The MUB40 Peptide for Use in Detecting Neutrophil-Mediated Inflammation Events
Summary
Here, we present a protocol to detect the presence of neutrophils in fixed/permeabilized histology sections and assess the activation state of live purified neutrophils. In particular, the MUB40 peptide binds lactoferrin present in neutrophil-specific and tertiary granules. Exposure of the granule contents through either permeabilization or neutrophil activation allows for the marking of neutrophils.
Abstract
Here, we provide a protocol involving the use of MUB40, a synthesized peptide with the ability to bind glycosylated lactoferrin stored at high concentrations in specific and tertiary granules of neutrophils. This protocol details how MUB40 conjugated directly to a fluorophore can be used to stain neutrophils in fixed/permeabilized tissues as well as how this can be used in live-cell imaging to assay for neutrophil activation and de-granulation. Neutrophil detection methods are limited to species-specific monoclonal antibodies, which are not always suitable for certain applications. MUB40 does not penetrate the cell membrane and is thus excluded from lactoferrin stored in non-activated/non-permeabilized neutrophils. MUB40 has the added benefit of recognizing lactoferrin from a broad host range, making it especially useful for comparing results in studies involving multiple research models, reducing the number of duplicate reagents, and simplifying protocols through single-step staining.
Introduction
Neutrophils are one of the primary arms of the innate immune system and are routinely recruited to the sites of inflammation around the body. The study of neutrophils has been largely impaired by their short lifespan in vitro (less than 8 h) and by limited detection tools under basal conditions or after activation. Here, we present a well-tested protocol for the broad and specific detection of mammalian neutrophils in fixed/permeabilized samples. We also provide a detailed protocol for staining live neutrophils with MUB40. Using the live neutrophil staining protocol, the timing and location of neutrophil activation can be pinpointed. This protocol is ideal for researchers who wish to study neutrophil activation or granule release. Beyond their antimicrobial functions, neutrophils are now appreciated as immunomodulatory cells involved in a wide range of diseases and immune responses (innate and adaptive)1,2. Neutrophils are present in most inflammatory tissues, at high numbers in infected tissues, in inflammatory tumors3, during IBS and Crohn's flare-ups4, and in areas of non-infectious inflammation such as the synovia of Rheumatoid Arthritis (RA) patients5. Neutrophils contain four classes of pre-formed granules named azurophil (α), specific (β1), tertiary (β2) granules, and secretory vesicles (γ)6. Upon migration to an inflammatory site, recruited neutrophils become activated and sequentially secrete granule contents (composed of adhesion, antimicrobial compounds and immunomodulatory molecules), which promotes further recruitment and contributes to infection resolution (but also to host tissue damage)7. While neutrophils are considered a critical aspect of innate immunity, to date there are few detection reagents available to study them, and even fewer that can be used to assess the activation state of living neutrophils.
Current methods of detecting neutrophils rely on monoclonal antibodies generated against cell-surface exposed antigens, such as Ly-6G2 or proteins specifically stored in neutrophil granules under basal conditions (e.g., myeloperoxidase, lactoferrin). Advantages of monoclonal antibodies include their strong binding, sensitivity, and versatility under various assay conditions. However, there are several downsides to using monoclonal antibodies and anti-Ly-6G in particular. These downsides include the specificity, as Ly-6G is present on a majority of myeloid cells in bone marrow and on all granulocytes including eosinophils; thus, deciphering neutrophils from eosinophils with this marker requires more complexes approaches8. Another frequent tradeoff with monoclonal antibodies is their often-limited host specificity range, making comparison studies with more than one animal species difficult. A third drawback of antibody detection methods, especially when using live cells or in vivo, is their potential to disrupt cell function or lead to cell activation. For example, the administration of anti-Ly-6G to mice is commonly applied to neutrophil depletion and transient neutropenia9. It has additionally been demonstrated that antibody injection may stimulate neutrophil antitumor function10. Finally, antibody detection of neutrophils does not reveal the activation state of the cell.
We have identified a 40-amino acid peptide called MUB40, which can be used in a number of assays to label neutrophils under in vitro or in vivo conditions as well as assay the activation status of live neutrophils11. MUB40 is derived from MUB7012, a domain of the Lactobacillus reuteri surface mucus-binding protein originally described for its ability to bind mucus13,14. MUB40 interacts with glycosylated lactoferrin that is present at high concentrations in neutrophil-specific and tertiary granules. MUB40 can be exposed to these granules through standard permeabilization steps present in histopathology or fluorescence-activated cell sorting (FACS) protocols for robust staining of fixed neutrophils. When live neutrophils are kept in an inactivated state, the MUB40 peptide is excluded from the granule contents, and cells do not stain positive with the marker. Upon activation, MUB40 can bind to exposed lactoferrin and lead to robust staining with the peptide. Thus, MUB40 can be used to determine the activation state of purified neutrophils, making it an attractive marker to follow the infectious process. The ability to bind to live activated neutrophils also allows MUB40 to be used as a tool to detect areas of neutrophil inflammation in live animal models (e.g., a mouse arthritis model15). The protocols in this manuscript detail how fluorescently labeled MUB40 can be used to reveal neutrophils in histology tissues and how to assay the activation of live purified neutrophils in vitro.
Protocol
All methods described here have been reviewed and approved by the French organizations CoRC (Comité de Recherche Clinique), CPP (Comité de Protection des Personnes), and CNIL (Commission Nationale Informatique et Liberté).
1. Staining Neutrophils in Histopathology Tissue with Fluorescently Labelled MUB40
- To obtain the tissue for histopathology, fix tissue/biopsy sections in a suitable volume of 4% paraformaldehyde (PFA) for 30 min up to 4 h depending on the thickness of the sample. Place the samples in a 4 °C refrigerator during fixation.
Note: Alternative fixation methods/timings can be substituted based on the user's unique needs.- Remove the PFA, add 16% sucrose solution with enough volume to completely cover the sample, and place the sample at 4 °C for 4 h up to overnight.
- Remove 16% sucrose solution and add 30% sucrose solution at a large enough volume to completely cover the sample, and place it at 4 °C for 4 h up to overnight.
- Remove the 30% sucrose solution and blot off excess solution from the tissue with a paper towel. Place the tissue section in optimal cutting temperature (OCT) media and snap-freeze in 2-methylbutane cooled in a dry ice-ethanol bath (-72 °C). Once frozen solid (after ~2 min), remove blocks from the 2-methylbutane and place them on dry ice until all blocks are finished and ready for long-term storage. Store the frozen blocks at -80 °C for long-term storage.
- The night before tissue blocks are to be cut, remove them from the -80 °C freezer and place them at -20 °C overnight to equilibrate.
- Using a cryostat, cut OCT-embedded tissue into 10- to 30 μm-thick slices and adsorb to high-quality glass slides.
Note: Thicker or thinner slices can be used depending on the experimental requirements. - With a wax pen or other hydrophobic method, draw a box around the tissue slice on the glass slide and let dry.
- Prepare the staining solution by performing a 1:1,000 dilution of MUB40-Cy5 (or other fluorescent MUB40version, 1 mg/mL stock solution) in 0.1% saponin-PBS solution. The optimal final MUB40 concentration should be 1 μg/mL.
Note: Lower final concentrations may be used (0.1-0.01 μg/mL) but may lead to lower signal intensity.
CAUTION: Saponin powder can cause breathing irritation. Make sure to weigh saponin in a cabinet or protected area. Alternative permeabilization methods can be used such as Triton. Add additional markers at manufacturer-recommended concentrations (e.g., 4′,6-diamidino-2-phenylindole (DAPI), phalloidin, fluorescent antibodies). - Gently dip the glass slide into a solution of 0.1% saponin-PBS three times.
- Carefully blot away excess liquid around the tissue section and add enough staining solution to completely cover the tissue section. Place the slide into a humidity chamber to avoid evaporation and incubate it for 1 h at room temperature. Keep it protected from light.
- Gently dip the slide into 0.1% saponin-PBS solution 3x. Then, gently dip the slide into PBS solution 3x. Finally, gently dip the slide into distilled H2O 3x. Carefully dry excess liquid from the slide.
- Add a small amount (~20 μL) of mounting medium (20 mM Tris pH 8.0, 0.5% N-propyl gallate, 90% glycerol) next to the tissue slice and gently lay down a glass coverslip on top of the glass slide. Gently and evenly press the coverslip onto the tissue section and, using a paper towel, carefully remove any mounting media that extrudes around the edges of the coverslip.
- Place the mounted slide in a dark cabinet for 24 h to allow the mounting media to solidify. Alternatively, place the mounted slide at 37 °C for 1 h.
- Using a cryostat, cut OCT-embedded tissue into 10- to 30 μm-thick slices and adsorb to high-quality glass slides.
- Image the tissue slices on a fluorescent microscope according to the desired acquisition method.
2. Visualization of Neutrophil Activation with Retro-Inverso-MUB40-Cy5 Peptide
- Obtain human neutrophils using the following protocol. Prepare the following solutions and place them in an anoxic cabinet overnight. Exposure to oxygen will begin to activate neutrophils and further induce cell death16,17.
Note: Neutrophils may be alternatively purified "on the bench" for MUB40-labeling experiments; however, be aware that oxygen exposure will begin to affect neutrophil activation.- Prepare the following solution:
Solution 1 [sodium chloride (NaCl) 0.9%]: add 4.5 g of NaCl to 500 mL of H2O. Filter the solution.
Solution 2 (dextran 6%): add 6 g of dextran in NaCl 0.9% solution to 100 mL.
Solution 3 (washing buffer): dissolve ethylenediaminetetraacetic acid (EDTA) in PBS pH 7.2 to a final EDTA concentration of 2 mM. Immediately prior to use, dissolve bovine serum albumin (BSA) in the PBS/EDTA solution such that the final BSA concentration is 10% w/v. - Centrifuge blood collection tubes at 650 x g for 20 min without breaks.
- Without disrupting separated blood, transfer the blood collection tubes to an anoxic cabinet and collect plasma fractions (top) in a 50 mL conical tube.
- Remove the plasma-containing tube from the anoxic cabinet and centrifuge at 2,900 x g for 20 min with breaks to pellet the platelets.
- Without disrupting the pelleted platelets, gently transfer the plasma tube back into the anoxic cabinet and pipet the platelet poor plasma into a fresh 50 mL tube (hereafter called "plasma").
- Prepare the following solution:
- Separation of red blood cells from leukocytes
- Using the tubes containing the red blood cells (RBCs) from step 2.1.3., combine the RBCs into a conical 50 mL tube (5 blood collection tube contents per 50 mL tube).
- Add NaCl 0.9% until the total volume reaches 44 mL. Add 6 mL of dextran 6% (last).
- Gently mix the blood/dextran mixture by inverting the tube 10-20 times. Let the tube sediment for at least 30 min.
- While sedimentation is occurring, prepare a Percoll gradient by adding 4.2 mL of Percoll solution to 5.8 mL of plasma in a 15 mL conical tube, and mix well by inverting the tube.
- Collect the top fraction of the dextran while trying to avoid pipetting red blood cells.
- Tighten the screw cap, remove the dextran tube from the anoxic chamber, and centrifuge the tube at 300 x g for 10 min with breaks.
- Carefully place the tube back into the anoxic chamber without disturbing the pelleted cells, and remove the liquid via pipetting.
- Gently re-suspend the cell pellet in 1 mL of plasma and very slowly add to the top of the Percoll solution. Be careful not to induce mixing of the layers when pipetting the cells on top.
- Carefully remove the Percoll solution tube from the anoxic chamber (avoiding mixing), and centrifuge at 800 x g for 20 min without breaks. Peripheral blood mononuclear cells (PBMCs) will remain on the Percoll solution surface, and neutrophils will pellet with the RBCs.
- Removal of contaminating RBCs
- Prepare 14 mL of washing buffer solution by adding 700 μL PBS + 10% BSA to 14 mL of washing buffer.
- Place the Percoll solution tube back into the anoxic chamber. Remove the Percoll and PBMCs via pipetting and re-suspend the pelleted cells in 1 mL of washing buffer solution. The pellet to be re-suspended will have a red color due to contaminating RBCs.
- Add 200 μL of anti-CD235a (glycophorin) magnetic beads to the re-suspended cells, gently mix, and incubate for a minimum of 15 min. This step loads the contaminating RBCs with magnetic beads so that they can be removed.
- Wash the separation column with 2 mL of washing buffer 3x.
- While holding a 15 mL conical tube under the separation column, slowly pipette the neutrophil/RBC mixture to the top of the column. Collect the cells in the 15 mL conical tube as they flow through. The flow-through should be cloudy and lose its red color due to the RBCs remaining bound to the column.
- Add 2 mL of washing buffer to the top of the column and collect in the same 15 mL conical tube. By the end of the 2 mL wash, the flow-through should be clear, signifying that all the neutrophils have been collected.
- Gently mix the tube to ensure even distribution of neutrophils and collect 10 μL for cell counting. Note the final volume for cell counting purposes. Enumerate the total number of cells with any standard cell counting method.
- Remove the neutrophil tube from the anoxic chamber, and centrifuge the neutrophils at 300 x g for 20 min with breaks.
- Place sedimented neutrophils back into the anoxic chamber and remove the washing buffer via pipetting. Re-suspend the neutrophil pellet in plasma with a maximum cell density of 107 PMN/mL.
- Enumeration of purified neutrophils and visualization using MUB40
- Add 1 μL of RI-MUB40-Cy5 (1mg/mL) to 1 mL of Rosewell Park Memorial Institute (RPMI) 1640 medium without phenol red. Mix by pipetting well.
- For the purposes of this protocol, the result has been outlined as if a potent neutrophil activating reagent, N-formylmethionyl-leucyl-phenylalanine (FMLP), is added. Add 1 μm of FMLP to the RPMI/RI-MUB40-Cy5 tube and mix via pipetting up and down.
NOTE: Each experimental procedure will be different from this point forward, depending on the questions being addressed. - Transfer the 1 mL RPMI/RI-MUB40-Cy5/FMLP mixture to a glass bottom microscopy dish. To this dish, add a volume of purified cells corresponding to 106 total neutrophils. Mix by gently pipetting.
- Place the dish into an inverted fluorescent microscope and start image acquisition. For example, it is recommended to acquire baseline images prior to the addition of a neutrophil-activating reagent such as FMLP or bacteria. In this example, start the acquisition of RPMI/RI-MUB40-Cy5/neutrophils, and at a later timepoint, pipette the neutrophil-activating reagent into the glass dish and continue image acquisition.
NOTE: Modifications to these steps can be made based on scientific needs. - Process acquired images/time-lapse movies as pertains to the specific microscope used.
Representative Results
Results of MUB40-stained tissues from histopathology slides typically reveal individual cells scattered throughout the tissue. MUB40 stains lactoferrin, which is present in neutrophil granules and compartmentalized. Thus, typically seen are punctate staining or several large separated areas of signal coming from individual neutrophils (Figure 1). It is helpful to add a second cell marker such as DAPI to help co-localize the MUB40 signal with the stained cell. The total number of detected cells depends upon the number of neutrophils present in the field of view and can vary dramatically depending upon the source and strength of the immune response. Shown here are representative images from purified fixed human neutrophils (Figure 1A) and from a tissue biopsy of an ulcerative colitis patient (Figure 1B). Also shown are images taken from a relatively low level of neutrophils in the lungs of mice infected with Klebsiella pneumoniae (Figure 1C) and a high level of neutrophils in the colon of a guinea pig infected with Shigella sonnei (Figure 1D).
Results using live, purified neutrophils typically show little-to-no cell staining at early time points, and gradually increase in RI-MUB40 staining over time as more neutrophils become activated. Shown here is a time course series of images from an experiment using purified human neutrophils infected with fluorescent S. sonnei (Figure 2). Depending on the strength of the activating signal, neutrophils may begin staining with MUB40 rapidly, so it is recommended to start acquiring images prior to the addition of activators. When cells become activated and RI-MUB40 positive, they should exhibit a similar staining profile to that of fixed and permeabilized cells, which give a punctate staining. The optimal concentration of MUB40 for both live and fixed cell staining is 1 μg/mL (Figure 3). Lower concentrations (0.1-0.01 μg/mL) can be used but result in lower signal intensity. It is also recommended to use a DIC image or other live-cell stain to differentiate which cells are displaying an RI-MUB40 signal.
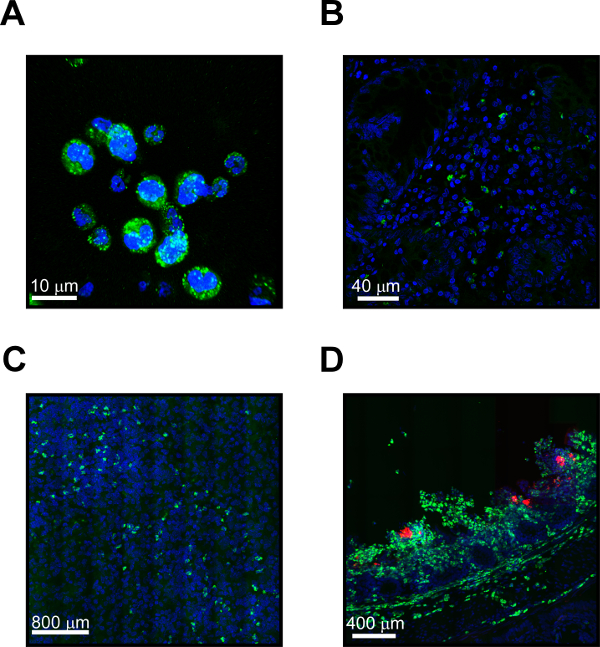
Figure 1: MUB40-stained neutrophils of human, mouse, and guinea pig origin. (A) Representative MUB40 staining (green) of neutrophils purified from healthy human donors. Cell nuclei are stained with DAPI (blue). (B) Staining of human neutrophils in tissue from an ulcerative colitis biopsy. Neutrophils are revealed with MUB40 (green) and cell nuclei are stained with DAPI (blue). (C) Histopathology from the lung of a mouse infected with Klebsiella pneumoniae. Mouse neutrophils are revealed in the tissue using MUB40 (green) and cell nuclei are stained with DAPI (blue). (D) Histopathology from the colon of a guinea pig infected with Shigella sonnei. Neutrophils responding to the infection are revealed in the tissue with MUB40 (green). S. sonnei bacteria (red) express the fluorescent dsRED protein. Cell nuclei are stained with DAPI (blue). Please click here to view a larger version of this figure.
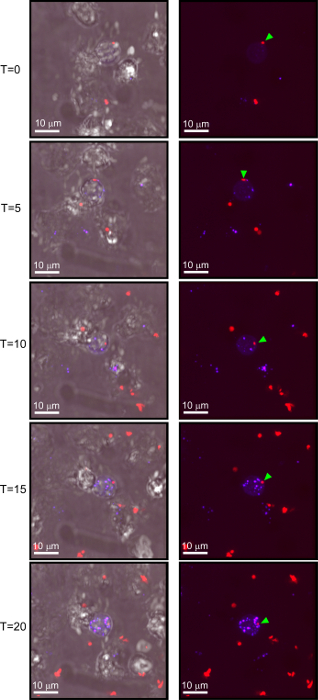
Figure 2: Live neutrophils stain with MUB40 only when activated. Selected images from a time lapse series involving purified human neutrophils infected with S. sonnei in the presence of RI-MUB40. In the first set of images (top) a neutrophil encounters a S. sonnei bacterium (red) shown with a green arrow. Over the time course, the bacterium is internalized by the neutrophil and digested. As the neutrophil becomes activated from the bacteria, it stains progressively stronger with RI-MUB40. Images on the left are fluorescent channels overlaid with DIC images. Images on the right are fluorescent channels only. Images were taken at 5-min intervals. Please click here to view a larger version of this figure.
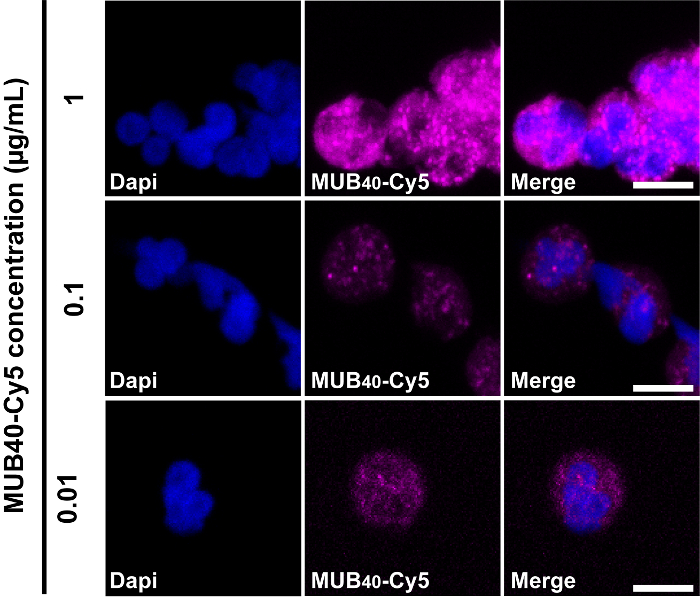
Figure 3: Effect of different MUB40 concentrations on neutrophil staining. Representative staining of fixed/permeabilized neutrophils with various concentrations of MUB40-Cy5. Purified human neutrophils were fixed and stained with the indicated concentrations of MUB40-Cy5. Cell nuclei are stained with DAPI (blue). Please click here to view a larger version of this figure.
Discussion
Here, two assays are described in which the MUB40 peptide is used to study neutrophil inflammation and activation. We show how MUB40 can reveal neutrophils present in histopathology sections or show their activation state using live purified neutrophils. The critical steps for using MUB40 as a staining reagent are the same as with any other fluorescent detection method. Care must be taken to ensure compatibility of fluorescent signals and that adequate washing steps have been taken to remove background staining. The most important considerations to achieve good staining results are the quality of the microscope, glass slides/cover slips, and the tissue sections. When viewing live purified neutrophils, the most important steps are in the purification process itself. Neutrophils are sensitive and can be activated by a number of different signals. In order to ensure that the experiment produces meaningful results, care must be taken to keep the purified neutrophils inactive until they are ready to be used. This can be achieved by performing the neutrophil purification protocol in an anoxic chamber to limit neutrophil exposure to oxygen.
Both a limitation and advantage of MUB40, depending upon the experimental question being addressed, is that MUB40 only stains activated neutrophils unless they are somehow permeabilized. This can be a huge advantage if the experiment calls for following inflammation in a live animal or purified cell, but it may be a disadvantage if the experiment requires detection of all live neutrophils regardless of activation state. A second potential limitation to this protocol is the fact that lactoferrin is present in various bodily secretions such as tears, milk, and mucus. For histopathology staining in which these secretions are maintained (e.g., colonic biopsies in which the mucus layer is intact), MUB40 will also stain the mucus layer along with any present neutrophils. In practice, this has yet to affect our analysis, but it should be noted as a potential signal source.
Currently, MUB40 is the only neutrophil biomarker that can differentiate between activated and non-activated live neutrophils. Also, MUB40, unlike antibody detection methods, does not appear to alter the function or survival of neutrophils in vitro or in vivo. For example, addition of specific antibodies against live neutrophils can be an activating signal affecting neutrophil function or survival in the host. This makes MUB40 a very useful reagent for research involving neutrophil activation or granule release in vitro or in vivo. Additionally, MUB40 has a broad host specificity range and can be directly conjugated to a number of different fluorophores, allowing for use in single-step staining assays across multiple hosts. Thus, MUB40 staining is comparable between mouse, human, and other mammalian targets, and it simplifies and reduces the number of reagents needed to detect neutrophils.
While this protocol focuses specifically on histopathology and live cell imaging using MUB40, we have also successfully used the peptide in FACS sorting and live animal in vivo imaging. We anticipate that MUB40 will be amenable to many different assays involving the detection of neutrophils or visualization of inflammatory events in the body, and that future studies and protocols will be developed to further leverage the spectrum of MUB40 uses, particularly using non-invasive imaging technics.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Fondation Laurette Fugain (LF-2015-15) (B.S.M.) and ANR JCJC grants (ANR-17-CE15-0012) (B.S.M.).
Materials
| MUB40-Cy5 | Benoit Marteyn | benoit.marteyn@pasteur.fr | Fluorescent MUB40 peptide (available with other conjugated fluorophores) |
| RI-MUB40-Cy5 | Benoit Marteyn | benoit.marteyn@pasteur.fr | Retro-inverso fluorescent MUB40 peptide. Synthesized with D-amino acids and resistant to proteases (available with other conjugated fluorophores) |
| Parformaldehyde | Sigma-Aldrich | 16005 | Fixative for histology |
| Sucrose | Sigma-Aldrich | S7903 | Solution used to remove excess PFA |
| Optimal Cutting Temperature Compound (OCT) | Sakura Finetek USA | 4583 | Used to freeze tissue before cryostat sectioning |
| 2-Methylbutane | Sigma-Aldrich | M32631 | Used to freeze tissue before cryostat sectioning |
| Ethanol | Sigma-Aldrich | 1.00974 | Used for dry ice bath to freeze tissue |
| Leica Cryostat | Leica Biosystems | CM1520 | Used to section tissues |
| Glass microscopy slides | Fisher Scientific | 12-518-101 | |
| Cover slips | Thor Labs | CG15CH | Hi precision coverslip |
| Dako pen | Sigma-Aldrich | Z377821 | Used to prevent liquid loss during tissue staining |
| Saponin | Sigma-Aldrich | 47036 | Used to permeabilize cells for IF staining |
| DAPI | Sigma-Aldrich | 10236276001 | Stains DNA |
| Alexa fluor 488 Phalloidin | Fisher Scientific | A12379 | Binds actin |
| Prolong Gold antifade Mountant | Fisher Scientific | P36930 | Prolongs IF signal and resistance to photobleaching |
| Fluorescent microscope | Various | Various | Used to image IF slides |
| Anoxic Cabinet | Various | Various | Used for the purification of live inactivated neutrophils |
| Sodium Chloride NaCL | Sigma-Aldrich | S7653 | |
| EDTA | Sigma-Aldrich | E9884 | Washing buffer component |
| MACS BSA buffer | Miltenyi Biotec | 130-091-376 | Washing buffer component |
| Percoll | Sigma-Aldrich | P4937 | Gradient for neutrophil purification |
| CD235a glycophorin magnetic microbeads | Miltenyi Biotec | 130-050-501 | Used to remove contaminating RBCs |
| LS columns | Miltenyi Biotec | 130-042-401 | Used in the removal of RBCs from neutrophils |
| RPMI-1640 without Phenol Red | Sigma-Aldrich | R8755 | Used for neutrophil assays |
Referências
- Nathan, C. Neutrophils and immunity: challenges and opportunities. Nature Reviews Immunology. 6 (3), 173-182 (2006).
- Nicolás-Ávila, J. &. #. 1. 9. 3. ;., Adrover, J. M., Hidalgo, A. Neutrophils in Homeostasis, Immunity, and Cancer. Immunity. 46 (1), 15-28 (2017).
- Coffelt, S. B., Wellenstein, M. D., de Visser, K. E. Neutrophils in cancer: neutral no more. Nature Reviews Cancer. 16 (7), 431-446 (2016).
- Wright, H. L., Moots, R. J., Bucknall, R. C., Edwards, S. W. Neutrophil function in inflammation and inflammatory diseases. Rheumatology. 49 (9), 1618-1631 (2010).
- Glennon-Alty, L., Hackett, A. P., Chapman, E. A., Wright, H. L. Neutrophils and redox stress in the pathogenesis of autoimmune disease. Free Radical Biology & Medicine. , (2018).
- Borregaard, N., Cowland, J. B. Granules of the human neutrophilic polymorphonuclear leukocyte. Blood. 89 (10), 3503-3521 (1997).
- Borregaard, N. Neutrophils, from marrow to microbes. Immunity. 33 (5), 657-670 (2010).
- Rose, S., Misharin, A., Perlman, H. A novel Ly6C/Ly6G-based strategy to analyze the mouse splenic myeloid compartment. Cytometry Part A: The Journal of the International Society for Analytical Cytology. 81 (4), 343-350 (2012).
- Shi, C., Hohl, T. M., Leiner, I., Equinda, M. J., Fan, X., Pamer, E. G. Ly6G+ neutrophils are dispensable for defense against systemic Listeria monocytogenes infection. Journal of Immunology. 187 (10), 5293-5298 (2011).
- Albanesi, M., Mancardi, D. A., et al. Neutrophils mediate antibody-induced antitumor effects in mice. Blood. 122 (18), 3160-3164 (2013).
- Anderson, M. C., Chaze, T., et al. MUB40 Binds to Lactoferrin and Stands as a Specific Neutrophil Marker. Cell Chemical biology. 25 (4), 483-493 (2018).
- Coïc, Y. -. M., Baleux, F., et al. Design of a specific colonic mucus marker using a human commensal bacterium cell surface domain. Journal of Biological Chemistry. 287 (19), 15916-15922 (2012).
- Roos, S., Jonsson, H. A high-molecular-mass cell-surface protein from Lactobacillus reuteri 1063 adheres to mucus components. Microbiology. 148, 433-442 (2002).
- Boekhorst, J., Helmer, Q., Kleerebezem, M., Siezen, R. J. Comparative analysis of proteins with a mucus-binding domain found exclusively in lactic acid bacteria. Microbiology. 152, 273-280 (2006).
- Mancardi, D. A., Jönsson, F., et al. Cutting Edge: The murine high-affinity IgG receptor FcγRIV is sufficient for autoantibody-induced arthritis. Journal of Immunology. 186 (4), 1899-1903 (2011).
- Walmsley, S. R., Print, C., et al. Hypoxia-induced neutrophil survival is mediated by HIF-1alpha-dependent NF-kappaB activity. Journal of Experimental Medicine. 201 (1), 105-115 (2005).
- Monceaux, V., Chiche-Lapierre, C., et al. Anoxia and glucose supplementation preserve neutrophil viability and function. Blood. 128 (7), 993-1002 (2016).

