Summary
Anastomotic leak or breakdown after surgery is a major cause of postoperative morbidity and mortality. Our surgery for creating a colonic anastomosis is a reliable and reproducible method for studying the healing mechanisms of anastomoses.
Abstract
Intestinal anastomoses are commonly performed in both elective and emergent operations. Even so, anastomotic leaks are a highly feared complications of colonic surgeries and can occur in up to 26% of surgical anastomoses, with mortality being up to 39% for patients with such a leak. Currently, there remains a paucity of data detailing the cellular mechanisms of anastomotic healing. Devising preventative strategies and treatment modalities for anastomotic leak could be greatly potentiated by a better understanding of appropriate anastomotic healing.
A murine model is ideal as previous studies have shown that the murine anastomosis is the most clinically similar to the human case as compared with other animal models. We offer an easily reproducible murine model of colonic anastomosis in mice that will allow for further illustration of anastomotic healing.
Introduction
A highly feared complication of surgical anastomosis of the intestine is anastomotic breakdown or leak. Anastomotic leak, which causes spillage of the intraluminal contents into the abdominal cavity, is life threatening and can quickly lead to intra-abdominal sepsis; shock rates vary from 0.3% to 5.5% following small bowel anastomoses, and rise to between 0.5% and 26% for colonic anastomoses1,2. Mortality rates following leaks can be as high as 39% due to the swift onset of sepsis and rapid progression to death following intraabdominal contamination3. Preventative strategies and treatment modalities are currently based on pathophysiology that is not fully understood.
Currently, anastomotic healing is often compared to the more widely studied cutaneous wound healing, which is proving to be a relatively poor facsimile. Healing occurs in a series of overlapping phases, starting with the initial lag phase. During days 0-4 after creation of the intestinal anastomosis, the lag phase is defined by an acute inflammatory response that clears the wound of cellular debris. Next, over days 3-4, fibroblastic proliferation and production of collagen typifies the fibrophasia phase. Finally, after day 10, a prolonged period of collagen remodeling is known as the maturation phase. It is important to note that anastomotic strength is quite low and depends on the extrinsic support of surgically placed staples or suture until collagen is deposited4. Understanding the role and timing each layer of the bowel wall plays in anastomotic wound healing and the involvement in inflammatory-mediated cell types such as macrophages, as well as elucidating surrogate markers to predict anastomotic failure or success could greatly reduce morbidity, mortality, and healthcare expenditure following colon operations5.
The murine model has been shown in previous studies to be the useful in mimicking human anastomosis6. While there are popular, proven and well-studied examples of the murine colonic anastomotic model, particularly the method described by Komen et al.7, these models favor ileocecal or ascending colocolonic anastomotic techniques8. Previous studies of human patients have demonstrated significant difference in leak rates between ileocecal, colocolonic and colorectal anastomoses, highlighting the need for further experimental models at varying anatomic locations. Additional, popularly used methods are aimed at developing a model which intentionally creates an anastomotic leak rather than elucidate the cellular mechanisms required for normal anastomotic wound healing9. Rat models have been attempted in the past but have rates of anastomotic dehiscence and/or signs of leak (i.e., abscess formation) significantly lower than humans and mice6,7. Additionally, more genetically modified experimental lines are available in mice than rats. This makes the rat model less useful for an anastomotic model. In addition, porcine and canine models have had less clinical investigation than the murine model6,7.
We propose a safe, technically simple, and easily and quickly reproducible procedure for the creation of colonic anastomoses in a murine model that should facilitate further investigation into the underreported mechanism of anastomotic wound healing10.
Protocol
The following protocol has been approved by the University of Oklahoma Health Sciences Center Institutional Animal Care and Use Committee (IACUC) and complies with all institutional ethical guidelines regarding the use of research animals. Additionally, all experiments were performed in accordance with institutional, state and federal regulations regarding experimental animal research.
NOTE: For this protocol, we used female and male congenic in-bred C57BL6/J mice at 12 to 72 weeks of age. All mice used for this procedure were kept at our non-barrier facility for at least one week prior to the operation to adapt to the local biome. Animals were allowed to feed up to time of operation and we used neither oral mechanical bowel preparation nor rectal flushing prior to procedure.
1) Creation of Anastomoses
- Autoclave the surgical instruments, warm the saline irrigation solution and thoroughly clean all operative surfaces with 70% ethanol.
- To induce anesthesia, place the mouse in an induction box that is connected to a vaporizer. Deliver 2-3% isoflurane at 1-2 L/min until mice are sedated.
- Place the animal on a warming device, such as a heating pad, to prevent hypothermia.
- Subcutaneously inject a one time dose of 0.1 mg/kg buprenorphine (3 µg for a 30 g mouse) for postoperative analgesia and a one time dose of 10 mg/kg of enrofloxacin for preoperative antibiotic prophylaxis.
NOTE: Though pre- and postoperative antibiotics are not required for this operation, institutional guidelines and the animal care and use committee at our institution recommended their use at our facility. - Place the anesthetic nose cone around the mouse's snout. Throughout the operation, carefully monitor the respiratory rate of the animal and adjust the anesthetic flow rate as needed. Maintain a respiratory rate at 30-40 breaths per minute.
- Use a hair clipper followed by a depilatory agent to remove the hair from the entire skin surface of the abdomen. Thoroughly remove the depilatory agent with gauze wetted with water or 70% ethanol. Thoroughly dry the animal.
- Prior to prepping the abdomen, tape the mouse's limbs to the heating pad or operative surface at the end of each leg to prevent the mouse from shifting during surgery. Disinfect the abdomen with chlorhexidine. Include the entire shaved area as well as the hair on the edges of the shaved area. Place a sterile drape over the mouse (Figure 1A).
- Prior to draping, cut a small ovoid or rectangular window into the drape to expose the prepped abdomen.
NOTE: The window should expose the entire abdomen of the mouse but cover the limbs, tail, head and thorax. We cut a 4 x 3 cm2 window for our operations (Figure 1B). - Make a small slit along the drape midline, from the bottom edge towards the ovoid or rectangular window, to allow access to the rectum during the procedure (Figure 1C).
- Prior to draping, cut a small ovoid or rectangular window into the drape to expose the prepped abdomen.
- Make a skin incision. Start along the midline of the lower abdomen and incise skin with scissors vertically to the xiphoid. Incise only the skin and avoid cutting the abdominal wall musculature (Figure 2A).
- Grasp the midline of the abdominal wall with forceps and lift away from the abdominal contents. Taking care not to injure intraabdominal structures, incise the length of the midline abdominal fascia vertically with sharp scissors. Extend the length of the skin incision (Figure 2B).
- When the intestines are exposed, locate the cecum and small bowel, and rotate the left-sided viscera medially to expose the descending and sigmoid colon. Coat the blunt probe tip with lubricating jelly and carefully advance the probe into the rectum to aid in the identification of the descending/sigmoid colon (Figure 3).
NOTE: Unlike humans, the mouse cecum is generally found in the left upper quadrant of the abdomen. Take care not to perforate the bowel wall with the probe in this step. If bowel wall is perforated, terminate the procedure and euthanize the mouse.- Avoid grasping the cecum with toothed or sharp forceps. Instead use atraumatic forceps, cotton tipped applicator, or gentle finger manipulation to rotate viscera.
- Use warmed sterile saline to ensure the exposed bowel is kept moist throughout the procedure.
- The colon will be on the left of midline and attached to the retroperitoneum via the mesentery. After identification, withdraw the blunt probe partially to facilitate colotomy.
- Perform transverse colotomy of the sigmoid colon.
- Gently grasp the sigmoid colon superior to the pelvic inlet with atraumatic forceps (Figure 4A).
- Using sharp micro-scissors, make a cut across the sigmoid colon perpendicular to the direction of the colon and extending across 80-90% the width of the colon. This will help prevent retraction of one end of the colon out of the surgical field, and facilitate repair (Figure 4B).
- If a mesenteric blood vessel is injured during the course of the visceral rotation or colotomy, gently hold pressure for up to 2 min with gauze or a cotton-tipped applicator. If bleeding persists after this time, place a figure-of-eight 8-0 monofilament suture around the point of bleeding.
NOTE: If the bowel shows signs of necrosis or injury after this, terminate the procedure and euthanize the mouse using a humane method approved by your institutional animal use committee.
- Repair the sigmoid colotomy.
- Using a Castroviejo needle driver, place 5-6 simple interrupted 8-0 nonabsorbable polypropylene stitches to repair the colostomy. Place sutures 1 mm apart from each other with 0.5 mm bites.
- Take care to avoid rolling the edges of bowel into the repair by taking slightly more serosa than mucosa with each stitch. Leave the tails 5 mm long. Take care not to accidentally catch surrounding bowel into the suture (Figure 4C).
NOTE: It is helpful to begin suturing at the near the mesenteric border of the colotomy, saving the anti-mesenteric-most portion of the colotomy for last. - As the repair progresses, advance the trans-anal blunt-tipped dissection probe to span the repair before the final sutures are placed. This will help prevent too narrow a repair and decrease rates of anastomotic stricture.
- Take care to avoid rolling the edges of bowel into the repair by taking slightly more serosa than mucosa with each stitch. Leave the tails 5 mm long. Take care not to accidentally catch surrounding bowel into the suture (Figure 4C).
- Using a Castroviejo needle driver, place 5-6 simple interrupted 8-0 nonabsorbable polypropylene stitches to repair the colostomy. Place sutures 1 mm apart from each other with 0.5 mm bites.
- Using a 10 mL syringe filled with warmed saline, irrigate the abdomen several times. If the surface underneath the mouse becomes soaked, dry the mouse or move the mouse to a dry surface in order to avoid hypothermia.
- Use a sterile gauze placed over the incision to soak up excess irrigation.
- At the final irrigation, fill the abdomen with sterile saline. Carefully advance a lubricated 18 G angiocatheter into the rectum and inflate with 0.5-1 cm3 of air. This will demonstrate any leaks in the anastomosis than can be repaired with another interrupted polypropylene suture using the sterile probe to span the anastomosis.
- Deflate the colon using the angiocatheter before removal.
- Make sure that there is no active bleeding. If there is, apply gentle pressure directly onto the point of bleeding for 1-2 min. Place hemostatic suture as described above if bleeding continues.
- Gently return the medialized viscera to its normal anatomic position (Figure 5A).
- Close the abdominal incision (Figure 5B).
- Using 4-0 absorbable braided sutures, place a running suture along the muscular layer of the abdominal wall starting at the top of the incision and finishing at the inferior aspect of the incision. Install a tie and cut the suture leaving 5 mm tails.
NOTE: Do not accidentally incorporate intestine in the abdominal wall closure. - Repeat with 4-0 monofilament suture to close the skin (Figure 5C).
- Using 4-0 absorbable braided sutures, place a running suture along the muscular layer of the abdominal wall starting at the top of the incision and finishing at the inferior aspect of the incision. Install a tie and cut the suture leaving 5 mm tails.
- Ensure that the entire animal is warm and dry, to prevent postoperative hypothermia.
- Carefully monitor the animal as it recovers from anesthesia. Administer 0.1 mg/kg buprenorphine every 72 h for 2-3 days after the procedure for pain control. During the recovery period, mice should be carefully examined daily for signs of abscess or peritoneal sepsis. Post-operative observation for pain and distress should include: restriction of movement including hunching type of posture, lack of eating or drinking, not proper grooming, excessive inflammation around wound, lack of normal healing at the site of incision, or not normal nesting. Any animal demonstrating any of these behaviors should be immediately euthanized by CO2 asphyxiation for a minimum of 10 min followed by bilateral thoracotomy.
- Mix 0.1 mg/mL of enrofloxacin with the mouse's drinking water for 7 days following surgery to prevent intra-abdominal infection or sepsis due to fecal contamination associated with the operation.
2) Harvesting Anastomotic Tissue
- Just before excising anastomotic tissue, euthanize the mouse using a humane method approved by the institutional animal use committee.
NOTE: Based on institutional policy, we euthanize our animals by placing them in a carbon dioxide chamber with 100% carbon dioxide gas infusion at 2 L/min for 5 min before performing bilateral thoracotomy. - Sterilize the abdomen with chlorhexidine followed by 70% ethanol.
- Re-incise the original midline incision with sharp scissors. Start at the lower pole of the previous incision and extend this cut superiorly to the xiphoid (Figure 6A).
- Using scissors, cut into the muscular layer, and after entering the peritoneal space, extend the cut to the top and bottom of the incision. Take care to avoid injuring the bowel when entering the abdomen or disrupting the anastomosis, as there may be bowel adhered to the incision or anastomotic site.
- Carefully perform a left medial visceral rotation exposing the repaired sigmoid colon (Figure 6B). There will likely be bowel to bowel adhesions surrounding the repair. Gently and bluntly dissect these adhesions away from the site of repair. Take great care not to disrupt the anastomosis.
- Identify the repair site by locating the tails of the polypropylene suture used for the repair. Using scissors and starting 1 cm proximal to the repair, transversely transect through the descending colon. Repeat this step 1 cm distal to site of anastomosis.
- Remove the sigmoidal segment by sharply dissecting the colon from the attached mesentery along the posterior border of the colon with fine scissors (Figure 7).
- Identify the mesenteric border of the colon specimen and use fine scissors to cut along the length of the mesenteric border in a longitudinal fashion. This will create a square or rectangular section of colon from the cylindrical specimen previously removed (Figure 8).
- Use a marking pen to demarcate the anti-mesenteric border of the colon prior to removal of the colon segment. After removal, make a longitudinal cut along the length of the colon opposite the marking. Fix, embed and cut sample tissue as needed for histology and/or immunostaining.
Representative Results
At 7 days following surgery, the anastomosis should be well healed. The anastomosis can be harvested at time points before and after seven days to better illustrate the stages of anastomotic healing. In Figure 9, the histological analysis shows a fibrotic (collagen visualized by trichrome staining) response mediated by smooth muscle alpha-actin positive myofibroblasts. However, we have found that results from histological analyses vary between mice, specifically the degree of re-epithelialization and concentration of myofibroblasts at the anastomotic site. We report mortality following this procedure to be less than 10%, with more than 90% of surviving mice having well-healed anastomoses at 7 days.
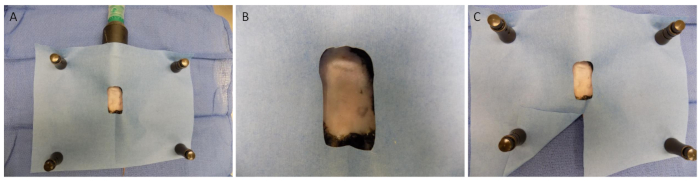
Figure 1: Draping and surgical preparation. (A) A Sterile drape is placed over the mouse after the abdomen has been prepped. (B) A small oval or rectangle is cut into the drape to expose the abdomen. (C) A small slit should be made along the midline of the drape near the ovoid or rectangular window, to allow access to the rectum during the procedure. Please click here to view a larger version of this figure.
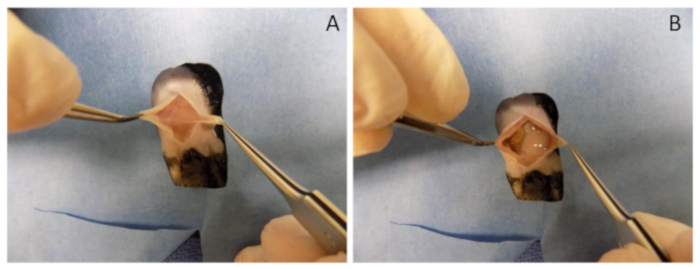
Figure 2: Incisions. (A) A skin incision is made while taking care not to injure the peritoneum.(B) The peritoneum is elevated away from the abdominal contents and incised exposing the intra-abdominal contents.
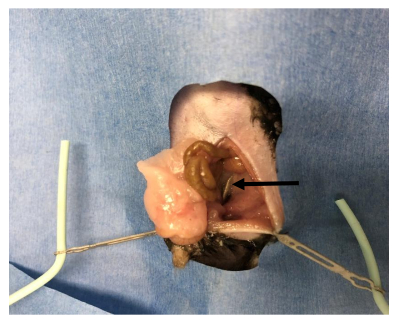
Figure 3: Probe placement. A blunt probe is gently placed in the rectum. After locating the cecum, the contents of the abdomen are rotated medially visualizing the rectal probe previously placed.
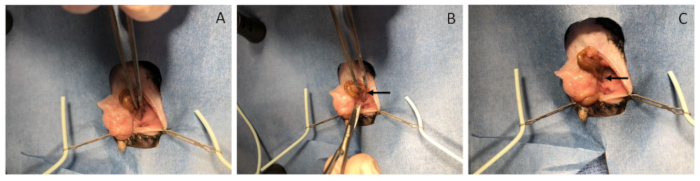
Figure 4: Colotomy and repair. (A) The sigmoid is grasped with atraumatic forceps. (B) Using sharp micro-scissors, make a cut across the sigmoid colon perpendicular to the direction of the colon and extending across 80-90% the width of the colon. (C) Using a Castro-Viejo needle driver, place 5-6 simple interrupted 8-0 polypropylene stitches to repair the colotomy. Sutures should be about 1 mm apart from each other with 0.5 mm bites. Please click here to view a larger version of this figure.
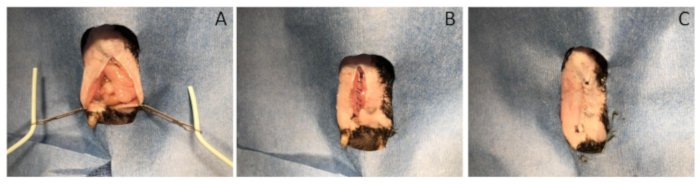
Figure 5: Wound closure. (A) The visceral organs are returned to their normal anatomic position. (B) The abdominal incision is closed with 4-0 braided suture. (C) The skin is closed with 4-0 monofilament suture. Please click here to view a larger version of this figure.
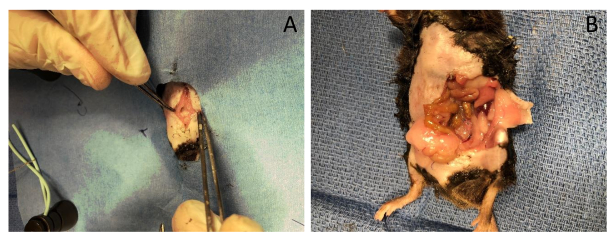
Figure 6: Incision post-surgery. (A) Re-open the original midline incision revealing the intra-abdominal contents. Be sure to extend the incision superiorly and laterally for better exposure to the sigmoid. (B) Rotate the viscera medially revealing the sigmoid anastomosis.
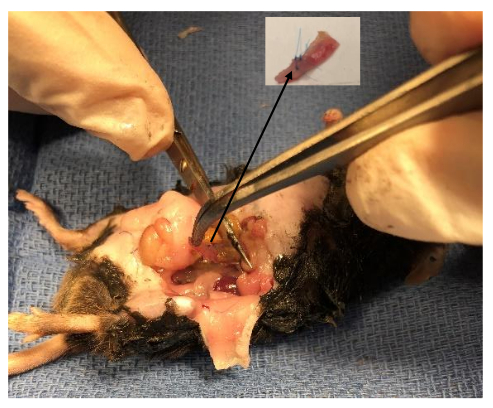
Figure 7: Harvesting the anastomosis. Remove the segment of bowel proximal and distal to the anastomosis by 1 cm on each side.
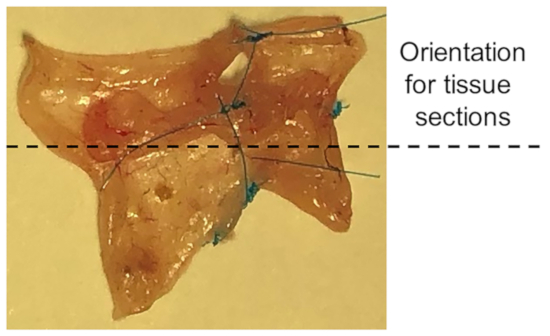
Figure 8: Orientation for tissue sections. Cut along the length of the mesenteric border in a longitudinal fashion. This leaves a rectangular specimen. This depicts specimen of sigmoid anastomosis.
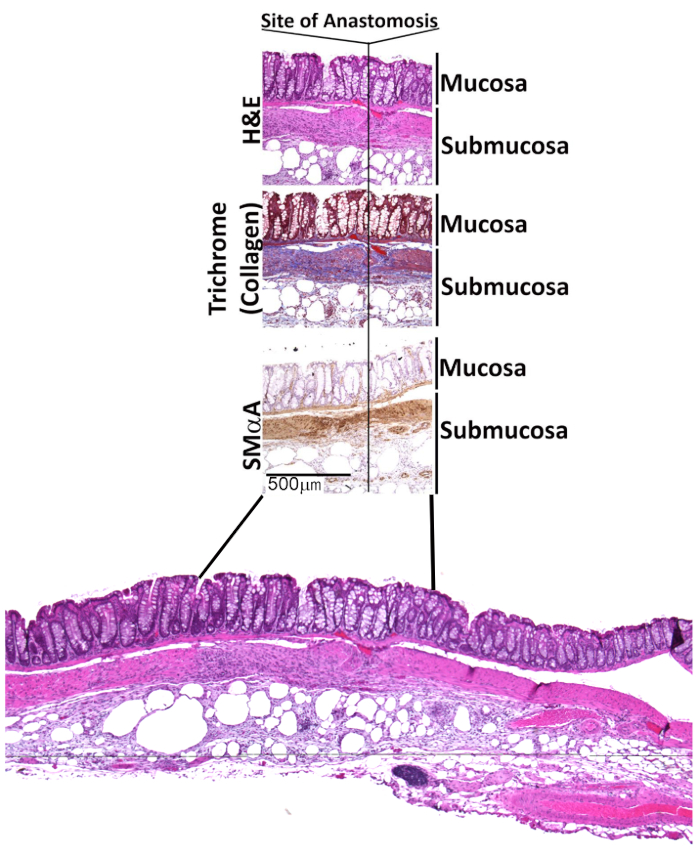
Figure 9: Histology and immunohistochemical analysis of a fully healed colonic anastomosis. Histological analysis shows fibrotic (collagen visualized by trichrome staining) response mediated by smooth muscle alpha-actin positive myofibroblasts.
Discussion
There are several key steps to ensure the success of and minimize the morbidity/mortality associated with this procedure. First, ensure careful handling of intestinal tissue and take care to prevent traction injury when rotating the viscera to expose the colon. Undue tension on the bowel or mesentery can injury the bowel or vasculature and cause bowel necrosis away from the site of anastomosis. Additionally, sharp forceps or forceps with teeth should not be used to handle the bowel.
Injury to the bowel separate from the site of planned anastomosis, either during visceral rotation or by perforation with the blunt probe, should result in euthanasia of the animal. In our experience, bowel necrosis or injury can lead to intra-abdominal sepsis or death, and a greatly increased inflammatory response that makes recovery of a healed anastomotic specimen prohibitively difficult.
Transection of the colon should span as much of the descending colon as possible, but care should be taken to not transect the bowel serosa in contact with the mesentery. In our experience, transection that includes the serosa-and especially the mesentery-can lead to retraction of the proximal section of the colon that makes anastomosis more difficult and can lead to further injury trying to recover the retracted segment.
Anastomosis can be repaired with 5-6 interrupted sutures. More may be needed for large mice. Sutures should be 1 mm apart. Place sutures on either side of the mesenteric border and then on alternating sides of the transection to meet in the middle at the anti-mesenteric border facilitates an easier and quicker anastomosis. Before the final suture, spanning the anastomosis with the blunt probe ensures a patent anastomosis and prevents "back-walling" the anastomosis, which would effectively ligate the colon.
Ensure hemostasis before abdominal closure. Despite postoperative antibiotic coverage, hemoperitoneum in the recovery period after intraabdominal contamination can lead to an unacceptably high rate of intra-abdominal infection and adhesive disease.
When first performing this procedure, several mice may be lost in the postoperative period. Our initial mortality rate approximated 30%. We have found this to be mainly due to anastomotic stricture leading to obstruction and bowel necrosis, breakdown of anastomotic repair, or bowel wall necrosis away from site of anastomosis likely due to ischemia after mesenteric injury. All of these can lead to intra-abdominal infection, sepsis and death. Once experienced in this procedure, operating surgeon should experience a mortality rate of under 10%. Evidence of subclinical anastomotic site leak, illustrated by purulence and dense adhesions at the site of anastomosis, were present in a further 10%. While this procedure can be done without the aid of magnification, its largest drawback as a reproducible experimental model is the relative technical challenge when compared to ileocecal or ascending colon anastomoses. We found surgical loupes exceedingly helpful in performing the anastomosis while minimizing trauma to the bowel wall. A surgical microscope may also be used, but we did not find that necessary.
Many techniques have been used to describe animal models for colonic anastomosis; particularly utilizing rat and rabbit models. We have found there to be a paucity of reproducible procedures of descending colon anastomosis in mouse models; we suspect this is due to the smaller size of mice increasing the technical difficulty of anastomotic procedures. However, mice have greater selection and wider availability of transgenic lines and antibodies, which to this point were unavailable to be used in a colon anastomosis model. We developed the current model to unify the mouse model with a model of colon anastomosis.
Our experience with this procedure includes both male and female mice of varying sizes and ages. At this point, we have not compared anastomotic wound healing results between ages, sizes or sexes of mice, though differences may certainly exist, and this model would allow that comparison.
We believe that this model can be easily and consistently reproduced and used to study anastomotic wound healing. Although we recommend first specimen harvests at seven days, earlier and later time points may be helpful in illustrating a timeline of anastomotic healing; demonstrating, for example, early migration of inflammatory cells and mediators into the anastomotic site and the deposition of collagen by fibroblasts in the following weeks.
Declarações
The authors have nothing to disclose.
Acknowledgements
None
Materials
| C57BL6/J mice | Jackson Labs | #00664 | |
| Fisherbrand Absorbent Underpads, 20" x 24" | Fisher Scientific | 14-206-62 | |
| Polylined Sterile Field, 18" x 24" | Busse Hospital Disposables | 696 | Cut a rectangular hole |
| Isothesia isoflurane | Henry Schein | 50033 | |
| Fisherbrand Sterile cotton gauze pad, 4" x 4" | Fisher Scientific | 22-415-469 | |
| Puralube petrolatum ophthalmic ointment, 1/8 oz. tube | Dechra Veterinary Products | NDC 17033-211-38 | |
| Nair depilatory cream | Church & Dwight Co. | 22339-05 | |
| Buprenex buprenorphine 0.3 mg/ml | Reckitt Benckiser Pharma Inc | NDC 12496-0757-5 | |
| 1 cc insulin syringe, 27 G | Becton Dickinson | 329412 | |
| Chloraprep Shampoo | Medline | APL82287 | |
| Webcol alcohol prep swabs | Covidien | 6818 | |
| BioGel PI surgical gloves | Mölnlycke Health Care | ALA42675Z | |
| Micro Forceps with teeth | Roboz | RS-5150 | |
| Fine scissors- sharp | Fine Science Tools | 14060-09 | |
| Straight serrated forceps | Fine Science Tools | 11050-10 | |
| Castro-Viejo needle driver | Fine Science Tools | 12565-14 | |
| 0.9% Sodium Chloride Irrigation | Baxter | BHL2F7121 | Warm to 37 °C prior to use |
| 10 ml syringe | Becton Dickinson | 309604 | |
| 4-0 Vicryl absorbable braided suture, 18", PS-2 needle | Henry Schein | 6546037 | |
| Blue monofilament suture 24” BV-1 needle | Henry Schein | 8305H | Usually comes double-armed. Cut the suture at the midway point to generate two usable sutures. |
| Cole-Parmer Microscissors, Standard Grade, Straight, 4". | Cole- Parmer | EW-10818-06 | |
| Medline Sterile lubricating jelly | Medline | MDS032273H |
Referências
- Murrell, Z. A., Stamos, M. J. Reoperation for anastomotic failure. Clinics in colon and rectal surgery. 19, 213-216 (2006).
- Shaban, F., Carney, K., McGarry, K., Holtham, S. Perforated Diverticulitis: To Anastomose or Not to Anastomose? A Systematic Review and Meta-Analysis. International journal of surgery (London, England). , (2018).
- Chambers, W. M., Mortensen, N. J. Postoperative leakage and abscess formation after colorectal surgery. Best practice, research. Clinical gastroenterology. 18, 865-880 (2004).
- Ashkanani, F., Krukowski, Z. H. Surgery – Oxford International Edition. Intestinal Anastomosis. 20, 104-107 (2002).
- Raptis, D., Pramateftakis, M. G., Kanellos, I. Our 20-year experience with experimental colonic anastomotic healing. Journal of medicine and life. 11, 5-14 (2018).
- Pommergaard, H. C., Rosenberg, J., Schumacher-Petersen, C., Achiam, M. P. Choosing the best animal species to mimic clinical colon anastomotic leakage in humans: a qualitative systematic review. European surgical research. Europaische chirurgische Forschung. Recherches chirurgicales europeennes. 47, 173-181 (2011).
- Komen, N., et al. Colorectal anastomotic leakage: a new experimental model. The Journal of surgical research. 155, 7-12 (2009).
- Perry, T., Borowiec, A., Dicken, B., Fedorak, R., Madsen, K. Murine ileocolic bowel resection with primary anastomosis. Journal of visualized experiments : JoVE. , e52106 (2014).
- Pommergaard, H. C., Achiam, M. P., Rosenberg, J. Colon anastomotic leakage: improving the mouse model. Surgery today. 44, 933-939 (2014).
- Bosmans, J. W. A. M., Jongen, A. C. H. M., Bouvy, N. D., Derikx, J. P. M. Colorectal anastomotic healing: why the biological processes that lead to anastomotic leakage should be revealed prior to conducting intervention studies. BMC Gastroenterology. 15, 180 (2015).

