual ROIs in the same set. Drag the red, outlined ROI to an area with a clear background to create a background ROI.
- Select Pointer Selection to position the ROIs to lie over each mouse's left lung, directly under the second rib.
- In the top toolbar, click Image Display.
- Check Overlay in the Image Display dialog to overlay the last x-ray image while maintaining the set ROI locations. If necessary, select Pointer Selection in the Navigation toolbar to adjust the positions of the ROIs and ensure adequate lung coverage in both images.
- In the Manual ROI dialog, select Template > Save to Template. Name the template and click OK.
- Close both images. Select No when prompted to save changes.
- Apply the ROI template to each X-ray image captured to analyze fluid clearance by opening all images taken during the study. Start by selecting an open file and click Manual ROIs > Template.
- Select the previously created ROI template from the drop down menu and click Apply to All Open Documents.
- Export Numerical ROI Data from the Images to a Spreadsheet
- In the upper left hand corner, click File > Export Data > ROI.
- Check As Displayed and Auto Open in Excel in the pop-up dialog.
- Select Export All Open Documents.
- Name the file and click Save.
- Close molecular imaging software.
Representative Results
The left panels in Figures 9 – 10 are of PN 10 mouse lungs imaged at baseline (pre-instilled). These images show successful instillation of saline challenges into the left lobe of the neonatal lungs. In Figure 9, the mouse lung was tracheally instilled with the saline solution defined above (see section 2.1). The middle and right panels of Figure 9 are X-ray images from the same mouse obtained 5 min and 2 hr post instillation; this animal had successfully cleared the saline challenge. Specifically, the X-ray intensity of this animals ROI increased from 187.67 to 515. Thus, there is a reverse correlation between pixel density and lung fluid volume; that is to say, the larger the relative value, the less fluid there is in the lungs. It may be helpful to understand that more X-ray energy is absorbed (hence a larger reported value) when there is less fluid attenuating the X-ray. In Figure 10, the PN 10 mouse lung was tracheally instilled with a compound containing oxidized glutathione (reconstituted in saline described in 2.1) that inhibited alveolar fluid clearance of the saline challenge by blocking epithelial sodium channel activity; the numerical value of this animal's ROI will decrease from the pre-instill and post-instilled X-ray imaged files, indicative of increasing X-ray opacity. Specifically, the net intensity of the animal approximately 5 min post-instillation was – 64, and decreased to – 182. Again, note the inverse relationship between the ROI pixel intensity and amount of fluid in the lungs; increased fluid in the upper left lobe of the lung attenuates X-ray absorbtion.
Evaluating net intensity of the ROI enables quantitative evaluation of changes in the rate of lung fluid clearance, albeit acquisition software also allows the investigators to express data in terms of g/cm3 if desired. Moreover, the investigators can use each animal as its own control and normalize all X-ray intensities to an initial time point (Io), such as t = 5 min and report net changes in X-ray opacity (i.e., a measure of change in lung fluid volumes).
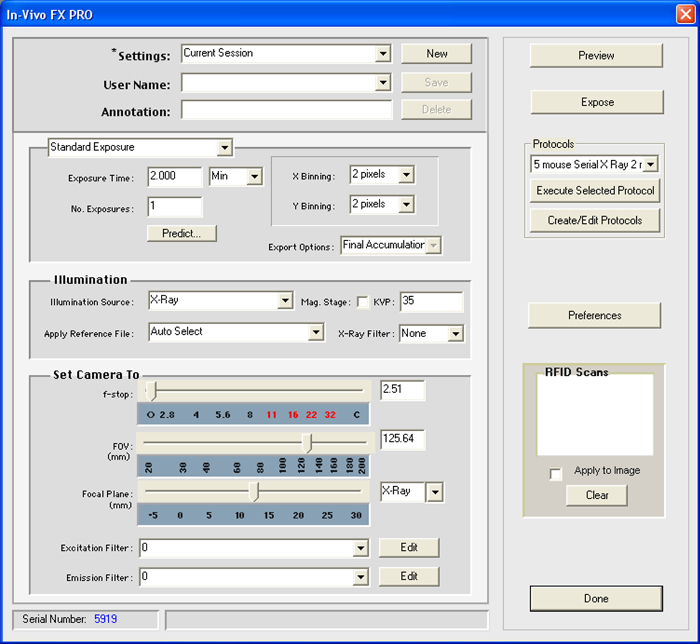
Figure 1. Exposure Settings. This screen shot illustrates the appropriate exposure settings utilized in this protocol. Please click here to view a larger version of this figure.
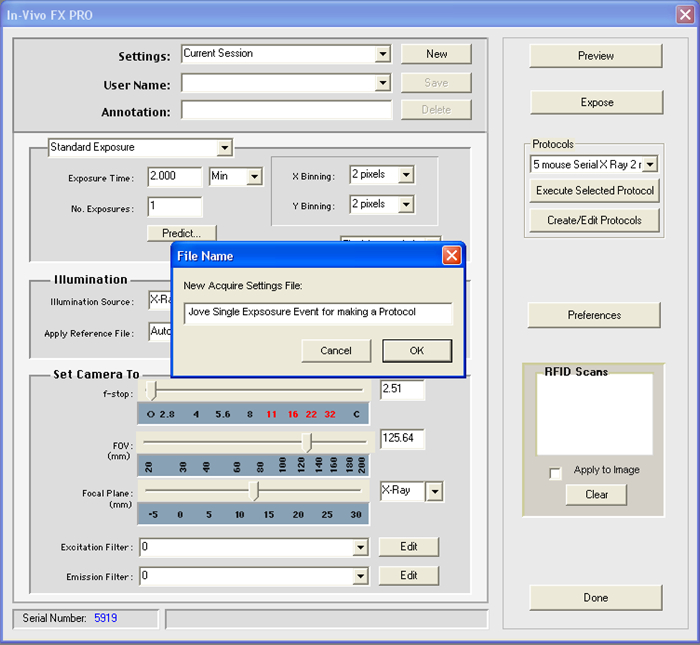
Figure 2. Settings File. This screen shot illustrates a key step in generating a file setting that will be used in a protocol. A pop up window (as shown) will request a new name for the acquisition settings file. Please click here to view a larger version of this figure.
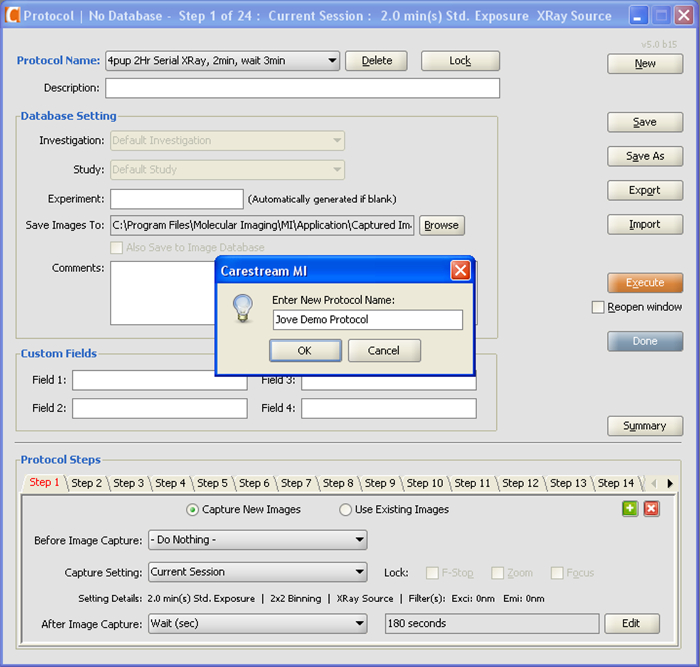
Figure 3. Imaging Protocol. This screen shot illustrates a key step in determining whether a new imaging protocol has been successfully created. A pop up window (as shown) will appear and a new protocol name will be requested for the generated protocol. Please click here to view a larger version of this figure.
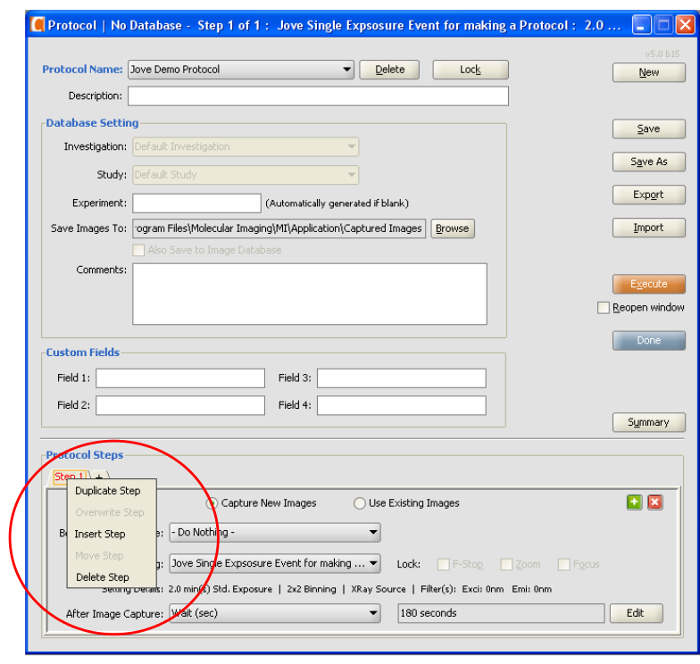
Figure 4. Protocol Steps. This screen shot illustrates a shortcut to duplicate an acquisition settings file, insert a new step, or to delete a step within an imaging protocol. Please click here to view a larger version of this figure.
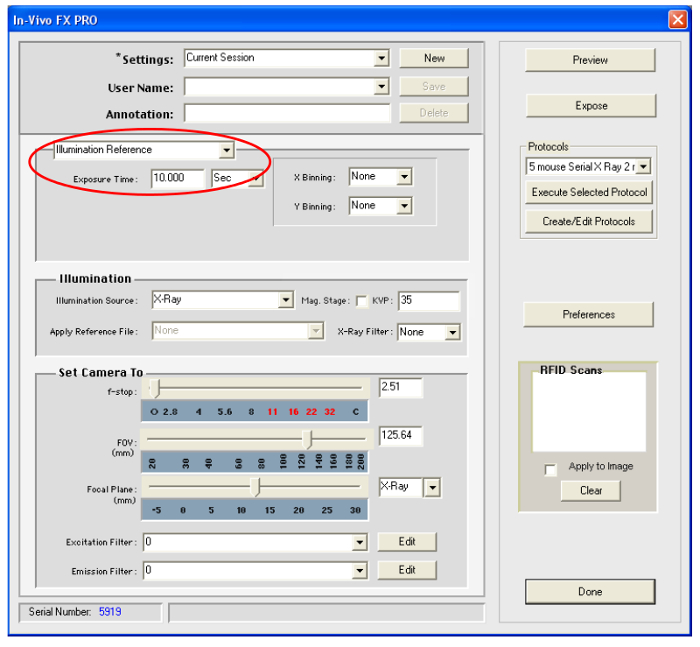
Figure 5. Illumination Reference. This screen shot exhibits the illumination reference command and appropriate settings in the animal imaging software appropriate for creating an illumination reference file. Please click here to view a larger version of this figure.
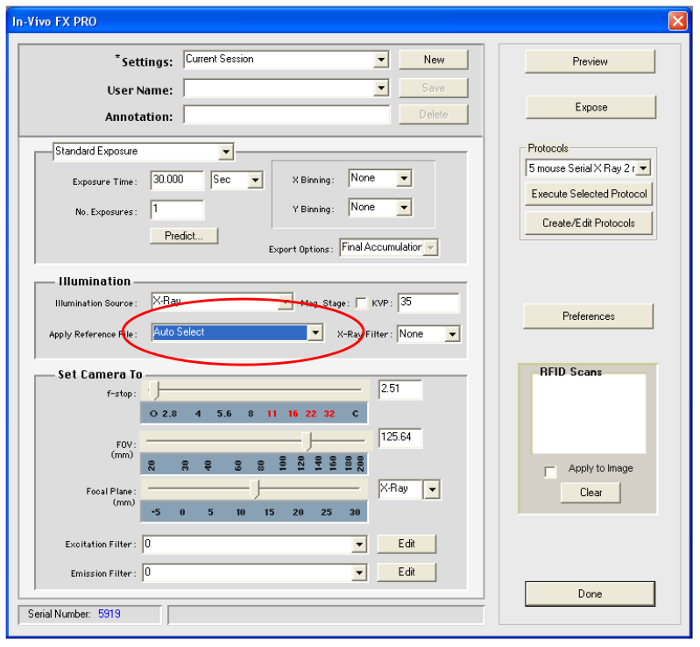
Figure 6. Auto Select. This screen shot exhibits the Auto Select function and appropriate settings in the animal imaging software appropriate for applying an illumination reference file. Please click here to view a larger version of this figure.
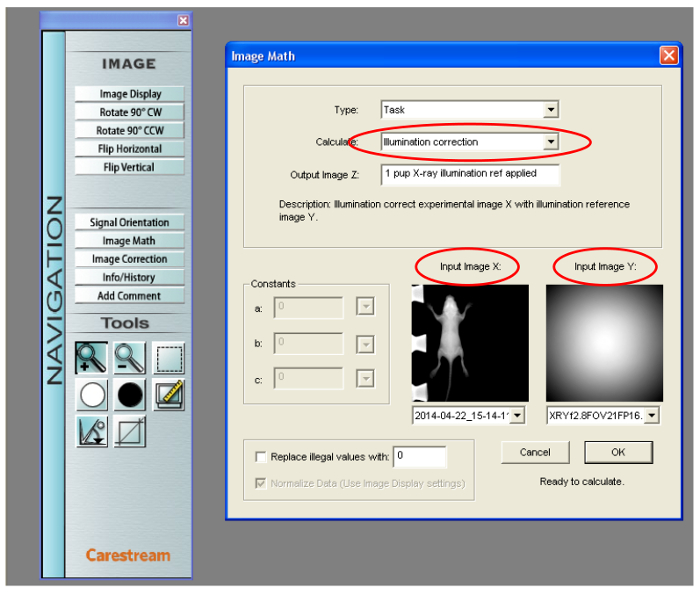
Figure 7. Illumination Correction. This screen shot illustrates the appropriate application of an illumination reference file generated after animal imaging. Please click here to view a larger version of this figure.
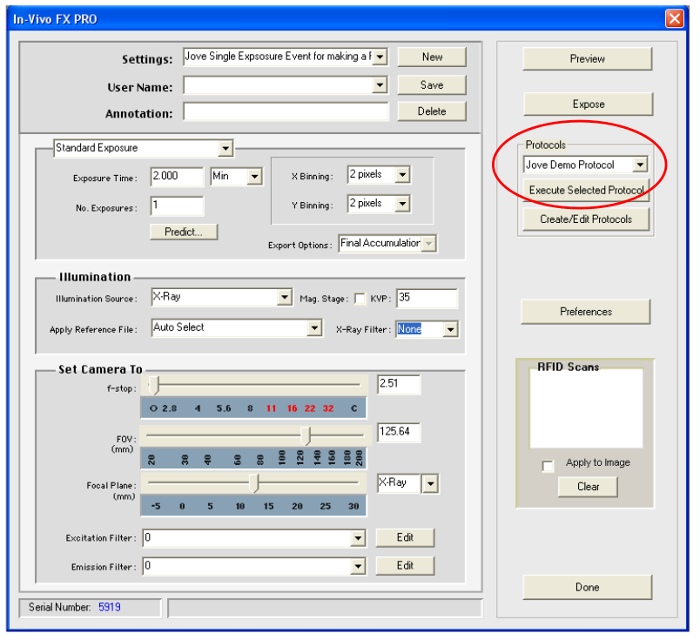
Figure 8. Execute Protocol. This screen shot illustrates how to execute a selected protocol. Please click here to view a larger version of this figure.
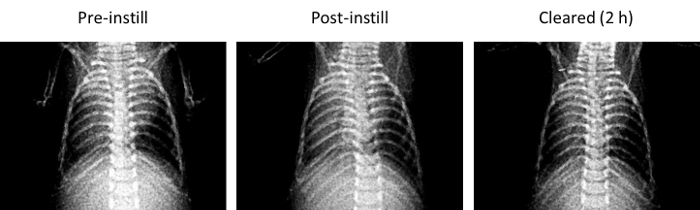
Figure 9. X-ray images of cleared lungs. Representative image of PN 10 lungs prior to receiving a saline challenge (pre-instill; left panel); 5 min post-instillation (middle panel), and 2 hr after the saline challenge had cleared from the otherwise healthy lung (right panel). Please click here to view a larger version of this figure.
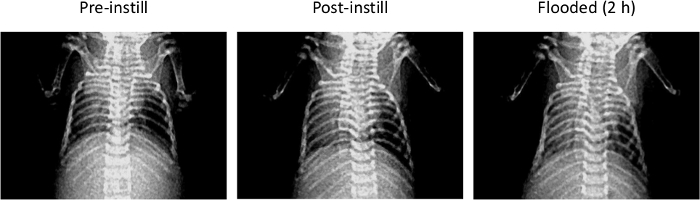
Figure 10. X-ray Images of Flooded Lungs. Representative image of PN 10 lungs prior to receiving a saline challenge (pre-instill; left panel) containing glutathione disulfide , which inhibits paracellular solute transport; 5 min post-instillation of glutathione disulfide (middle panel), and 2 hr after inhibiting paracellular transport which leads to alveolar flooding (right panel). Please click here to view a larger version of this figure.
| No filter= | 10 sec exposure |
| 0.1mm = | 15 sec exposure |
| 0.2mm = | 20 sec exposure |
| 0.4mm = | 30 sec exposure |
| 0.8mm= | 30 sec exposure |
| The size of the X-ray filter correlates to a specific exposure time for creating an illumination reference file. | |
Table 1. Illumination Reference File. This file reports the appropriate exposure times for generating illumination reference files based on X-ray filters used in imaging studies.
Discussion
Using X-ray imaging, clear images of neonatal lungs can be analyzed for lung fluid volumes. We7,3,11, and others10, have successfully utilized X-ray imaging to determine dynamic changes in lung fluid volume in freely breathing anesthetized animal models, and this technique holds great promise to advance the study of neonatal lung injury. One major advantage in using our approach to assess lung fluid volume (as opposed to x-ray phase constrast10 for example) is that up to five PN10 mouse pups can be simultaneously studied using an imaging system that is common place in research facilities and cores.
Instilling an appropriate lung fluid volume, as to not drown or collapse the lung, is critical to the successful implementation of this protocol and may need to be experimentally explored before X-ray imaging protocols can be applied. The sensitivity of this assay allows for the detection of very small volumes of instilled saline via X-ray detection. We have been able to discern differences in the X-ray opacity of neonatal lungs instilled with 10 μl volumes of saline solution. The difference in X-ray opacities are even more pronounced when sodium channel inhibitors are introduced into the alveolar airspace because the saline challenges cannot be absorbed and the lungs continue to secrete fluid into the airspace. In the event that an inappropriately high volume of saline is introduced, placing animals into the imaging chamber with oxygen flowing into the chamber via the anesthesia ports can facilitate respiration in flooded lungs.
Our results using X-ray imaging are comparable to alveolar fluid clearances measured using more conventional approaches, such as lung wet-to-dry weight ratios and Evan's Blue for the determination of protein concentration4. We now demonstrate that this approach can be applied to the neonatal mouse pup. This X-ray imaging technique for determining lung fluid volumes can easily be combined with additional imaging modalities. For instance, fluorescent markers or bioluminescent probes can be simultaneously instilled into the alveoli and assessed. (Detection of fluorescent and luminescent probes has been described8, and is beyond the scope of this report). The ability to co-register the volume of lung fluid (using X-ray imaging) alongside the ability to detect fluorescent biomarkers is one of several advantages of using this dynamic assay and the commercial system for measuring lung fluid clearance. Other benefits of utilizing this approach for determining clearance and relative lung fluid volume includes the ability to conduct longitudinal studies (thus decreasing the number of animals needed to achieve statistically significant observations), and the ability to detect small changes in lung fluid volume in freely breathing, anesthetized, neonatal mouse pups. One limitation of using an in vivo imaging approach, however, is that the anesthesia can alter the distribution of gas and blood flow within the lungs. Mismatches in ventilation and perfusion (V/Q) and shunting have been shown to increase under anesthesia in healthy adult volunteers12, thus reducing oxygenation of the body. This adverse effect, however, can be compensated for by increasing the inspired oxygen concentration. From a technical viewpoint, variability between imaging systems in X-ray flux energy may require optimization of each system prior to performing imaging studies. For example, on a system with an X-ray source with more flux and/or a detector with superior quantum efficiency, a higher f/stop and lower binning state might provide better image quality when assessing small change in X-ray impedance.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work is supported by a grant awarded to MNH by Children’s Healthcare of Atlanta, the APS 2014 Short-Term Research Education Program to Increase Diversity in Health-Related Research (STRIDE) fellowship awarded to PT, and the University of Notre Dame Integrated Imaging Facility.
Materials
| Preclinical Imaging System (In- Vivo MS FX PRO) | Bruker; Billerica, MA | |
| Ketamine | Ketaset; Fort Dodge Animal Health, IA | 26637-411-01 |
| Xylazine | Lloyd Laboratories; Shenandoah, IA | 4821 |
| Dulbecco's Phosphate Buffered Saline (with Calcium and Magnesium) | Lonza; Walkersville, MD | 17-513F |
| Sodium chloride | Amresco; Solon, OH | 241 |
| Potassuim chloride | Fisher Scientific; Fair Lawn, NJ | P217-3 |
| Calcium chloride | Sigma-Aldrich; St. Loius, MO | C5080 |
| HEPES | Sigma-Aldrich; St. Loius, MO | H3375 |
| 0.3 mL insulin syringe with 31Gx5/16" (8mm) needle | BD Insulin Syringe; Franklin Lakes, NJ | 328438 |
Referências
- Institute of Laboratory Animal Resources. . Guide for the Care and Use of Laboratory Animals. , (2011).
- Helms, M. N., Torres-Gonzalez, E., Goodson, P., Rojas, M. Direct tracheal instillation of solutes into mouse lung. J Vis Exp. , (2010).
- Hilgendorff, A., Reiss, I., Ehrhardt, H., Eickelberg, O., Alvira, C. M. Chronic lung disease in the preterm infant. Lessons learned from animal models. Am J Respir Cell Mol Biol. 50, 233-245 (2014).
- Goodson, P., et al. Nadph oxidase regulates alveolar epithelial sodium channel activity and lung fluid balance in vivo via O2- signaling. Am J Physiol Lung Cell Mol Physiol. 302, L410-L419 (2012).
- McNeilly, T. N., Tennant, P., Lujan, L., Perez, M., Harkiss, G. D. Differential infection efficiencies of peripheral lung and tracheal tissues in sheep infected with Visna/maedi virus via the respiratory tract. J Gen Virol. 88, 670-679 (2007).
- Starcher, B., Williams, I. A method for intratracheal instillation of endotoxin into the lungs of mice. Lab Anim. 23, 234-240 (1989).
- Downs, C. A., Kumar, A., Kreiner, L. H., Johnson, N. M., Helms, M. N. H2O2 regulates lung ENaC via ubiquitin-like protein Nedd8. J Biol Chem. 288, 8136-8145 (2013).
- Gammon, S. T., et al. Preclinical anatomical, molecular, and functional imaging of the lung with multiple modalities. Am J Physiol Lung Cell Mol Physiol. 306, L897-L914 (2014).
- Takemura, Y., et al. Cholinergic regulation of epithelial sodium channels in rat alveolar type 2 epithelial cells. Am J Physiol Lung Cell Mol Physiol. 304, L428-L437 (2013).
- Lewis, R. A., et al. Dynamic imaging of the lungs using x-ray phase contrast. Phys Med Biol. 50, 5031-5040 (2005).
- Nyren, S., Radell, P., Mure, M., Petersson, J., Jacobsson, H., Lindahl, S. G., Sanchez-Crespo, A. Inhalation anesthesia increases V/Q heterogeneity during spontaneous breathing in healthy subjects. Anesthesiology. 113 (6), 1370-1375 (2010).

