Synthesis of Core-shell Lanthanide-doped Upconversion Nanocrystals for Cellular Applications
Summary
A protocol is presented for the synthesis of core-shell lanthanide-doped upconversion nanocrystals (UCNs) and their cellular applications for channel protein regulation upon near-infrared (NIR) light illumination.
Abstract
Lanthanide-doped upconversion nanocrystals (UCNs) have attracted much attention in recent years based on their promising and controllable optical properties, which allow for the absorption of near-infrared (NIR) light and can subsequently convert it into multiplexed emissions that span over a broad range of regions from the UV to the visible to the NIR. This article presents detailed experimental procedures for high-temperature co-precipitation synthesis of core-shell UCNs that incorporate different lanthanide ions into nanocrystals for efficiently converting deep-tissue penetrable NIR excitation (808 nm) into a strong blue emission at 480 nm. By controlling the surface modification with biocompatible polymer (polyacrylic acid, PAA), the as-prepared UCNs acquires great solubility in buffer solutions. The hydrophilic nanocrystals are further functionalized with specific ligands (dibenzyl cyclooctyne, DBCO) for localization on the cell membrane. Upon NIR light (808 nm) irradiation, the upconverted blue emission can effectively activate the light-gated channel protein on the cell membrane and specifically regulate the cation (e.g., Ca2+) influx in the cytoplasm. This protocol provides a feasible methodology for the synthesis of core-shell lanthanide-doped UCNs and subsequent biocompatible surface modification for further cellular applications.
Introduction
In recent years, lanthanide-doped upconversion nanocrystals (UCNs) have been widely used as an alternative to conventional organic dyes and quantum dots in biomedical applications, which are mainly based on their outstanding chemical and optical properties, including great biocompatibility, high resistance to photobleaching, and narrow-bandwidth emission1,2,3. More importantly, they can serve as a promising nanotransducer with excellent tissue penetration depth in vivo to convert near-infrared (NIR) excitation into a broad range of emissions from the UV, visible, and the NIR regions through a multi-photon upconversion process4,5. These unique properties make lanthanide-doped UCNs serve as a particularly promising vector for biological sensing, biomedical imaging, and diseases theranostics6,7,8.
The general components of UCNs are mainly based on the doped lanthanide ions in the insulating host matrix containing a sensitizer (e.g., Yb3+, Nd3+) and an activator (e.g., Tm3+, Er3+, Ho3+) within the crystal homogeneously9. The different optical emission from the nanocrystals is attributed to the localized electronic transition within the 4f orbitals of the lanthanide dopants due to their ladder-like arranged energy level10. Therefore, it is critical to precisely control the size and morphology of synthesized UCNs with multicomponent lanthanide dopants. By right, some promising methods have been well established for the preparation of lanthanide-doped UCNs, including thermal decomposition, high-temperature co-precipitation, hydrothermal synthesis, sol-gel processing, etc.11,12,13 Among these approaches, the high-temperature co-precipitation method is one of the most popular and convenient strategies for UCNs synthesis, which can be strictly controlled to prepare desired high-quality nanocrystals with uniform shape and size distribution in a relatively short reaction time and low-cost14. However, most nanostructures synthesized by this method are mainly capped with hydrophobic ligands such as oleic acid and oleylamine, which generally hinder their further bioapplication due to the limited of hydrophobic ligand solubility in aqueous solution15. Therefore, it is necessary to perform suitable surface modification techniques to prepare biocompatible UCNs in biological applications in vitro and in vivo.
Herein, we present the detailed experimental procedure for the synthesis of core-shell UCNs nanostructures through the high-temperature co-precipitation method and a feasible modification technique to functionalize biocompatible polymer on UCNs surface for further cellular applications. This UCNs nanoplatform incorporates three lanthanide ions (Yb3+, Nd3+, and Tm3+) into the nanocrystals to acquire strong blue emission (~480 nm) upon NIR light excitation at 808 nm, which has greater penetration depth in living tissue. It is well known that Nd3+-doped UCNs display minimized water absorption and overheating effects at this spectral window (808 nm) as compared to conventional UCNs upon 980 nm irradiation16,17,18. Moreover, to utilize the UCNs in biological systems, the hydrophobic ligands (oleic acid) on the surface of UCNs are firstly removed by sonication in acid solution19. Then the ligand-free UCNs are further modified with a biocompatible polymer (polyacrylic acid, PAA) to acquire great solubility in aqueous solutions20. Furthermore, as a proof-of-concept in cellular applications, the hydrophilic UCNs are further functionalized with molecular ligands (dibenzyl cyclooctyne, DBCO) for specific localization on the N3-tagged cell membrane. Upon NIR light (808 nm) irradiation, the upconverted blue emission at 480 nm can effectively activate a light-gated channel protein, channelrhodopsins-2 (ChR2), on cell the surface and thus facilitate cation (e.g., Ca2+ ion) influx across the membrane of living cells.
This video protocol provides a feasible methodology for lanthanide-doped UCNs synthesis, biocompatible surface modification, and UCNs bioapplication in living cells. Any differences in the synthesis techniques and chemical reagents used in nanocrystal growth will influence the size distribution, morphology, and upconversion luminescence (UCL) spectra of final UCNs nanostructures used in cell experiments. This detailed video protocol is prepared to help new researchers in this field to improve the reproducibility of UCNs with the high-temperature co-precipitation method and avoid the most common mistakes in UCNs biocompatible surface modification for further cellular applications.
Protocol
Caution: Please consult all relevant material safety data sheets (MSDS) before use. Please use all appropriate safety practices when performing the synthesis of UCNs at a high temperature (~290 °C), including the use of engineering controls (fume hood) and personal protective equipment (e.g., safety goggles, gloves, lab coat, full length pants, and closed-toe shoes).
1. Synthesis of NaYF4:Yb/Tm/Nd(30/0.5/1%)@NaYF4:Nd(20%) core-shell nanocrystals
- Synthesis of NaYF4:Yb/Tm/Nd(30/0.5/1%) core nanostructure
- Preparation of the RE(CH3CO2)3, NaOH, NH4F methanol stock solution
NOTE: The rare-earth (RE) lanthanide ions include Yttrium (Y), Ytterbium (Yb), Thulium (Tm), and Neodymium (Nd).- Dissolve 500 mg Yttrium(III) acetate hydrate in 5 mL methanol (100 mg/mL), 250 mg Ytterbium (III) acetate hydrate in 5 mL methanol (50 mg/mL), 10 mg Thulium (III) acetate hydrate in 1 mL methanol (10 mg/mL), and 10 mg Neodymium (III) acetate hydrate in 1 mL methanol (10 mg/mL) in glass vials with an ultrasonic cleaning bath for 2 min.
- Combine 400 mg NaOH in a 50 mL centrifuge tube with 20 mL methanol with ultrasonic cleaning bath to prepare NaOH stock solution (20 mg/mL).
- Combine 600 mg NH4F in a 50 mL centrifuge tube with 30 mL methanol with ultrasonic cleaning bath to prepare NH4F stock solution (20 mg/mL).
NOTE: Place the stock solution of lanthanide complexes, NaOH, and NH4F in glass vials, seal with parafilm and store them in a refrigerator at ~4 °C until needed. The prepared methanol stock solutions are replaced once every 2 weeks.
- Preparation of NaYF4:Yb/Tm/Nd core nanostructure
- Pipette 3 mL of oleic acid and 7 mL of 1-octadecene into a 50-mL three-neck flask.
- Combine 1.089 mL of the Y(CH3CO2)3 stock solution, 0.608 mL of the Yb(CH3CO2)3 stock solution, 83.6 µL of the Tm(CH3CO2)3 stock solution, and 128.5 µL of the Nd(CH3CO2)3 stock solution into the flask.
- Fit a thermometer (0 – 360 °C range) in the flask and let its tip touch the solution. Place the flask in a glass container with phenylmethyl silicone oil.
- Heat the solution to 100 °C with a hot plate to evaporate off the methanol. Connect the flask to a Schlenk line with a dual vacuum/gas manifold to remove the residual methanol and keep the reaction mixture under vacuum for 2 – 3 min while stirring.
- Increase the temperature to 150 °C and keep this temperature for 60 min. Maintain a stirring speed of 700 rpm during the synthesis.
NOTE: Set a moderate stirring speed to avoid splashing the solution. - Stop the hot plate and keep the flask at room temperature to allow the solution cool down slowly.
NOTE: The protocol can be paused here. - Combine 2 mL of NaOH-methanol stock solution and 2.965 mL of NH4F-methanol stock solution into a 15 mL centrifuge tube. Tighten the tube with its cap and mix the solution by powerful vortexing for 5 s.
- Add the NaOH-NH4F mixture slowly into the flask by a glass pipette over 5 min.
- Increase the temperature of the mixture to 50 °C and keep this temperature for 30 min.
NOTE: Set the temperature at no more than 50 °C because the high temperature will promote crystal nucleation and growth. - Increase the temperature (~100 °C) to evaporate off the methanol and connect the flask to a Schlenk line with a dual vacuum/gas manifold to remove the residual methanol and keep the reaction mixture under vacuum for 2 – 3 min.
- Switch the position of the stopcock to fill the flask with nitrogen.
- Increase the temperature to 290 °C at a heating rate of ~5 °C/min. Keep the reaction mixture at 290 °C for 1.5 h.
- Stop the hot plate and remove the flask to allow the reaction mixture to cool down slowly in room temperature while stirring.
CAUTION: Be careful of the hot plate with high temperature (>400 °C) to avoid severe burns upon skin contact. - Transfer the mixture in the flask into a 50 mL centrifuge tube. Rinse the flask with 30 mL of ethanol and transfer the solution to the centrifuge tube.
- Centrifuge the product at 4,000 × g for 8 min at room temperature and discard the supernatant.
- Add 10 mL of hexane to the centrifuge tube and re-disperse the product with sonication (60 kHz, 240 W) for 2 min.
- Add 30 mL of ethanol to the tube. Spin down the product at 4,000 × g for 8 min and then discard the supernatant.
- Re-disperse the solid products in the bottom of centrifuge tube with 5 mL hexane. Store the solution in a refrigerator at ~4 °C for a next step.
NOTE: The upconversion emission of core-shell UCNs is tested by irradiating the solutions with an 808 nm laser (2 W).
- Preparation of the RE(CH3CO2)3, NaOH, NH4F methanol stock solution
- Preparation of NaYF4:Yb/Tm/Nd(30/0.5/1%)@NaYF4:Nd(20%) core-shell nanocrystals
- Combine 3 mL of oleic acid and 7 mL of 1-octadecene into a 50-mL three-neck flask. Add 1.082 mL of the Y(CH3CO2)3 stock solution and 2.87 mL of the Nd(CH3CO2)3 stock solution into the flask.
- Add the obtained core nanostructure (80 mg in 5 mL hexane from step 1.1.2.18) into the flask while stirring.
- Fit a thermometer (0 – 360 °C range) in the flask and let its tip touch the mixture. Place the flask in a glass container with phenylmethyl silicone oil.
- Heat the mixture at 100 °C on the top of hot plate to evaporate off the methanol and hexane. Connect the flask to a Schlenk line with a dual vacuum/gas manifold to remove the residual solvent and keep the reaction mixture under vacuum for 2 – 3 min while stirring.
- Increase the temperature to 150 °C and keep it for 60 min. Maintain the stirring speed at 700 rpm in the synthesis.
NOTE: Set a moderate stirring speed to avoid splashing the solution. - Stop the hot plate and remove the flask to allow the solution to cool down slowly at room temperature.
NOTE: The protocol can be paused here. - Pipette 2 mL of NaOH-methanol stock solution and 2.965 mL of NH4F-methanol stock solution into a 15 mL centrifuge tube. Tighten the tube with its cap and mix the solution by powerful vortexing for 5 s.
- Add the mixture into the flask by a glass pipette slowly over 5 min.
- Increase the temperature of the mixture to 50 °C and keep it at 50 °C for 30 min.
NOTE: Set the temperature no higher than 50 °C because the high temperature will promote crystal nucleation and growth. - Increase the temperature (~100 °C) to evaporate off the methanol and connect the flask to a Schlenk line with a dual vacuum/gas manifold to remove the residual methanol, keeping the reaction mixture under vacuum for 2 – 3 min.
- Switch the position of the stopcock to fill the flask with nitrogen.
- Increase the temperature to 290 °C at a heating rate of ~5 °C/min. Keep the reaction mixture at 290 °C for 1.5 h.
- Stop the hot plate and remove the flask to allow the reaction mixture to cool down slowly at room temperature while stirring.
CAUTION: Be careful of the hot plate with high temperature (>400 °C) to avoid severe burns upon skin contact. - Transfer the mixture in the flask into a 50 mL centrifuge tube. Rinse the flask with 30 mL of ethanol and transfer the solution to the centrifuge tube.
- Centrifuge the product at 4,000 × g for 8 min at room temperature and discard the supernatant.
- Add 10 mL of hexane to the centrifuge tube and re-disperse the product with sonication (60 kHz, 240 W) for 2 min.
- Add 30 mL of ethanol to the tube. Spin down the product at 4,000 × g for 8 min and then discard the supernatant.
- Re-disperse the solid products in the bottom of centrifuge tube with 5 mL hexane. Store the solution in a refrigerator at ~4 °C until needed.
NOTE: The upconversion emission of core-shell UCNs is tested by irradiating the solutions with an 808 nm laser (2 W).
2. Synthesis of Biocompatible UCNs Nanostructures
- Preparation of ligand-free UCNs nanoparticle
- Combine 30 mL ethanol with the as-prepared oleate-capped UCNs solution (step 1.2.18) in a 50 mL centrifuge tube. Centrifuge the mixture at 4,000 × g for 8 min at room temperature and discard the supernatant.
- Combine 10 mL acid aqueous solution (pH = 4) adjusted by HCl (0.1 M) with the precipitate in 50 mL centrifuge tube and dissolve the precipitate by sonication (60 kHz, 240 W) for 30 min.
- Transfer the solution in a glass vial with vigorous stirring for 2 h.
- Extract the aqueous solution with 30 mL diethyl ether to remove the oleic acid, repeat three times.
- Add 10 mL water in the combined ether layers with gentle shaking.
- Collect the aqueous phase together (20 mL) and add 20 mL acetone with vortexing for 5 s.
- Spin down the product at 35,000 × g for 10 min and then discard the supernatant. Dissolve the precipitate in 2 mL water.
- Preparation of polymer modified UCNs (PAA-UCNs)
- Combine 200 mg of polyacrylic acid (PAA, Mw = 1,800) with 20 mL water by sonication (60 kHz, 240 W) for 20 min. Add the aforementioned ligand-free UCNs in the PAA solution with vigorous stirring.
- Adjust the pH value to 7.4 by NaOH solution (1 M) with sonication (60 kHz, 240 W) for 30 min. Keep the mixture for 24 h at room temperature while stirring.
- Collect the precipitate by centrifugation at 35,000 × g for 10 min. Re-suspend the product in 10 mL water by sonication (60 kHz, 240 W) for 5 min and centrifuge again at 35,000 × g for 10 min. Repeat this step three times.
- Re-disperse the product in 8 mL water by sonication (60 kHz, 240 W) for 5 min. Store the solution in a refrigerator at ~4 °C until needed.
- Preparation of functional DBCO-UCNs nanoparticle
- Spin down the as-prepared PAA@UCNs (1 mg) by centrifugation at 35,000 × g for 10 min. Re-suspend the precipitate in 1 mL dry DMF by sonication (60 kHz, 240 W) for 1 min and centrifuge again at 35,000 × g for 10 min. Repeat this step twice.
- Dissolve the precipitate in 200 µL dry DMF in a glass vial. Add HOBT (12.2 mg), EDC (14 mg), DBCO-NH2 (5 mg), and DIPEA (16 µL) in the vial with magnetic stirring for 24 h.
- Collect the product by centrifugation at 35,000 × g for 10 min. Remove the supernatant and re-suspend the precipitate in 1 mL DMSO and centrifuge again at 35,000 × g for 10 min. Repeat this step three times.
- Disperse the product in 0.2 mL DMSO and store in a refrigerator at ~ 4 °C before use.
3. Bioapplications of DBCO-UCNs in the Regulation of Membrane Channels in Living Cells
- Culture the HEK293 cells in Dulbecco's Modified Eagle's Medium (DMEM) supplemented with 10% FBS, 100 units/mL penicillin, 100 µg/mL streptomycin and maintain in a humidified incubator with 5% CO2 at 37 °C. Seed 1×105 cells in 1 mL DMEM/well in a 12-well plate and keep it in the incubator for 24 h.
- Combine the plasmid (pCAGGS-ChR2-Venus, 1 µg) with P3000 transfection reagent (2 µL) in Eagle's Minimum Essential Media (MEM) (100 µL) in microcentrifuge tube A, and add transfection reagent (1.5 µL) in MEM (100 µL) in tube B.
- Incubate the mixture of solutions in tubes A and B for 10 min at room temperature.
- Combine the solution in tube A and B with 400 µL MEM. Wash the cells with 1 mL serum-free DMEM twice.
- Add the 600 µL transfection mixture into each well of a 12-well plate and incubate the cells at 37 °C incubator for 4 h. Remove the medium and wash twice with 1 mL DMEM.
- Add 1 mL DMEM containing 1 µL Ac4ManNAz (50 mM in DMSO) in the well and keep it in the incubator for 2 days.
- Remove the medium and wash once with 1 mL PBS. Add 1 mL Trypsin-EDTA (0.25%) solution in each well of the 12-well plate and incubate at 37 °C until cells have detached (~2 min). Re-culture the cells in a confocal dish at 1×105 cells/well in 1 mL DMEM for overnight.
- Remove the medium and add 1 mL fresh DMEM with 2 µL DBCO-UCNs (50 mg/mL) in the dish for 2 h at 37 °C. For confocal imaging, wash cells twice with DMEM and add 1 µL Rhod-N3 (10 mM) for 30 min.
- For intracellular calcium analysis, incubate the cells with the mixture of 1 µL Rhod-3 AM (10 mM), 10 µL Probenecid (250 mM), and 10 µL loading buffer (Pluronic surfactant polyols,0.1% w/v)) in 1 mL DMEM medium for 30 min in the dark.
- Wash the cells with serum-free DMEM and irradiate with 808 nm NIR light at the dosage of 0.8 W/cm2 for 20 min (5 min break after 5 min illumination).
- Record the imaging results on the confocal microscope (laser source: 561 nm laser; detector range: 610/75 nm; lens: 100x 1.4 NA oil, exposure time: 100 ms). Add 1 mL Trypsin-EDTA (0.25%) solution in each well and incubate at 37 °C until cells have detached (~2 min). Re-suspend the cells in 1 mL PBS for flow cytometry (FCM) analysis (Ex: 561 nm, Em: 582/15 nm).
Representative Results
The schematic synthesis process of core-shell lanthanide-doped UCNs are shown in Figure 1 and Figure 2. The transmission electron microscopy (TEM) and high-resolution transmission electron microscopy (HRTEM) images of core and core-shell UCNs nanostructures were collected respectively (Figure 1). The ligand-free UCNs are prepared by removing the hydrophobic oleic acid on the surface of UCNs in acid solution, and modified with hydrophilic polymer (PAA). Then the COOH-capped PAA-UCNs are functionalized with DBCO-NH2 by an amide condensation reaction. The TEM images of ligand-free UCNs, PAA-UCNs and DBCO-UCNs are shown in Figure 2. The dynamic light scattering (DLS), zeta potential results and upconversion luminescence (UCL) spectra of DBCO-UCNs are collected respectively (Figure 3). The fourier transform infrared (FTIR) spectroscopy of DBCO-NH2, PAA-UCNs and DBCO-UCNs are presented in Figure 4.
In cell experiments, the successful expression of ChR2 on the cell membrane is confirmed by the obvious green fluorescent protein (GFP) marker (Venus) using confocal microscopy (Figure 5A). The DBCO-UCNs are specifically localized on the surface of ChR2-expressing HEK293 cells by click reaction (Figure 5A). The intracellular calcium analysis upon NIR light stimulation is also confirmed by confocal imaging and flow cytometry (FCM) based on a standard fluorescent Ca2+ indicator, Rhod-3 AM (Figure 5B, C).
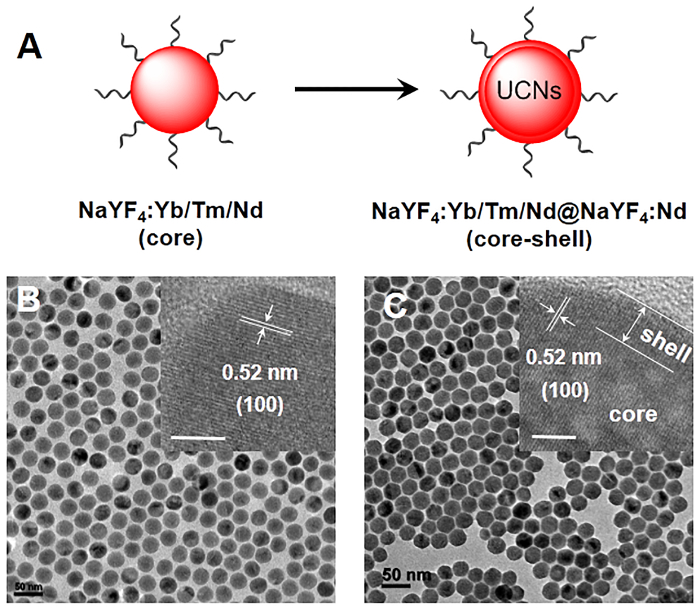
Figure 1. Synthesis of core-shell lanthanide-doped UCNs. (A) Schematic illustration of the synthesis process of UCNs. (B, C) TEM of core (NaYF4:Yb/Tm/Nd (30/0.5/1%)) and core-shell (NaYF4:Yb/Tm/Nd (30/0.5/1%)@NaYF4:Nd (20%)) UCNs nanostructures. Scale bar: 50 nm. Inset: HRTEM images of core and core-shell UCNs nanostructures. Scale bar: 5 nm. Please click here to view a larger version of this figure.
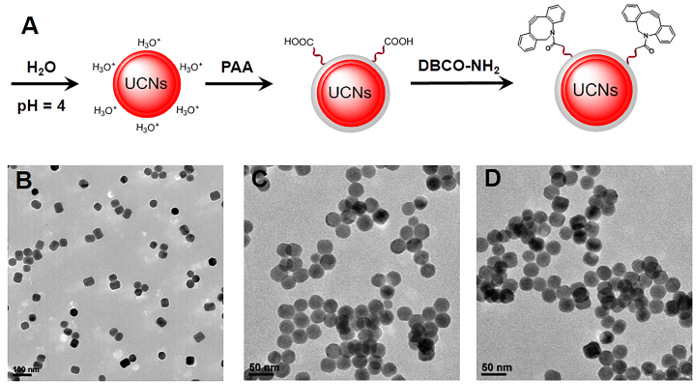
Figure 2. Synthesis of functional UCNs nanostructures. (A) Schematic illustration of the synthesis process of ligand-free UCNs, PAA-UCNs and DBCO-UCNs, respectively. (B, C, D) TEM images of ligand-free UCNs, PAA-UCNs and DBCO-UCNs. Scale bar: 100 nm in panel B and 50 nm in panel C/D. Please click here to view a larger version of this figure.
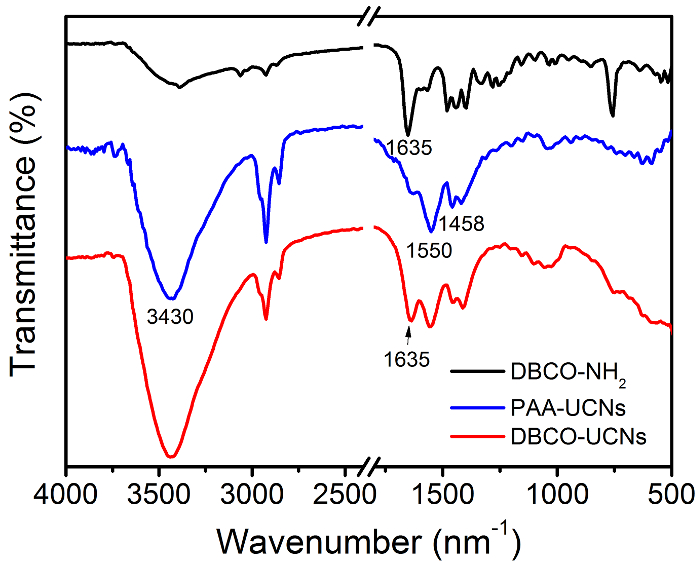
Figure 3. FTIR spectroscopy of DBCO-NH2, PAA-UCNs and DBCO-UCNs, respectively. Please click here to view a larger version of this figure.
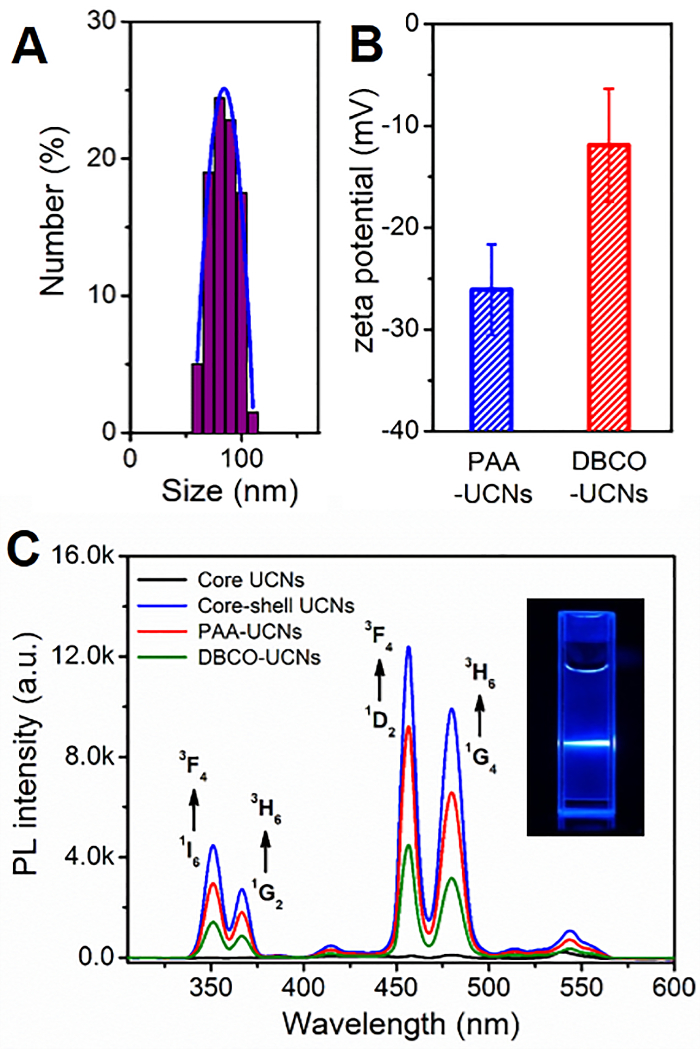
Figure 4. Characterization of DBCO-UCNs in buffer solution. (A) DLS results of DBCO-UCNs. (B) Zeta potential results of PAA-UCNs and DBCO-UCNs (mean ± SD). (C) UCL spectra of core and core-shell UCNs in hexane (1 mg/mL), PAA-UCNs and DBCO-UCNs in water (1 mg/mL) separately (Ex: 808 nm). Inset: luminescence photograph of UCNs with 808 nm irradiation. Please click here to view a larger version of this figure.
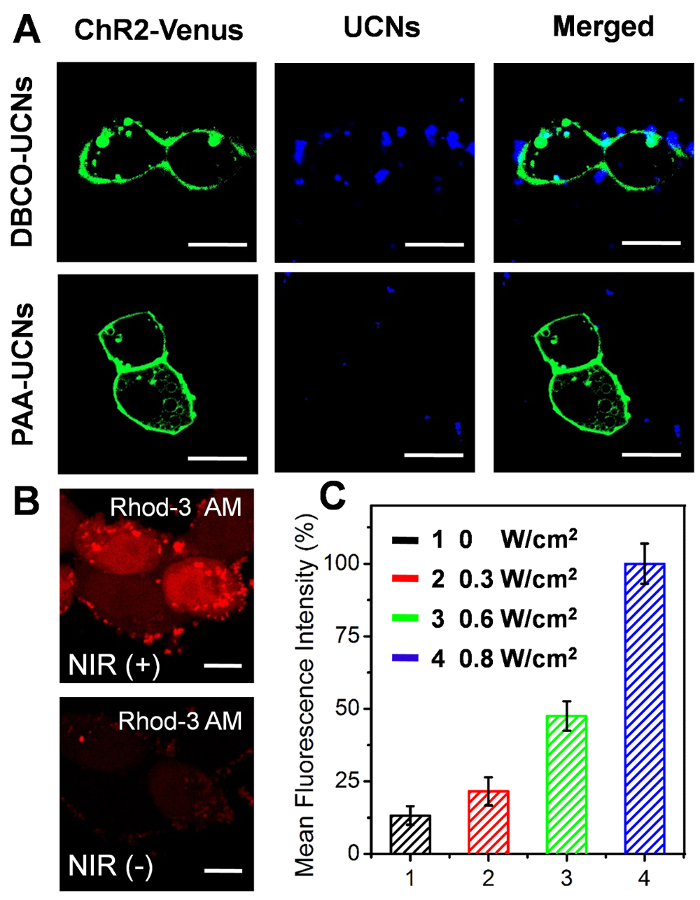
Figure 5. Cellular applications of DBCO-UCNs upon NIR light illumination. (A) Confocal imaging of ChR2-expressing N3-tagged HEK293 cells incubated with DBCO-UCNs (top) and PAA-UCNs (bottom) for 2 h at 100 µg/mL. Green: ChR2-Venus (Ex: 488 nm, Em: 530/50 nm), blue: UCNs (Ex: 543 nm, Em: 580/50 nm). Scale bar: 20 µm. (B) Cellular Ca2+ imaging with Rhod-3 AM before and after NIR-light irradiation (0.8 W/cm2 for 20 min). Ex: 561 nm, Em: 610/75 nm. Scale bar: 10 µm. (C) Quantitative FCM analysis of the normalized fluorescence intensity of cellular Ca2+ with different light dosages illumination (mean ± SD). Please click here to view a larger version of this figure.
Discussion
This article has presented a method for the synthesis of core-shell lanthanide-doped upconversion nanocrystals (UCNs) and their surface modification with functional moieties for cellular applications. This novel nanomaterial possesses outstanding optical properties, which can emit UV and visible light upon NIR light excitation through a multi-photon upconversion process. In this protocol, the core-shell UCNs nanostructures (NaYF4:Yb/Tm/Nd (30/0.5/1%)@NaYF4:Nd (20%)) are prepared by a high-temperature co-precipitation method in a mixture of oleic acid and 1-octadecene. TEM images demonstrate the morphology of these core and core-shell nanocrystals are spherical in shape with a diameter of 20 nm and 30 nm respectively, implying a shell thickness of about 5 nm (Figure 1). The core-shell architecture is also revealed by the high resolution TEM images in the inset of Figure 1, which shows similar lattice fringes with that of the core UCNs nanostructures. Moreover, a typical d-spacing around 0.52 nm agrees well with the spacing of the (100) plane of β-NaYF4, indicating that all the core and core-shell UCNs nanostructures are highly crystallized and maintain the same crystal structure. Furthermore, the upconversion luminescence spectra demonstrate the core-shell nanocrystals emit strong blue emission with a much higher intensity at 480 nm than the core particles upon excitation with a diode laser to achieve a continuous wave at 808 nm (Figure 4C). The enhanced emission of core-shell UCNs is attributed to the suppressed surface quenching effect of Yb3+, Nd3+, and Tm3+ ions that are embedded in the interior layer of the core-shell nanocrystals21. These results strongly confirm the successful preparation of lanthanide-doped core-shell UCNs nanomaterial in this proposal.
There are several critical steps in the high-temperature co-precipitation synthesis of these nanocrystals. First, the added volume of lanthanide ion stock solution in the synthesis process of the core and core-shell UCNs should be strictly controlled (step 1.1.2.2). High level doping ions will result in a concentration-related cross-relaxation or quenching effect due to ion-ion interaction in the nanocrystals21. Second, the temperature should be kept at less than 50 °C to ensure complete nucleation after NaOH and NH4F are added into the flask (step 1.1.2.9 and step 1.2.9), which is critical to ensure a uniform morphology by promoting crystal growth based on Ostwald ripening effects22. Third, the size and optical properties of core-shell nanoparticles can be principally controlled by adjusting the amount of lanthanide ions in the precursors of the shell (Y3+ and Nd3+) and as-prepared core particles (step 1.2.1). It is also important that the morphology of core-shell nanocrystals is based on the anisotropic shell growth of different types of core particles, which is useful for modulating the different optical emissions of UCNs upon NIR light illumination23.
In addition, it is also a considerable challenge to effectively convert hydrophobic UCNs to hydrophilic UCNs nanoparticles for further biological applications. Though some methods have been reported to improve the biocompatibility of UCNs in buffer solution, including ligand exchange, silica coating, oleate-capping ligand oxidation, etc., they suffer from unexpected aggregation, time consuming, and complex procedures24,25,26. Herein, we develop a simple approach to obtain water-dispersible ligand-free UCNs nanostructures by removing the oleic acid on the surface of oleate-capped UCNs in an acid water solution (pH = 4). The pH value adjusted by HCl is critical to control the release of oleate ligand and to affect the luminescence of UCNs. More importantly, a biocompatible polymer (polyacrylic acid, PAA) with a large number of carboxyl groups can link with the lanthanide ions on the surface of UCNs by coordination interaction, which will provide more functional groups for further chemical modification. Therefore, we functionalize the DBCO-NH2 moieties on the surface of UCNs for further specific N3-tagged cell membrane localization. The TEM images of ligand-free UCNs, PAA-UCNs, and DBCO-UCNs in Figure 2 demonstrate the great dispersity and solubility of these nanostructures in buffer solution after surface modification. In addition, fourier transform infrared (FTIR) spectroscopy was performed to characterize the surface modification of DBCO groups on UCNs nanoplatform. As shown in Figure 3, the strong band around 3,430 cm-1 is due to the H-O stretching vibration of the carboxyl groups, and the two bands centered at 1550 cm-1 and 1458 cm-1 are associated with the asymmetric and symmetric stretching vibration modes of the carboxylate anions, indicating the successful coating of COOH group-containing polymers (PAA) on the UCNP surface. After reaction with DBCO-NH2 moieties, an obvious band at 1,635 cm-1 is present based on the C=C stretching vibration on the aromatic ring of DBCO groups, which is consistent with the FTIR spectrum of DBCO-NH2 moieties at the same wavenumber. Moreover, the dynamic light scattering (DLS) result indicates the increased hydrodynamic diameter of DBCO-UCNs in aqueous solution (96.4±10.4 nm) (Figure 4A). The zeta potential results also indicate the negative surface of PAA-UCNs (-26.1±4.4 mV) and DBCO-UCNs (-11.9±5.6 mV) respectively (Figure 4B), demonstrating the great solubility and stability of these nanostructures in buffer solution. In addition, the UCL spectra of PAA-UCNs and DBCO-UCNs demonstrate that they can emit similar upconverted emission at 480 nm upon 808 nm light irradiation (Figure 4C), which can be utilized for further NIR light-mediated photoactivation in biological systems.
Furthermore, as a proof-of-concept, in order to demonstrate the bioapplication of functionalized UCNs in living cells, a light-gated channel protein (ChR2) is engineered on the cell surface to regulate the cellular functions by mediating the influx of cation ions (e.g., Ca2+) in the cytoplasm27,28,29. The successful expression of ChR2 on the membrane of the HEK293 cell line is confirmed by the presence of the green fluorescent protein (GFP) marker (Venus) using confocal microscopy (Figure 5A). Moreover, the localization of UCNs on the N3-tagged cell membrane is obviously visualized on the cell surface (blue) after 2 h incubation, which can be attributed to fact that the residual DBCO groups on the surface of UCNs nanostructures are stained with an azide-containing fluorescent dye (5-carboxytetramethylrhodamine-azide, Rhod-N3) through the copper-free bioorthogonal click reaction. Furthermore, NIR light (808 nm) is utilized to irradiate the localized UCNs nanotransducer on the cell surface, which can activate the light-gated ChR2 channel proteins and facilitate Ca2+ ion influx across the membrane. As shown in Figure 5B, a significant increase of red fluorescence in the cytosol is observed by a standard fluorescent Ca2+ indicator, Rhod-3 AM, while no apparent fluorescence increment is recorded in the absence of light illumination. The quantitative flow cytometry (FCM) analysis in Figure 5C also demonstrates that such NIR-light-responsive fluorescence enhancement is light-dose-dependent, clearly suggesting that the biocompatible DBCO-UCNs can effectively regulate the channel proteins on the cell membrane upon NIR-light irradiation.
In summary, based on the aforementioned results, we anticipate that the present protocol not only provides detailed experimental procedures for high-temperature co-precipitation synthesis of core-shell UCNs nanostructures, but also develops a simple technique for biocompatible surface modification of UCNs for further NIR light-mediated photoactivation in biological applications. More importantly, based on the rationale behind this method, the optical properties of UCNs can further be adjusted by doping different kinds of lanthanide ions (e.g., Erbium (III), Holmium (III), etc.) in the crystals to emit UV, green, red, and NIR light upon 808 nm light irradiation30. Moreover, the UCNs surface can be also modified with various functional groups (e.g., peptide, protein, lipid, targeting ligand, etc.) for further biomedical applications31,32. These advantages make biocompatible UCNs nanomaterial a suitable candidate for the study of physiological processes in vitro and in vivo, and may thus act as personalized nanomedicine for clinical theranostics in the future.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was partially supported by NTU-AIT-MUV NAM/16001, RG110/16 (S), (RG 11/13) and (RG 35/15) awarded in Nanyang Technological University, Singapore and National Natural Science Foundation of China (NSFC) (No. 51628201).
Materials
| 1-Octadecene | Sigma Aldrich | O806 | Technical grade |
| oleic acid | Sigma Aldrich | 364525 | Technical grade |
| Methanol | Fisher Scientific | A412 | Technical grade |
| Ethanol | Fisher Scientific | A405 | Technical grade |
| Acetone | Fisher Scientific | A18 | Technical grade |
| Hexane | Sigma Aldrich | H292 | Technical grade |
| Thulium (III) acetate hydrate (Tm(CH3CO2)3) | Sigma Aldrich | 367702 | 99.9% trace metals basis |
| Neodymium (III) acetate hydrate (Nd(CH3CO2)3) | Sigma Aldrich | 325805 | 99.9% trace metals basis |
| Ytterbium (III) acetate hydrate (Yb(CH3CO2)3) | Sigma Aldrich | 326011 | 99.9% trace metals basis |
| Yttrium(III) acetate hydrate (Y(CH3CO2)3) | Sigma Aldrich | 326046 | 99.9% trace metals basis |
| Sodium hydroxide (NaOH) | Sigma Aldrich | S5881 | reagent grade |
| Ammonium fluoride (NH4F) | Sigma Aldrich | 338869 | ACS reagent |
| Hydrogen chloride (HCl) | Fisher Scientific | A144 | reagent grade |
| polyacrylic acid (PAA) | Sigma Aldrich | 323667 | average Mw 1800 |
| 1-Hydroxybenzotriazole hydrate (HOBT) | Sigma Aldrich | 54802 | ACS reagent |
| 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDC) | Sigma Aldrich | E7750 | commercial grade |
| Dibenzocyclooctyne-amine (DBCO-NH2) | Sigma Aldrich | 761540 | ACS reagent |
| N,N-Diisopropylethylamine (DIPEA) | Sigma Aldrich | D125806 | ACS reagent |
| Dimethyl sulfoxide (DMSO) | Fisher Scientific | BP231 | Technical grade |
| HEK293 cell line | ATCC | CRL-1573 | human embryonic kidney |
| Fetal Bovine Serum (FBS) | Sigma Aldrich | F1051 | ACS reagent |
| Penicillin-Streptomycin | Thermo Fisher | 15140122 | 10,000 U/mL |
| plasmid (pCAGGS-ChR2-Venus) | Addgene | 15753 | Plasmid sent as bacteria in agar stab |
| Dulbecco's Modified Eagle Medium (DMEM) | Thermo Fisher | 11965092 | High glucose |
| opti-Modified Eagle Medium (MEM) | Thermo Fisher | 51985034 | Reduced Serum Media |
| Lipofectamine 3000 Transfection Reagent | Thermo Fisher | L3000015 | Lipid-Based Transfection |
| N-Azidoacetylmannosamine, Acetylated (Ac4ManNAz) | Sigma Aldrich | A7605 | ACS reagent |
| Trypsin-EDTA (0.25%) | Thermo Fisher | 25200056 | Phenol red |
| Rhod-3 AM Calcium Imaging Kit | Thermo Fisher | R10145 | Fluorescence dye |
| 5-carboxytetramethylrhodamine-azide (Rhod-N3) | Sigma Aldrich | 760757 | Azide-fluor 545 |
| Confical dish | ibidi GmbH | 81158 | Glass Bottom, 35 mm |
| 50 ml conical centrifuge tubes | Greiner Bio-One | 227261 | Polypropylene |
| 15 ml conical centrifuge tubes | Greiner Bio-One | 188271 | Polypropylene |
| 1.5 ml conical microcentrifuge tubes | Greiner Bio-One | 616201 | Polypropylene |
| Phenylmethyl silicone oil | Clearco Products | 63148-52-7 | Less than 320 degrees Celsius |
| Glass thermometer | GH Zeal | L0111/10 | From -10 to 360 degrees Celsius |
| 12-well plate | Sigma Aldrich | Z707775 | Polystyrene |
Referências
- Wang, F., et al. Tuning upconversion through energy migration in core-shell nanoparticles. Nat Mater. 10 (12), 968-973 (2011).
- Liu, Y., et al. Amplified stimulated emission in upconversion nanoparticles for super-resolution nanoscopy. Nature. 543 (7644), 229-233 (2017).
- Fan, W., Bu, W., Shi, J. On The Latest Three-Stage Development of Nanomedicines based on Upconversion Nanoparticles. Adv Mater. 28 (24), 3987-4011 (2016).
- Zhu, X., et al. Temperature-feedback upconversion nanocomposite for accurate photothermal therapy at facile temperature. Nat Commun. 7, 10437-10446 (2016).
- Li, W., Wang, J., Ren, J., Qu, X. Near-infrared upconversion controls photocaged cell adhesion. J Am Chem Soc. 136 (6), 2248-2251 (2014).
- Min, Y., Li, J., Liu, F., Yeow, E. K., Xing, B. Near-infrared light-mediated photoactivation of a platinum antitumor prodrug and simultaneous cellular apoptosis imaging by upconversion-luminescent nanoparticles. Angew Chem Int Ed. 53 (4), 1012-1016 (2014).
- Yang, D., Ma, P., Hou, Z., Cheng, Z., Li, C., Lin, J. Current advances in lanthanide ion (Ln(3+))-based upconversion nanomaterials for drug delivery. Chem Soc Rev. 44 (6), 1416-1448 (2015).
- Wang, C., Cheng, L., Liu, Z. Upconversion nanoparticles for photodynamic therapy and other cancer therapeutics. Theranostics. 3 (5), 317-330 (2013).
- Li, L. L., et al. Biomimetic surface engineering of lanthanide-doped upconversion nanoparticles as versatile bioprobes. Angew Chem Int Ed. 51 (25), 6121-6125 (2012).
- Wang, J., Ming, T., Jin, Z., Wang, J., Sun, L. D., Yan, C. H. Photon energy upconversion through thermal radiation with the power efficiency reaching 16%. Nat Commun. 5, 5669-5678 (2014).
- Zou, W., Visser, C., Maduro, J. A., Pshenichnikov, M. S., Hummelen, J. C. Broadband dye-sensitized upconversion of near-infrared light. Nat Photonics. 6 (8), 560-564 (2012).
- Liu, Y., Tu, D., Zhu, H., Li, R., Luo, W., Chen, X. A strategy to achieve efficient dual-mode luminescence of Eu(3+) in lanthanides doped multifunctional NaGdF(4) nanocrystals. Adv Mater. 22 (30), 3266-3271 (2010).
- Min, Y., Li, J., Liu, F., Padmanabhan, P., Yeow, E. K., Xing, B. Recent Advance of Biological Molecular Imaging Based on Lanthanide-Doped Upconversion-Luminescent Nanomaterials. Nanomaterials. 4 (1), 129-154 (2014).
- Li, X., Zhang, F., Zhao, D. Lab on upconversion nanoparticles: optical properties and applications engineering via designed nanostructure. Chem Soc Rev. 44 (6), 1346-1378 (2015).
- Gu, Z., Yan, L., Tian, G., Li, S., Chai, Z., Zhao, Y. Recent advances in design and fabrication of upconversion nanoparticles and their safe theranostic applications. Adv Mater. 25 (28), 3758-3779 (2013).
- Dong, H., Sun, L. D., Yan, C. H. Energy transfer in lanthanide upconversion studies for extended optical applications. Chem Soc Rev. 44 (6), 1608-1634 (2015).
- Ai, X., et al. In vivo covalent cross-linking of photon-converted rare-earth nanostructures for tumour localization and theranostics. Nat Commun. 7, 10432-10440 (2016).
- Lu, S., et al. Multifunctional Nano-Bioprobes Based on Rattle-Structured Upconverting Luminescent Nanoparticles. Angew Chem Int Ed. 54 (27), 7915-7919 (2015).
- Bogdan, N., Vetrone, F., Ozin, G. A., Capobianco, J. A. Synthesis of ligand-free colloidally stable water dispersible brightly luminescent lanthanide-doped upconverting nanoparticles. Nano Lett. 11 (2), 835-840 (2011).
- Zheng, W., Huang, P., Tu, D., Ma, E., Zhu, H., Chen, X. Lanthanide-doped upconversion nano-bioprobes: electronic structures, optical properties, and biodetection. Chem Soc Rev. 44 (6), 1379-1415 (2015).
- Chen, X., Peng, D., Ju, Q., Wang, F. Photon upconversion in core-shell nanoparticles. Chem Soc Rev. 44 (6), 1318-1330 (2015).
- Wang, F., Deng, R., Liu, X. Preparation of core-shell NaGdF4 nanoparticles doped with luminescent lanthanide ions to be used as upconversion-based probes. Nat Protoc. 9 (7), 1634-1644 (2014).
- Chen, G., Agren, H., Ohulchanskyy, T. Y., Prasad, P. N. Light upconverting core-shell nanostructures: nanophotonic control for emerging applications. Chem Soc Rev. 44 (6), 1680-1713 (2015).
- Yang, Y., et al. In vitro and in vivo uncaging and bioluminescence imaging by using photocaged upconversion nanoparticles. Angew Chem Int Ed. 51 (13), 3125-3129 (2012).
- Sedlmeier, A., Gorris, H. H. Surface modification and characterization of photon-upconverting nanoparticles for bioanalytical applications. Chem Soc Rev. 44 (6), 1526-1560 (2015).
- Hu, M., et al. Near infrared light-mediated photoactivation of cytotoxic Re(I) complexes by using lanthanide-doped upconversion nanoparticles. Dalton Trans. 45 (36), 14101-14108 (2016).
- Nagel, G., et al. Channelrhodopsin-2, a directly light-gated cation-selective membrane channel. Proc Natl Acad Sci USA. 100 (24), 13940-13945 (2003).
- Ai, X., et al. Remote Regulation of Membrane Channel Activity by Site-Specific Localization of Lanthanide-Doped Upconversion Nanocrystals. Angew Chem Int Ed. 56 (11), 3031-3035 (2017).
- Xie, R., et al. In vivo metabolic labeling of sialoglycans in the mouse brain by using a liposome-assisted bioorthogonal reporter strategy. Proc Natl Acad Sci USA. 113 (19), 5173-5178 (2016).
- Bansal, A., Zhang, Y. Photocontrolled nanoparticle delivery systems for biomedical applications. Acc Chem Res. 47 (10), 3052-3060 (2014).
- Yang, Y., Aw, J., Xing, B. Nanostructures for NIR light-controlled therapies. Nanoscale. 9 (11), 3698-3718 (2017).
- Ai, X., Mu, J., Xing, B. Recent Advances of Light-Mediated Theranostics. Theranostics. 6 (13), 2439-2457 (2016).

