Fabrication of Decellularized Cartilage-derived Matrix Scaffolds
Summary
Decellularized cartilage-derived scaffolds can be used as a scaffold to guide cartilage repair and as a means to regenerate osteochondral tissue. This paper describes the decellularization process in detail and provides suggestions to use these scaffolds in in vitro settings.
Abstract
Osteochondral defects lack sufficient intrinsic repair capacity to regenerate functionally sound bone and cartilage tissue. To this extent, cartilage research has focused on the development of regenerative scaffolds. This article describes the development of scaffolds that are completely derived from natural cartilage extracellular matrix, coming from an equine donor. Potential applications of the scaffolds include producing allografts for cartilage repair, serving as a scaffold for osteochondral tissue engineering, and providing in vitro models to study tissue formation. By decellularizing the tissue, the donor cells are removed, but many of the natural bioactive cues are thought to be retained. The main advantage of using such a natural scaffold in comparison to a synthetically produced scaffold is that no further functionalization of polymers is required to drive osteochondral tissue regeneration. The cartilage-derived matrix scaffolds can be used for bone and cartilage tissue regeneration in both in vivo and in vitro settings.
Introduction
Articular cartilage defects in the knee caused by traumatic events can lead to discomfort, and above all can have a large impact on the lives of the young and active population1,2,3. Moreover, cartilage damage at a young age may lead to a more rapid onset of osteoarthritis later in life4. Currently, the only salvage therapy for generalized osteoarthritis of the knee is joint replacement surgery. As cartilage is a hypocellular, aneural, and avascular tissue, its regenerative capacity is severely limited. Therefore, regenerative medicine approaches are sought after to aid and stimulate the regenerative capacity of the native tissue. For this purpose, scaffolds are designed and used as either a cell-carrier or as an inductive material that incites differentiation and regeneration of tissue by the body's native cells5.
Decellularized scaffolds have been widely studied within regenerative medicine6. It has had some success, for example, in aiding the regeneration of skin7, abdominal structures8, and tendons9. The advantage of using decellularized scaffolds is their natural origin and their capacity to retain bioactive cues that both attract and induce cell differentiation into the appropriate lineage required for tissue repair6,10. Moreover, since extracellular matrix (ECM) is a natural biomaterial, and decellularization prevents a potential immune response by removing cellular or genetic content, issues regarding biocompatibility and biodegradability are overcome.
Cartilage-derived matrix (CDM) scaffolds have shown great chondrogenic potential in in vitro experiments when seeded with mesenchymal stromal cells11. In addition, these scaffolds have shown the potential to form bone tissue through endochondral ossification on ectopic locations in in vivo settings12. As CDM scaffolds guide the formation of both bone and cartilage tissue, these scaffolds may hold potential for osteochondral defect repair in addition to cartilage repair.
This article describes a protocol adapted from Yang et al. (2010)13 for the production of decellularized CDM scaffolds from equine stifle cartilage. These scaffolds are rich in collagen type II and devoid of cells, and do not contain any glycosaminoglycans (GAGs) after decellularization. Both in vitro and in vivo experiments on (osteo)chondral defect repair can be conducted using these scaffolds.
Protocol
For this protocol, equine stifle cartilage was obtained from horses that had died from other causes than osteoarthritis. Tissue was obtained with permission of the owners, in line with the institutional ethical regulations.
NOTE: This protocol describes the fabrication of scaffolds from decellularized equine cartilage, which can be used for applications such as in vitro tissue culture platforms or for in vivo implantation in regenerative medicine strategies. The enzymatic treatment steps must be performed in the described chronological order.
1. Harvesting of Articular Cartilage from Donor (Cadaveric) Joints
- Ahead of the harvesting step, prepare 1 L of cartilage washing solution, consisting of sterile phosphate-buffered saline (PBS), supplemented with 100 U/mL penicillin, 100 µg/mL streptomycin, and 1% (v/v) Amphotericin B.
- Wear gloves and a lab coat during the entire harvesting procedure, since the donor can carry pathogens.
- Obtain an intact (cadaveric) joint for cartilage isolation.
NOTE: At this point, the skin around the joint is still intact. Equine stifle cartilage obtained from horse was used in this protocol, but other joints from other species can also be used. - Remove the skin that is located around the joint area of interest by using a scalpel and a surgical tweezer. In addition, remove excessive fat and muscle tissue if necessary, without damaging the underlying joint. Optionally, transfer the prepared cadaver to a biological safety cabinet.
- To perform an arthrotomy, i.e., a separation of the joint, first define the location where the joint articulates by performing extension and flexion of the joint.
- Carefully remove more layers of fat, muscles, and tendons with a scalpel and surgical tweezer to open up the joint area.
- Make an incision to reach the joint cavity.
NOTE: The joint cavity is filled with synovial fluid, which can drip from the cavity when performing a correct incision. - Continue to open up the joint completely by cutting through the tendons that keep the joint together.
- Inspect the cartilage for any macroscopic damage.
NOTE: If the articular cartilage does not have a glossy and smooth appearance or if evident blistering, clefts, or defects are present, discard the cartilage. - Use a sterile scalpel to remove the cartilage from the subchondral bone. Cut all the way down to the subchondral bone to also remove the deep zone of the cartilage (Figure 1). To prevent the cartilage from drying out, regularly drip cartilage washing solution on the cartilage.
NOTE: At this time, the size of the cartilage slices does not matter. - Collect the cartilage slices in 50 mL tubes containing previously prepared cartilage washing solution (Figure 2A).
- After collecting the cartilage from all the areas of interest, snap freeze the cartilage slices in liquid nitrogen for 5 min.
- Transfer the cartilage slices into 50 mL tubes and immediately lyophilize the cartilage slices for 24 h in a freeze dryer. Set the freeze dryer at approximately 0.090 mbar while the ice condenser is -51 °C (Figure 2B).
- Store the cartilage slices in a dry place at room temperature until further use.
NOTE: Up to the point of scaffold formation, the solutions and cartilage do not have to be prepared and treated in a sterile way.
2. Creating Decellularized Cartilage Particles
- Submerse the lyophilized cartilage slices in liquid nitrogen.
- Directly grind the samples either manually or by a milling machine. When grinding the cartilage slices by hand, use a mortar and pestle and grind the slices for approximately 45 min until they are pulverized (Figure 2C and 2D). When grinding automatically, use a milling machine at its pre-set speed for a few seconds up to a minute, until the snap-frozen cartilage slices are pulverized.
NOTE: Pre-cool the mortar or the grinding compartment of the milling machine by adding liquid nitrogen. When using a milling machine, make sure that all particles are ground, and that no particles stay in the bottom of the grinding compartment. - Sieve the cartilage particles to get rid of larger parts by using a sieve with a 0.71 mm mesh size.
- Store the particles in a dry place at room temperature.
3. Enzymatic Decellularization with Trypsin 0.25%-EDTA
- Prepare the digestion solution, consisting of 0.25% trypsin with EDTA supplemented with 100 U/mL penicillin, 100 μg/mL streptomycin, and 1% (v/v) Amphotericin B. Store the solution at 4 °C.
NOTE: Trypin is a protease that degrades proteins residing in the cartilage tissue. This enzymatic step will open up the dense cartilage structure. - Fill the 50 mL tubes with the pulverized cartilage particles up to a volume of approximately 7.5 mL per tube.
- Incubate the cartilage particles with the prepared digestion solution at 37 °C for 24 h in total, while refreshing the digestion solution every 4 h.
- First, add 30 mL of the digestion solution to each 50 mL tube that contains cartilage particles.
- Then, resuspend the particles by using a vortex or pipet so that all particles are exposed to the enzymatic solution.
- Incubate the samples for 4 h at 37 °C on a roller.
- To refresh the digestion solution, centrifuge the tubes for 20 min at 3113 x g to cause sedimentation of the cartilage particles. Discard the supernatant.
NOTE: The supernatant will become clearer with every trypsin incubation period. - Repeat step 3.3.1–3.3.4 until the particles have been incubated with the digestion solution for 6 cycles of 4 h.
- After the final cycle, remove the supernatant after centrifuging and wash the particles in 30 mL of cartilage washing solution twice. Centrifuge the particles for 20 min at 3113 x g between each of the washes.
4. Enzymatic Decellularization with Nuclease Treatment
- Prepare a 10 mM Tris-HCl solution at pH 7.5 in deionized water.
- Add 50 U/mL deoxyribonuclease and 1 U/mL ribonuclease A to the Tris-HCl buffer, to obtain the nuclease solution.
NOTE: This step is performed to specifically degrade deoxyribonucleases and ribonucleases. - To remove the cartilage washing solution from the cartilage particles (step 3.4), centrifuge for 20 min at 3,113 x g, and remove the supernatant.
- Add 30 mL of the nuclease solution to the cartilage particles and stir the particles through the solution, making sure that all particles are exposed to the nuclease solution.
- Incubate the samples on a roller for 4 h at 37 °C.
- Remove the nuclease solution by centrifuging the cartilage particles for 20 min at 3,113 x g and discard the supernatant.
- Wash the samples by adding 30 mL of 10 mM Tris-HCl solution without the deoxyribonuclease and ribonuclease A to the cartilage particles. Resuspend the particles and leave the samples on a roller for 20 h at room temperature.
5. Detergent Decellularization
- Make the detergent solution by dissolving 1% (v/v) octoxynol-1 in PBS.
- Remove the Tris-HCl solution from the cartilage particles by centrifuging for 20 min at 3,113 x g and discarding the supernatant.
- Add 30 mL of the prepared 1% detergent solution to the cartilage particles. Resuspend the cartilage particles gently to avoid foaming of the solution.
NOTE: This step breaks down cellular membranes. - Incubate the samples on a roller for 24 h at room temperature on a roller.
- Remove the detergent solution by centrifugation for 20 min at 3,113 x g and discard the supernatant.
- To remove all of the remnants of the decellularization solutions, wash the cartilage particles in 6 cycles of 8 h in 30 mL of cartilage washing solution. Perform washing on a roller at room temperature.
- To change to the next wash, centrifuge the suspension for 20 min at 3,113 x g, discard the supernatant and add fresh cartilage washing solution.
- Leave the last wash in the tubes and store the decellularized cartilage particles at -80 °C.
6. Creating Scaffolds from the Decellularized Particles
- If the particles have been stored at -80 °C, thaw the closed tubes that contain the cartilage particles in warm water before creating the scaffolds.
- Transfer the cartilage particles with a small ladle to a cylindrical mold, for example, a plastic vial with a diameter of 8 mm and a height of 2 cm.
- When placing the cartilage particles into the plastic mold, press all the air-bubbles out to avoid cavities in the scaffold, and fill the mold until the edge.
NOTE: It will be more difficult to take the scaffolds out of a metal mold after scaffold formation, which can lead to cracks in the scaffold. - Freeze the molds with cartilage particles for 10 min at -20 °C.
- Lyophilize the cartilage scaffolds within their molds for 24 h in a freeze dryer.
- After lyophilization, take the scaffold out of the mold (Figure 3) and cross-link them with ultraviolet (UV) light at 30 cm distance and 365 nm overnight.
- In order to use the scaffolds for in vitro cell culture or in vivo implantation, sterilize the scaffolds with, for example, ethylene oxide (EtO) gas.
NOTE: EtO sterilization is performed by an external party.
7. Characterization of the Decellularized Scaffolds with Histological Stainings
NOTE: To ensure complete decellularization and to visualize the remaining natural character of the cartilage, perform several histological stainings before using the scaffolds in any experiment, including hematoxylin and eosin (H&E) staining to ensure decellularization, Safranin-O staining to visualize residual GAG presence, collagen type I immunohistochemistry to differentiate between collagen content, and collagen type II immunohistochemistry to differentiate between collagen content.
- Cut the scaffolds in thin slices of approximately 3 mm with a scalpel.
NOTE: If the scaffolds are cut in larger or smaller sizes, the durations of the dehydration cycles need to be adapted. - Embed the scaffolds in a drop of 4% (w/v) alginate and induce cross-linking by adding a similar volume of 3.7% non-buffered formalin that contains 20 mM CaCl2.
NOTE: Alginate embedding makes the scaffold slices more rigid compared to the washing steps prior to paraffin embedding. In case the scaffolds have been cell-seeded or in vivo implanted, alginate embedding is not necessary, as the composition of the scaffolds will be resistant enough due to ECM incorporation by the cells. - Dehydrate the samples by placing them in a graded ethanol series, starting with one-hour cycles of 70%, 96%, 96%, 100%, and 100% ethanol, followed by two 1 h cycles of xylene, and ending with two 1 h cycles of paraffin at 60 °C.
- After dehydration, paraffin-embed the samples in a mold and cool the samples down.
- Cut the samples with a microtome in slices of 5 μm thick.
- Before staining, rehydrate the samples by placing them in a returned graded ethanol series. Start with two washes of xylene for 5 min, followed by 2 washes of 100%, 95%, and 70% ethanol for 3 min per wash. Finally, wash the samples 3 times in water for 2 min.
NOTE: In case of using alginate to process samples for paraffin embedding, make sure to wash off the alginate with 10 mM citric acid prior to rehydration of the paraffin sections. - Stain the samples with hematoxylin and eosin, Safranin-O, collagen I, and collagen II as previous described11.
8. Characterization of the Decellularized Scaffolds with Quantitative Analyses
- Obtain papain digests of the scaffolds as described previously11.
- Perform an assay with a fluorescence-based DNA quantification kit to measure the double-strand DNA content of the scaffolds to ensure complete decellularization. Follow the protocol provided by the manufacturer. Express the amount of DNA per dry weight of the scaffold.
- Perform a dimethylmethylene blue assay to quantify the remainder of the GAGs within the scaffold, as described previously11. Express the amount in GAG per DNA.
9. Seeding of the Decellularized Scaffolds
- Cut sterilized scaffolds from step 6.7 into 3 mm thick slices.
- Transfer the scaffolds in separate wells of a 6-well plate.
- Rehydrate scaffolds with cell culture medium by pipetting 1 mL of medium on top of the scaffold and let it soak for 30 min. Use either chondrocyte or mesenchymal stem cell (MSC) expansion medium.
Note: Use the same medium for scaffold rehydration as for cell culture of the cells that will be seeded. Chondrocyte expansion medium consists of Dulbecco's modified media (DMEM) with 10% heat-inactivated fetal bovine serum (FBS), 100 U/mL penicillin, 100 μg/mL streptomycin, and 10 ng/mL fibroblast growth factor-2 (FGF-2). MSC expansion medium consists of minimum essential medium alpha (a-MEM) with 10% heat-inactivated FBS, 0.2 mM L-ascorbic acid 2-phosphate, 100 U/mL penicillin, 100 μg/mL streptomycin, and 10 ng/mL FGF-2. - Prepare 3 x 106 cells in 100 μL of medium, which is the required volume for each scaffold with a diameter of 8 mm and a height of 3 mm.
NOTE: Multiple cell types from different species can be used for seeding. When using chondrocytes or mesenchymal stromal cells, isolate the cells as previously described11. When using chondrocytes, make sure that they have not been expanded past the P1 passage in order to minimize the number of dedifferentiated chondrocytes. When using mesenchymal stromal cells, they must be tested on their ability for multi-lineage differentiation, as previously described14. - Pipet 50 μL of prepared cell suspension on top of the pre-soaked scaffold and incubate the scaffold for 1 h at 37 °C.
- Carefully turn the scaffold upside down, pipet the remaining 50 μL of cell suspension on this side of the scaffold and incubate for 1 h at 37 °C.
- After incubation, add 3 mL of medium to the wells, and culture the cell-seeded scaffolds at 37 °C. Handle the culture plate gently to avoid detachment of the cells.
- Culture the scaffolds for the period of time that is required for the experiment and change the medium 2–3 times a week. Pipet slowly and as far away from the scaffold as possible.
- After culturing of the cell-seeded scaffold, cut the scaffolds in half and process them for both histology and biochemical analyses.
Representative Results
Decellularization of CDM scaffolds must always be confirmed using histological stainings as well as using DNA quantification to measure the amount of DNA remnants. Insufficient decellularization might lead to undesired immunological responses that influence the results in in vivo settings15,16,17. For this specific decellularization method, DNA was below the detection range, which started at 13.6 ± 2.3 ng/mg DNA/dry weight (n = 3). Full decellularization using this protocol will lead to the production of a scaffold that is rich in collagen type II (Figure 4C) and has no cells (Figure 4A) or GAGs (Figure 4B). Healthy equine cartilage is displayed as comparison, stained for Safranin-O (Figure 5A) and collagen II (Figure 5B).
The scaffold must display a macroscopically homogeneous porosity. Air bubbles will lead to easily detectable large holes in the scaffold and are, therefore, carefully removed. These large holes in the scaffold may have a detrimental impact on the mechanical properties and lead to inhomogeneous cell attachment upon seeding. Successful production of the scaffold also involves a freeze-drying step lasting for at least 24 h; this will lead to a scaffold that has a white appearance (Figure 3). In case of insufficient lyophilization, the scaffolds will have a yellowish color and no clear pores can be observed.
In order for the scaffold to be used for in vivo applications and in vitro cell-seeding, cell integration with the scaffold, as well as cellular functionality, must be shown. Here, scaffolds were seeded with MSCs that produced ECM after 4 (Figure 6A-D) and 6 (Figure 6E–H) weeks of in vitro culturing. Formation of GAG, collagen II, and a peripheral collagen I, as well as the presence of cells, was shown. Additionally, the specificity of collagen II is displayed in Figure 5B, where cartilage but not bone is stained positive for collagen II in an osteochondral plug.
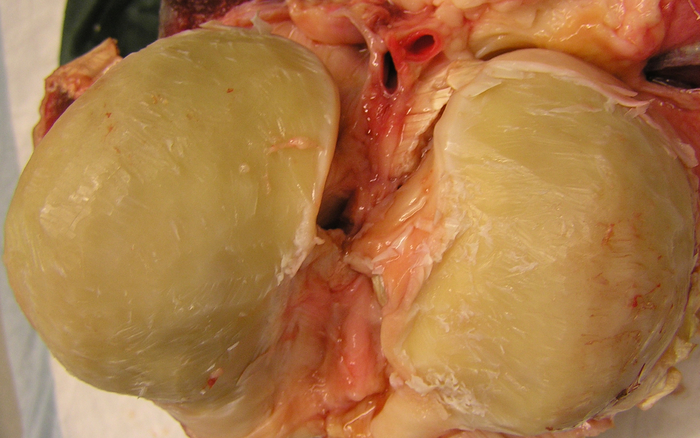
Figure 1: Equine knee after removing full-thickness cartilage. The cartilage is removed from the condyles using a scalpel until the calcified cartilage layer that cannot be cut using a scalpel is reached. Please click here to view a larger version of this figure.
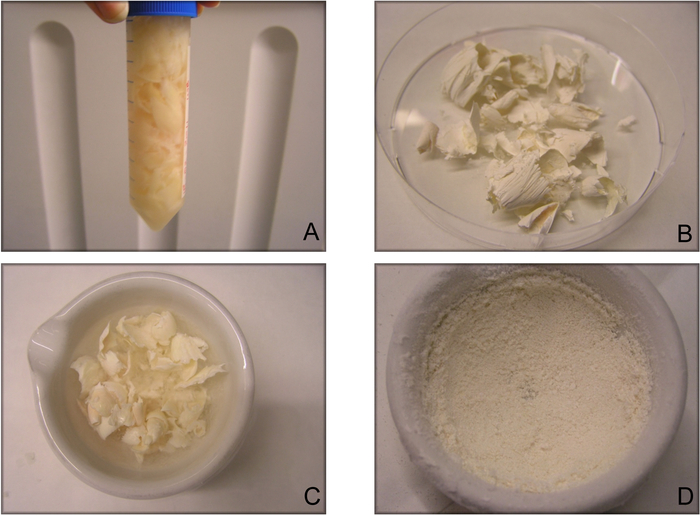
Figure 2: Sequential steps in creating decellularized cartilage-derived matrix particles. (A) Cartilage slices that have been removed from the condyles are washed in an antibiotic-infused solution. (B) Cartilage slices are lyophilized, and now have a white and paper-like appearance. (C) Snap-freezing of the lyophilized cartilage is performed right before (D) pulverizing cartilage particles by hand-milling using a mortar and pestle. Step D can also be done with automatic milling. This figure has been modified from Benders et al.11. Please click here to view a larger version of this figure.

Figure 3: The final product, a decellularized cartilage-derived matrix scaffold. This cylindrical scaffold is 2 cm high and has a diameter of 8 mm. The scaffold has a clear porous structure. The left and right picture display a scaffold from two different angles. Note that no large holes are present at the surface of the scaffold as all of the air bubbles were removed prior to lyophilization. This figure has been modified from Benders et al.11. Please click here to view a larger version of this figure.
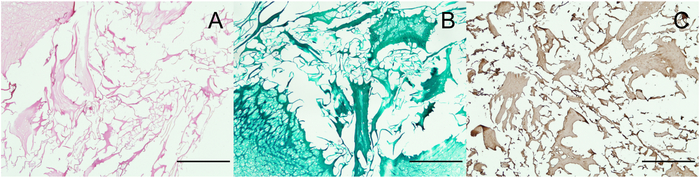
Figure 4: Histological characterization of the scaffold. (A) H&E staining shows ECM particles of different sizes and the absence of cells. (B) Safranin-O staining shows that no GAGs have been retained in the decellularization process. (C) Collagen type II immunolocalization reveals that the decellularized particles are rich in collagen type II. Scale bars = 500 µm. This figure has been modified from Benders et al.11. Please click here to view a larger version of this figure.
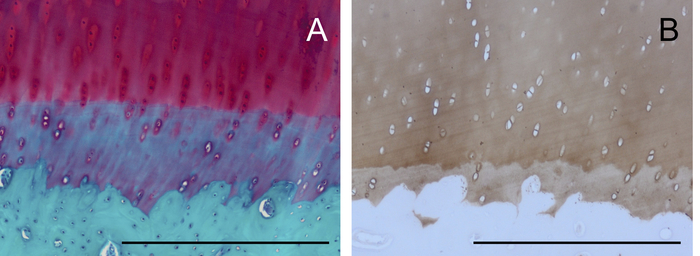
Figure 5: Histological representation of healthy equine cartilage. (A) An osteochondral plug stained with Safranin-O and Fast Green shows bone without GAGs (green), cartilage with GAGs (red), and the calcified cartilage layer in between. (B) A collagen II-stained osteochondral plug stains cartilage but not bone. Scale bars = 500 µm. Please click here to view a larger version of this figure.
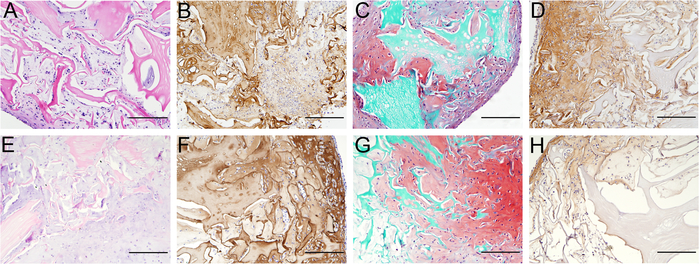
Figure 6: Neo-matrix formation on the scaffold after 4 and 6 weeks of culture using mesenchymal stromal cells. After 4 (A-D) and 6 (E-H) weeks of culture, newly formed matrix is rich in cells (A+E), collagen II (B+F), and GAGs (C+G), as can be observed with H & E, collagen II, and Safranin-O stainings, respectively. IN addition, collagen I is present after both 4 (D) and 6 (H) weeks in the periphery of the scaffold. Cell density as well as the amount of matrix deposition is higher at the periphery. Scale bars = 500 µm. This figure has been modified from Benders et al.11. Please click here to view a larger version of this figure.
Discussion
The ECM of articular cartilage is very dense and quite resilient to different enzymatic treatments. The multi-step decellularization protocol described in this article addresses such resistance and successfully generates decellularized matrices. To achieve that, the process spans over several days. Many decellularization processes have been proposed for different types of tissues18, and this article describes a protocol suitable for the decellularization of cartilage. In this protocol, it is, however, necessary to follow the enzymatic treatment with the detergent steps in order to remove all cells. The amount of DNA is diminished remarkably in the first few steps involving the treatment with trypsin, and leaving out these steps will not result in proper decellularization11.
Note that this protocol is based on the decellularization of equine cartilage tissue. The activity of enzyme solutions used was found sufficient for the adequate removal of the equine chondrocytes. However, despite the conservation of the matrix composition across species, the protocol may have to be adjusted for decellularization of cartilage from other animals due to the differences in the amount of naturally residing chondrocytes19. For example, cartilage of smaller animals is known to have a higher cell content, and may, therefore, require a more aggressive decellularization process. A particular reason for choosing equine cartilage to create decellularized scaffolds is that equine and human cartilage show clear resemblance in thickness, cell density, and biochemical make-up20.
To ensure a reproducible product, several assessment criteria may be important to determine whether complete decellularization has been achieved. In this protocol, both an H&E staining and biochemical quantification were used to evaluate the residual amount of DNA in the end product. Other researchers have also proposed to determine the size of the remaining DNA, with a maximum of 200 bp in length for quality control21, or a residual DNA amount of less than 50 ng/mg dry tissue weight18,22. Regardless, alterations to the protocol must always be followed up with histological evaluation and quantitative assays to determine the effect of decellularization, as well as the remaining ECM products.
The main limitation of this protocol is that, the thorough decellularization procedure involving the exposure to trypsin leads to extensive loss of GAGs. Even though trypsin does not cleave GAGs, the reason for the loss of GAGs can be the opening up of the cartilage tissue by trypsin cleaving proteins that anchor or encapsulate GAGs. ECM components like GAGs are important for retaining water in articular cartilage, and therefore play a significant role in the biomechanical resilience of the tissue4. Protocols that aim to reduce the loss of GAGs throughout the decellularization process will affect the thoroughness of the decellularization process.
After decellularization, cell integration and function have been shown in cell-seeded scaffolds. Previous research has shown that matrix production by chondrocytes on this scaffold is unsatisfactory, especially when compared to the abundant matrix deposition by mesenchymal stromal cells11. As cartilage-like tissue is deposited on the scaffold, this new matrix is generally first deposited in the periphery of the scaffold before invading the rest of the scaffolds. This can be clearly observed on the histological slices where a cell-rich periphery, rather than a cell-rich center, is often seen (Figure 6). However, this effect may be reduced when using perfusion bioreactors for cell seeding and to enhance nutrient exchange. As matrix deposition occurs, the scaffolds will also assume a glossier appearance, becoming more mechanically consistent and less brittle. As such, they can be easily cut using a scalpel without falling apart. The properties of the newly formed matrix can be evaluated using both histological stainings and quantitative assays. As no GAGs are left after the decellularization process, all of the GAGs that can be quantitatively measured will be a product of neosynthesis.
The scaffolds produced using this decellularization protocol provide an off-the-shelf solution and can be implanted without the necessity of cell-seeding prior to implantation. However, when applied as a treatment for (osteo)chondral defects, the biomechanical properties will have to be enhanced to diminish the chance of indentation of the construct in the early phases of articular loading. Co-implantation with protective layers on top, or other reinforcement strategies may need to be implemented. In the future, further refinement of the protocol may enhance the regenerative potential of the scaffold. For instance, the retention of GAGs, the preservation of collagen fiber orientation, or combination with other biomaterials to reinforce these scaffolds may be beneficial to allow for an improved and smoothly regenerated articular surface. Consequently, these scaffolds may play a role as components of the next generation of regenerative grafts for the treatment of (osteo)chondral defects.
Declarações
The authors have nothing to disclose.
Acknowledgements
The authors would like to acknowledge W. Boot for assistance in the production of the scaffolds. K.E.M. Benders is supported by the Alexandre Suerman Stipendium from the University Medical Center. R. Levato and J. Malda are supported by the Dutch Arthritis Foundation (grant agreements CO-14-1-001 and LLP-12, respectively).
Materials
| Cadaveric joint | This can be obtained as rest material from the local butcher or veterinary center. | ||
| Sterile phosphate-buffered saline (PBS) | |||
| Penicillin-Streptomycin | Gibco | 15140 | |
| Amphotericin B | Thermo Fischer Scientific | 15290026 | |
| Liquid nitrogen | |||
| Trypsin-EDTA (0.25%), phenol red | Thermo Fischer Scientific | 25200072 | |
| Tris-HCl pH 7.5 | |||
| Deoxyribonuclease I from bovine pancreas | Sigma-Aldrich | DN25 | |
| Ribonuclease A from bovine pancreas | Sigma-Aldrich | R6513 | |
| Triton X-100 (octoxynol-1) | Sigma-Aldrich | X100 | |
| Papain | Sigma-Aldrich | P3125 | |
| Trisodium citrate dihydrate | Sigma-Aldrich | S4641 | |
| Alginate | Sigma-Aldrich | 180947 | |
| Formalin | |||
| CaCl2 | |||
| Ethanol | |||
| Xylene | |||
| Paraffin | |||
| Ethylene oxide sterilization | Synergy Health, Venlo, the Netherlands | ||
| Multipotent Stromal cells/chondrocytes from equine donors | MSCs and chondrocytes can be isolated from donor joints that are rest material, coming from the local butcher or veterinary center. | ||
| MEM alpha | Thermo Fischer Scientific | 22561 | |
| L-ascorbic acid 2-phosphate | Sigma-Aldrich | A8960 | |
| DMEM | Thermo Fischer Scientific | 41965 | |
| Heat inactivated bovine serum albumin | Sigma-Aldrich | 10735086001 | |
| Fibroblast growth factor-2 (FGF-2) | R & D Systems | 233-FB | |
| DNA quantification kit (Quant-iT PicoGreen dsDNA Reagent) | Thermo Fischer Scientific | P7581 | |
| 1,9-Dimethyl-Methylene Blue zinc chloride double salt | Sigma-Aldrich | 341088 | |
| Freeze-dryer | SALMENKIPP | ALPHA 1-2 LD plus | |
| Analytical mill | IKA | A 11 basic | |
| mortar/pestle | Haldenwanger 55/0A | ||
| Roller plate | CAT | RM5 | |
| Centrifuge (for 50 mL tubes) | Eppendorf | 5810R | |
| Capsule (cylindric mold) | TAAB | 8 mm flat | |
| Superlight S UV | Lumatec | 2001AV | |
| Incubator | |||
| Microtome | |||
| Sieve (mesh size 0.71 mm) | VWR | 34111229 | |
| Scalpel | |||
| Scalpel holder | |||
| Small laddle |
Referências
- Dunlop, D. D., et al. Risk factors for functional decline in older adults with arthritis. Arthritis and rheumatism. 52 (4), 1274-1282 (2005).
- Fitzpatrick, K., Tokish, J. M. A military perspective to articular cartilage defects. The journal of knee surgery. 24 (3), 159-166 (2011).
- Flanigan, D. C., Harris, J. D., Trinh, T. Q., Siston, R. A., Brophy, R. H. Prevalence of chondral defects in athletes’ knees: a systematic review. Medicine and science in sports and exercise. 42 (10), 1795-1801 (2010).
- Martel-Pelletier, J., Boileau, C., Pelletier, J. P., Roughley, P. J. Cartilage in normal and osteoarthritis conditions. Best practice & research. Clinical rheumatology. 22 (2), 351-384 (2008).
- Vinatier, C., et al. Cartilage tissue engineering: towards a biomaterial-assisted mesenchymal stem cell therapy. Current stem cell research & therapy. 4 (4), 318-329 (2009).
- Taylor, D. A., Sampaio, L. C., Ferdous, Z., Gobin, A. S., Taite, L. J. Decellularized matrices in regenerative medicine. Acta biomaterialia. 74, 74-89 (2018).
- Vashi, C. Clinical Outcomes for Breast Cancer Patients Undergoing Mastectomy and Reconstruction with Use of DermACELL, a Sterile, Room Temperature Acellular Dermal Matrix. Plastic Surgery International. 2014 (704323), 1-7 (2014).
- Satterwhite, T. S., et al. Abdominal wall reconstruction with dual layer cross-linked porcine dermal xenograft: the "Pork Sandwich" herniorraphy. Journal of plastic, reconstructive & aesthetic surgery : JPRAS. 65 (3), 333-341 (2012).
- Martinello, T., et al. Successful recellularization of human tendon scaffolds using adipose-derived mesenchymal stem cells and collagen gel. Journal of tissue engineering and regenerative medicine. 8 (8), 612-619 (2014).
- Benders, K. E., et al. Extracellular matrix scaffolds for cartilage and bone regeneration. Trends in biotechnology. 31 (3), 169-176 (2013).
- Benders, K. E., et al. Multipotent Stromal Cells Outperform Chondrocytes on Cartilage-Derived Matrix Scaffolds. Cartilage. 5 (4), 221-230 (2014).
- Gawlitta, D., et al. Decellularized cartilage-derived matrix as substrate for endochondral bone regeneration. Tissue engineering. Part A. 21 (3-4), 694-703 (2015).
- Yang, Z., et al. Fabrication and repair of cartilage defects with a novel acellular cartilage matrix scaffold. Tissue engineering. Part C, Methods. 16 (5), 865-876 (2010).
- Pittenger, M. F., et al. Multilineage potential of adult human mesenchymal stem cells. Science. 284 (5411), 143-147 (1999).
- Meyer, S. R., et al. Decellularization reduces the immune response to aortic valve allografts in the rat. The Journal of thoracic and cardiovascular surgery. 130 (2), 469-476 (2005).
- Brown, B. N., Valentin, J. E., Stewart-Akers, A. M., McCabe, G. P., Badylak, S. F. Macrophage phenotype and remodeling outcomes in response to biologic scaffolds with and without a cellular component. Biomaterials. 30 (8), 1482-1491 (2009).
- Keane, T. J., Londono, R., Turner, N. J., Badylak, S. F. Consequences of ineffective decellularization of biologic scaffolds on the host response. Biomaterials. 33 (6), 1771-1781 (2012).
- Crapo, P. M., Gilbert, T. W., Badylak, S. F. An overview of tissue and whole organ decellularization. Biomaterials. 32 (12), 3233-3243 (2011).
- Malda, J., et al. Of mice, men and elephants: the relation between articular cartilage thickness and body mass. PloS One. 8 (2), e57683 (2013).
- Malda, J., et al. Comparative study of depth-dependent characteristics of equine and human osteochondral tissue from the medial and lateral femoral condyles. Osteoarthritis and Cartilage. 20 (10), 1147-1151 (2012).
- Londono, R., Badylak, S. F. Biologic scaffolds for regenerative medicine: mechanisms of in vivo remodeling. Annals of biomedical engineering. 43 (3), 577-592 (2015).
- Gilbert, T. W. Strategies for tissue and organ decellularization. Journal of cellular biochemistry. 113 (7), 2217-2222 (2012).

